ADC Histogram Features of Breast Cancer Brain Metastases as Candidate Imaging Biomarkers of Primary Tumor ER, PR, Ki-67, and Luminal Status
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design and Ethics
2.2. Patient Selection
2.3. MRI Acquisition
2.4. Lesion Identification and Selection

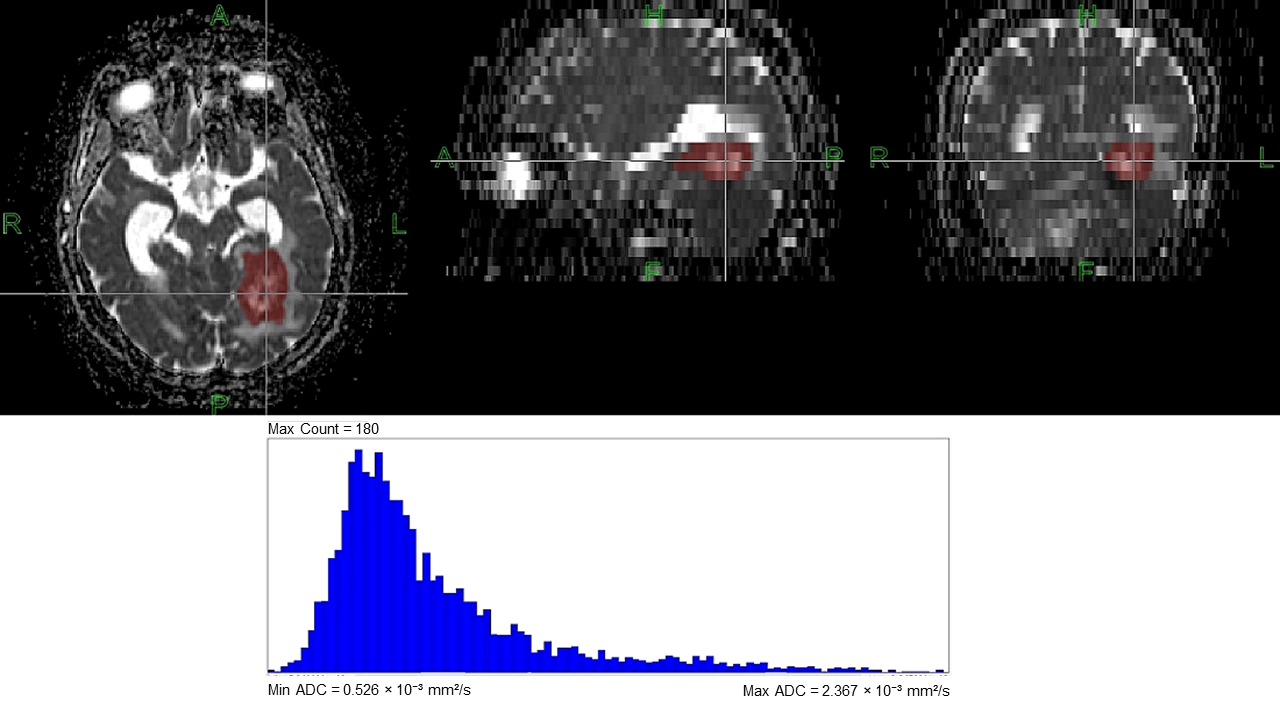
2.5. Post-Processing and ADC Histogram Analysis
2.6. Pathological Evaluation
2.7. Statistical Analysis
3. Results
3.1. Cohort Characteristics
3.2. Group Comparisons
3.3. Interobserver Agreement
3.4. Logistic Regression and ROC Performance
3.4.1. ER Prediction
3.4.2. PR Prediction
3.4.3. Ki-67 Prediction
3.4.4. Luminality Prediction
4. Discussion
4.1. ER and PR Prediction: Emphasis on Low-Percentile Diffusion Metrics
4.2. HER2: Lack of Discriminatory Value for ADC Histograms in BCBM
4.3. Ki-67: Biological Interpretation and the Role of Percentile Selection
4.4. Luminality: ADC10-Based Discrimination of Luminal vs. Non-Luminal Breast Cancer Brain Metastases
4.5. Clinical Relevance: Toward Non-Invasive Biomarker Estimation in BCBM
4.6. Thresholds, AUC, and Pperformance Reporting
4.7. Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ADC | apparent diffusion coefficient |
| AIC | Akaike information criterion |
| ASCO/CAP | American Society of Clinical Oncology/College of American Pathologists |
| AUC | area under the receiver operating characteristic curve |
| BCBM | breast cancer brain metastases |
| CI | confidence interval |
| DWI | diffusion-weighted imaging |
| ER | estrogen receptor |
| FLAIR | fluid-attenuated inversion recovery |
| HER2 | human epidermal growth factor receptor 2 |
| ICC | intraclass correlation coefficient |
| IQR | interquartile range |
| Ki-67 | Ki-67 proliferation index |
| log_volume | log-transformed lesion volume |
| MRI | magnetic resonance imaging |
| N | negative (biomarker status) |
| OR | odds ratio |
| P | positive (biomarker status) |
| p_ER | model-predicted probability of ER positivity |
| p_ki67 | model-predicted probability of Ki-67 positivity |
| p_N | model-predicted probability of non-luminal status |
| p_PR | model-predicted probability of PR positivity |
| PR | progesterone receptor |
| ROC | receiver operating characteristic |
| ROI | region of interest |
| T1WI | T1-weighted imaging |
| T2WI | T2-weighted imaging |
| VOI | volume of interest |
References
- Cho, S.; Joo, B.; Park, M.; Ahn, S.J.; Suh, S.H.; Park, Y.W.; Ahn, S.S.; Lee, S.-K. A Radiomics-Based Model for Potentially More Accurate Identification of Subtypes of Breast Cancer Brain Metastases. Yonsei Med. J. 2023, 64, 573–580. [Google Scholar] [CrossRef]
