Single Domain Antibodies as New Biomarker Detectors
Abstract
1. Introduction
2. Classes of Diagnostic Tests that Target Biomarkers
2.1. Non-Infectious Diseases and Non-Diseases
2.1.1. Diagnostics for Cancer
2.1.2. Diagnostics for Pregnancy
2.1.3. Diagnostics for Envenomation
2.2. Infectious Diseases
2.2.1. Diagnostics for Viral Disease
2.2.2. Diagnostics for Bacterial Disease
2.2.3. Diagnostics for Parasitic Diseases
3. Currently Available Antibody Binders for Detection of Biomarkers
3.1. Monoclonal Antibodies
3.2. Limitations of Conventional Monoclonal Antibodies
4. Phage Display Technology for New Biomarker Binder Discovery
5. Natural Single Domain Antibodies
5.1. VHH Heavy Chain Domain in Camelids
5.2. VNAR Heavy Chain Domain in Sharks
6. Use of Different Binders for Specific Applications
6.1. Applications of Camelids VHH Domain or Nanobodies®
6.2. Applications of Shark VNAR Domain
7. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Hendriksen, I.C.; Ferro, J.; Montoya, P.; Chhaganlal, K.D.; Seni, A.; Gomes, E.; Silamut, K.; Lee, S.J.; Lucas, M.; Chotivanich, K.; et al. Diagnosis, clinical presentation, and in-hospital mortality of severe malaria in HIV-coinfected children and adults in Mozambique. Clin. Infect. Dis. 2012, 55, 1144–1153. [Google Scholar] [CrossRef] [PubMed]
- Murphy, J.F. Reducing misdiagnosis in clinical practice. Ir. Med. J. 2009, 102, 68. [Google Scholar] [PubMed]
- Zegarra Montes, L.Z.; Sanchez Mejia, A.A.; Loza Munarriz, C.A.; Gutierrez, E.C. Semen and urine culture in the diagnosis of chronic bacterial prostatitis. Int. Braz. J. Urol. 2008, 34, 30–37; discussion 38–40. [Google Scholar] [CrossRef] [PubMed]
- Olson, W.C.; Smolkin, M.E.; Farris, E.M.; Fink, R.J.; Czarkowski, A.R.; Fink, J.H.; Chianese-Bullock, K.A.; Slingluff, C.L., Jr. Shipping blood to a central laboratory in multicenter clinical trials: Effect of ambient temperature on specimen temperature, and effects of temperature on mononuclear cell yield, viability and immunologic function. J. Transl. Med. 2011, 9, 26. [Google Scholar] [CrossRef] [PubMed]
- Wilson, M.L. General principles of specimen collection and transport. Clin. Infect. Dis. 1996, 22, 766–777. [Google Scholar] [CrossRef] [PubMed]
- Wongsrichanalai, C.; Barcus, M.J.; Muth, S.; Sutamihardja, A.; Wernsdorfer, W.H. A review of malaria diagnostic tools: Microscopy and rapid diagnostic test (RDT). Am. J. Trop. Med. Hyg. 2007, 77, 119–127. [Google Scholar] [PubMed]
- Thomson, S.; Lohmann, R.C.; Crawford, L.; Dubash, R.; Richardson, H. External quality assessment in the examination of blood films for malarial parasites within Ontario, Canada. Arch. Pathol. Lab. Med. 2000, 124, 57–60. [Google Scholar] [PubMed]
- Johnston, S.P.; Pieniazek, N.J.; Xayavong, M.V.; Slemenda, S.B.; Wilkins, P.P.; da Silva, A.J. PCR as a confirmatory technique for laboratory diagnosis of malaria. J. Clin. Microbiol. 2006, 44, 1087–1089. [Google Scholar] [CrossRef] [PubMed]
- Hulka, B.S. Overview of biological markers. In Biological Markers in Epidemiology; Oxford University Press: New York, NY, USA, 1990; pp. 3–15. [Google Scholar]
- Tiernan, J.P.; Perry, S.L.; Verghese, E.T.; West, N.P.; Yeluri, S.; Jayne, D.G.; Hughes, T.A. Carcinoembryonic antigen is the preferred biomarker for in vivo colorectal cancer targeting. Br. J. Cancer 2013, 108, 662–667. [Google Scholar] [CrossRef] [PubMed]
- Watabe-Rudolph, M.; Song, Z.; Lausser, L.; Schnack, C.; Begus-Nahrmann, Y.; Scheithauer, M.O.; Rettinger, G.; Otto, M.; Tumani, H.; Thal, D.R.; et al. Chitinase enzyme activity in CSF is a powerful biomarker of Alzheimer disease. Neurology 2012, 78, 569–577. [Google Scholar] [CrossRef] [PubMed]
- Dawson, S.J.; Tsui, D.W.; Murtaza, M.; Biggs, H.; Rueda, O.M.; Chin, S.F.; Dunning, M.J.; Gale, D.; Forshew, T.; Mahler-Araujo, B.; et al. Analysis of circulating tumor DNA to monitor metastatic breast cancer. N. Engl. J. Med. 2013, 368, 1199–1209. [Google Scholar] [CrossRef] [PubMed]
- Niedbala, R.S.; Mauck, C.; Harrison, P.; Doncel, G.F. Biomarker discovery: Validation and decision-making in product development. Sex. Transm. Dis. 2009, 36, S76–S80. [Google Scholar] [CrossRef] [PubMed]
- Rai, A.J.; Zhang, Z.; Rosenzweig, J.; Shih Ie, M.; Pham, T.; Fung, E.T.; Sokoll, L.J.; Chan, D.W. Proteomic approaches to tumor marker discovery. Arch. Pathol. Lab. Med. 2002, 126, 1518–1526. [Google Scholar] [PubMed]
- Lopez, M.M.; Bertolini, E.; Olmos, A.; Caruso, P.; Gorris, M.T.; Llop, P.; Penyalver, R.; Cambra, M. Innovative tools for detection of plant pathogenic viruses and bacteria. Int. Microbiol. 2003, 6, 233–243. [Google Scholar] [CrossRef] [PubMed]
- Kerner, J.; Lee, K.; Tandler, B.; Hoppel, C.L. VDAC proteomics: Post-translation modifications. Biochim. Biophys. Acta 2012, 1818, 1520–1525. [Google Scholar] [CrossRef] [PubMed]
- Demerdash, Z.A.; Diab, T.M.; Aly, I.R.; Mohamed, S.H.; Mahmoud, F.S.; Zoheiry, M.K.; Mansour, W.A.; Attia, M.E.; El-Bassiouny, A.E. Diagnostic efficacy of monoclonal antibody based sandwich enzyme linked immunosorbent assay (ELISA) for detection of Fasciola gigantica excretory/secretory antigens in both serum and stool. Parasites Vectors 2011, 4, 176. [Google Scholar] [CrossRef] [PubMed]
- Fujiwara, K.; Yoshizaki, Y.; Shin, M.; Miyazaki, T.; Saita, T.; Nagata, S. Immunocytochemistry for vancomycin using a monoclonal antibody that reveals accumulation of the drug in rat kidney and liver. Antimicrob. Agents Chemother. 2012, 56, 5883–5891. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.; Belov, L.; Solomon, M.J.; Chan, C.; Clarke, S.J.; Christopherson, R.I. Colorectal cancer cell surface protein profiling using an antibody microarray and fluorescence multiplexing. J. Vis. Exp. 2011. [Google Scholar] [CrossRef] [PubMed]
- Raponi, S.; de Propris, M.S.; Intoppa, S.; Milani, M.L.; Vitale, A.; Elia, L.; Perbellini, O.; Pizzolo, G.; Foa, R.; Guarini, A. Flow cytometric study of potential target antigens (CD19, CD20, CD22, CD33) for antibody-based immunotherapy in acute lymphoblastic leukemia: Analysis of 552 cases. Leuk. Lymphoma 2011, 52, 1098–1107. [Google Scholar] [CrossRef] [PubMed]
- Zandian, M.; Jungbauer, A. An immunoaffinity column with a monoclonal antibody as ligand for human follicle stimulating hormone. J. Sep. Sci. 2009, 32, 1585–1591. [Google Scholar] [CrossRef] [PubMed]
- Vora, A.J.; Nuttall, P.; James, V. Screening plasma HAV antibody sources. Lancet 1991, 338, 62. [Google Scholar] [CrossRef]
- Spillner, E.; Braren, I.; Greunke, K.; Seismann, H.; Blank, S.; du Plessis, D. Avian IgY antibodies and their recombinant equivalents in research, diagnostics and therapy. Biologicals 2012, 40, 313–322. [Google Scholar] [CrossRef] [PubMed]
- Hanly, W.C.; Artwohl, J.E.; Bennett, B.T. Review of Polyclonal Antibody Production Procedures in Mammals and Poultry. ILAR J. 1995, 37, 93–118. [Google Scholar] [CrossRef] [PubMed]
- Gaciarz, A.; Veijola, J.; Uchida, Y.; Saaranen, M.J.; Wang, C.; Horkko, S.; Ruddock, L.W. Systematic screening of soluble expression of antibody fragments in the cytoplasm of E. coli. Microb. Cell Fact. 2016, 15, 22. [Google Scholar] [CrossRef] [PubMed]
- Zielonka, S.; Empting, M.; Grzeschik, J.; Konning, D.; Barelle, C.J.; Kolmar, H. Structural insights and biomedical potential of IgNAR scaffolds from sharks. MAbs 2015, 7, 15–25. [Google Scholar] [CrossRef] [PubMed]
- Saerens, D.; Ghassabeh, G.H.; Muyldermans, S. Single-domain antibodies as building blocks for novel therapeutics. Curr. Opin. Pharmacol. 2008, 8, 600–608. [Google Scholar] [CrossRef] [PubMed]
- O’Kennedy, R.; Roben, P. Antibody engineering: An overview. Essays Biochem. 1991, 26, 59–75. [Google Scholar] [PubMed]
- Ahmad, Z.A.; Yeap, S.K.; Ali, A.M.; Ho, W.Y.; Alitheen, N.B.; Hamid, M. scFv antibody: Principles and clinical application. Clin. Dev. Immunol. 2012, 2012, 980250. [Google Scholar] [CrossRef] [PubMed]
- Tu, B.; Ziemann, R.N.; Tieman, B.C.; Hawksworth, D.J.; Tyner, J.; Scheffel, J.; Pinkus, M.S.; Brophy, S.E.; Werneke, J.M.; Gutierrez, R.; et al. Generation and characterization of chimeric antibodies against NS3, NS4, NS5, and core antigens of hepatitis C virus. Clin. Vaccine Immunol. 2010, 17, 1040–1047. [Google Scholar] [CrossRef] [PubMed]
- Karu, A.E.; Bell, C.W.; Chin, T.E. Recombinant Antibody Technology. ILAR J. 1995, 37, 132–141. [Google Scholar] [CrossRef]
- Azzazy, H.M.; Highsmith, W.E., Jr. Phage display technology: Clinical applications and recent innovations. Clin. Biochem. 2002, 35, 425–445. [Google Scholar] [CrossRef]
- Hoogenboom, H.R. Selecting and screening recombinant antibody libraries. Nat. Biotechnol. 2005, 23, 1105–1116. [Google Scholar] [CrossRef] [PubMed]
- Dick, H.M. Single domain antibodies. Br. Med. J. 1990, 300, 959–960. [Google Scholar] [CrossRef]
- Diaz, M.; Stanfield, R.L.; Greenberg, A.S.; Flajnik, M.F. Structural analysis, selection, and ontogeny of the shark new antigen receptor (IgNAR): Identification of a new locus preferentially expressed in early development. Immunogenetics 2002, 54, 501–512. [Google Scholar] [CrossRef] [PubMed]
- Stijlemans, B.; Caljon, G.; Natesan, S.K.; Saerens, D.; Conrath, K.; Perez-Morga, D.; Skepper, J.N.; Nikolaou, A.; Brys, L.; Pays, E.; et al. High affinity nanobodies against the Trypanosome brucei VSG are potent trypanolytic agents that block endocytosis. PLoS Pathog. 2011, 7, e1002072. [Google Scholar] [CrossRef] [PubMed]
- Shao, C.Y.; Secombes, C.J.; Porter, A.J. Rapid isolation of IgNAR variable single-domain antibody fragments from a shark synthetic library. Mol. Immunol. 2007, 44, 656–665. [Google Scholar] [CrossRef] [PubMed]
- Herrin, B.R.; Alder, M.N.; Roux, K.H.; Sina, C.; Ehrhardt, G.R.; Boydston, J.A.; Turnbough, C.L., Jr.; Cooper, M.D. Structure and specificity of lamprey monoclonal antibodies. Proc. Natl. Acad. Sci. USA 2008, 105, 2040–2045. [Google Scholar] [CrossRef] [PubMed]
- Akazawa-Ogawa, Y.; Takashima, M.; Lee, Y.H.; Ikegami, T.; Goto, Y.; Uegaki, K.; Hagihara, Y. Heat-induced irreversible denaturation of the camelid single domain VHH antibody is governed by chemical modifications. J. Biol. Chem. 2014, 289, 15666–15679. [Google Scholar] [CrossRef] [PubMed]
- Kovaleva, M.; Ferguson, L.; Steven, J.; Porter, A.; Barelle, C. Shark variable new antigen receptor biologics—A novel technology platform for therapeutic drug development. Expert Opin. Biol. Ther. 2014, 14, 1527–1539. [Google Scholar] [CrossRef] [PubMed]
- Tucker, J.D.; Bu, J.; Brown, L.B.; Yin, Y.P.; Chen, X.S.; Cohen, M.S. Accelerating worldwide syphilis screening through rapid testing: A systematic review. Lancet Infect. Dis. 2010, 10, 381–386. [Google Scholar] [CrossRef]
- Suppiah, J.; Thimma, J.S.; Cheah, S.H.; Vadivelu, J. Development and evaluation of polymerase chain reaction assay to detect Burkholderia genus and to differentiate the species in clinical specimens. FEMS Microbiol. Lett. 2010, 306, 9–14. [Google Scholar] [CrossRef] [PubMed]
