Role of Airwave Oscillometry in Patients with Combined Fibrosis–Emphysema Syndrome (CPFE) with Preserved FEV1/FVC Ratio
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Airwave Oscillometry (AOS)
2.3. Definition of the Criteria for Small-Airway Disease (SAD)
2.4. Statistical Analysis
3. Results
4. Discussion
Study Limitations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Cottin, V.; Selman, M.; Inoue, Y.; Wong, A.W.; Corte, T.J.; Flaherty, K.R.; Han, M.K.; Jacob, J.; Johannson, K.A.; Kitaichi, M.; et al. Syndrome of Combined Pulmonary Fibrosis and Emphysema An Official ATS/ERS/JRS/ALAT Research Statement. Am. J. Respir. Crit. Care Med. 2022, 206, E7–E41. [Google Scholar] [CrossRef]
- Cottin, V.; Cordier, J.-F. The Syndrome of Combined Pulmonary Fibrosis and Emphysema. Chest 2009, 136, 1–2. [Google Scholar] [CrossRef]
- Jen, R.; Ryerson, C.J. Combined pulmonary fibrosis and emphysema. Curr. Respir. Care Rep. 2013, 2, 254–259. [Google Scholar] [CrossRef]
- Ryerson, C.J.; Hartman, T.; Elicker, B.M.; Ley, B.; Lee, J.S.; Abbritti, M.; Jones, K.D.; King, T.E.; Ryu, J.; Collard, H.R. Clinical Features and Outcomes in Combined Pulmonary Fibrosis and Emphysema in Idiopathic Pulmonary Fibrosis. Chest 2013, 144, 234–240. [Google Scholar] [CrossRef] [PubMed]
- Manjunath, K.S.; Udnur, H. HRCT diagnosis of combined pulmonary fibrosis and emphysema in a patient of chronic obstructive pulmonary disease with pulmonary hypertension and clinical or radiograph suspicion of pulmonary fibrosis. BJR Case Rep. 2016, 2, 20150070. [Google Scholar] [CrossRef]
- Lin, H.; Jiang, S. Combined pulmonary fibrosis and emphysema (CPFE): An entity different from emphysema or pulmonary fibrosis alone. J. Thorac. Dis. 2015, 7, 767–779. [Google Scholar]
- Kitaguchi, Y.; Fujimoto, K.; Hanaoka, M.; Honda, T.; Hotta, J.; Hirayama, J. Pulmonary function impairment in patients with combined pulmonary fibrosis and emphysema with and without airflow obstruction. Int. J. COPD 2014, 9, 805–811. [Google Scholar] [CrossRef] [PubMed]
- Çiftci, F.; Gülpınar, B.; Atasoy, Ç.; Kayacan, O.; Saryal, S. Combined pulmonary fibrosis and emphysema: How does cohabitation affect respiratory functions? Adv. Med. Sci. 2019, 64, 285–291. [Google Scholar] [CrossRef]
- Bodlet, A.; Maury, G.; Jamart, J.; Dahlqvist, C. Influence of radiological emphysema on lung function test in idiopathic pulmonary fibrosis. Respir. Med. 2013, 107, 1781–1788. [Google Scholar] [CrossRef] [PubMed]
- Kitaguchi, Y.; Fujimoto, K.; Hayashi, R.; Hanaoka, M.; Honda, T.; Kubo, K. Annual changes in pulmonary function in combined pulmonary fibrosis and emphysema: Over a 5-year follow-up. Respir. Med. 2013, 107, 1986–1992. [Google Scholar] [CrossRef]