- Heitkamp, A.; Madesta, F.; Amberg, S.; Wahaj, S.; Schröder, T.; Bechstein, M.; Meyer, L.; Broocks, G.; Hanning, U.; Gauer, T.; et al. Discordant and Converting Receptor Expressions in Brain Metastases from Breast Cancer: MRI-Based Non-Invasive Receptor Status Tracking. Cancers 2023, 15, 2880. [Google Scholar] [CrossRef]
- Luo, X.; Xie, H.; Yang, Y.; Zhang, C.; Zhang, Y.; Li, Y.; Yang, Q.; Wang, D.; Luo, Y.; Mai, Z.; et al. Radiomic Signatures for Predicting Receptor Status in Breast Cancer Brain Metastases. Front. Oncol. 2022, 12, 878388. [Google Scholar] [CrossRef]
- Horvat, J.V.; Bernard-Davila, B.; Helbich, T.H.; Zhang, M.; Morris, E.A.; Thakur, S.B.; Ochoa-Albiztegui, R.E.; Leithner, D.; Marino, M.A.; Baltzer, P.A.; et al. Diffusion-weighted Imaging (DWI) with Apparent Diffusion Coefficient (ADC) Mapping as a Quantitative Imaging Biomarker for Prediction of Immunohistochemical Receptor Status, Proliferation Rate, and Molecular Subtypes of Breast Cancer. J. Magn. Reson. Imaging 2019, 50, 836–846. [Google Scholar] [CrossRef] [PubMed]
- Iima, M.; Kataoka, M.; Honda, M.; Le Bihan, D. Diffusion-Weighted MRI for the Assessment of Molecular Prognostic Biomarkers in Breast Cancer. Korean J. Radiol. 2024, 25, 623–633. [Google Scholar] [CrossRef]
- Meyer, H.-J.; Wienke, A.; Surov, A. Diffusion-Weighted Imaging of Different Breast Cancer Molecular Subtypes: A Systematic Review and Meta-Analysis. Breast Care 2022, 17, 47–54. [Google Scholar] [CrossRef] [PubMed]
- Surov, A.; Chang, Y.-W.; Li, L.; Martincich, L.; Partridge, S.C.; Kim, J.Y.; Wienke, A. Apparent Diffusion Coefficient Cannot Predict Molecular Subtype and Lymph Node Metastases in Invasive Breast Cancer: A Multicenter Analysis. BMC Cancer 2019, 19, 1043. [Google Scholar] [CrossRef]
- Ahn, S.J.; Park, M.; Bang, S.; Cho, E.; Ahn, S.G.; Suh, S.H.; Lee, J.-M. Apparent Diffusion Coefficient Histogram in Breast Cancer Brain Metastases May Predict Their Biological Subtype and Progression. Sci. Rep. 2018, 8, 9947. [Google Scholar] [CrossRef] [PubMed]
- Michoux, N.F.; Ceranka, J.W.; Vandemeulebroucke, J.; Peeters, F.; Lu, P.; Absil, J.; Triqueneaux, P.; Liu, Y.; Collette, L.; Willekens, I.; et al. Repeatability and Reproducibility of ADC Measurements: A Prospective Multicenter Whole-Body-MRI Study. Eur. Radiol. 2021, 31, 4514–4527. [Google Scholar] [CrossRef]
- On behalf of the EUSOBI International Breast Diffusion-Weighted Imaging Working Group; Baltzer, P.; Mann, R.M.; Iima, M.; Sigmund, E.E.; Clauser, P.; Gilbert, F.J.; Martincich, L.; Partridge, S.C.; Patterson, A.; et al. Diffusion-Weighted Imaging of the Breast—A Consensus and Mission Statement from the EUSOBI International Breast Diffusion-Weighted Imaging Working Group. Eur. Radiol. 2020, 30, 1436–1450. [Google Scholar] [CrossRef]