- Saetiew, C.; Limpaiboon, T.; Jearanaikoon, P.; Daduang, S.; Pientong, C.; Kerdsin, A.; Daduang, J. Rapid detection of the most common high-risk human papillomaviruses by loop-mediated isothermal amplification. J. Virol. Methods 2011, 178, 22–30. [Google Scholar] [CrossRef] [PubMed]
- Tessitore, A.; Gaggiano, A.; Cicciarelli, G.; Verzella, D.; Capece, D.; Fischietti, M.; Zazzeroni, F.; Alesse, E. Serum biomarkers identification by mass spectrometry in high-mortality tumors. Int. J. Proteom. 2013, 2013, 125858. [Google Scholar] [CrossRef] [PubMed]
- Balmer, O.; Tostado, C. New fluorescence markers to distinguish co-infecting Trypanosoma brucei strains in experimental multiple infections. Acta Trop. 2006, 97, 94–101. [Google Scholar] [CrossRef] [PubMed]
- Petersen, E.R.B.; Olsen, D.A.; Christensen, H.; Hansen, S.B.; Christensen, C.; Brandslund, I. Rhodopsin in plasma from patients with diabetic retinopathy—Development and validation of digital ELISA by Single Molecule Array (Simoa) technology. J. Immunol. Methods 2017, 446, 60–69. [Google Scholar] [CrossRef] [PubMed]
- Ndao, M. Diagnosis of parasitic diseases: Old and new approaches. Interdiscip. Perspect. Infect. Dis. 2009, 2009, 278246. [Google Scholar] [CrossRef] [PubMed]
- Tangpukdee, N.; Duangdee, C.; Wilairatana, P.; Krudsood, S. Malaria diagnosis: A brief review. Korean J. Parasitol. 2009, 47, 93–102. [Google Scholar] [CrossRef] [PubMed]
- Esquivel-Velazquez, M.; Ostoa-Saloma, P.; Morales-Montor, J.; Hernandez-Bello, R.; Larralde, C. Immunodiagnosis of neurocysticercosis: Ways to focus on the challenge. J. Biomed. Biotechnol. 2011, 2011, 516042. [Google Scholar] [CrossRef] [PubMed]
- Hang, V.T.; Nguyet, N.M.; Trung, D.T.; Tricou, V.; Yoksan, S.; Dung, N.M.; van Ngoc, T.; Hien, T.T.; Farrar, J.; Wills, B.; et al. Diagnostic accuracy of NS1 ELISA and lateral flow rapid tests for dengue sensitivity, specificity and relationship to viraemia and antibody responses. PLoS Negl. Trop. Dis. 2009, 3, e360. [Google Scholar] [CrossRef] [PubMed]
- Houze, S.; Boutron, I.; Marmorat, A.; Dalichampt, M.; Choquet, C.; Poilane, I.; Godineau, N.; le Guern, A.S.; Thellier, M.; Broutier, H.; et al. Performance of rapid diagnostic tests for imported malaria in clinical practice: Results of a national multicenter study. PLoS ONE 2013, 8, e75486. [Google Scholar] [CrossRef] [PubMed]
- Masanja, M.I.; McMorrow, M.; Kahigwa, E.; Kachur, S.P.; McElroy, P.D. Health workers’ use of malaria rapid diagnostic tests (RDTs) to guide clinical decision making in rural dispensaries, Tanzania. Am. J. Trop. Med. Hyg. 2010, 83, 1238–1241. [Google Scholar] [CrossRef] [PubMed]
- Ng, W.Y.; Tiong, C.C.; Jacob, E. Maltose interference-free test strips for blood glucose testing at point-of-care: A laboratory performance evaluation. Diabetes Technol. Ther. 2010, 12, 889–893. [Google Scholar] [CrossRef] [PubMed]
- Lafleur, L.; Stevens, D.; McKenzie, K.; Ramachandran, S.; Spicar-Mihalic, P.; Singhal, M.; Arjyal, A.; Osborn, J.; Kauffman, P.; Yager, P.; et al. Progress toward multiplexed sample-to-result detection in low resource settings using microfluidic immunoassay cards. Lab. Chip 2012, 12, 1119–1127. [Google Scholar] [CrossRef] [PubMed]
- Sheppard, C.L.; Guiver, M.; Hartley, J.; Harrison, T.G.; George, R.C. Use of a multiplexed immunoassay for detection of serotype-specific Streptococcus pneumoniae antigen in pleural fluid and cerebrospinal fluid specimens. J. Med. Microbiol. 2011, 60, 1879–1881. [Google Scholar] [CrossRef] [PubMed]
- Taniuchi, M.; Verweij, J.J.; Noor, Z.; Sobuz, S.U.; Lieshout, L.; Petri, W.A., Jr.; Haque, R.; Houpt, E.R. High throughput multiplex PCR and probe-based detection with Luminex beads for seven intestinal parasites. Am. J. Trop. Med. Hyg. 2011, 84, 332–337. [Google Scholar] [CrossRef] [PubMed]
- Van Doorn, H.R.; Kinh, N.; Tuan, H.M.; Tuan, T.A.; Minh, N.N.; Bryant, J.E.; Hang, V.; Uyen le, T.T.; Thinh le, Q.; Anh, T.; et al. Clinical validation of a point-of-care multiplexed in vitro immunoassay using monoclonal antibodies (the MSD influenza test) in four hospitals in Vietnam. J. Clin. Microbiol. 2012, 50, 1621–1625. [Google Scholar] [CrossRef] [PubMed]
- Balboni, I.; Limb, C.; Tenenbaum, J.D.; Utz, P.J. Evaluation of microarray surfaces and arraying parameters for autoantibody profiling. Proteomics 2008, 8, 3443–3449. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization (WHO). Noncommunicable Diseases—Fact Sheet Updated June 2017; Media Centre: Geneva, Switzerland, 2017. [Google Scholar]
- Collier, A.; Ghosh, S.; McGlynn, B.; Hollins, G. Prostate cancer, androgen deprivation therapy, obesity, the metabolic syndrome, type 2 diabetes, and cardiovascular disease: A review. Am. J. Clin. Oncol. 2012, 35, 504–509. [Google Scholar] [CrossRef] [PubMed]
- Van Horn, L. What do cancer, diabetes, and cardiovascular disease have in common? J. Am. Diet. Assoc. 2009, 109, 1329. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization (WHO). World Cancer Report 2008; World Health Organization (WHO): Geneva, Switzerland, 2008; p. 512. [Google Scholar]
- Ferlay, J.; Bray, F.; Pisani, P.; Parkin, D.M. Cancer, Incidence, Mortality and Prevalence Worldwide IARC CancerBase; Version 2.0; IARC Press: Lyon, France, 2004. [Google Scholar]
- Li, X.; Scarfe, A.; King, K.; Fenton, D.; Butts, C.; Winget, M. Timeliness of cancer care from diagnosis to treatment: A comparison between patients with breast, colon, rectal or lung cancer. Int. J. Qual. Health Care 2013, 25, 197–204. [Google Scholar] [CrossRef] [PubMed]
- Gretzer, M.B.; Partin, A.W. PSA markers in prostate cancer detection. Urol. Clin. N. Am. 2003, 30, 677–686. [Google Scholar] [CrossRef]
- Crawford, N.P.; Colliver, D.W.; Galandiuk, S. Tumor markers and colorectal cancer: Utility in management. J. Surg. Oncol. 2003, 84, 239–248. [Google Scholar] [CrossRef] [PubMed]
- Cheung, K.L.; Robertson, F.R. Objective measurement of remission and progression in metastatic breast cancer by the use of serum tumour markers. Minerva Chir. 2003, 58, 297–303. [Google Scholar] [PubMed]
- Hanna, W.M.; Kahn, H.J.; Pienkowska, M.; Blondal, J.; Seth, A.; Marks, A. Defining a test for HER-2/neu evaluation in breast cancer in the diagnostic setting. Mod. Pathol. 2001, 14, 677–685. [Google Scholar] [CrossRef] [PubMed]
- Trompetas, V.; Panagopoulos, E.; Priovolou-Papaevangelou, M.; Ramantanis, G. Giant benign true cyst of the spleen with high serum level of CA 19-9. Eur. J. Gastroenterol. Hepatol. 2002, 14, 85–88. [Google Scholar] [CrossRef] [PubMed]
- Anderiesz, C.; Quinn, M.A. Screening for ovarian cancer. Med. J. Aust. 2003, 178, 655–656. [Google Scholar] [PubMed]
- Wayner, E.A.; Quek, S.I.; Ahmad, R.; Ho, M.E.; Loprieno, M.A.; Zhou, Y.; Ellis, W.J.; True, L.D.; Liu, A.Y. Development of an ELISA to detect the secreted prostate cancer biomarker AGR2 in voided urine. Prostate 2012, 72, 1023–1034. [Google Scholar] [CrossRef] [PubMed]
- Cheng, H.Y.; Lai, L.J.; Ko, F.H. Rapid and sensitive detection of rare cancer cells by the coupling of immunomagnetic nanoparticle separation with ELISA analysis. Int. J. Nanomed. 2012, 7, 2967–2973. [Google Scholar] [CrossRef] [PubMed]
- Tritschler, S.; Scharf, S.; Karl, A.; Tilki, D.; Knuechel, R.; Hartmann, A.; Stief, C.G.; Zaak, D. Validation of the diagnostic value of NMP22 BladderChek test as a marker for bladder cancer by photodynamic diagnosis. Eur. Urol. 2007, 51, 403–407; discussion 407–408. [Google Scholar] [CrossRef] [PubMed]
- Yang, M.; Javadi, A.; Li, H.; Gong, S. Ultrasensitive immunosensor for the detection of cancer biomarker based on graphene sheet. Biosens. Bioelectron. 2010, 26, 560–565. [Google Scholar] [CrossRef] [PubMed]
- Adams, A.A.; Okagbare, P.I.; Feng, J.; Hupert, M.L.; Patterson, D.; Gottert, J.; McCarley, R.L.; Nikitopoulos, D.; Murphy, M.C.; Soper, S.A. Highly efficient circulating tumor cell isolation from whole blood and label-free enumeration using polymer-based microfluidics with an integrated conductivity sensor. J. Am. Chem. Soc. 2008, 130, 8633–8641. [Google Scholar] [CrossRef] [PubMed]
- Kashani-Sabet, M.; Rangel, J.; Torabian, S.; Nosrati, M.; Simko, J.; Jablons, D.M.; Moore, D.H.; Haqq, C.; Miller, J.R., 3rd; Sagebiel, R.W. A multi-marker assay to distinguish malignant melanomas from benign nevi. Proc. Natl. Acad. Sci. USA 2009, 106, 6268–6272. [Google Scholar] [CrossRef] [PubMed]
- Moraes, G.S.; Amaral Cristovam, R.; Savaris, R.F. Comparative analysis of the accuracy of urinary hCG tests in vitro. Rev. Assoc. Med. Bras. 2011, 57, 516–522. [Google Scholar] [CrossRef] [PubMed]
- Taylor, A.E.; Khoury, R.H.; Crowley, W.F., Jr. A comparison of 13 different immunometric assay kits for gonadotropins: Implications for clinical investigation. J. Clin. Endocrinol. Metab. 1994, 79, 240–247. [Google Scholar] [PubMed]
- Coons, S.J. A look at the purchase and use of home pregnancy-test kits. Am. Pharm. 1989, NS29, 46–48. [Google Scholar] [PubMed]
- Grudzinskas, J.G.; Lenton, E.A.; Gordon, Y.B.; Kelso, I.M.; Jeffrey, D.; Sobowale, O.; Chard, T. Circulating levels of pregnancy-specific beta1-glycoprotein in early pregnancy. Br. J. Obstet. Gynaecol. 1977, 84, 740–742. [Google Scholar] [CrossRef] [PubMed]
- Bohn, H.; Kraus, W.; Winckler, W. New soluble placental tissue proteins: Their isolation, characterization, localization and quantification. Placenta Suppl. 1982, 4, 67–81. [Google Scholar] [PubMed]
- Whyte, A.; Heap, R.B. Reproductive immunology. Early pregnancy factor. Nature 1983, 304, 121–122. [Google Scholar] [CrossRef] [PubMed]
- Morton, H.; Rolfe, B.; Clunie, G.J. An early pregnancy factor detected in human serum by the rosette inhibition test. Lancet 1977, 1, 394–397. [Google Scholar] [CrossRef]
- Snyder, J.A.; Haymond, S.; Parvin, C.A.; Gronowski, A.M.; Grenache, D.G. Diagnostic considerations in the measurement of human chorionic gonadotropin in aging women. Clin. Chem. 2005, 51, 1830–1835. [Google Scholar] [CrossRef] [PubMed]
- Matsui, H.; Kihara, M.; Usui, H.; Tate, S.; Mitsuhashi, A.; Shozu, M. Comparison of 2 commercially available human chorionic gonadotropin immunoassays used in the management of gestational trophoblastic neoplasia. J. Reprod. Med. 2009, 54, 631–635. [Google Scholar] [PubMed]
- Vaitukaitis, J.L. Development of the home pregnancy test. Ann. N. Y. Acad. Sci. 2004, 1038, 220–222. [Google Scholar] [CrossRef] [PubMed]
- Lapthorn, A.J.; Harris, D.C.; Littlejohn, A.; Lustbader, J.W.; Canfield, R.E.; Machin, K.J.; Morgan, F.J.; Isaacs, N.W. Crystal structure of human chorionic gonadotropin. Nature 1994, 369, 455–461. [Google Scholar] [CrossRef] [PubMed]