- Turan, M.O.; Karasu, S.; Anar, C.; Sertogullarindan, B. Comparison of emphysematous COPD patients with or without Combined Pulmonary Fibrosis and Emphysema (CPFE). Eur. Respir. J. 2023, 62, PA3462. [Google Scholar] [CrossRef]
- Petraglia, L.; Komici, K.; Bencivenga, L.; Rengo, G.; Pagliaro, R.; Sciattarella, A.; Ferrara, N.; Bianco, A.; Guerra, G. The association between muscle strength and z scores of pulmonary function. Sci. Rep. 2025, 15, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Calaras, D.; Mathioudakis, A.G.; Lazar, Z.; Corlateanu, A. Combined Pulmonary Fibrosis and Emphysema: Comparative Evidence on a Complex Condition. Biomedicines 2023, 11, 1636. [Google Scholar] [CrossRef]
- Jankowich, M.D.; Rounds, S.I.S. Combined pulmonary fibrosis and emphysema syndrome: A review. Chest 2012, 141, 222–231. [Google Scholar] [CrossRef]
- Pagliaro, R.; Aronne, L.; Fomez, R.; Ferri, V.; Montella, A.; Zamparelli, S.S.; Bianco, A.; Perrotta, F. High-Flow Nasal Cannula System in Respiratory Failure Associated with Interstitial Lung Diseases: A Systematic Review and Narrative Synthesis. J. Clin. Med. 2024, 13, 2956. [Google Scholar] [CrossRef]
- Pagliaro, R.; Simeon, V.; Notizia, L.; Arena, S.; Mariniello, D.F.; Stella, G.M.; Bianco, A.; Perrotta, F.; Aronne, L. The Efficacy of High-Flow Nasal Cannula (HFNC) Treatment in Patients with Chronic Type II Respiratory Failure Secondary to COPD. J. Clin. Med. 2026, 15, 1924. [Google Scholar] [CrossRef]
- Lee, J.H.; Lim, C.-M.; Koh, Y.; Hong, S.-B.; Song, J.-W.; Huh, J.W. High-flow nasal cannula oxygen therapy in idiopathic pulmonary fibrosis patients with respiratory failure. J. Thorac. Dis. 2020, 12, 966–972. [Google Scholar] [CrossRef]
- Pagliaro, R.; Della Monica, P.; D’agnano, V.; Schiattarella, A.; D’orologio, A.; Medusa, P.M.; Stella, G.M.; Colapietra, F.; Perrotta, F.; Bianco, A.; et al. Is Immunotherapy a Contraindication for Treating Lung Cancer Patients with Interstitial Lung Diseases? A Review of the Literature. J. Clin. Med. 2026, 15, 996. [Google Scholar] [CrossRef]
- Vitale, M.; Pagliaro, R.; Viscardi, G.; Pastore, L.; Castaldo, G.; Perrotta, F.; Campbell, S.F.; Bianco, A.; Scialò, F. Unraveling Resistance in Lung Cancer Immunotherapy: Clinical Milestones, Mechanistic Insights, and Future Strategies. Int. J. Mol. Sci. 2025, 26, 9244. [Google Scholar] [CrossRef] [PubMed]
- Akagi, T.; Matsumoto, T.; Harada, T.; Tanaka, M.; Kuraki, T.; Fujita, M.; Watanabe, K. Coexistent emphysema delays the decrease of vital capacity in idiopathic pulmonary fibrosis. Respir. Med. 2009, 103, 1209–1215. [Google Scholar] [CrossRef] [PubMed]
