2. Materials and Methods
2.1. Study Design and Setting
This was a single-center, prospective observational study conducted in the ED of a tertiary care hospital between 1 March 2022 and 1 November 2022. Consecutive adult patients (≥18 years) presenting to the ED who fulfilled the Sepsis-3 diagnostic criteria [
17] at admission were screened for eligibility. The study was approved by the institutional review board, and written informed consent was obtained from all participants or their legal representatives.
A priori sample size calculation was performed for the primary comparison of baseline RRI
0 between transient and non-transient SA-AKI groups. Because no prior emergency department–based study had reported an effect estimate for this specific comparison, the calculation was informed by previously published data from critically ill patients showing a large between-group difference in renal resistive index (0.618 ± 0.081 vs. 0.701 ± 0.073; Cohen’s d ≈ 1.04) [
13]. To avoid overestimating the expected effect, a more conservative large effect size (Cohen’s d = 0.80) was assumed. Using a two-sided α of 0.05, 80% power, and a 1:1 allocation ratio, the minimum required sample size was estimated as 52 patients.
2.2. Study Population
The study included adult patients (≥18 years) who presented to the ED and met Sepsis-3 clinical criteria at admission [
17]. Management during ED care was based on contemporary sepsis treatment principles consistent with the 2021 Surviving Sepsis Campaign guidelines [
1]. To isolate sepsis-related alterations in renal perfusion and minimize confounding, patients with conditions or treatments known to substantially affect renal blood flow were excluded. Exclusion criteria included pregnancy; trauma; referral from another healthcare facility after initiation of treatment; ED length of stay less than 4 h; known advanced chronic kidney disease (CKD) (documented pre-admission eGFR < 30 mL/min/1.73 m
2 or known stage 4–5 CKD); diabetes mellitus; essential hypertension; renal artery stenosis; advanced liver failure or hepatorenal syndrome; the use of renin–angiotensin–aldosterone system inhibitors, angiotensin-converting enzyme inhibitors, angiotensin receptor blockers, or diuretics; the absence of urinary catheterization with unknown urine output during the first 4 h; requirement for renal replacement therapy; postrenal acute kidney injury; and cardiac arrest within the first 4 h of ED admission despite successful resuscitation. A flow diagram illustrating patient screening, exclusions, and final enrollment is presented in
Figure 1.
2.3. Definition of SA-AKI and Renal Recovery Patterns
Acute kidney injury (AKI) was defined according to the Kidney Disease: Improving Global Outcomes (KDIGO) criteria [
18]. SA-AKI was operationally defined as AKI occurring in temporal association with sepsis, in the absence of alternative primary renal or postrenal causes [
19]. Patients with SA-AKI were classified as having transient or non-transient kidney injury based on renal recovery. Transient SA-AKI was defined as complete normalization of serum creatinine and urine output within 72 h of diagnosis, whereas non-transient SA-AKI was defined as persistence of KDIGO criteria beyond 72 h [
20,
21]. Serum creatinine was used as the primary parameter for renal function. eGFR was not included in descriptive or comparative analyses because its calculation is unreliable in acute, non–steady-state conditions. Therefore, renal function variables presented in the study reflect acute renal status at presentation rather than confirmed pre-morbid baseline kidney function.
2.4. Data Collection
Baseline demographic, clinical, laboratory, and hemodynamic variables were recorded at ED admission using previously prepared data collection forms. These variables included age, sex, known comorbidities, medication history, vital signs at presentation, and mean arterial pressure (MAP), which was specifically recorded as a baseline hemodynamic parameter because systemic hemodynamic status may influence ultrasonographic renal perfusion measurements, along with laboratory tests (including complete blood count, kidney function tests, venous blood gas analysis, and coagulation parameters), urine output, ultrasonographic findings, KDIGO classifications, Sequential Organ Failure Assessment (SOFA), quick Sequential Organ Failure Assessment (qSOFA), Simplified Acute Physiology Score III (SAPS III), source of infection, and clinical outcomes at days 7 and 28.