- Alfthan, H.; Schroder, J.; Fraser, R.; Koskimies, A.; Halila, H.; Stenman, U.H. Choriogonadotropin and its beta subunit separated by hydrophobic-interaction chromatography and quantified in serum during pregnancy by time-resolved immunofluorometric assays. Clin. Chem. 1988, 34, 1758–1762. [Google Scholar] [PubMed]
- Spona, J.; Dancoine, F.; Lorenz, U.; Valente, D.; Wasley, G.D. Early detection of pregnancy by new beta-hCG monoclonal urine test. Gynecol. Obstet. Investig. 1985, 19, 6–10. [Google Scholar] [CrossRef]
- Bastian, L.A.; Nanda, K.; Hasselblad, V.; Simel, D.L. Diagnostic efficiency of home pregnancy test kits. A meta-analysis. Arch. Fam. Med. 1998, 7, 465–469. [Google Scholar] [CrossRef] [PubMed]
- Cole, L.A.; Khanlian, S.A. The need for a quantitative urine hCG assay. Clin. Biochem. 2009, 42, 676–683. [Google Scholar] [CrossRef] [PubMed]
- De Medeiros, S.F.; Amato, F.; Matthews, C.D.; Norman, R.J. Comparison of specific immunoassays for detection of the β-core human chorionic gonadotrophin fragment in body fluids. J. Endocrinol. 1992, 135, 161–174. [Google Scholar] [CrossRef] [PubMed]
- Norman, R.J.; Lowings, C.; Chard, T. Dipstick method for human chorionic gonadotropin suitable for emergency use on whole blood and other fluids. Lancet 1985, 1, 19–20. [Google Scholar] [CrossRef]
- Chow, S.N.; Ouyang, P.C.; Chu, C.T.; Lee, C.Y. Rapid and simple immunoassays for measurement of human chorionic gonadotropin using monoclonal antibodies. J. Formos. Med. Assoc. 1990, 89, 792–798. [Google Scholar] [PubMed]
- Spyropoulou, I.; Karamalegos, C.; Bolton, V.N. A prospective randomized study comparing the outcome of in vitro fertilization and embryo transfer following culture of human embryos individually or in groups before embryo transfer on day 2. Hum. Reprod. 1999, 14, 76–79. [Google Scholar] [CrossRef] [PubMed]
- Holl, K.; Lundin, E.; Kaasila, M.; Grankvist, K.; Afanasyeva, Y.; Hallmans, G.; Lehtinen, M.; Pukkala, E.; Surcel, H.M.; Toniolo, P.; et al. Effect of long-term storage on hormone measurements in samples from pregnant women: The experience of the Finnish Maternity Cohort. Acta Oncol. 2008, 47, 406–412. [Google Scholar] [CrossRef] [PubMed]
- Karalliedde, L. Animal toxins. Br. J. Anaesth. 1995, 74, 319–327. [Google Scholar] [CrossRef] [PubMed]
- Pollyea, D.A.; George, T.I.; Corless, C.; Gotlib, J. When yellow jackets attack: Recurrent and severe anaphylactic reactions to insect bites and stings. Am. J. Hematol. 2009, 84, 843–846. [Google Scholar] [CrossRef] [PubMed]
- Nowatzki, J.; Sene, R.V.; Paludo, K.S.; Rizzo, L.E.; Souza-Fonseca-Guimaraes, F.; Veiga, S.S.; Nader, H.B.; Franco, C.R.; Trindade, E.S. Brown spider (Loxosceles intermedia) venom triggers endothelial cells death by anoikis. Toxicon 2012, 60, 396–405. [Google Scholar] [CrossRef] [PubMed]
- Cruz, L.S.; Vargas, R.; Lopes, A.A. Snakebite envenomation and death in the developing world. Ethn. Dis. 2009, 19, S42–S46. [Google Scholar]
- Girish, K.S.; Kemparaju, K. Overlooked issues of snakebite management: Time for strategic approach. Curr. Top. Med. Chem. 2011, 11, 2494–2508. [Google Scholar] [CrossRef] [PubMed]
- Theakston, R.D.; Lloyd-Jones, M.J.; Reid, H.A. Micro-ELISA for detecting and assaying snake venom and venom-antibody. Lancet 1977, 2, 639–641. [Google Scholar] [CrossRef]
- Rial, A.; Morais, V.; Rossi, S.; Massaldi, H. A new ELISA for determination of potency in snake antivenoms. Toxicon 2006, 48, 462–466. [Google Scholar] [CrossRef] [PubMed]
- Cox, J.C.; Moisidis, A.V.; Shepherd, J.M.; Drane, D.P.; Jones, S.L. A novel format for a rapid sandwich EIA and its application to the identification of snake venoms. J. Immunol. Methods 1992, 146, 213–218. [Google Scholar] [CrossRef]
- Sutherland, S.K. Antivenom use in Australia. Premedication, adverse reactions and the use of venom detection kits. Med. J. Aust. 1992, 157, 734–739. [Google Scholar] [PubMed]
- Sutherland, S.K.; Leonard, R.L. Snakebite deaths in Australia 1992–1994 and a management update. Med. J. Aust. 1995, 163, 616–618. [Google Scholar] [PubMed]
- Trevett, A.J.; Lalloo, D.G.; Nwokolo, N.C.; Theakston, D.G.; Naraqi, S.; Warrell, D.A. Venom detection kits in the management of snakebite in Central province, Papua New Guinea. Toxicon 1995, 33, 703–705. [Google Scholar] [CrossRef]
- Tibballs, J.; Kuruppu, S.; Hodgson, W.C.; Carroll, T.; Hawdon, G.; Sourial, M.; Baker, T.; Winkel, K. Cardiovascular, haematological and neurological effects of the venom of the Papua New Guinean small-eyed snake (Micropechis ikaheka) and their neutralisation with CSL polyvalent and black snake antivenoms. Toxicon 2003, 42, 647–655. [Google Scholar] [CrossRef] [PubMed]
- Ong, R.K.; Swindells, K.; Mansfield, C.S. Prospective determination of the specificity of a commercial snake venom detection kit in urine samples from dogs and cats. Aust. Vet. J. 2010, 88, 222–224. [Google Scholar] [CrossRef] [PubMed]
- Van Dong, L.; Quyen le, K.; Eng, K.H.; Gopalakrishnakone, P. Immunogenicity of venoms from four common snakes in the South of Vietnam and development of ELISA kit for venom detection. J. Immunol. Methods 2003, 282, 13–31. [Google Scholar] [CrossRef]
- Le Dong, V.; Selvanayagam, Z.E.; Gopalakrishnakone, P.; Eng, K.H. A new avidin-biotin optical immunoassay for the detection of β-bungarotoxin and application in diagnosis of experimental snake envenomation. J. Immunol. Methods 2002, 260, 125–136. [Google Scholar] [CrossRef]
- Ho, M.; Warrell, M.J.; Warrell, D.A.; Bidwell, D.; Voller, A. A critical reappraisal of the use of enzyme-linked immunosorbent assays in the study of snake bite. Toxicon 1986, 24, 211–221. [Google Scholar] [CrossRef]
- Selvanayagam, Z.E.; Gopalakrishnakone, P. Tests for detection of snake venoms, toxins and venom antibodies: Review on recent trends (1987–1997). Toxicon 1999, 37, 565–586. [Google Scholar] [CrossRef]
- Bonacini, M.; Lin, H.J.; Hollinger, F.B. Effect of coexisting HIV-1 infection on the diagnosis and evaluation of hepatitis C virus. J. Acquir. Immune Defic. Syndr. 2001, 26, 340–344. [Google Scholar] [CrossRef] [PubMed]
- Alvarez-Fernandez, M.; Potel-Alvarellos, C.; Alvarez-Garcia, P.; Otero-Varela, M.I.; Constenla-Carames, L. An outbreak of negative rapid agglutination test meticillin-resistant Staphylococcus aureus. J. Hosp. Infect. 2008, 69, 194–195. [Google Scholar] [CrossRef] [PubMed]
- Abate, K. Modern-day malaria: An overview of this lingering threat. Adv. Nurse Pract 2008, 16, 67–68. [Google Scholar] [PubMed]
- Bygbjerg, I.C. Double burden of noncommunicable and infectious diseases in developing countries. Science 2012, 337, 1499–1501. [Google Scholar] [CrossRef] [PubMed]
- Zaidi, A.K.; Awasthi, S.; deSilva, H.J. Burden of infectious diseases in South Asia. Br. Med. J. 2004, 328, 811–815. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization (WHO). World Health Organization Fact. Sheets: Infectious Diseases; World Health Organization (WHO): Geneva, Switzerland, 2010. [Google Scholar]
- Pultorak, E.; Wong, W.; Rabins, C.; Mehta, S.D. Economic burden of sexually transmitted infections: Incidence and direct medical cost of Chlamydia, gonorrhea, and syphilis among Illinois adolescents and young adults, 2005–2006. Sex. Transm. Dis. 2009, 36, 629–636. [Google Scholar] [CrossRef] [PubMed]
- Mao, S.P.; Shao, B.R. Schistosomiasis control in the people’s Republic of China. Am. J. Trop. Med. Hyg. 1982, 31, 92–99. [Google Scholar] [CrossRef] [PubMed]
- Qiu, S.; Xu, X.; Wang, Y.; Yang, G.; Wang, Z.; Wang, H.; Zhang, L.; Liu, N.; Chen, C.; Liu, W.; et al. Emergence of resistance to fluoroquinolones and third-generation cephalosporins in Shigella flexneri subserotype 1c isolates from China. Clin. Microbiol. Infect. 2012, 18, E95–E98. [Google Scholar] [CrossRef] [PubMed]
- The Joint United Nations Programme on HIV/AIDS (UNAIDS). Global HIV/AIDS Response Progress Report 2011: Epidemic Update and Health Sector Progress towards Universal Access; World Health Organization, UNAIDS: Geneva, Switzerland, 2011; p. 229. [Google Scholar]
- Goldsby, R.A.; Kindt, T.J.; Osborne, B.A. AIDS and Other Immunodeficiencies, 4th ed.; W.H. Freeman and Company: New York, NY, USA, 2000; pp. 467–496. [Google Scholar]
- Piwowar-Manning, E.M.; Tustin, N.B.; Sikateyo, P.; Kamwendo, D.; Chipungu, C.; Maharaj, R.; Mushanyu, J.; Richardson, B.A.; Hillier, S.; Brooks Jackson, J. Validation of rapid HIV antibody tests in 5 African countries. J. Int. Assoc. Physicians AIDS Care 2010, 9, 170–172. [Google Scholar] [CrossRef] [PubMed]
- Ferreira Junior, O.C.; Ferreira, C.; Riedel, M.; Widolin, M.R.; Barbosa-Junior, A. Evaluation of rapid tests for anti-HIV detection in Brazil. AIDS 2005, 19, S70–S75. [Google Scholar] [CrossRef] [PubMed]
- Gurtler, L. Difficulties and strategies of HIV diagnosis. Lancet 1996, 348, 176–179. [Google Scholar] [CrossRef]
- Kivuyo, S.L.; Johannessen, A.; Troseid, M.; Kasubi, M.J.; Gundersen, S.G.; Naman, E.; Mushi, D.; Ngowi, B.J.; Mfinanga, G.S.; Bruun, J.N. p24 antigen detection on dried blood spots is a feasible and reliable test for infant HIV infection in rural Tanzania. Int. J. STD AIDS 2011, 22, 719–721. [Google Scholar] [CrossRef] [PubMed]
- Connell, J.A.; Parry, J.V.; Mortimer, P.P.; Duncan, J. Novel assay for the detection of immunoglobulin G antihuman immunodeficiency virus in untreated saliva and urine. J. Med. Virol. 1993, 41, 159–164. [Google Scholar] [CrossRef] [PubMed]
- Gershy-Damet, G.M.; Koffi, K.; Abouya, L.; Sasson-Morokro, M.; Brattegaard, K.; Parry, J.V.; de Cock, K.M.; Mortimer, P.P. Salivary and urinary diagnosis of human immunodeficiency viruses 1 and 2 infection in Cote d’Ivoire, using two assays. Trans. R. Soc. Trop. Med. Hyg. 1992, 86, 670–671. [Google Scholar] [CrossRef]
- Gallo, D.; George, J.R.; Fitchen, J.H.; Goldstein, A.S.; Hindahl, M.S. Evaluation of a system using oral mucosal transudate for HIV-1 antibody screening and confirmatory testing. OraSure HIV Clinical Trials Group. JAMA 1997, 277, 254–258. [Google Scholar] [CrossRef] [PubMed]
- Respess, R.A.; Rayfield, M.A.; Dondero, T.J. Laboratory testing and rapid HIV assays: Applications for HIV surveillance in hard-to-reach populations. AIDS 2001, 15, S49–S59. [Google Scholar] [CrossRef] [PubMed]
- Larson, B.A.; Schnippel, K.; Ndibongo, B.; Xulu, T.; Brennan, A.; Long, L.; Fox, M.P.; Rosen, S. Rapid point-of-care CD4 testing at mobile HIV testing sites to increase linkage to care: An evaluation of a pilot program in South Africa. J. Acquir. Immune Defic. Syndr. 2012, 61, e13–e17. [Google Scholar] [CrossRef] [PubMed]
- Kanshana, S.; Simonds, R.J. National program for preventing mother-child HIV transmission in Thailand: Successful implementation and lessons learned. AIDS 2002, 16, 953–959. [Google Scholar] [CrossRef] [PubMed]
- US Food and Drug Administation (USFDA). US Food and Drug Administation Center for Biologics Evaluation and Research. Available online: http://www.fda.gov/cber/products/testkits.htm (accessed on 15 February 2017).