- Whisenant, J.G.; Romanoff, J.; Rahbar, H.; Kitsch, A.E.; Harvey, S.M.; Moy, L.; DeMartini, W.B.; Dogan, B.E.; Yang, W.T.; Wang, L.C.; et al. Factors Affecting Image Quality and Lesion Evaluability in Breast Diffusion-Weighted MRI: Observations from the ECOG-ACRIN Cancer Research Group Multisite Trial (A6702). J. Breast Imaging 2021, 3, 44–56. [Google Scholar] [CrossRef] [PubMed]
- Sharma, U.; Sah, R.G.; Agarwal, K.; Parshad, R.; Seenu, V.; Mathur, S.R.; Hari, S.; Jagannathan, N.R. Potential of Diffusion-Weighted Imaging in the Characterization of Malignant, Benign, and Healthy Breast Tissues and Molecular Subtypes of Breast Cancer. Front. Oncol. 2016, 6, 126. [Google Scholar] [CrossRef] [PubMed]
- Meng, L.; Ma, P. Apparent Diffusion Coefficient Value Measurements with Diffusion Magnetic Resonance Imaging Correlated with the Expression Levels of Estrogen and Progesterone Receptor in Breast Cancer: A Meta-Analysis. J. Cancer Res. Ther. 2016, 12, 36–42. [Google Scholar] [CrossRef]
- Feng, W.; Gao, Y.; Lu, X.-R.; Xu, Y.-S.; Guo, Z.-Z.; Lei, J.-Q. Correlation between Molecular Prognostic Factors and Magnetic Resonance Imaging Intravoxel Incoherent Motion Histogram Parameters in Breast Cancer. Magn. Reson. Imaging 2022, 85, 262–270. [Google Scholar] [CrossRef] [PubMed]
- Choi, S.Y.; Chang, Y.-W.; Park, H.J.; Kim, H.J.; Hong, S.S.; Seo, D.Y. Correlation of the Apparent Diffusion Coefficiency Values on Diffusion-Weighted Imaging with Prognostic Factors for Breast Cancer. Br. J. Radiol. 2012, 85, e474–e479. [Google Scholar] [CrossRef]
- Tezcan, S.; Ozturk, F.U.; Uslu, N.; Akcay, E.Y. The Role of Combined Diffusion-Weighted Imaging and Dynamic Contrast-Enhanced MRI for Differentiating Malignant From Benign Breast Lesions Presenting Washout Curve. Can. Assoc. Radiol. J. 2021, 72, 460–469. [Google Scholar] [CrossRef]
- Martincich, L.; Deantoni, V.; Bertotto, I.; Redana, S.; Kubatzki, F.; Sarotto, I.; Rossi, V.; Liotti, M.; Ponzone, R.; Aglietta, M.; et al. Correlations between Diffusion-Weighted Imaging and Breast Cancer Biomarkers. Eur. Radiol. 2012, 22, 1519–1528. [Google Scholar] [CrossRef]
- Ren, C.; Zou, Y.; Zhang, X.; Li, K. Diagnostic Value of Diffusion-weighted Imaging-derived Apparent Diffusion Coefficient and Its Association with Histological Prognostic Factors in Breast Cancer. Oncol. Lett. 2019, 18, 3295–3303. [Google Scholar] [CrossRef]