- Nathan, S.D.; Yang, M.; Morgenthien, E.A.; Stauffer, J.L. FVC variability in patients with IPF and role of 6-min walk test to predict further change. Eur. Respir. J. 2020, 55, 1902151. [Google Scholar] [CrossRef]
- Sharpe, A.L.; Reibman, J.; Oppenheimer, B.W.; Goldring, R.M.; Liu, M.; Shao, Y.; Bohart, I.; Kwok, B.; Weinstein, T.; Addrizzo-Harris, D.; et al. Role of small airway dysfunction in unexplained exertional dyspnoea. ERJ Open Res. 2023, 9. [Google Scholar] [CrossRef]
- Kraemer, R.; Matthys, H. Small airway disease assessed by parameters of small airway dysfunction in patients with asthma, asthma-chronic obstructive pulmonary disease-overlap, and chronic obstructive pulmonary disease. Explor. Asthma Allergy 2024, 2, 85–96. [Google Scholar] [CrossRef]
- Agrawal, A.; Desiraju, K. Impulse oscillometry: The state-of-art for lung function testing. Lung India 2016, 33, 410–416. [Google Scholar] [CrossRef]
- D’Agnano, V.; Mariniello, D.F.; Pagliaro, R.; Far, M.S.; Schiattarella, A.; Scialò, F.; Stella, G.; Matera, M.G.; Cazzola, M.; Bianco, A.; et al. Sirtuins and Cellular Senescence in Patients with Idiopathic Pulmonary Fibrosis and Systemic Autoimmune Disorders. Drugs 2024, 84, 491–501. [Google Scholar] [CrossRef] [PubMed]
- Grabowska, W.; Sikora, E.; Bielak-Zmijewska, A. Sirtuins, a promising target in slowing down the ageing process. Biogerontology 2017, 18, 447–476. [Google Scholar] [CrossRef]
- Scialò, F.; Pagliaro, R.; Gelzo, M.; Matera, M.G.; D’aGnano, V.; Zamparelli, S.S.; Castaldo, G.; Cazzola, M.; Bianco, A.; Perrotta, F. Fatty Acids Dysregulation Correlates with Lung Function in Idiopathic Pulmonary Fibrosis. Lung 2025, 203, 1–10. [Google Scholar] [CrossRef]
- Nigro, E.; D’aGnano, V.; Pagliaro, R.; Mallardo, M.; Bianco, A.; Picone, C.; D’eRrico, A.G.; Daniele, A.; Perrotta, F. Exploring the role of serum adiponectin and its holigomerization in fibrotic interstitial lung diseases: Results from a cross-sectional study. BMC Pulm. Med. 2025, 25, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Mazzarella, G.; Esposito, V.; Bianco, A.; Ferraraccio, F.; Prati, M.; Lucariello, A.; Manente, L.; Mezzogiorno, A.; De Luca, A. Inflammatory effects on human lung epithelial cells after exposure to diesel exhaust micron sub particles (PM1.0) and pollen allergens. Environ. Pollut. 2012, 161, 64–69. [Google Scholar] [CrossRef]