- Christiansen, C.B.; Jessen, T.E.; Nielsen, C.; Staun-Olsen, P. False negative anti-HIV-1/HIV-2 ELISAs in acute HIV-2 infection. Vox Sang. 1996, 70, 144–147. [Google Scholar] [CrossRef] [PubMed]
- Parpia, Z.A.; Elghanian, R.; Nabatiyan, A.; Hardie, D.R.; Kelso, D.M. p24 antigen rapid test for diagnosis of acute pediatric HIV infection. J. Acquir. Immune Defic. Syndr. 2010, 55, 413–419. [Google Scholar] [CrossRef] [PubMed]
- Bourlet, T.; Pretis, C.; Pillet, S.; Lesenechal, M.; Piche, J.; Pozzetto, B. Comparative evaluation of the VIDAS HIV DUO Ultra assay for combined detection of HIV-1 antigen and antibodies to HIV. J. Virol. Methods 2005, 127, 165–167. [Google Scholar] [CrossRef] [PubMed]
- Kariuki, S.; Revathi, G.; Kiiru, J.; Mengo, D.M.; Mwituria, J.; Muyodi, J.; Munyalo, A.; Teo, Y.Y.; Holt, K.E.; Kingsley, R.A.; et al. Typhoid in Kenya is associated with a dominant multidrug-resistant Salmonella enterica serovar Typhi haplotype that is also widespread in Southeast Asia. J. Clin. Microbiol. 2010, 48, 2171–2176. [Google Scholar] [CrossRef] [PubMed]
- Parry, C.M.; Hien, T.T.; Dougan, G.; White, N.J.; Farrar, J.J. Typhoid fever. N. Engl. J. Med. 2002, 347, 1770–1782. [Google Scholar] [CrossRef] [PubMed]
- Pratap, C.B.; Patel, S.K.; Shukla, V.K.; Tripathi, S.K.; Singh, T.B.; Nath, G. Drug resistance in Salmonella enterica serotype Typhi isolated from chronic typhoid carriers. Int. J. Antimicrob. Agents 2012, 40, 279–280. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization (WHO). Background Document: The Diagnosis, Treatment and Prevention of Typhoid Fever; WHO Documentary: Geneva, Switzerland, 2003; Volume WHO/V and B/03.07. [Google Scholar]
- Pu, S.J.; Huang, H.S. Diagnostic value of a single Widal test. Chin. J. Microbiol. Immunol. 1985, 18, 256–263. [Google Scholar]
- Brodie, J. Antibodies and the Aberdeen typhoid outbreak of 1964. I. The Widal reaction. J. Hyg. 1977, 79, 161–180. [Google Scholar] [CrossRef] [PubMed]
- Jhaveri, K.N.; Nandwani, S.K.; Mehta, P.K.; Surati, R.R.; Parmar, B.D. False positive modified Widal test in acute malaria. J. Assoc. Physicians India 1995, 43, 754–755. [Google Scholar] [PubMed]
- Parry, C.M.; Hoa, N.T.; Diep, T.S.; Wain, J.; Chinh, N.T.; Vinh, H.; Hien, T.T.; White, N.J.; Farrar, J.J. Value of a single-tube widal test in diagnosis of typhoid fever in Vietnam. J. Clin. Microbiol. 1999, 37, 2882–2886. [Google Scholar] [PubMed]
- Pandya, M.; Pillai, P.; Deb, M. Rapid diagnosis of typhoid fever by detection of Barber protein and Vi antigen of Salmonella serotype typhi. J. Med. Microbiol. 1995, 43, 185–188. [Google Scholar] [CrossRef] [PubMed]
- Gopalakrishnan, V.; Sekhar, W.Y.; Soo, E.H.; Vinsent, R.A.; Devi, S. Typhoid fever in Kuala Lumpur and a comparative evaluation of two commercial diagnostic kits for the detection of antibodies to Salmonella typhi. Singapore Med. J. 2002, 43, 354–358. [Google Scholar] [PubMed]
- Keddy, K.H.; Sooka, A.; Letsoalo, M.E.; Hoyland, G.; Chaignat, C.L.; Morrissey, A.B.; Crump, J.A. Sensitivity and specificity of typhoid fever rapid antibody tests for laboratory diagnosis at two sub-Saharan African sites. Bull. World Health Organ. 2011, 89, 640–647. [Google Scholar] [CrossRef] [PubMed]
- Oracz, G.; Feleszko, W.; Golicka, D.; Maksymiuk, J.; Klonowska, A.; Szajewska, H. Rapid diagnosis of acute Salmonella gastrointestinal infection. Clin. Infect. Dis. 2003, 36, 112–115. [Google Scholar] [CrossRef] [PubMed]
- Feleszko, W.; Maksymiuk, J.; Oracz, G.; Golicka, D.; Szajewska, H. The TUBEX typhoid test detects current Salmonella infections. J. Immunol. Methods 2004, 285, 137–138. [Google Scholar] [CrossRef] [PubMed]
- Tam, F.C.; Lim, P.L. The TUBEX typhoid test based on particle-inhibition immunoassay detects IgM but not IgG anti-O9 antibodies. J. Immunol. Methods 2003, 282, 83–91. [Google Scholar] [CrossRef] [PubMed]
- Choo, K.E.; Oppenheimer, S.J.; Ismail, A.B.; Ong, K.H. Rapid serodiagnosis of typhoid fever by dot enzyme immunoassay in an endemic area. Clin. Infect. Dis. 1994, 19, 172–176. [Google Scholar] [CrossRef] [PubMed]
- Ismail, A.; Kader, Z.S.; Kok-Hai, O. Dot enzyme immunosorbent assay for the serodiagnosis of typhoid fever. Southeast Asian J. Trop. Med. Public Health 1991, 22, 563–566. [Google Scholar] [PubMed]
- Ismail, A.; Hai, O.K.; Kader, Z.A. Demonstration of an antigenic protein specific for Salmonella typhi. Biophys. Res. Commun. 1991, 181, 301–305. [Google Scholar] [CrossRef]
- Jackson, A.A.; Ismail, A.; Ibrahim, T.A.; Kader, Z.S.; Nawi, N.M. Retrospective review of dot enzyme immunoassay test for typhoid fever in an endemic area. Southeast Asian J. Trop. Med. Public Health 1995, 26, 625–630. [Google Scholar] [PubMed]
- Choo, K.E.; Davis, T.M.; Ismail, A.; Ong, K.H. Longevity of antibody responses to a Salmonella typhi-specific outer membrane protein: Interpretation of a dot enzyme immunosorbent assay in an area of high typhoid fever endemicity. Am. J. Trop. Med. Hyg. 1997, 57, 656–659. [Google Scholar] [CrossRef] [PubMed]
- Choo, K.E.; Davis, T.M.; Ismail, A.; Tuan Ibrahim, T.A.; Ghazali, W.N. Rapid and reliable serological diagnosis of enteric fever: Comparative sensitivity and specificity of Typhidot and Typhidot-M tests in febrile Malaysian children. Acta Trop. 1999, 72, 175–183. [Google Scholar] [CrossRef]
- House, D.; Wain, J.; Ho, V.A.; Diep, T.S.; Chinh, N.T.; Bay, P.V.; Vinh, H.; Duc, M.; Parry, C.M.; Dougan, G.; et al. Serology of typhoid fever in an area of endemicity and its relevance to diagnosis. J. Clin. Microbiol. 2001, 39, 1002–1007. [Google Scholar] [CrossRef] [PubMed]
- Olsen, S.J.; Pruckler, J.; Bibb, W.; Nguyen, T.M.; Tran, M.T.; Sivapalasingam, S.; Gupta, A.; Phan, T.P.; Nguyen, T.C.; Nguyen, V.C.; et al. Evaluation of rapid diagnostic tests for typhoid fever. J. Clin. Microbiol. 2004, 42, 1885–1889. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization (WHO). World Malaria Report 2012; WHO Press: Geneva, Switzerland, 2012; p. 288. [Google Scholar]
- Singh, B.; Kim Sung, L.; Matusop, A.; Radhakrishnan, A.; Shamsul, S.S.; Cox-Singh, J.; Thomas, A.; Conway, D.J. A large focus of naturally acquired Plasmodium knowlesi infections in human beings. Lancet 2004, 363, 1017–1024. [Google Scholar] [CrossRef]
- Lee, K.S.; Cox-Singh, J.; Brooke, G.; Matusop, A.; Singh, B. Plasmodium knowlesi from archival blood films: Further evidence that human infections are widely distributed and not newly emergent in Malaysian Borneo. Int. J. Parasitol. 2009, 39, 1125–1128. [Google Scholar] [CrossRef] [PubMed]
- Sulistyaningsih, E.; Fitri, L.E.; Loscher, T.; Berens-Riha, N. Diagnostic difficulties with Plasmodium knowlesi infection in humans. Emerg. Infect. Dis. 2010, 16, 1033–1034. [Google Scholar] [CrossRef] [PubMed]
- Van den Eede, P.; van, H.N.; van Overmeir, C.; Vythilingam, I.; Duc, T.N.; Hung le, X.; Manh, H.N.; Anne, J.; D’Alessandro, U.; Erhart, A. Human Plasmodium knowlesi infections in young children in central Vietnam. Malar. J. 2009, 8, 249. [Google Scholar] [CrossRef] [PubMed]
- Snow, R.W.; Guerra, C.A.; Noor, A.M.; Myint, H.Y.; Hay, S.I. The global distribution of clinical episodes of Plasmodium falciparum malaria. Nature 2005, 434, 214–217. [Google Scholar] [CrossRef] [PubMed]
- Bell, D.; Wongsrichanalai, C.; Barnwell, J.W. Ensuring quality and access for malaria diagnosis: How can it be achieved? Nat. Rev. Microbiol. 2006, 4, S7–S20. [Google Scholar] [CrossRef] [PubMed]
- Murray, C.K.; Bell, D.; Gasser, R.A.; Wongsrichanalai, C. Rapid diagnostic testing for malaria. Trop. Med. Int. Health 2003, 8, 876–883. [Google Scholar] [CrossRef] [PubMed]
- Van den Ende, J.; Jacobs, J.; Bisoffi, Z. Utility of point-of-care malaria rapid diagnostic tests. Am. J. Trop. Med. Hyg. 2010, 83, 207. [Google Scholar] [PubMed]
- Bisoffi, Z.; Gobbi, F.; Angheben, A.; van den Ende, J. The role of rapid diagnostic tests in managing malaria. PLoS Med. 2009, 6, e1000063. [Google Scholar] [CrossRef] [PubMed]
- Howard, R.J.; Uni, S.; Aikawa, M.; Aley, S.B.; Leech, J.H.; Lew, A.M.; Wellems, T.E.; Rener, J.; Taylor, D.W. Secretion of a malarial histidine-rich protein (Pf HRP II) from Plasmodium falciparum-infected erythrocytes. J. Cell Biol. 1986, 103, 1269–1277. [Google Scholar] [CrossRef] [PubMed]
- Rock, E.P.; Marsh, K.; Saul, A.J.; Wellems, T.E.; Taylor, D.W.; Maloy, W.L.; Howard, R.J. Comparative analysis of the Plasmodium falciparum histidine-rich proteins HRP-I, HRP-II and HRP-III in malaria parasites of diverse origin. Parasitology 1987, 95 Pt 2, 209–227. [Google Scholar] [CrossRef] [PubMed]
- Parra, M.E.; Evans, C.B.; Taylor, D.W. Identification of Plasmodium falciparum histidine-rich protein 2 in the plasma of humans with malaria. J. Clin. Microbiol. 1991, 29, 1629–1634. [Google Scholar] [PubMed]
- Aslan, G.; Ulukanligil, M.; Seyrek, A.; Erel, O. Diagnostic performance characteristics of rapid dipstick test for Plasmodium vivax malaria. Mem. Inst. Oswaldo Cruz 2001, 96, 683–686. [Google Scholar] [CrossRef] [PubMed]
- Moody, A. Rapid diagnostic tests for malaria parasites. Clin. Microbiol. Rev. 2002, 15, 66–78. [Google Scholar] [CrossRef] [PubMed]
- Meier, B.; Dobeli, H.; Certa, U. Stage-specific expression of aldolase isoenzymes in the rodent malaria parasite Plasmodium berghei. Mol. Biochem. Parasitol. 1992, 52, 15–27. [Google Scholar] [CrossRef]
- Cloonan, N.; Fischer, K.; Cheng, Q.; Saul, A. Aldolase genes of Plasmodium species. Mol. Biochem. Parasitol. 2001, 113, 327–330. [Google Scholar] [CrossRef]
- Aguilar, R.; Machevo, S.; Menendez, C.; Bardaji, A.; Nhabomba, A.; Alonso, P.L.; Mayor, A. Comparison of placental blood microscopy and the ICT HRP2 rapid diagnostic test to detect placental malaria. Trans. R. Soc. Trop. Med. Hyg. 2012, 106, 573–575. [Google Scholar] [CrossRef] [PubMed]
- Forney, J.R.; Magill, A.J.; Wongsrichanalai, C.; Sirichaisinthop, J.; Bautista, C.T.; Heppner, D.G.; Miller, R.S.; Ockenhouse, C.F.; Gubanov, A.; Shafer, R.; et al. Malaria rapid diagnostic devices: Performance characteristics of the ParaSight F device determined in a multisite field study. J. Clin. Microbiol. 2001, 39, 2884–2890. [Google Scholar] [CrossRef] [PubMed]
- Mueller, I.; Betuela, I.; Ginny, M.; Reeder, J.C.; Genton, B. The sensitivity of the OptiMAL rapid diagnostic test to the presence of Plasmodium falciparum gametocytes compromises its ability to monitor treatment outcomes in an area of Papua New Guinea in which malaria is endemic. J. Clin. Microbiol. 2007, 45, 627–630. [Google Scholar] [CrossRef] [PubMed]
- Soto Tarazona, A.; Solari Zerpa, L.; Mendoza Requena, D.; Llanos-Cuentas, A.; Magill, A. Evaluation of the rapid diagnostic test OptiMAL for diagnosis of malaria due to Plasmodium vivax. Braz. J. Infect. Dis. 2004, 8, 151–155. [Google Scholar] [CrossRef] [PubMed]
- Playford, E.G.; Walker, J. Evaluation of the ICT malaria P.f/P.v and the OptiMal rapid diagnostic tests for malaria in febrile returned travellers. J. Clin. Microbiol. 2002, 40, 4166–4171. [Google Scholar] [CrossRef] [PubMed]
- Piper, R.; Lebras, J.; Wentworth, L.; Hunt-Cooke, A.; Houze, S.; Chiodini, P.; Makler, M. Immunocapture diagnostic assays for malaria using Plasmodium lactate dehydrogenase (pLDH). Am. J. Trop. Med. Hyg. 1999, 60, 109–118. [Google Scholar] [CrossRef] [PubMed]
- Tjitra, E.; Suprianto, S.; Dyer, M.; Currie, B.J.; Anstey, N.M. Field evaluation of the ICT malaria P.f/P.v immunochromatographic test for detection of Plasmodium falciparum and Plasmodium vivax in patients with a presumptive clinical diagnosis of malaria in eastern Indonesia. J. Clin. Microbiol. 1999, 37, 2412–2417. [Google Scholar] [PubMed]
- World Health Organization (WHO). List of Known Commercially Available Antigen-Detecting Malaria RDTs. Available online: http://www.wpro.who.int/malaria/sites/rdt/ (accessed on 12 April 2017).
