Subcutaneous and Mediastinal Emphysema Followed by Group A Beta-Hemolytic Streptococci Mediastinitis. A Complicated Course after Adenotonsillectomy: Case Report
Abstract
1. Introduction
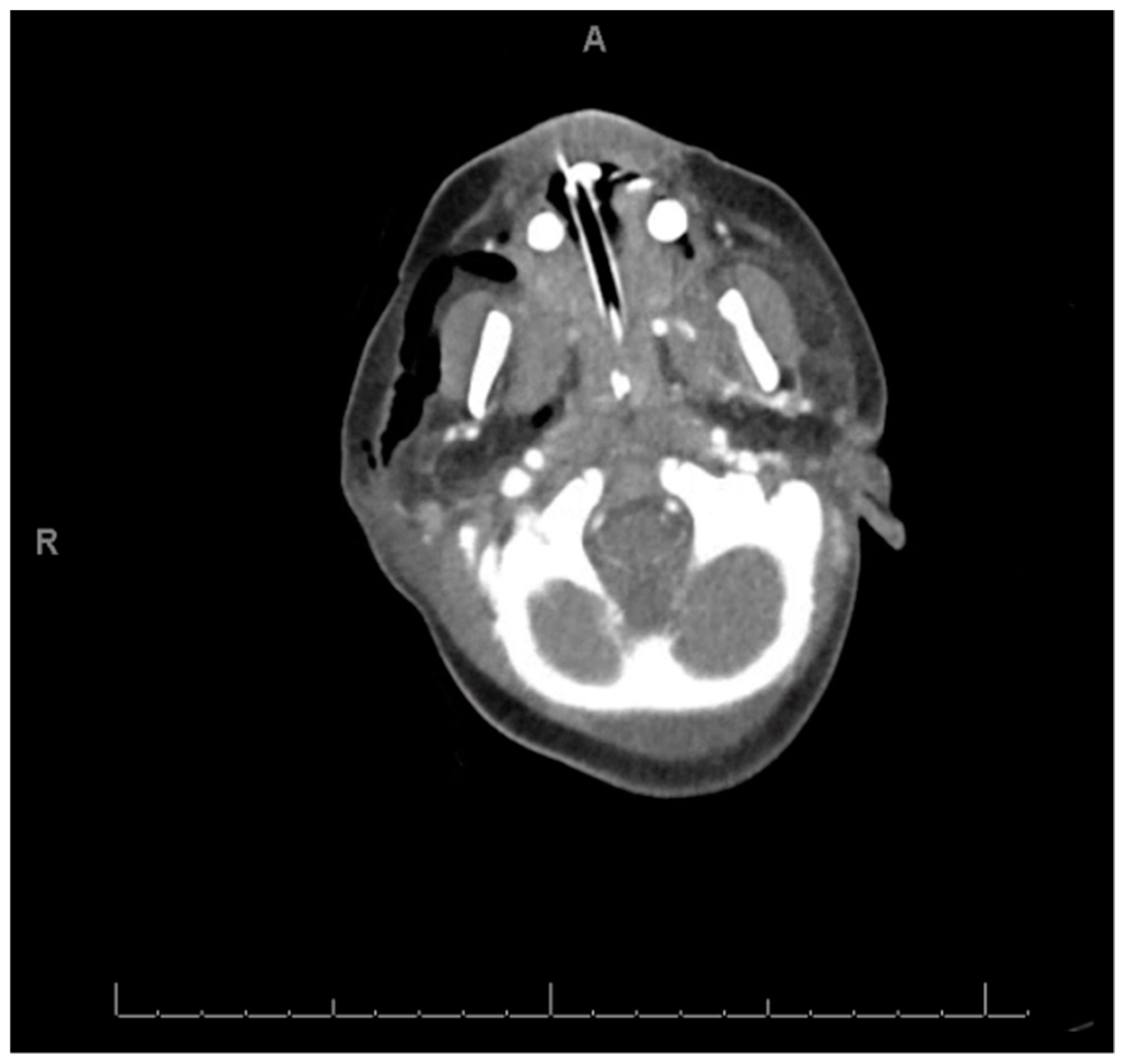
2. Case Report
3. Discussion
4. Conclusions
Consent for Publication
Author Contributions
Funding
Conflicts of Interest
Appendix A
References
- Greig, S. Current perspectives on the role of tonsillectomy. J. Paediatr. Child Health 2017, 53, 1065–1070. [Google Scholar] [CrossRef] [PubMed]
- Heiser, C.; Basile, N.; Giger, R.; Cao Van, H.; Guinand, N.; Hörmann, K.; Stuck, B.A. Taste Disturbance Following Tonsillectomy—A Prospective Study. Laryngoscope 2010, 120, 2119–2124. [Google Scholar] [CrossRef] [PubMed]
- Heiser, C.; Basile, N.; Giger, R.; Cao Van, H.; Guinand, N.; Hörmann, K.; Stuck, B.A. Taste Disorders after Tonsillectomy: A long-term follow-up. Laryngoscope 2012, 122, 1265–1266. [Google Scholar] [CrossRef] [PubMed]
- De Luca Canto, G.; Pacheco-Pereira, C.; Aydinoz, S.; Bhattacharjee, R.; Tan, H.; Kheirandish-Gozal, L.; Flores-Mir, C.; Gozal, D. Adenotonsillectomy Complications: A Meta-analysis. Pediatrics 2015, 136, 702–718. [Google Scholar] [CrossRef] [PubMed]
- Kendrick, D.; Gibbin, K. An audit of the complications of paediatric tonsillectomy, adenoidectomy and adenotonsillectomy. Clin. Otolaryngol. Allied Sci. 1993, 18, 115–117. [Google Scholar] [CrossRef] [PubMed]
- Richtlijn Ziekten van Adenoïd en Tonsillen in de Tweede lijn 2014; Nederlandse Vereniging voor KNO-heelkunde en Heelkunde van het Hoofd-Halsgebied: Utrecht, The Netherlands, 2014.
- Mistry, D.; Kelly, G. Consent for tonsillectomy. Clin. Otolaryngol. Allied Sci. 2004, 29, 362–368. [Google Scholar] [CrossRef] [PubMed]
- Timmers-Raaijmaakers, B.C.M.S.; Wolfs, T.F.W.; Jansen, N.J.G.; Bos, A.P.; van Vught, A.J. Invasive group A streptococcal infection after tonsillectomy. Pediatr. Infect. Dis. J. 2003, 22, 929–930. [Google Scholar] [CrossRef]
- Saravakos, P.; Taxeidis, M.; Kastanioudakis, I.; Reichel, O. Subcutaneous emphysema as a complication of tonsillectomy: Systematic literature review and case report. Iran. J. Otorhinolaryngol. 2018, 30, 3–10. [Google Scholar]
- Gillot, C.; Tombu, S.; Crestani, V.; Huvelle, P.; Moreau, P. Subcutaneous emphysema and mediastinitis: Unusual complications of tonsillectomy. B-ENT 2005, 1, 197–200. [Google Scholar]