- Pagliaro, R.; Scialò, F.; Schiattarella, A.; Cianci, R.; Campbell, S.F.M.; Perrotta, F.; Bianco, A.; Castaldo, G. Mechanisms of Lung Cancer Development in Cystic Fibrosis Patients: The Role of Inflammation, Oxidative Stress, and Lung Microbiome Dysbiosis. Biomolecules 2025, 15, 828. [Google Scholar] [CrossRef]
- D’Agnano, V.; Perrotta, F.; Fomez, R.; Carrozzo, V.M.; Schiattarella, A.; Zamparelli, S.S.; Pagliaro, R.; Bianco, A.; Mariniello, D.F. Pharmacological Treatment of Interstitial Lung Diseases: A Novel Landscape for Inhaled Agents. Pharmaceutics 2024, 16, 1391. [Google Scholar] [CrossRef] [PubMed]
- Perrotta, F.; Zamparelli, S.S.; D’agnano, V.; Montella, A.; Fomez, R.; Pagliaro, R.; Schiattarella, A.; Cazzola, M.; Bianco, A.; Mariniello, D.F. Genomic Profiling for Predictive Treatment Strategies in Fibrotic Interstitial Lung Disease. Biomedicines 2024, 12, 1384. [Google Scholar] [CrossRef] [PubMed]
- Stanojevic, S.; Kaminsky, D.A.; Miller, M.R.; Thompson, B.; Aliverti, A.; Barjaktarevic, I.; Cooper, B.G.; Culver, B.; Derom, E.; Hall, G.L.; et al. ERS/ATS technical standard on interpretive strategies for routine lung function tests. Eur. Respir. J. 2022, 60, 2101499. [Google Scholar] [CrossRef]
- Sarkar, S.; Jadhav, U.; Ghewade, B.; Sarkar, S.; Wagh, P. Oscillometry in Lung Function Assessment: A Comprehensive Review of Current Insights and Challenges. Cureus 2023, 15, e47935. [Google Scholar] [CrossRef]
- King, G.G.; Bates, J.; Berger, K.I.; Calverley, P.; De Melo, P.L.; Dellacà, R.L.; Farre, R.; Hall, G.; Ioan, I.; Irvin, C.G.; et al. Technical standards for respiratory oscillometry. Eur. Respir. J. 2020, 55, 1900753. [Google Scholar] [CrossRef]
- Tirelli, C.; Mira, S.; Italia, M.; Maggioni, S.; Intravaia, C.; Zava, M.; Contino, S.; Parazzini, E.M.; Mondoni, M. Applications of Forced Oscillatory Technique in Obstructive and Restrictive Pulmonary Diseases: A Concise State of the Art. J. Clin. Med. 2025, 14, 5718. [Google Scholar] [CrossRef]
- Kuo, C.R.; Jabbal, S.; Lipworth, B. I Say IOS You Say AOS: Comparative Bias in Respiratory Impedance Measurements. Lung 2019, 197, 473–481. [Google Scholar] [CrossRef]
- Janson, C.; Malinovschi, A.; Amaral, A.F.S.; Accordini, S.; Bousquet, J.; Buist, A.S.; Canonica, G.W.; Dahlén, B.; Garcia-Aymerich, J.; Gnatiuc, L.; et al. Bronchodilator reversibility in asthma and COPD: Findings from three large population studies. Eur. Respir. J. 2019, 54, 1900561. [Google Scholar] [CrossRef]
- Tuomisto, L.E.; Ilmarinen, P.; Lehtimäki, L.; Tommola, M.; Kankaanranta, H. Immediate bronchodilator response in FEV1 as a diagnostic criterion for adult asthma. Eur. Respir. J. 2019, 53, 1800904. [Google Scholar] [CrossRef] [PubMed]