- Conroy, P.J.; Hearty, S.; Leonard, P.; O’Kennedy, R.J. Antibody production, design and use for biosensor-based applications. Semin. Cell Dev. Biol. 2009, 20, 10–26. [Google Scholar] [CrossRef] [PubMed]
- Newcombe, C.; Newcombe, A.R. Antibody production: Polyclonal-derived biotherapeutics. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2007, 848, 2–7. [Google Scholar] [CrossRef] [PubMed]
- Milstein, C. 12th Sir Hans Krebs Lecture. From antibody diversity to monoclonal antibodies. Eur. J. Biochem. 1981, 118, 429–436. [Google Scholar] [CrossRef] [PubMed]
- Torrance, L.; Ziegler, A.; Pittman, H.; Paterson, M.; Toth, R.; Eggleston, I. Oriented immobilisation of engineered single-chain antibodies to develop biosensors for virus detection. J. Virol. Methods 2006, 134, 164–170. [Google Scholar] [CrossRef] [PubMed]
- Kohler, G.; Milstein, C. Continuous cultures of fused cells secreting antibody of predefined specificity. Nature 1975, 256, 495–497. [Google Scholar] [CrossRef] [PubMed]
- Lipman, N.S.; Jackson, L.R.; Trudel, L.J.; Weis-Garcia, F. Monoclonal versus polyclonal antibodies: Distinguishing characteristics, applications, and information resources. ILAR J. 2005, 46, 258–268. [Google Scholar] [CrossRef] [PubMed]
- Yokoyama, W.M. Production of monoclonal antibody supernatant and ascites fluid. Curr. Protoc. Mol. Biol. 2008. [Google Scholar] [CrossRef]
- Jackson, L.R.; Trudel, L.J.; Fox, J.G.; Lipman, N.S. Monoclonal antibody production in murine ascites. I. Clinical and pathologic features. Lab. Anim. Sci. 1999, 49, 70–80. [Google Scholar] [PubMed]
- Hendriksen, C.F.; de Leeuw, W. Production of monoclonal antibodies by the ascites method in laboratory animals. Res. Immunol. 1998, 149, 535–542. [Google Scholar] [CrossRef]
- Li, F.; Vijayasankaran, N.; Shen, A.Y.; Kiss, R.; Amanullah, A. Cell culture processes for monoclonal antibody production. MAbs 2010, 2, 466–479. [Google Scholar] [CrossRef] [PubMed]
- Dewar, V.; Voet, P.; Denamur, F.; Smal, J. Industrial implementation of in vitro production of monoclonal antibodies. ILAR J. 2005, 46, 307–313. [Google Scholar] [CrossRef] [PubMed]
- Janeway, C.A.; Travers, J.P.; WAlport, M.; Schlomchik, M. Immunology, 5th ed.; Garland Science: New York, NY, USA, 2001. [Google Scholar]
- Wesolowski, J.; Alzogaray, V.; Reyelt, J.; Unger, M.; Juarez, K.; Urrutia, M.; Cauerhff, A.; Danquah, W.; Rissiek, B.; Scheuplein, F.; et al. Single domain antibodies: Promising experimental and therapeutic tools in infection and immunity. Med. Microbiol. Immunol. 2009, 198, 157–174. [Google Scholar] [CrossRef] [PubMed]
- Braden, B.C.; Souchon, H.; Eisele, J.L.; Bentley, G.A.; Bhat, T.N.; Navaza, J.; Poljak, R.J. Three-dimensional structures of the free and the antigen-complexed Fab from monoclonal anti-lysozyme antibody D44.1. J. Mol. Biol. 1994, 243, 767–781. [Google Scholar] [CrossRef]
- Murray, C.K.; Gasser, R.A., Jr.; Magill, A.J.; Miller, R.S. Update on rapid diagnostic testing for malaria. Clin. Microbiol. Rev. 2008, 21, 97–110. [Google Scholar] [CrossRef] [PubMed]
- Guire, P.E. Stability issues for protein-based in vitro diagnostic products. IVD Technol. 1999, 5, 50–54. [Google Scholar]
- Chiodini, P.L.; Bowers, K.; Jorgensen, P.; Barnwell, J.W.; Grady, K.K.; Luchavez, J.; Moody, A.H.; Cenizal, A.; Bell, D. The heat stability of Plasmodium lactate dehydrogenase-based and histidine-rich protein 2-based malaria rapid diagnostic tests. Trans. R. Soc. Trop. Med. Hyg. 2007, 101, 331–337. [Google Scholar] [CrossRef] [PubMed]
- Jorgensen, P.; Chanthap, L.; Rebueno, A.; Tsuyuoka, R.; Bell, D. Malaria rapid diagnostic tests in tropical climates: The need for a cool chain. Am. J. Trop. Med. Hyg. 2006, 74, 750–754. [Google Scholar] [PubMed]
- Ward, E.S.; Gussow, D.; Griffiths, A.D.; Jones, P.T.; Winter, G. Binding activities of a repertoire of single immunoglobulin variable domains secreted from E. coli. Nature 1989, 341, 544–546. [Google Scholar] [CrossRef] [PubMed]
- Barthelemy, P.A.; Raab, H.; Appleton, B.A.; Bond, C.J.; Wu, P.; Wiesmann, C.; Sidhu, S.S. Comprehensive analysis of the factors contributing to the stability and solubility of autonomous human VH domains. J. Biol. Chem. 2008, 283, 3639–3654. [Google Scholar] [CrossRef] [PubMed]
- Huang, L.; Reekmans, G.; Saerens, D.; Friedt, J.M.; Frederix, F.; Francis, L.; Muyldermans, S.; Campitelli, A.; van Hoof, C. Prostate-specific antigen immunosensing based on mixed self-assembled monolayers, camel antibodies and colloidal gold enhanced sandwich assays. Biosens. Bioelectron. 2005, 21, 483–490. [Google Scholar] [CrossRef] [PubMed]
- Hudson, P.J.; Souriau, C. Engineered antibodies. Nat. Med. 2003, 9, 129–134. [Google Scholar] [CrossRef] [PubMed]
- Jostock, T.; Vanhove, M.; Brepoels, E.; van Gool, R.; Daukandt, M.; Wehnert, A.; Van Hegelsom, R.; Dransfield, D.; Sexton, D.; Devlin, M.; et al. Rapid generation of functional human IgG antibodies derived from Fab-on-phage display libraries. J. Immunol. Methods 2004, 289, 65–80. [Google Scholar] [CrossRef] [PubMed]
- McCafferty, J.; Griffiths, A.D.; Winter, G.; Chiswell, D.J. Phage antibodies: Filamentous phage displaying antibody variable domains. Nature 1990, 348, 552–554. [Google Scholar] [CrossRef] [PubMed]
- Hoogenboom, H.R. Overview of antibody phage-display technology and its applications. Methods Mol. Biol. 2002, 178, 1–37. [Google Scholar] [PubMed]
- Hoogenboom, H.R.; de Bruine, A.P.; Hufton, S.E.; Hoet, R.M.; Arends, J.W.; Roovers, R.C. Antibody phage display technology and its applications. Immunotechnology 1998, 4, 1–20. [Google Scholar] [CrossRef]
- Iason, G. The role of plant secondary metabolites in mammalian herbivory: Ecological perspectives. Proc. Nutr. Soc. 2005, 64, 123–131. [Google Scholar] [CrossRef] [PubMed]
- Griffiths, A.D.; Malmqvist, M.; Marks, J.D.; Bye, J.M.; Embleton, M.J.; McCafferty, J.; Baier, M.; Holliger, K.P.; Gorick, B.D.; Hughes-Jones, N.C.; et al. Human anti-self antibodies with high specificity from phage display libraries. EMBO J. 1993, 12, 725–734. [Google Scholar] [PubMed]
- Winter, G.; Griffiths, A.D.; Hawkins, R.E.; Hoogenboom, H.R. Making antibodies by phage display technology. Annu. Rev. Immunol. 1994, 12, 433–455. [Google Scholar] [CrossRef] [PubMed]
- Hill, J.E.; Hemmingsen, S.M.; Goldade, B.G.; Dumonceaux, T.J.; Klassen, J.; Zijlstra, R.T.; Goh, S.H.; van Kessel, A.G. Comparison of ileum microflora of pigs fed corn-, wheat-, or barley-based diets by chaperonin-60 sequencing and quantitative PCR. Appl. Environ. Microbiol. 2005, 71, 867–875. [Google Scholar] [CrossRef] [PubMed]
- Neri, D.; Petrul, H.; Roncucci, G. Engineering recombinant antibodies for immunotherapy. Cell Biophys. 1995, 27, 47–61. [Google Scholar] [CrossRef] [PubMed]
- Xia, M.S.; Hu, C.H.; Xu, Z.R. Effects of copper-bearing montmorillonite on growth performance, digestive enzyme activities, and intestinal microflora and morphology of male broilers. Poult. Sci. 2004, 83, 1868–1875. [Google Scholar] [CrossRef] [PubMed]
- Paul, S.S.; Kamra, D.N.; Sastry, V.R.; Sahu, N.P.; Agarwal, N. Effect of anaerobic fungi on in vitro feed digestion by mixed rumen microflora of buffalo. Reprod. Nutr. Dev. 2004, 44, 313–319. [Google Scholar] [CrossRef] [PubMed]
- Hume, M.E.; Poole, T.L.; Pultz, N.J.; Hanrahan, J.A.; Donskey, C.J. Inhibition of vancomycin-resistant enterococcus by continuous-flow cultures of human stool microflora with and without anaerobic gas supplementation. Curr. Microbiol. 2004, 48, 364–367. [Google Scholar] [CrossRef] [PubMed]
- Franklin, M.A.; Mathew, A.G.; Vickers, J.R.; Clift, R.A. Characterization of microbial populations and volatile fatty acid concentrations in the jejunum, ileum, and cecum of pigs weaned at 17 vs. 24 days of age. J. Anim. Sci. 2002, 80, 2904–2910. [Google Scholar] [CrossRef] [PubMed]
- Engberg, R.M.; Hedemann, M.S.; Jensen, B.B. The influence of grinding and pelleting of feed on the microbial composition and activity in the digestive tract of broiler chickens. Br. Poult. Sci. 2002, 43, 569–579. [Google Scholar] [CrossRef] [PubMed]
- Abbady, A.Q.; Al-Mariri, A.; Zarkawi, M.; Al-Assad, A.; Muyldermans, S. Evaluation of a nanobody phage display library constructed from a Brucella-immunised camel. Vet. Immunol. Immunopathol. 2011, 142, 49–56. [Google Scholar] [CrossRef] [PubMed]
- Dolk, E.; van der Vaart, M.; Lutje Hulsik, D.; Vriend, G.; de Haard, H.; Spinelli, S.; Cambillau, C.; Frenken, L.; Verrips, T. Isolation of llama antibody fragments for prevention of dandruff by phage display in shampoo. Appl. Environ. Microbiol. 2005, 71, 442–450. [Google Scholar] [CrossRef] [PubMed]
- Dooley, H.; Flajnik, M.F.; Porter, A.J. Selection and characterization of naturally occurring single-domain (IgNAR) antibody fragments from immunized sharks by phage display. Mol. Immunol. 2003, 40, 25–33. [Google Scholar] [CrossRef]
- Xu, G.; Tasumi, S.; Pancer, Z. Yeast surface display of lamprey variable lymphocyte receptors. Methods Mol. Biol. 2011, 748, 21–33. [Google Scholar] [CrossRef] [PubMed]
- Hussack, G.; Arbabi-Ghahroudi, M.; Mackenzie, C.R.; Tanha, J. Isolation and characterization of Clostridium difficile toxin-specific single-domain antibodies. Methods Mol. Biol. 2012, 911, 211–239. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Regula, L.K.; Stewart, A.; Lai, J.R. Synthetic Fab fragments that bind the HIV-1 gp41 heptad repeat regions. Biochem. Biophys. Res. Commun. 2011, 413, 611–615. [Google Scholar] [CrossRef] [PubMed]
- Shui, X.; Huang, J.; Li, Y.H.; Xie, P.L.; Li, G.C. Construction and selection of human Fab antibody phage display library of liver cancer. Hybridoma 2009, 28, 341–347. [Google Scholar] [CrossRef] [PubMed]
- Sowa, K.M.; Cavanagh, D.R.; Creasey, A.M.; Raats, J.; McBride, J.; Sauerwein, R.; Roeffen, W.F.; Arnot, D.E. Isolation of a monoclonal antibody from a malaria patient-derived phage display library recognising the Block 2 region of Plasmodium falciparum merozoite surface protein-1. Mol. Biochem. Parasitol. 2001, 112, 143–147. [Google Scholar] [CrossRef]
- Yang, G.H.; Yoon, S.O.; Jang, M.H.; Hong, H.J. Affinity maturation of an anti-hepatitis B virus PreS1 humanized antibody by phage display. J. Microbiol. 2007, 45, 528–533. [Google Scholar] [PubMed]