- Panerari, A.C.; Soter, A.C.; Silva, F.L.; Oliveira, L.F.; Neves, M.D.; Cedin, A.C. Onset of subcutaneous emphysema and pneumomediastinum after tonsillectomy: A case report. Braz. J. Otorhinolaryngol. 2005, 71, 94–96. [Google Scholar] [CrossRef]
- Kim, J.P.; Park, J.J.; Kang, H.S.; Song, M.S. Subcutaneous emphysema and pneumomediastinum after tonsillectomy. Am. J. Otolaryngol. 2010, 31, 212–215. [Google Scholar] [CrossRef] [PubMed]
- Tran, D.D.; Littlefield, P.D. Late presentation of subcutaneous emphysema and pneumomediastinum following elective tonsillectomy. Am. J. Otolaryngol. 2015, 36, 299–302. [Google Scholar] [CrossRef] [PubMed]
- Yelnoorkar, S.; Issing, W. Cervicofacial Surgical Emphysema following Tonsillectomy. Case Rep. Otolaryngol. 2014, 2014, 746152. [Google Scholar] [CrossRef] [PubMed]
- Miman, M.C.; Ozturan, O.; Durmus, M.; Kalcioglu, M.; Gedik, E. Cervical subcutaneous emphysema: An usual complication of adenotonsillectomy. Paediatr. Anaesth. 2001, 11, 491–493. [Google Scholar] [CrossRef] [PubMed]
- Sonne, J.E.; Kim, S.B.; Frank, D.K. Cervical necrotizing fasciitis as a complication of tonsillectomy. Otolaryngol. Head Neck Surg. 2001, 125, 670–672. [Google Scholar] [CrossRef] [PubMed]
- Shine, N.P.; Sader, C.; Coates, H. Cervicofacial emphysema and pneumomediastinum following pediatric adenotonsillectomy: A rare complication. Int. J. Pediatr. Otorhinolaryngol. 2005, 69, 1579–1582. [Google Scholar] [CrossRef] [PubMed]
- Piotrowski, S.; Jesikiewicz, D. Mediastinal emphysema as a complication of the tonsilloadenotomy in child. Otolaryngol. Pol. 2009, 63, 528–531. [Google Scholar] [CrossRef]
- Ero, O.; Aydin, E. A Rare Complication of Tonsillectomy: Subcutaneous Emphysema. Turk. Arch. Otorhinolaryngol. 2016, 54, 172–174. [Google Scholar]
- Frampton, S.J.; Ward, M.J.A.; Sunkaraneni, V.S.; Ismail-Koch, H.; Sheppard, Z.A.; Salib, R.J.; Jain, P.K. Guillotine versus dissection tonsillectomy: Randomized controlled trial. J. Laryngol. Otol. 2012, 126, 1142–1149. [Google Scholar] [CrossRef]
- Smith, S. Tonsillotomy: An alternative surgical option to total tonsillectomy in children with obstructive sleep apnoea. Aus. Fam. Physician 2016, 45, 894–896. [Google Scholar]
- Hung, M.H.; Shih, P.Y.; Yang, Y.M.; Lan, J.Y.; Fan, S.Z.; Jeng, C.S. Cervicofacial subcutaneous emphysema following tonsillectomy: Implications for anesthesiologists. Acta Anaesthesiol. Taiwan 2009, 47, 134–137. [Google Scholar] [CrossRef]