- Bozdağ, M.; Er, A.; Ekmekçi, S. Differentiation of Brain Metastases Originating from Lung and Breast Cancers Using Apparent Diffusion Coefficient Histogram Analysis and the Relation of Histogram Parameters with Ki-67. Neuroradiol. J. 2022, 35, 370–377. [Google Scholar] [CrossRef]
- Zhang, B.; Zhou, F.; Zhou, Q.; Xue, C.; Ke, X.; Zhang, P.; Han, T.; Deng, L.; Jing, M.; Zhou, J. Whole-Tumor Histogram Analysis of Multi-Parametric MRI for Differentiating Brain Metastases Histological Subtypes in Lung Cancers: Relationship with the Ki-67 Proliferation Index. Neurosurg. Rev. 2023, 46, 218. [Google Scholar] [CrossRef]
- Surov, A.; Meyer, H.J.; Wienke, A. Associations between Apparent Diffusion Coefficient (ADC) and KI 67 in Different Tumors: A Meta-Analysis. Part 1: ADCmean. Oncotarget 2017, 8, 75434–75444. [Google Scholar] [CrossRef] [PubMed]
- Surov, A.; Clauser, P.; Chang, Y.-W.; Li, L.; Martincich, L.; Partridge, S.C.; Kim, J.Y.; Meyer, H.J.; Wienke, A. Can Diffusion-Weighted Imaging Predict Tumor Grade and Expression of Ki-67 in Breast Cancer? A Multicenter Analysis. Breast Cancer Res. 2018, 20, 58. [Google Scholar] [CrossRef]
- Zhou, Y.; Lin, G.; Chen, W.; Chen, Y.; Shi, C.; Peng, Z.; Chen, L.; Cai, S.; Pan, Y.; Chen, M.; et al. Multiparametric MRI-Based Interpretable Machine Learning Radiomics Model for Distinguishing Between Luminal and Non-Luminal Tumors in Breast Cancer: A Multicenter Study. Acad. Radiol. 2025, 32, 3801–3812. [Google Scholar] [CrossRef] [PubMed]
- Mosquera, C.; Ferrer, L.; Milone, D.H.; Luna, D.; Ferrante, E. Class Imbalance on Medical Image Classification: Towards Better Evaluation Practices for Discrimination and Calibration Performance. Eur. Radiol. 2024, 34, 7895–7903. [Google Scholar] [CrossRef]
- Buda, M.; Maki, A.; Mazurowski, M.A. A Systematic Study of the Class Imbalance Problem in Convolutional Neural Networks. Neural Netw. 2018, 106, 249–259. [Google Scholar] [CrossRef]
- Liu, S.; Roemer, F.; Ge, Y.; Bedrick, E.J.; Li, Z.-M.; Guermazi, A.; Sharma, L.; Eaton, C.; Hochberg, M.C.; Hunter, D.J.; et al. Comparison of Evaluation Metrics of Deep Learning for Imbalanced Imaging Data in Osteoarthritis Studies. Osteoarthr. Cartil. 2023, 31, 1242–1248. [Google Scholar] [CrossRef]
- Richardson, E.; Trevizani, R.; Greenbaum, J.A.; Carter, H.; Nielsen, M.; Peters, B. The Receiver Operating Characteristic Curve Accurately Assesses Imbalanced Datasets. Patterns 2024, 5, 100994. [Google Scholar] [CrossRef]
- Esposito, C.; Landrum, G.A.; Schneider, N.; Stiefl, N.; Riniker, S. GHOST: Adjusting the Decision Threshold to Handle Imbalanced Data in Machine Learning. J. Chem. Inf. Model. 2021, 61, 2623–2640. [Google Scholar] [CrossRef]