- Carmen, S.; Jermutus, L. Concepts in antibody phage display. Brief Funct. Genomic Proteomic 2002, 1, 189–203. [Google Scholar] [CrossRef] [PubMed]
- Brichta, J.; Hnilova, M.; Viskovic, T. Generation of hapten-specific recombinant antibodies: Antibody phage display technology: A review. Vet. Med. 2005, 50, 231–252. [Google Scholar]
- Flajnik, M.F. Comparative analyses of immunoglobulin genes: Surprises and portents. Nat. Rev. Immunol. 2002, 2, 688–698. [Google Scholar] [CrossRef] [PubMed]
- Hamers-Casterman, C.; Atarhouch, T.; Muyldermans, S.; Robinson, G.; Hamers, C.; Songa, E.B.; Bendahman, N.; Hamers, R. Naturally occurring antibodies devoid of light chains. Nature 1993, 363, 446–448. [Google Scholar] [CrossRef] [PubMed]
- Goldman, E.R.; Anderson, G.P.; Conway, J.; Sherwood, L.J.; Fech, M.; Vo, B.; Liu, J.L.; Hayhurst, A. Thermostable llama single domain antibodies for detection of botulinum A neurotoxin complex. Anal. Chem. 2008, 80, 8583–8591. [Google Scholar] [CrossRef] [PubMed]
- Perez, J.M.; Renisio, J.G.; Prompers, J.J.; van Platerink, C.J.; Cambillau, C.; Darbon, H.; Frenken, L.G. Thermal unfolding of a llama antibody fragment: A two-state reversible process. Biochemistry 2001, 40, 74–83. [Google Scholar] [CrossRef] [PubMed]
- Dumoulin, M.; Conrath, K.; van Meirhaeghe, A.; Meersman, F.; Heremans, K.; Frenken, L.G.; Muyldermans, S.; Wyns, L.; Matagne, A. Single-domain antibody fragments with high conformational stability. Protein Sci. 2002, 11, 500–515. [Google Scholar] [CrossRef] [PubMed]
- Harmsen, M.M.; de Haard, H.J. Properties, production, and applications of camelid single-domain antibody fragments. Appl. Microbiol. Biotechnol. 2007, 77, 13–22. [Google Scholar] [CrossRef] [PubMed]
- Muyldermans, S.; Atarhouch, T.; Saldanha, J.; Barbosa, J.A.; Hamers, R. Sequence and structure of VH domain from naturally occurring camel heavy chain immunoglobulins lacking light chains. Protein Eng. 1994, 7, 1129–1135. [Google Scholar] [CrossRef] [PubMed]
- Muyldermans, S. Single domain camel antibodies: Current status. J. Biotechnol. 2001, 74, 277–302. [Google Scholar] [CrossRef]
- Dumoulin, M.; Last, A.M.; Desmyter, A.; Decanniere, K.; Canet, D.; Larsson, G.; Spencer, A.; Archer, D.B.; Sasse, J.; Muyldermans, S.; et al. A camelid antibody fragment inhibits the formation of amyloid fibrils by human lysozyme. Nature 2003, 424, 783–788. [Google Scholar] [CrossRef] [PubMed]
- Vance, D.J.; Tremblay, J.M.; Mantis, N.J.; Shoemaker, C.B. Stepwise engineering of heterodimeric single domain camelid VHH antibodies that passively protect mice from ricin toxin. J. Biol. Chem. 2013, 288, 36538–36547. [Google Scholar] [CrossRef] [PubMed]
- De Genst, E.; Silence, K.; Decanniere, K.; Conrath, K.; Loris, R.; Kinne, J.; Muyldermans, S.; Wyns, L. Molecular basis for the preferential cleft recognition by dromedary heavy-chain antibodies. Proc. Natl. Acad. Sci. USA 2006, 103, 4586–4591. [Google Scholar] [CrossRef] [PubMed]
- Desmyter, A.; Transue, T.R.; Ghahroudi, M.A.; Thi, M.H.; Poortmans, F.; Hamers, R.; Muyldermans, S.; Wyns, L. Crystal structure of a camel single-domain VH antibody fragment in complex with lysozyme. Nat. Struct. Biol. 1996, 3, 803–811. [Google Scholar] [CrossRef] [PubMed]
- Vincke, C.; Loris, R.; Saerens, D.; Martinez-Rodriguez, S.; Muyldermans, S.; Conrath, K. General strategy to humanize a camelid single-domain antibody and identification of a universal humanized nanobody scaffold. J. Biol. Chem. 2009, 284, 3273–3284. [Google Scholar] [CrossRef] [PubMed]
- Line, B.R.; Breyer, R.J.; McElvany, K.D.; Earle, D.C.; Khazaeli, M.B. Evaluation of human anti-mouse antibody response in normal volunteers following repeated injections of fanolesomab (NeutroSpec), a murine anti-CD15 IgM monoclonal antibody for imaging infection. Nucl. Med. Commun. 2004, 25, 807–811. [Google Scholar] [CrossRef] [PubMed]
- Dmitriev, O.Y.; Lutsenko, S.; Muyldermans, S. Nanobodies as Probes for Protein Dynamics in Vitro and in Cells. J. Biol. Chem. 2016, 291, 3767–3775. [Google Scholar] [CrossRef] [PubMed]
- Van Brussel, A.S.; Adams, A.; Oliveira, S.; Dorresteijn, B.; El Khattabi, M.; Vermeulen, J.F.; van der Wall, E.; Mali, W.P.; Derksen, P.W.; van Diest, P.J.; et al. Hypoxia-Targeting Fluorescent Nanobodies for Optical Molecular Imaging of Pre-Invasive Breast Cancer. Mol. Imaging Biol. 2016, 18, 535–544. [Google Scholar] [CrossRef] [PubMed]
- Deckers, N.; Saerens, D.; Kanobana, K.; Conrath, K.; Victor, B.; Wernery, U.; Vercruysse, J.; Muyldermans, S.; Dorny, P. Nanobodies, a promising tool for species-specific diagnosis of Taenia solium cysticercosis. Int. J. Parasitol. 2009, 39, 625–633. [Google Scholar] [CrossRef] [PubMed]
- Roovers, R.C.; Laeremans, T.; Huang, L.; De Taeye, S.; Verkleij, A.J.; Revets, H.; de Haard, H.J.; van Bergen en Henegouwen, P.M. Efficient inhibition of EGFR signaling and of tumour growth by antagonistic anti-EFGR Nanobodies. Cancer Immunol. Immunother. 2007, 56, 303–317. [Google Scholar] [CrossRef] [PubMed]
- Franco, E.J.; Sonneson, G.J.; DeLegge, T.J.; Hofstetter, H.; Horn, J.R.; Hofstetter, O. Production and characterization of a genetically engineered anti-caffeine camelid antibody and its use in immunoaffinity chromatography. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2010, 878, 177–186. [Google Scholar] [CrossRef] [PubMed]
- Ladenson, R.C.; Crimmins, D.L.; Landt, Y.; Ladenson, J.H. Isolation and characterization of a thermally stable recombinant anti-caffeine heavy-chain antibody fragment. Anal. Chem. 2006, 78, 4501–4508. [Google Scholar] [CrossRef] [PubMed]
- Diaz, M.; Greenberg, A.S.; Flajnik, M.F. Somatic hypermutation of the new antigen receptor gene (NAR) in the nurse shark does not generate the repertoire: Possible role in antigen-driven reactions in the absence of germinal centers. Proc. Natl. Acad. Sci. USA 1998, 95, 14343–14348. [Google Scholar] [CrossRef] [PubMed]
- Greenberg, A.S.; Avila, D.; Hughes, M.; Hughes, A.; McKinney, E.C.; Flajnik, M.F. A new antigen receptor gene family that undergoes rearrangement and extensive somatic diversification in sharks. Nature 1995, 374, 168–173. [Google Scholar] [CrossRef] [PubMed]
- Roux, K.H.; Greenberg, A.S.; Greene, L.; Strelets, L.; Avila, D.; McKinney, E.C.; Flajnik, M.F. Structural analysis of the nurse shark (new) antigen receptor (NAR): Molecular convergence of NAR and unusual mammalian immunoglobulins. Proc. Natl. Acad. Sci. USA 1998, 95, 11804–11809. [Google Scholar] [CrossRef] [PubMed]
- Dooley, H.; Stanfield, R.L.; Brady, R.A.; Flajnik, M.F. First molecular and biochemical analysis of in vivo affinity maturation in an ectothermic vertebrate. Proc. Natl. Acad. Sci. USA 2006, 103, 1846–1851. [Google Scholar] [CrossRef] [PubMed]
- Nuttall, S.D.; Krishnan, U.V.; Hattarki, M.; De Gori, R.; Irving, R.A.; Hudson, P.J. Isolation of the new antigen receptor from wobbegong sharks, and use as a scaffold for the display of protein loop libraries. Mol. Immunol. 2001, 38, 313–326. [Google Scholar] [CrossRef]
- Liu, J.L.; Anderson, G.P.; Delehanty, J.B.; Baumann, R.; Hayhurst, A.; Goldman, E.R. Selection of cholera toxin specific IgNAR single-domain antibodies from a naive shark library. Mol. Immunol. 2007, 44, 1775–1783. [Google Scholar] [CrossRef] [PubMed]
- Ohtani, M.; Hikima, J.; Jung, T.S.; Kondo, H.; Hirono, I.; Aoki, T. Construction of an artificially randomized IgNAR phage display library: Screening of variable regions that bind to hen egg white lysozyme. Mar. Biotechnol. 2013, 15, 56–62. [Google Scholar] [CrossRef] [PubMed]
- Camacho-Villegas, T.; Mata-Gonzalez, T.; Paniagua-Solis, J.; Sanchez, E.; Licea, A. Human TNF cytokine neutralization with a vNAR from Heterodontus francisci shark: A potential therapeutic use. MAbs 2013, 5, 80–85. [Google Scholar] [CrossRef] [PubMed]
- Streltsov, V.A.; Varghese, J.N.; Carmichael, J.A.; Irving, R.A.; Hudson, P.J.; Nuttall, S.D. Structural evidence for evolution of shark Ig new antigen receptor variable domain antibodies from a cell-surface receptor. Proc. Natl. Acad. Sci. USA 2004, 101, 12444–12449. [Google Scholar] [CrossRef] [PubMed]
- Goodchild, S.A.; Dooley, H.; Schoepp, R.J.; Flajnik, M.; Lonsdale, S.G. Isolation and characterisation of Ebolavirus-specific recombinant antibody fragments from murine and shark immune libraries. Mol. Immunol. 2011, 48, 2027–2037. [Google Scholar] [CrossRef] [PubMed]
- Barelle, C.; Gill, D.S.; Charlton, K. Shark novel antigen receptors--the next generation of biologic therapeutics? Adv. Exp. Med. Biol. 2009, 655, 49–62. [Google Scholar] [CrossRef] [PubMed]
- Stanfield, R.L.; Dooley, H.; Flajnik, M.F.; Wilson, I.A. Crystal structure of a shark single-domain antibody V region in complex with lysozyme. Science 2004, 305, 1770–1773. [Google Scholar] [CrossRef] [PubMed]
- Muyldermans, S.; Cambillau, C.; Wyns, L. Recognition of antigens by single-domain antibody fragments: The superfluous luxury of paired domains. Trends Biochem. Sci. 2001, 26, 230–235. [Google Scholar] [CrossRef]
- Streltsov, V.A.; Carmichael, J.A.; Nuttall, S.D. Structure of a shark IgNAR antibody variable domain and modeling of an early-developmental isotype. Protein Sci. 2005, 14, 2901–2909. [Google Scholar] [CrossRef] [PubMed]
- Griffiths, K.; Dolezal, O.; Parisi, K.; Angerosa, J.; Dogovski, C.; Barraclough, M.; Sanalla, A.; Casey, J.; González, I.; Perugini, M.; et al. Shark Variable New Antigen Receptor (VNAR) Single Domain Antibody Fragments: Stability and Diagnostic Applications. Antibodies 2013, 2, 66–81. [Google Scholar] [CrossRef]
- Flajnik, M.F.; Deschacht, N.; Muyldermans, S. A case of convergence: Why did a simple alternative to canonical antibodies arise in sharks and camels? PLoS Biol. 2011, 9, e1001120. [Google Scholar] [CrossRef] [PubMed]
- Pays, E.; Vanhamme, L.; Perez-Morga, D. Antigenic variation in Trypanosoma brucei: Facts, challenges and mysteries. Curr. Opin. Microbiol. 2004, 7, 369–374. [Google Scholar] [CrossRef] [PubMed]