- Ley, B.; Ryerson, C.J.; Vittinghoff, E.; Ryu, J.; Tomassetti, S.; Lee, J.S.; Poletti, V.; Buccioli, M.; Elicker, B.M.; Jones, K.D.; et al. A Multidimensional Index and Staging System for Idiopathic Pulmonary Fibrosis. Ann. Intern. Med. 2012, 156, 684–691. [Google Scholar] [CrossRef]
- Cottini, M.; Bondi, B.; Bagnasco, D.; Braido, F.; Passalacqua, G.; Licini, A.; Lombardi, C.; Berti, A.; Comberiati, P.; Landi, M.; et al. Impulse oscillometry defined small airway dysfunction in asthmatic patients with normal spirometry: Prevalence, clinical associations, and impact on asthma control. Respir. Med. 2023, 218, 107391. [Google Scholar] [CrossRef]
- Lazarinis, N.; Evangelia, F.; Anders, L.; Bossios, A. Small airways disease in chronic obstructive pulmonary disease. Expert Rev. Respir. Med. 2024, 18, 539–552. [Google Scholar] [CrossRef]
- Mariniello, D.F.; D’agnano, V.; Cennamo, D.; Conte, S.; Quarcio, G.; Notizia, L.; Pagliaro, R.; Schiattarella, A.; Salvi, R.; Bianco, A.; et al. Comorbidities in COPD: Current and Future Treatment Challenges. J. Clin. Med. 2024, 13, 743. [Google Scholar] [CrossRef]
- Huang, Y.; Zhang, X.; Wang, J.; Bao, W.; Lv, C.; Zhang, Y.; Tian, X.; Zhou, Y.; Zhang, M. Role of impulse oscillometry in chronic obstructive pulmonary disease and asthma-chronic obstructive pulmonary disease overlap. Clin. Transl. Allergy 2025, 15, e70057. [Google Scholar] [CrossRef]
- Karayama, M.; Inui, N.; Mori, K.; Kono, M.; Hozumi, H.; Suzuki, Y.; Furuhashi, K.; Hashimoto, D.; Enomoto, N.; Fujisawa, T.; et al. Respiratory impedance is correlated with morphological changes in the lungs on three-dimensional CT in patients with COPD. Sci. Rep. 2017, 7, 41709. [Google Scholar] [CrossRef] [PubMed]
- Shaikh, S.B.; Prabhu, A.; Bhandary, Y.P. Targeting anti-aging protein sirtuin (Sirt) in the diagnosis of idiopathic pulmonary fibrosis. J. Cell. Biochem. 2019, 120, 6878–6885. [Google Scholar] [CrossRef] [PubMed]
- Cottini, M.; Licini, A.; Lombardi, C.; Berti, A. Clinical Characterization and Predictors of IOS-Defined Small-Airway Dysfunction in Asthma. J. Allergy Clin. Immunol. Pract. 2020, 8, 997–1004.e2. [Google Scholar] [CrossRef]
- Su, Z.Q.; Guan, W.-J.; Li, S.-Y.; Ding, M.; Chen, Y.; Jiang, M.; Chen, X.-B.; Zhong, C.-H.; Tang, C.-L.; Zhong, N.-S. Significances of spirometry and impulse oscillometry for detecting small airway disorders assessed with endobronchial optical coherence tomography in COPD. Int. J. COPD 2018, 13, 3031–3044. [Google Scholar] [CrossRef]