- Stewart, A.E.; Brewster, D.F.; Bernstein, P.E. Subcutaneous emphysema and pneumomediastinum complication tonsillectomy. Arch. Otolaryngol. Head Neck Surg. 2004, 130, 1324–1327. [Google Scholar] [CrossRef] [PubMed]
- Ferguson, C.C.; McGarry, G.P.; Beckman, I.H.; Brder, M. Surgical emphysema complicating tonsillectomy and dental extraction. Can. Med. Assoc. J. 1955, 72, 847–848. [Google Scholar] [PubMed]
- Pratt, L.W.; Hornberger, H.R.; Moore, V.J. Mediastinal emphysema complicating tonsillectomy and adenoidectomy. Ann. Otol. Rhinol. Laryngol. 1962, 71, 158–169. [Google Scholar] [CrossRef] [PubMed]
- Klug, T.E.; Henriksen, J.; Rusan, M.; Fuursted, K.; Ovesen, T. Bacteremia during quinsy and elective tonsillectomy: An evaluation of antibiotic prophylaxis recommendations for patients undergoing tonsillectomy. J. Cardiovasc. Pharmacol. Ther. 2012, 17, 298–302. [Google Scholar] [CrossRef] [PubMed]
- Low, D.E. Toxic shock syndrome: Major advances in pathogenesis, but not treatment. Crit. Care Clin. 2013, 29, 651–675. [Google Scholar] [CrossRef]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Duvekot, A.; van Heesch, G.; Veder, L. Subcutaneous and Mediastinal Emphysema Followed by Group A Beta-Hemolytic Streptococci Mediastinitis. A Complicated Course after Adenotonsillectomy: Case Report. Diagnostics 2019, 9, 11. https://doi.org/10.3390/diagnostics9010011
Duvekot A, van Heesch G, Veder L. Subcutaneous and Mediastinal Emphysema Followed by Group A Beta-Hemolytic Streptococci Mediastinitis. A Complicated Course after Adenotonsillectomy: Case Report. Diagnostics. 2019; 9(1):11. https://doi.org/10.3390/diagnostics9010011
Chicago/Turabian StyleDuvekot, Anne, Gwen van Heesch, and Laura Veder. 2019. "Subcutaneous and Mediastinal Emphysema Followed by Group A Beta-Hemolytic Streptococci Mediastinitis. A Complicated Course after Adenotonsillectomy: Case Report" Diagnostics 9, no. 1: 11. https://doi.org/10.3390/diagnostics9010011
APA StyleDuvekot, A., van Heesch, G., & Veder, L. (2019). Subcutaneous and Mediastinal Emphysema Followed by Group A Beta-Hemolytic Streptococci Mediastinitis. A Complicated Course after Adenotonsillectomy: Case Report. Diagnostics, 9(1), 11. https://doi.org/10.3390/diagnostics9010011