- Saerens, D.; Stijlemans, B.; Baral, T.N.; Nguyen Thi, G.T.; Wernery, U.; Magez, S.; de Baetselier, P.; Muyldermans, S.; Conrath, K. Parallel selection of multiple anti-infectome Nanobodies without access to purified antigens. J. Immunol. Methods 2008, 329, 138–150. [Google Scholar] [CrossRef] [PubMed]
- Hernandez, M.; Beltran, C.; Garcia, E.; Fragoso, G.; Gevorkian, G.; Fleury, A.; Parkhouse, M.; Harrison, L.; Sotelo, J.; Sciutto, E. Cysticercosis: Towards the design of a diagnostic kit based on synthetic peptides. Immunol. Lett. 2000, 71, 13–17. [Google Scholar] [CrossRef]
- Dorny, P.; Brandt, J.; Zoli, A.; Geerts, S. Immunodiagnostic tools for human and porcine cysticercosis. Acta Trop. 2003, 87, 79–86. [Google Scholar] [CrossRef]
- Garcia, H.H.; Harrison, L.J.; Parkhouse, R.M.; Montenegro, T.; Martinez, S.M.; Tsang, V.C.; Gilman, R.H. A specific antigen-detection ELISA for the diagnosis of human neurocysticercosis. The Cysticercosis Working Group in Peru. Trans. R. Soc. Trop. Med. Hyg. 1998, 92, 411–414. [Google Scholar] [CrossRef]
- Anderson, G.P.; Goldman, E.R. TNT detection using llama antibodies and a two-step competitive fluid array immunoassay. J. Immunol. Methods 2008, 339, 47–54. [Google Scholar] [CrossRef] [PubMed]
- Goldman, E.R.; Anderson, G.P.; Liu, J.L.; Delehanty, J.B.; Sherwood, L.J.; Osborn, L.E.; Cummins, L.B.; Hayhurst, A. Facile generation of heat-stable antiviral and antitoxin single domain antibodies from a semisynthetic llama library. Anal. Chem. 2006, 78, 8245–8255. [Google Scholar] [CrossRef] [PubMed]
- Hmila, I.; Abdallah, R.B.; Saerens, D.; Benlasfar, Z.; Conrath, K.; Ayeb, M.E.; Muyldermans, S.; Bouhaouala-Zahar, B. VHH, bivalent domains and chimeric Heavy chain-only antibodies with high neutralizing efficacy for scorpion toxin AahI’. Mol. Immunol. 2008, 45, 3847–3856. [Google Scholar] [CrossRef] [PubMed]
- Strokappe, N.; Szynol, A.; Aasa-Chapman, M.; Gorlani, A.; Forsman Quigley, A.; Hulsik, D.L.; Chen, L.; Weiss, R.; de Haard, H.; Verrips, T. Llama antibody fragments recognizing various epitopes of the CD4bs neutralize a broad range of HIV-1 subtypes A, B and C. PLoS ONE 2012, 7, e33298. [Google Scholar] [CrossRef] [PubMed]
- Vanlandschoot, P.; Stortelers, C.; Beirnaert, E.; Ibanez, L.I.; Schepens, B.; Depla, E.; Saelens, X. Nanobodies(R): New ammunition to battle viruses. Antivir. Res. 2011, 92, 389–407. [Google Scholar] [CrossRef] [PubMed]
- Pant, N.; Marcotte, H.; Hermans, P.; Bezemer, S.; Frenken, L.; Johansen, K.; Hammarstrom, L. Lactobacilli producing bispecific llama-derived anti-rotavirus proteins in vivo for rotavirus-induced diarrhea. Future Microbiol. 2011, 6, 583–593. [Google Scholar] [CrossRef] [PubMed]
- Ryan, S.; Kell, A.J.; van Faassen, H.; Tay, L.L.; Simard, B.; MacKenzie, R.; Gilbert, M.; Tanha, J. Single-domain antibody-nanoparticles: Promising architectures for increased Staphylococcus aureus detection specificity and sensitivity. Bioconjug.Chem. 2009, 20, 1966–1974. [Google Scholar] [CrossRef] [PubMed]
- Kenanova, V.; Wu, A.M. Tailoring antibodies for radionuclide delivery. Expert Opin. Drug Deliv. 2006, 3, 53–70. [Google Scholar] [CrossRef] [PubMed]
- Huang, L.; Gainkam, L.O.; Caveliers, V.; Vanhove, C.; Keyaerts, M.; De Baetselier, P.; Bossuyt, A.; Revets, H.; Lahoutte, T. SPECT imaging with 99mTc-labeled EGFR-specific nanobody for in vivo monitoring of EGFR expression. Mol. Imaging Biol. 2008, 10, 167–175. [Google Scholar] [CrossRef] [PubMed]
- Pleschberger, M.; Saerens, D.; Weigert, S.; Sleytr, U.B.; Muyldermans, S.; Sara, M.; Egelseer, E.M. An S-layer heavy chain camel antibody fusion protein for generation of a nanopatterned sensing layer to detect the prostate-specific antigen by surface plasmon resonance technology. Bioconjug. Chem. 2004, 15, 664–671. [Google Scholar] [CrossRef] [PubMed]
- Pruszynski, M.; Koumarianou, E.; Vaidyanathan, G.; Revets, H.; Devoogdt, N.; Lahoutte, T.; Zalutsky, M.R. Targeting breast carcinoma with radioiodinated anti-HER2 Nanobody. Nucl. Med. Biol. 2013, 40, 52–59. [Google Scholar] [CrossRef] [PubMed]
- Vaneycken, I.; Devoogdt, N.; van Gassen, N.; Vincke, C.; Xavier, C.; Wernery, U.; Muyldermans, S.; Lahoutte, T.; Caveliers, V. Preclinical screening of anti-HER2 nanobodies for molecular imaging of breast cancer. FASEB J. 2011, 25, 2433–2446. [Google Scholar] [CrossRef] [PubMed]
- Keyaerts, M.; Xavier, C.; Heemskerk, J.; Devoogdt, N.; Everaert, H.; Ackaert, C.; Vanhoeij, M.; Duhoux, F.P.; Gevaert, T.; Simon, P.; et al. Phase I Study of 68Ga-HER2-Nanobody for PET/CT Assessment of HER2 Expression in Breast Carcinoma. J. Nucl. Med. 2016, 57, 27–33. [Google Scholar] [CrossRef] [PubMed]
- Anderson, G.P.; Moreira, S.C.; Charles, P.T.; Medintz, I.L.; Goldman, E.R.; Zeinali, M.; Taitt, C.R. TNT detection using multiplexed liquid array displacement immunoassays. Anal. Chem. 2006, 78, 2279–2285. [Google Scholar] [CrossRef] [PubMed]
- Tillib, S.V.; Ivanova, T.I.; Lyssuk, E.Y.; Larin, S.S.; Kibardin, A.V.; Korobko, E.V.; Vikhreva, P.N.; Gnuchev, N.V.; Georgiev, G.P.; Korobko, I.V. Nanoantibodies for detection and blocking of bioactivity of human vascular endothelial growth factor A(165). Biochemistry 2012, 77, 659–665. [Google Scholar] [CrossRef] [PubMed]
- Minaeian, S.; Rahbarizadeh, F.; Zarkesh-Esfahani, S.H.; Ahmadvand, D.; Broom, O.J. Neutralization of human papillomavirus by specific nanobodies against major capsid protein L1. J. Microbiol. Biotechnol. 2012, 22, 721–728. [Google Scholar] [CrossRef] [PubMed]
- Vosjan, M.J.; Vercammen, J.; Kolkman, J.A.; Stigter-van Walsum, M.; Revets, H.; van Dongen, G.A. Nanobodies targeting the hepatocyte growth factor: Potential new drugs for molecular cancer therapy. Mol. Cancer Ther. 2012, 11, 1017–1025. [Google Scholar] [CrossRef] [PubMed]
- Smolarek, D.; Hattab, C.; Hassanzadeh-Ghassabeh, G.; Cochet, S.; Gutierrez, C.; de Brevern, A.G.; Udomsangpetch, R.; Picot, J.; Grodecka, M.; Wasniowska, K.; et al. A recombinant dromedary antibody fragment (VHH or nanobody) directed against human Duffy antigen receptor for chemokines. Cell Mol. Life Sci. 2010, 67, 3371–3387. [Google Scholar] [CrossRef] [PubMed]
- Thys, B.; Saerens, D.; Schotte, L.; De Bleeser, G.; Muyldermans, S.; Hassanzadeh-Ghassabeh, G.; Rombaut, B. A simple quantitative affinity capturing assay of poliovirus antigens and subviral particles by single-domain antibodies using magnetic beads. J. Virol. Methods 2011, 173, 300–305. [Google Scholar] [CrossRef] [PubMed]
- Thys, B.; Schotte, L.; Muyldermans, S.; Wernery, U.; Hassanzadeh-Ghassabeh, G.; Rombaut, B. in vitro antiviral activity of single domain antibody fragments against poliovirus. Antivir. Res. 2010, 87, 257–264. [Google Scholar] [CrossRef] [PubMed]
- Ahmadvand, D.; Rasaee, M.J.; Rahbarizadeh, F.; Kontermann, R.E.; Sheikholislami, F. Cell selection and characterization of a novel human endothelial cell specific nanobody. Mol. Immunol. 2009, 46, 1814–1823. [Google Scholar] [CrossRef] [PubMed]
- Ahmadvand, D.; Rasaee, M.J.; Rahbarizadeh, F.; Mohammadi, M. Production and characterization of a high-affinity nanobody against human endoglin. Hybridoma 2008, 27, 353–360. [Google Scholar] [CrossRef] [PubMed]
- Abbady, A.Q.; Al-Daoude, A.; Al-Mariri, A.; Zarkawi, M.; Muyldermans, S. Chaperonin GroEL a Brucella immunodominant antigen identified using Nanobody and MALDI-TOF-MS technologies. Vet. Immunol. Immunopathol. 2012, 146, 254–263. [Google Scholar] [CrossRef] [PubMed]
- Goldman, E.R.; Anderson, G.P.; Bernstein, R.D.; Swain, M.D. Amplification of immunoassays using phage-displayed single domain antibodies. J. Immunol. Methods 2010, 352, 182–185. [Google Scholar] [CrossRef] [PubMed]
- Swain, M.D.; Anderson, G.P.; Zabetakis, D.; Bernstein, R.D.; Liu, J.L.; Sherwood, L.J.; Hayhurst, A.; Goldman, E.R. Llama-derived single-domain antibodies for the detection of botulinum A neurotoxin. Anal. Bioanal. Chem. 2010, 398, 339–348. [Google Scholar] [CrossRef] [PubMed]
- Leung, K. 99mTc(CO)3-Anti-carcinoembryonic antigen (CEA) humanized CEA5 graft nanobody. In Molecular Imaging and Contrast Agent Database (MICAD); Bethesda: Rockyville, MD, USA, 2004. [Google Scholar]
- Sukhanova, A.; Even-Desrumeaux, K.; Kisserli, A.; Tabary, T.; Reveil, B.; Millot, J.M.; Chames, P.; Baty, D.; Artemyev, M.; Oleinikov, V.; et al. Oriented conjugates of single-domain antibodies and quantum dots: Toward a new generation of ultrasmall diagnostic nanoprobes. Nanomedicine 2012, 8, 516–525. [Google Scholar] [CrossRef] [PubMed]
- Vaneycken, I.; Govaert, J.; Vincke, C.; Caveliers, V.; Lahoutte, T.; de Baetselier, P.; Raes, G.; Bossuyt, A.; Muyldermans, S.; Devoogdt, N. in vitro analysis and in vivo tumor targeting of a humanized, grafted nanobody in mice using pinhole SPECT/micro-CT. J. Nucl. Med. 2010, 51, 1099–1106. [Google Scholar] [CrossRef] [PubMed]
- Leung, K. 99mTc(CO)3-Anti-vascular cell adhesion molecule-1 nanobody cAbVCAM1-5. In Molecular Imaging and Contrast Agent Database (MICAD); Bethesda: Rockyville, MD, USA, 2004. [Google Scholar]
- Leung, K. Microbubbles conjugated with anti-vascular cell adhesion molecule-1 nanobody cAbVCAM1-5. In Molecular Imaging and Contrast Agent Database (MICAD); Bethesda: Rockyville, MD, USA, 2004. [Google Scholar]
- Broisat, A.; Hernot, S.; Toczek, J.; De Vos, J.; Riou, L.M.; Martin, S.; Ahmadi, M.; Thielens, N.; Wernery, U.; Caveliers, V.; et al. Nanobodies targeting mouse/human VCAM1 for the nuclear imaging of atherosclerotic lesions. Circ. Res. 2012, 110, 927–937. [Google Scholar] [CrossRef] [PubMed]
- Schmitz, K.R.; Bagchi, A.; Roovers, R.C.; van Bergen en Henegouwen, P.M.; Ferguson, K.M. Structural evaluation of EGFR inhibition mechanisms for nanobodies/VHH domains. Structure 2013, 21, 1214–1224. [Google Scholar] [CrossRef] [PubMed]
- Roovers, R.C.; Vosjan, M.J.; Laeremans, T.; el Khoulati, R.; de Bruin, R.C.; Ferguson, K.M.; Verkleij, A.J.; van Dongen, G.A.; van Bergen en Henegouwen, P.M. A biparatopic anti-EGFR nanobody efficiently inhibits solid tumour growth. Int. J. Cancer 2011, 129, 2013–2024. [Google Scholar] [CrossRef] [PubMed]
- Hmila, I.; Saerens, D.; Ben Abderrazek, R.; Vincke, C.; Abidi, N.; Benlasfar, Z.; Govaert, J.; El Ayeb, M.; Bouhaouala-Zahar, B.; Muyldermans, S. A bispecific nanobody to provide full protection against lethal scorpion envenoming. FASEB J. 2010, 24, 3479–3489. [Google Scholar] [CrossRef] [PubMed]