- Xu, W.J.; Shang, W.-Y.; Feng, J.-M.; Song, X.-Y.; Li, L.-Y.; Xie, X.-P.; Wang, Y.-M.; Liang, B.-M. Machine learning for accurate detection of small airway dysfunction-related respiratory changes: An observational study. Respir. Res. 2024, 25, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Linardatos, P.; Papastefanopoulos, V.; Kotsiantis, S. Explainable ai: A review of machine learning interpretability methods. Entropy 2021, 23, 18. [Google Scholar] [CrossRef]
- Li, L.Y.; Yan, T.-S.; Yang, J.; Li, Y.-Q.; Fu, L.-X.; Lan, L.; Liang, B.-M.; Wang, M.-Y.; Luo, F.-M. Impulse oscillometry for detection of small airway dysfunction in subjects with chronic respiratory symptoms and preserved pulmonary function. Respir. Res. 2021, 22, 1–10. [Google Scholar] [CrossRef]
- Huang, C.H.; Chou, K.T.; Perng, D.W.; Hsiao, Y.H.; Huang, C.W. Using Machine Learning with Impulse Oscillometry Data to Develop a Predictive Model for Chronic Obstructive Pulmonary Disease and Asthma. J. Pers. Med. 2024, 14, 398. [Google Scholar] [CrossRef] [PubMed]
- Fanizzi, A.; Fadda, F.; Maddalo, M.; Saponaro, S.; Lorenzon, L.; Ubaldi, L.; Lambri, N.; Giuliano, A.; Loi, E.; Signoriello, M.; et al. Developing an ensemble machine learning study: Insights from a multi-center proof-of-concept study. PLoS ONE 2024, 19, e0303217. [Google Scholar] [CrossRef]
- Zhou, Y.; Zhou, Y.; Tong, Z.; Zhu, X.; Wu, C.; Zhou, Y.; Dong, Z. Deciphering the cellular and molecular landscape of pulmonary fibrosis through single-cell sequencing and machine learning. J. Transl. Med. 2025, 23, 3. [Google Scholar] [CrossRef]
- Zaidan, M.F.; Reddy, A.P.; Duarte, A. Impedance Oscillometry: Emerging Role in the Management of Chronic Respiratory Disease. Curr. Allergy Asthma Rep. 2018, 18, 3. [Google Scholar] [CrossRef] [PubMed]
- Chiu, H.-Y.; Hsiao, Y.-H.; Su, K.-C.; Lee, Y.-C.; Ko, H.-K.; Perng, D.-W. Small Airway Dysfunction by Impulse Oscillometry in Symptomatic Patients with Preserved Pulmonary Function. J. Allergy Clin. Immunol. Pract. 2020, 8, 229–235.e3. [Google Scholar] [CrossRef]
- Salvi, S.; Brashier, B. Measuring lung function using sound waves: Role of the forced oscillation technique and impulse oscillometry system. Breathe 2015, 11, 57–65. [Google Scholar] [CrossRef]
- Pisi, R.; Aiello, M.; Zanini, A.; Tzani, P.; Paleari, D.; Marangio, E.; Spanevello, A.; Nicolini, G.; Chetta, A. Small airway dysfunction and flow and volume bronchodilator responsiveness in patients with chronic obstructive pulmonary disease. Int. J. COPD 2015, 10, 1191–1197. [Google Scholar]
- Toumpanakis, D.; Karagiannis, K.; Paredi, P.; Bikov, A.; Bonifazi, M.; Lota, H.K.; Kalsi, H.; Minelli, C.; Dikaios, N.; Kastis, G.A.; et al. Contribution of Peripheral Airways Dysfunction to Poor Quality of Life in Sarcoidosis. Chest 2025, 168, 423–434. [Google Scholar] [CrossRef] [PubMed]