- Van Den Goorbergh, R.; Van Smeden, M.; Timmerman, D.; Van Calster, B. The Harm of Class Imbalance Corrections for Risk Prediction Models: Illustration and Simulation Using Logistic Regression. J. Am. Med. Inform. Assoc. 2022, 29, 1525–1534. [Google Scholar] [CrossRef]
- Rajaraman, S.; Ganesan, P.; Antani, S. Deep Learning Model Calibration for Improving Performance in Class-Imbalanced Medical Image Classification Tasks. PLoS ONE 2022, 17, e0262838. [Google Scholar] [CrossRef] [PubMed]
- Walsh, R.; Tardy, M. A Comparison of Techniques for Class Imbalance in Deep Learning Classification of Breast Cancer. Diagnostics 2022, 13, 67. [Google Scholar] [CrossRef] [PubMed]

| Characteristic | Overall (n = 72) |
|---|---|
| Patient characteristics | |
| Age, years | 54.0 (15.25) [33–77] |
| Lesion imaging characteristics | |
| Maximum diameter, mm | 18.3 (19.05) [3.2–83.7] |
| Lesion volume | 1.102 (6.599) [0.046–114.09] |
| ADC min | 0.593 (0.194) [0.381–1.060] |
| ADC 1st percentile | 0.681 (0.219) [0.400–1.120] |
| ADC 5th percentile | 0.747 (0.267) [0.400–1.210] |
| ADC 10th percentile | 0.800 (0.262) [0.463–1.360] |
| ADC 25th percentile | 0.885 (0.370) [0.500–1.660] |
| ADC 50th percentile | 1.006 (0.468) [0.500–2.550] |
| ADC 75th percentile | 1.113 (0.657) [0.600–2.900] |
| ADC 90th percentile | 1.298 (0.887) [0.600–2.990] |
| ADC 95th percentile | 1.418 (1.007) [0.672–3.100] |
| ADC 99th percentile | 1.633 (1.076) [0.672–3.330] |
| ADC max | 1.637 (1.130) [0.672–4.090] |
| Skewness | 0.426 (0.739) [−0.583–2.010] |
| Kurtosis | −0.522 (1.300) [−1.514–4.870] |
| Entropy | 3.859 (0.539) [2.138–4.420] |
| Biopsy/pathology characteristics | |
| Biopsy type | |
| —Excisional | 36 (50.0%) |
| —Incisional | 36 (50.0%) |
| Histopathologic subtype | |
| —IDC (invasive ductal carcinoma) | 66 (91.7%) |
| —ILC (invasive lobular carcinoma) | 3 (4.2%) |
| —Mixed | 3 (4.2%) |
| Histologic grade | |
| —Grade 1 | 2 (2.8%) |
| —Grade 2 | 20 (27.8%) |
| —Grade 3 | 50 (69.4%) |
| Estrogen receptor (ER) | |
| —Negative | 32 (44.4%) |
| —Positive | 40 (55.6%) |
| Progesterone receptor (PR) | |
| —Negative | 33 (45.8%) |
| —Positive | 39 (54.2%) |
| Human epidermal growth factor receptor 2 (HER2) | |
| —Negative | 40 (55.6%) |
| —Positive | 32 (44.4%) |
| Molecular subtype | |
| —HER2-enriched | 13 (18.1%) |
| —Luminal B—HER2-negative | 23 (31.9%) |
| —Luminal B—HER2-positive | 20 (27.8%) |
| —Triple-negative | 16 (22.2%) |
| Ki-67 | |
| —Negative | 9 (12.5%) |
| —Positive | 63 (87.5%) |