- Altintas, I.; Kok, R.J.; Schiffelers, R.M. Targeting epidermal growth factor receptor in tumors: From conventional monoclonal antibodies via heavy chain-only antibodies to nanobodies. Eur. J. Pharm. Sci. 2012, 45, 399–407. [Google Scholar] [CrossRef] [PubMed]
- Chopra, A. [99mTc]Epidermal growth factor receptor-specific nanobody. In Molecular Imaging and Contrast Agent Database (MICAD); Bethesda: Rockyville, MD, USA, 2004. [Google Scholar]
- Friedman, M.; Stahl, S. Engineered affinity proteins for tumour-targeting applications. Biotechnol. Appl. Biochem. 2009, 53, 1–29. [Google Scholar] [CrossRef] [PubMed]
- Hultberg, A.; Temperton, N.J.; Rosseels, V.; Koenders, M.; Gonzalez-Pajuelo, M.; Schepens, B.; Ibanez, L.I.; Vanlandschoot, P.; Schillemans, J.; Saunders, M.; et al. Llama-derived single domain antibodies to build multivalent, superpotent and broadened neutralizing anti-viral molecules. PLoS ONE 2011, 6, e17665. [Google Scholar] [CrossRef] [PubMed]
- Narum, D.L.; Thomas, A.W. Differential localization of full-length and processed forms of PF83/AMA-1 an apical membrane antigen of Plasmodium falciparum merozoites. Mol. Biochem. Parasitol. 1994, 67, 59–68. [Google Scholar] [CrossRef]
- Nuttall, S.D.; Humberstone, K.S.; Krishnan, U.V.; Carmichael, J.A.; Doughty, L.; Hattarki, M.; Coley, A.M.; Casey, J.L.; Anders, R.F.; Foley, M.; et al. Selection and affinity maturation of IgNAR variable domains targeting Plasmodium falciparum AMA1. Proteins 2004, 55, 187–197. [Google Scholar] [CrossRef] [PubMed]
- Henderson, K.A.; Streltsov, V.A.; Coley, A.M.; Dolezal, O.; Hudson, P.J.; Batchelor, A.H.; Gupta, A.; Bai, T.; Murphy, V.J.; Anders, R.F.; et al. Structure of an IgNAR-AMA1 complex: Targeting a conserved hydrophobic cleft broadens malarial strain recognition. Structure 2007, 15, 1452–1466. [Google Scholar] [CrossRef] [PubMed]
- Slots, J.; Ting, M. Actinobacillus actinomycetemcomitans and Porphyromonas gingivalis in human periodontal disease: Occurrence and treatment. Periodontol. 2000 1999, 20, 82–121. [Google Scholar] [CrossRef] [PubMed]
- Kadowaki, T.; Nakayama, K.; Okamoto, K.; Abe, N.; Baba, A.; Shi, Y.; Ratnayake, D.B.; Yamamoto, K. Porphyromonas gingivalis proteinases as virulence determinants in progression of periodontal diseases. J. Biochem. 2000, 128, 153–159. [Google Scholar] [CrossRef] [PubMed]
- Aduse-Opoku, J.; Davies, N.N.; Gallagher, A.; Hashim, A.; Evans, H.E.; Rangarajan, M.; Slaney, J.M.; Curtis, M.A. Generation of lys-gingipain protease activity in Porphyromonas gingivalis W50 is independent of Arg-gingipain protease activities. Microbiology 2000, 146, 1933–1940. [Google Scholar] [CrossRef] [PubMed]
- Nuttall, S.D.; Krishnan, U.V.; Doughty, L.; Nathanielsz, A.; Ally, N.; Pike, R.N.; Hudson, P.J.; Kortt, A.A.; Irving, R.A. A naturally occurring NAR variable domain binds the Kgp protease from Porphyromonas gingivalis. FEBS Lett. 2002, 516, 80–86. [Google Scholar] [CrossRef]
- Papaneri, A.B.; Wirblich, C.; Cooper, K.; Jahrling, P.B.; Schnell, M.J.; Blaney, J.E. Further characterization of the immune response in mice to inactivated and live rabies vaccines expressing Ebola virus glycoprotein. Vaccine 2012, 30, 6136–6141. [Google Scholar] [CrossRef] [PubMed]
- Kondratowicz, A.S.; Maury, W.J. Ebolavirus: A brief review of novel therapeutic targets. Future Microbiol. 2012, 7, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Fausther-Bovendo, H.; Mulangu, S.; Sullivan, N.J. Ebolavirus vaccines for humans and apes. Curr. Opin. Virol. 2012, 2, 324–329. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.L.; Anderson, G.P.; Goldman, E.R. Isolation of anti-toxin single domain antibodies from a semi-synthetic spiny dogfish shark display library. BMC Biotechnol. 2007, 7, 78. [Google Scholar] [CrossRef] [PubMed]
- Nuttall, S.D.; Krishnan, U.V.; Doughty, L.; Pearson, K.; Ryan, M.T.; Hoogenraad, N.J.; Hattarki, M.; Carmichael, J.A.; Irving, R.A.; Hudson, P.J. Isolation and characterization of an IgNAR variable domain specific for the human mitochondrial translocase receptor Tom70. Eur. J. Biochem. 2003, 270, 3543–3554. [Google Scholar] [CrossRef] [PubMed]
- Walsh, R.; Nuttall, S.; Revill, P.; Colledge, D.; Cabuang, L.; Soppe, S.; Dolezal, O.; Griffiths, K.; Bartholomeusz, A.; Locarnini, S. Targeting the hepatitis B virus precore antigen with a novel IgNAR single variable domain intrabody. Virology 2011, 411, 132–141. [Google Scholar] [CrossRef] [PubMed]
- Streltsov, V.A.; Varghese, J.N.; Masters, C.L.; Nuttall, S.D. Crystal structure of the amyloid-beta p3 fragment provides a model for oligomer formation in Alzheimer’s disease. J. Neurosci. 2011, 31, 1419–1426. [Google Scholar] [CrossRef] [PubMed]
- Bojalil, R.; Mata-Gonzalez, M.T.; Sanchez-Munoz, F.; Yee, Y.; Argueta, I.; Bolanos, L.; Amezcua-Guerra, L.M.; Camacho-Villegas, T.A.; Sanchez-Castrejon, E.; Garcia-Ubbelohde, W.J.; et al. Anti-tumor necrosis factor VNAR single domains reduce lethality and regulate underlying inflammatory response in a murine model of endotoxic shock. BMC Immunol. 2013, 14, 17. [Google Scholar] [CrossRef] [PubMed]
- Konning, D.; Rhiel, L.; Empting, M.; Grzeschik, J.; Sellmann, C.; Schroter, C.; Zielonka, S.; Dickgiesser, S.; Pirzer, T.; Yanakieva, D.; et al. Semi-synthetic vNAR libraries screened against therapeutic antibodies primarily deliver anti-idiotypic binders. Sci. Rep. 2017, 7, 9676. [Google Scholar] [CrossRef] [PubMed]
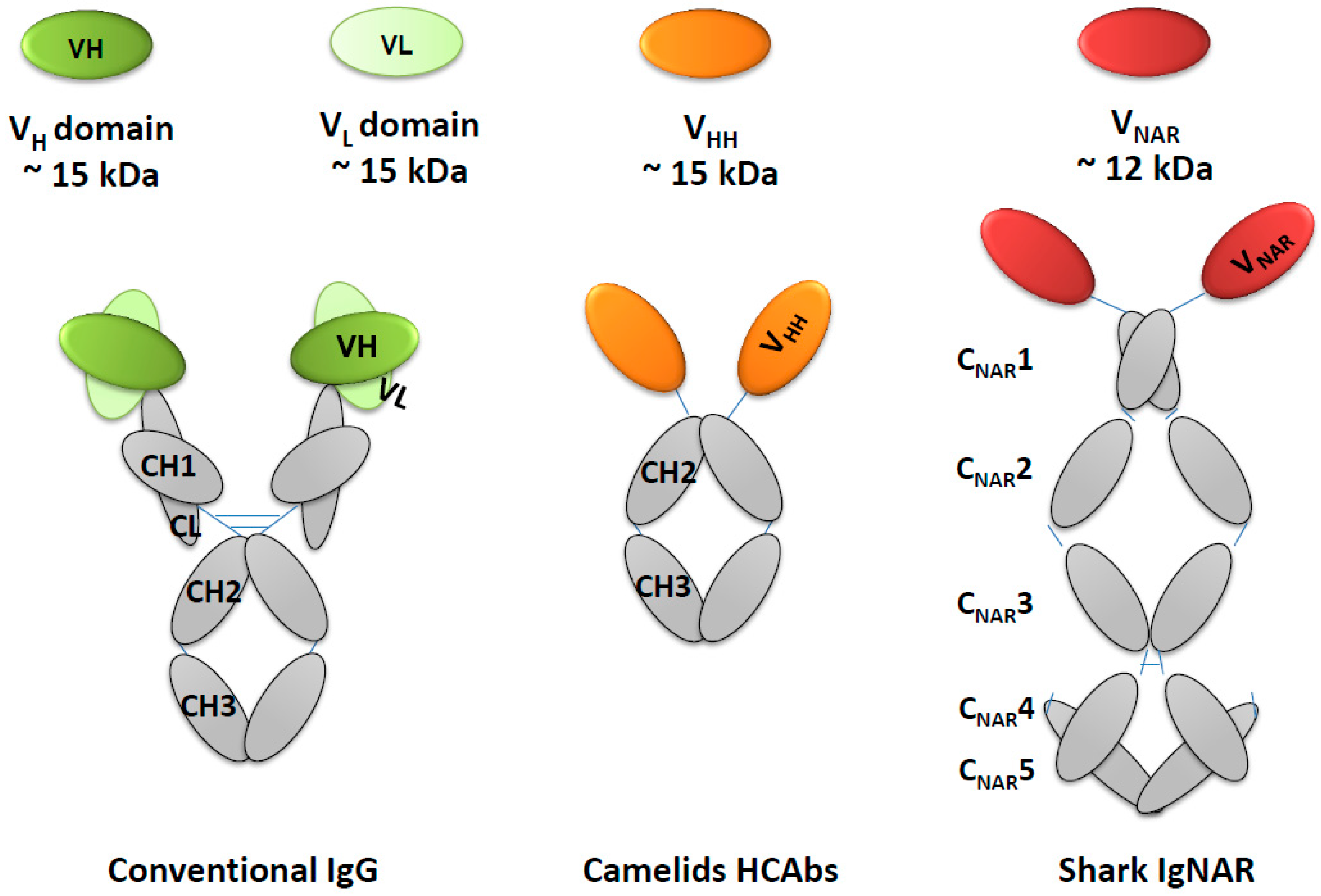
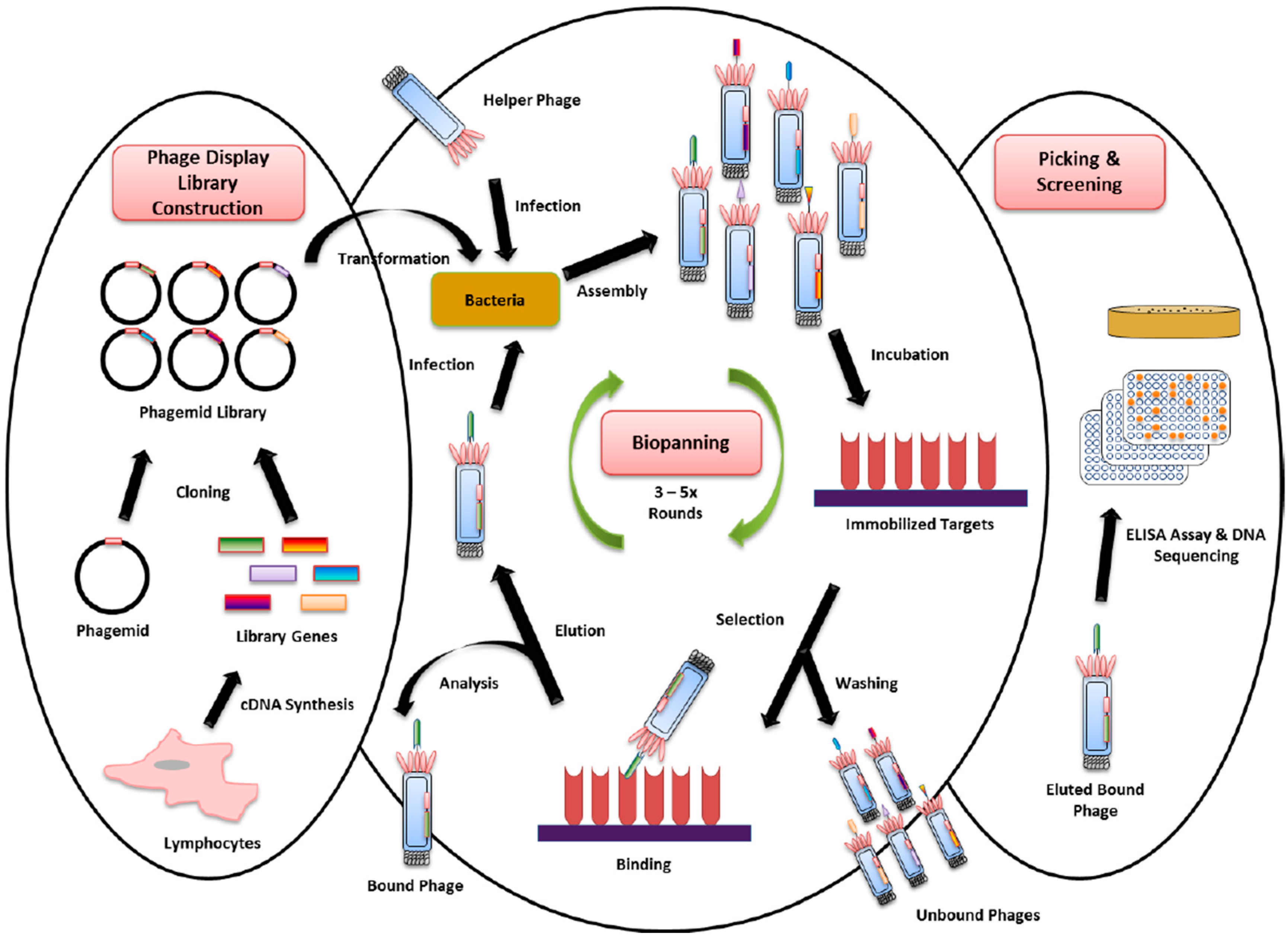
| Characteristics | Monoclonal Antibody | Polyclonal Antibodies | Recombinant Antibodies |
|---|---|---|---|
| Cost to produce | +++ | + | ++ |
| Difficulty of production | +++ | + | ++ |
| Skills or training | +++ | + | ++ |
| Time scale | +++ | + | ++ |
| Specificity and affinity | +++ | + | +++ |
| Amounts | + | ++ | +++ |
| Commercial availability | ++ | +++ | + |
| Variability | + | +++ | + |
| Target Antigens | Diseases | Applications | Reference |
|---|---|---|---|
| HER2 | Breast cancer | Diagnostic | [284,285,286] |
| TNT | Explosive | Diagnostic | [274,287] |
| Ts14 glycoprotein | T. solium cysticercosis | Diagnostic | [249] |
| LMM, ES, CSE, TSB, LLGPs, VF of T. solium | Neurocysticercosis | Immunodiagnosis | [249] |
| VEGF-A165 | Neoangiogenesis | Diagnostic and therapeutic | [288] |
| HPV-16 L1 protein | Cervical cancer | Diagnostic and therapeutic | [289,290] |
| DARC | Malaria (by P. vivax) | Diagnostic or therapeutic | [291] |
| Poliovirus type 1 Sabin strain particles | Poliomyelitis | Diagnostic and therapeutic | [292,293] |
| CD105 | Angiogenesis related tumors | Diagnostic and therapeutic | [294,295] |
| HSP-60 | Brucellosis (Livestock) | Diagnostic and vaccine | [222,296] |
| Caffeine carboxylate KLH | Beverages | Detection and separation | [251,252] |
| SEB | Toxin | Sensor and diagnostic | [275] |
| Ricin | Toxin | Sensor and diagnostic | [275] |
| BoNT/A | Toxin | Sensor and diagnostic | [235,297,298] |
| CEA | Colon cancer | In vivo imaging | [299,300,301] |
| VCAM1 | Atherosclerotic plaques | Molecular imaging | [302,303,304] |
| EGFR | Tumours | Detection and imaging | [305,306] |
| Scorpion AahII | Toxin | Neutralizing and therapeutic | [276,307] |
| Heat-killed B. melitensis Riv1 | Brucellosis | Diagnostic, therapeutic and vaccination | [282,308,309,310] |
| RSV protein F | Acute lower respiratory tract | Therapeutic | [311] |
| vWF | Thrombosis | Therapeutic | www.ablynx.com |
| TNFα, IL-6R, IgE | Rheumatoid arthritis | Therapeutic | www.ablynx.com |
| RANKL | Bone metastasis | Therapeutic | www.ablynx.com |
| RSV | bronchiolitis and pneumonia | Therapeutic | www.ablynx.com |
| DR5 | Solid tumors | Therapeutic | www.ablynx.com |
| Not stated | Alzheimer’s disease | Therapeutic | www.ablynx.com |
| Target Antigens | Diseases | Applications | Reference |
|---|---|---|---|
| AMA1 (P. falciparum) | Malaria | Diagnostic | [313,314] |
| Zaire ebolavirus viral nucleoprotein | Ebolavirus Haemorrhagic Fever | Diagnostic | [262] |
| Cholera toxin | Toxin | Diagnostic | [258] |
| Tom70 | Human immunosilent target processes | Detection | [323] |
| BoNT/A | Toxin | Sensor and diagnostic | [322] |
| Ricin | Toxin | Sensor and diagnostic | [322] |
| SEB | Toxin | Sensor and diagnostic | [322] |
| HBeAg | Hepatitis B virus | Immunolocalization and diagnostic | [324] |
| Kgp protease (P. gingivalis) | Periodontal disease | Neutralization | [318] |
| Nonfibrillar oligomer formation | Alzheimer’s disease | Modelling | [325] |
| rhTNFα | pro-inflammatory cytokine | Therapeutic | [260,326] |
| mAb idiotope | Cancer | Therapeutic | [327] |
| GPCR’s ion channels | Therapeutic | www.adalta.com.au | |
| Anti-thrombotic drug targets | Cardiovacular disease | Diagnostic and therapeutic | www.adalta.com.au |
| Idiopathic pulmonary fibrosis | Inflammation | Therapeutic | www.adalta.com.au |
| Multiple sclerosis | Central neuron system disease | Therapeutic | www.ossianix.com |
| Botulinum toxin light chain protease | Gastrointestinal tract | Therapeutic | www.ossianix.com |
| Myostatin | Neurological disease | Therapeutic | www.ossianix.com |
| Uveitis | Eye inflammatory | Therapeutic | www.elasmogen.com |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Leow, C.H.; Fischer, K.; Leow, C.Y.; Cheng, Q.; Chuah, C.; McCarthy, J. Single Domain Antibodies as New Biomarker Detectors. Diagnostics 2017, 7, 52. https://doi.org/10.3390/diagnostics7040052
Leow CH, Fischer K, Leow CY, Cheng Q, Chuah C, McCarthy J. Single Domain Antibodies as New Biomarker Detectors. Diagnostics. 2017; 7(4):52. https://doi.org/10.3390/diagnostics7040052
Chicago/Turabian StyleLeow, Chiuan Herng, Katja Fischer, Chiuan Yee Leow, Qin Cheng, Candy Chuah, and James McCarthy. 2017. "Single Domain Antibodies as New Biomarker Detectors" Diagnostics 7, no. 4: 52. https://doi.org/10.3390/diagnostics7040052
APA StyleLeow, C. H., Fischer, K., Leow, C. Y., Cheng, Q., Chuah, C., & McCarthy, J. (2017). Single Domain Antibodies as New Biomarker Detectors. Diagnostics, 7(4), 52. https://doi.org/10.3390/diagnostics7040052