- Papapostolou, G.; Nasr, A.; Jarenbäck, L.; Romberg, K.; Tunsäter, A.; Ankerst, J.; Bjermer, L.; Tufvesson, E. Peripheral Airway Obstruction in Association with Symptoms and Quality of Life in Asthma. J. Asthma Allergy 2025, 18, 491–505. [Google Scholar] [CrossRef]
- Barkas, G.I.; Daniil, Z.; Kotsiou, O.S. The Role of Small Airway Disease in Pulmonary Fibrotic Diseases. J. Pers. Med. 2023, 13, 1600. [Google Scholar] [CrossRef] [PubMed]
- Bickel, S.; Popler, J.; Lesnick, B.; Eid, N. Impulse Oscillometry: Interpretation and Practical Applications. Chest 2014, 146, 841–847. [Google Scholar] [CrossRef] [PubMed]
- Ong-Salvador, R.; Laveneziana, P.; de Jongh, F. ERS/ATS Global Lung Function Initiative normal values and classifying severity based on z-scores instead of per cent predicted. Breathe 2024, 20, 230227. [Google Scholar] [CrossRef]
- Bednarek, M.; Grabicki, M.; Piorunek, T.; Batura-Gabryel, H. Current place of impulse oscillometry in the assessment of pulmonary diseases. Respir. Med. 2020, 170, 105952. [Google Scholar] [CrossRef] [PubMed]

| CFPE Patients | n.52 | ||
|---|---|---|---|
| Gender | Pulmonary Function Test Parameters | ||
| Male | 44 (84.6%) | FEV1% Pre-bd | 88.73 ± 17.81 |
| Female | 8 (15.4%) | FEV1 (l) Pre-bd | 2.18 ± 0.45 |
| Age (Years) | FVC% Pre-bd | 81.08 ± 17.08 | |
| 70.8 (71.40 ± 3.93) | FVC (l) Pre-bd | 2.59 ± 0.51 | |
| Smoking History | FEV1/FVC Pre-bd | 84.26 ± 6.45 | |
| Former | 52 (100%) | FEF 75% pre-bd | 74.93 ± 14.36 |
| mMMRC | FEF 50% pre-bd | 67.66 ± 20.91 | |
| 0–1 | 24 (46.6%) | FEF 25% pre-bd | 79.89 ± 20.42 |
| ≥2 | 28 (53.4%) | DLCO% pre-bd | 37.45 ± 17.10 |
| GAP Stages | TLC% pre-bd | 66.92 ± 12.91 | |
| Stage I (0–3) | 18 (27.3%) | TLC (l) pre-bd | 3.88 ± 0.96 |
| Stage II (4–5) | 26 (54.5%) | Airwave Oscillometry Parameters | |
| Stage III (6–8) | 8 (18.2%) | R5 pre-bd (kPa·s·L−1) | 3.21 ± 1.01 |
| COPD treatment | R5–19 pre-bd (kPa·s·L−1) | 0.07 ± 0.06 | |
| None | 28 (53.8%) | ∆R5–19 (kPa·s·L−1) | 0.20 ± 1.85 |
| LAMA | 14 (26.9%) | Ax pre-bd (kPa·L−1) | 10.40 ± 4.93 |
| LABA-LAMA | 10 (19.2%) | ∆Ax (kPa·L−1) | −0.01 ± 0.22 |
| Antifibrotics | Arterial Blood Gas Analysis Parameters | ||
| None | 16 (30.8%) | Ph | 7.44 ± 0.02 |
| Pirfenidone | 17 (32.7%) | pO2 (mmHg) | 77.94 ± 9.40 |
| Nintedanib | 19 (36.5%) | pCO2 (mmHg) | 38.17 ± 3.27 |
| Oxygen Supplementation | HCO3− (mmol/L) | 26.00 ± 2.28 | |
| Rest/Intermittent Oxygen | 34 (65.4%) | Lac (mmol/L) | 1.06 ± 0.70 |
| SAD-Group n = 21 pts | Non-SAD Group n = 31 pts | p-Value | |
|---|---|---|---|