| Lymphovascular invasion (LVI) | |
| —Negative | 11 (15.3%) |
| —Positive | 61 (84.7%) |
| ER Model (ER = P vs. N): Age + log_volume + ADC10x10 | |||
|---|---|---|---|
| Predictor | OR | 95% CI | p-value |
| Age | 0.989 | 0.935–1.046 | 0.697 |
| log_volume | 1.137 | 0.790–1.637 | 0.489 |
| ADC10x10 | 0.441 | 0.289–0.672 | <0.001 |
| PR model (PR = P vs. N): age + log_volume + ADC10x10 | |||
| Age | 0.991 | 0.938–1.046 | 0.734 |
| log_volume | 1.086 | 0.761–1.549 | 0.649 |
| ADC10x10 | 0.478 | 0.321–0.713 | <0.001 |
| Ki-67 model (Ki-67 = P vs. N): age + log_volume + ADC75x10 | |||
| Age | 0.928 | 0.831–1.040 | 0.182 |
| log_volume | 0.590 | 0.246–1.410 | 0.237 |
| ADC75x10 | 3.095 | 1.323–7.240 | 0.009 |
| Luminality model (N vs. L): age + log_volume + ADC10x10 | |||
| Age | 1.007 | 0.952–1.065 | 0.811 |
| log_volume | 0.816 | 0.564–1.181 | 0.281 |
| ADC10x10 | 2.251 | 1.490–3.400 | <0.001 |
| Outcome | Predictor (Model Probability) | AUC (95% CI) | Youden Optimal Cutoff | Sensitivity | Specificity | Accuracy |
|---|---|---|---|---|---|---|
| ER (P vs. N) | p_ER | 0.847 (0.752–0.942) | 0.671 | 0.725 | 0.906 | 0.806 |
| PR (P vs. N) | p_PR | 0.819 (0.720–0.918) | 0.647 | 0.692 | 0.848 | 0.764 |
| Ki-67 (P vs. N) | p_ki67 | 0.905 (0.820–0.989) | 0.814 | 0.857 | 0.889 | 0.861 |
| Luminality (N vs. L) | p_N | 0.832 (0.730–0.935) | 0.382 | 0.828 | 0.814 | 0.819 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Saygılı Öz, D.; Savran, B.; Çiledağ, N.; Ünal, Ö.; Karabulut, B. ADC Histogram Features of Breast Cancer Brain Metastases as Candidate Imaging Biomarkers of Primary Tumor ER, PR, Ki-67, and Luminal Status. Diagnostics 2026, 16, 1154. https://doi.org/10.3390/diagnostics16081154
Saygılı Öz D, Savran B, Çiledağ N, Ünal Ö, Karabulut B. ADC Histogram Features of Breast Cancer Brain Metastases as Candidate Imaging Biomarkers of Primary Tumor ER, PR, Ki-67, and Luminal Status. Diagnostics. 2026; 16(8):1154. https://doi.org/10.3390/diagnostics16081154
Chicago/Turabian StyleSaygılı Öz, Diba, Burcu Savran, Nazan Çiledağ, Özkan Ünal, and Berna Karabulut. 2026. "ADC Histogram Features of Breast Cancer Brain Metastases as Candidate Imaging Biomarkers of Primary Tumor ER, PR, Ki-67, and Luminal Status" Diagnostics 16, no. 8: 1154. https://doi.org/10.3390/diagnostics16081154
APA StyleSaygılı Öz, D., Savran, B., Çiledağ, N., Ünal, Ö., & Karabulut, B. (2026). ADC Histogram Features of Breast Cancer Brain Metastases as Candidate Imaging Biomarkers of Primary Tumor ER, PR, Ki-67, and Luminal Status. Diagnostics, 16(8), 1154. https://doi.org/10.3390/diagnostics16081154