| Age (yrs) | 72.40 ± 3.73 | 72.29 ± 4.60 | 0.623 |
| Gender (Male) | 16 | 28 | 0.531 |
| GAP Index | 3.75 ± 1.39 | 4.71 ± 1.54 | 0.160 |
| mMRC (≥2) | 12 | 16 | 0.856 |
| Oxygen Supplementation | 7 | 25 | 0.070 |
| Use of bronchodilators | 16 | 8 | 0.015 |
| Use of antifibrotics | 16 | 20 | 0.856 |
| Pre-BD | Post-BD | |||||
|---|---|---|---|---|---|---|
| SAD | Non-SAD | p-Value | SAD | Non-SAD | p-Value | |
| FEV1 L | 2.32 ± 0.37 | 2.17 ± 0.29 | 0.113 | 2.30 ± 0.27 | 2.20 ± 0.32 | 0.210 |
| FEV1% | 90.43 ± 9.69 | 87.26 ± 13.87 | 0.737 | 85.47 ± 8.72 | 86.58 ± 12.66 | 0.711 |
| FVC L | 2.87 ± 0.30 | 2.72 ± 0.42 | 0.126 | 2.90 ± 0.31 | 2.81 ± 0.42 | 0.411 |
| FVC% | 80.55 ± 9.75 | 79.33 ± 12.85 | 0.641 | 85.66 ± 9.16 | 80.94 ± 14.58 | 0.157 |
| FEV1/FVC | 82.70 ± 4.75 | 81.90 ± 6.40 | 0.753 | 80.65 ± 6.40 | 82.75 ± 5.80 | 0.690 |
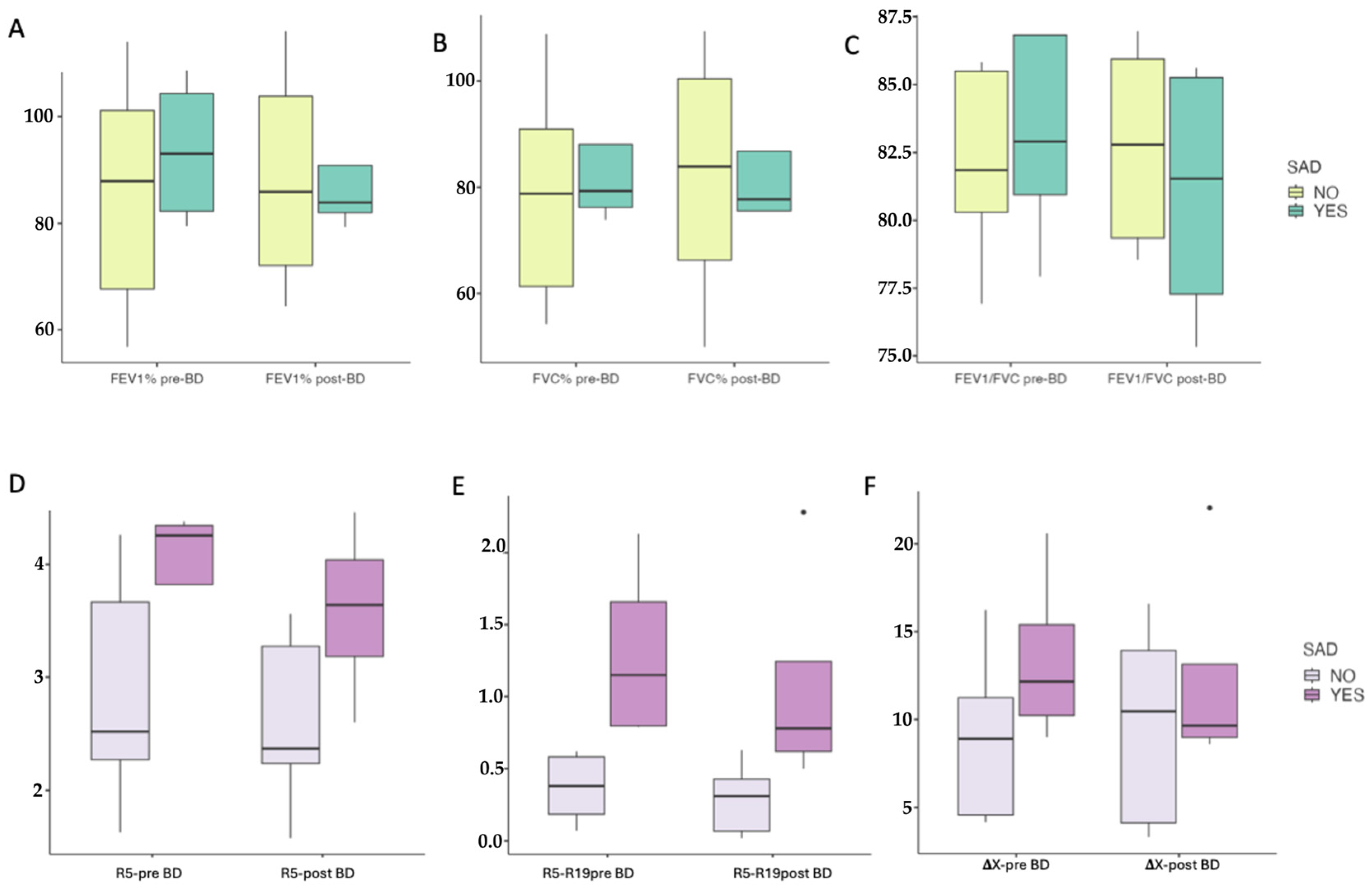
| R5 | 3.21 ± 0.88 | 2.81 ± 0.65 | 0.012 | 3.03 ± 0.81 | 2.73 ± 0.87 | <0.001 |
| R5-R19 | 0.97 ± 0.57 | 0.51 ± 0.24 | <0.001 | 0.97 ± 0.57 | 0.51 ± 0.29 | 0.002 |
| AX | 11.78 ± 4.22 | 9.33 ± 4.16 | 0.024 | 11.53 ± 4.65 | 9.85 ± 3.32 | 0.026 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Pagliaro, R.; Scialò, F.; Mariniello, D.F.; D’Agnano, V.; Palma, M.I.; Campbell, S.F.; Ora, J.; Cerqua, F.S.; Stella, G.M.; Bianco, A.; et al. Role of Airwave Oscillometry in Patients with Combined Fibrosis–Emphysema Syndrome (CPFE) with Preserved FEV1/FVC Ratio. Diagnostics 2026, 16, 1159. https://doi.org/10.3390/diagnostics16081159
Pagliaro R, Scialò F, Mariniello DF, D’Agnano V, Palma MI, Campbell SF, Ora J, Cerqua FS, Stella GM, Bianco A, et al. Role of Airwave Oscillometry in Patients with Combined Fibrosis–Emphysema Syndrome (CPFE) with Preserved FEV1/FVC Ratio. Diagnostics. 2026; 16(8):1159. https://doi.org/10.3390/diagnostics16081159
Chicago/Turabian StylePagliaro, Raffaella, Filippo Scialò, Domenica Francesca Mariniello, Vito D’Agnano, Maria Ilaria Palma, Susan F. Campbell, Josuel Ora, Francesco Saverio Cerqua, Giulia Maria Stella, Andrea Bianco, and et al. 2026. "Role of Airwave Oscillometry in Patients with Combined Fibrosis–Emphysema Syndrome (CPFE) with Preserved FEV1/FVC Ratio" Diagnostics 16, no. 8: 1159. https://doi.org/10.3390/diagnostics16081159
APA StylePagliaro, R., Scialò, F., Mariniello, D. F., D’Agnano, V., Palma, M. I., Campbell, S. F., Ora, J., Cerqua, F. S., Stella, G. M., Bianco, A., & Perrotta, F. (2026). Role of Airwave Oscillometry in Patients with Combined Fibrosis–Emphysema Syndrome (CPFE) with Preserved FEV1/FVC Ratio. Diagnostics, 16(8), 1159. https://doi.org/10.3390/diagnostics16081159