2.5. Ultrasonographic Assessment of Renal Perfusion
Renal ultrasonography was performed to assess early intrarenal perfusion dynamics during the initial phase of sepsis. All examinations were conducted by a single emergency physician with more than four years of experience in emergency ultrasonography and formal certification in basic and advanced ultrasound applications. A single-operator approach was intentionally adopted to ensure internal consistency of image acquisition and measurement technique. The operator was blinded to laboratory results, AKI classification, and subsequent renal outcomes at the time of image acquisition.
All ultrasound examinations were performed using a Mindray M5 ultrasound system (UMT-200 version; Mindray, Mahwah, NJ, USA) with a convex transducer, following standardized acquisition protocols based on the American College of Emergency Physicians renal ultrasound guidelines [
10].
Power Doppler ultrasonography was preferred over conventional color Doppler imaging for its superior sensitivity in detecting low-velocity blood flow, particularly within the renal cortex and arcuate vessels [
15]. Examples are provided in
Supplementary Materials S1–S6.
2.6. SPDUS Technique
The order of ultrasonographic measurements was pre-specified in the study protocol. The SPDUS was obtained first to standardize the global assessment of intrarenal perfusion, prior to the repeated vessel targeting, spectral waveform acquisition, and transducer manipulation required for RRI measurement. Because both assessments were completed within the same brief examination window using a fixed sequence, the likelihood of ordering-related bias was considered minimal. SPDUS assessment was performed bilaterally using the same semiquantitative four-grade scoring system. The right kidney was typically examined first for orientation and image acquisition, followed by evaluation of the contralateral kidney using the same technique. Each kidney was initially localized in the transverse plane by scanning between the mid-axillary and anterior axillary lines. After centering the kidney within the acoustic window, the probe was rotated 90 degrees to obtain a coronal view, in which the renal hilum was optimally visualized with minimal rib shadowing. Power Doppler mode was then activated to assess the distribution of intrarenal blood flow. Because the SPDUS grade was identical in both kidneys, a single patient-level SPDUS was recorded for statistical analysis.
All SPDUS images were acquired at end-expiration to reduce respiratory motion artifacts. Renal perfusion was graded using a previously described four-grade semiquantitative scoring system [
15]: Grade 0 indicated absence of identifiable intrarenal vessels; Grade 1 indicated visualization of a small number of vessels confined to the hilar region; Grade 2 indicated visualization of hilar and interlobar vessels in most of the renal parenchyma; and Grade 3 indicated visualization of renal vessels extending to the arcuate arteries throughout the entire field of view.
2.7. RRI Technique
Following the SPDUS assessment, RRI measurements were obtained from both kidneys using a standardized posterolateral approach. Each kidney was first identified in the transverse plane and subsequently examined in the longitudinal plane. Color Doppler mode was used to identify an interlobar or arcuate artery, after which pulsed-wave Doppler was applied to obtain spectral waveforms.
After visualization of stable waveforms across three consecutive cardiac cycles, peak systolic flow rate (PSFR) and end-diastolic flow rate (EDFR) were measured. The RRI was calculated using the standard formula (PSFR − EDFR)/PSFR. For each kidney, three measurements were obtained from the same arterial level, and the average value was calculated. The mean RRI of both kidneys was used for analysis.
2.8. Timing of Measurements and Dynamic Assessment
Baseline ultrasonographic measurements (SPDUS
0 and RRI
0) were obtained within the first 6–10 min following ED admission and completed within 5 min to avoid delays in routine care. Follow-up measurements (SPDUS
4 and RRI
4) were performed at the 4th hour of ED stay to explore short-term changes in renal perfusion following standard sepsis management in the ED, which included early intravenous fluid resuscitation, administration of appropriate antimicrobial therapy, hemodynamic monitoring, source-directed supportive care, and vasopressor treatment when required according to the patient’s clinical status [
1]. After completion of the ED evaluation and treatment, patients were admitted to the ICU or the inpatient ward according to clinical severity and organ support requirements. Although initial ED management followed standard sepsis care principles, subsequent inpatient management was not protocolized by the study and was determined by the treating teams.
2.9. Statistical Analysis
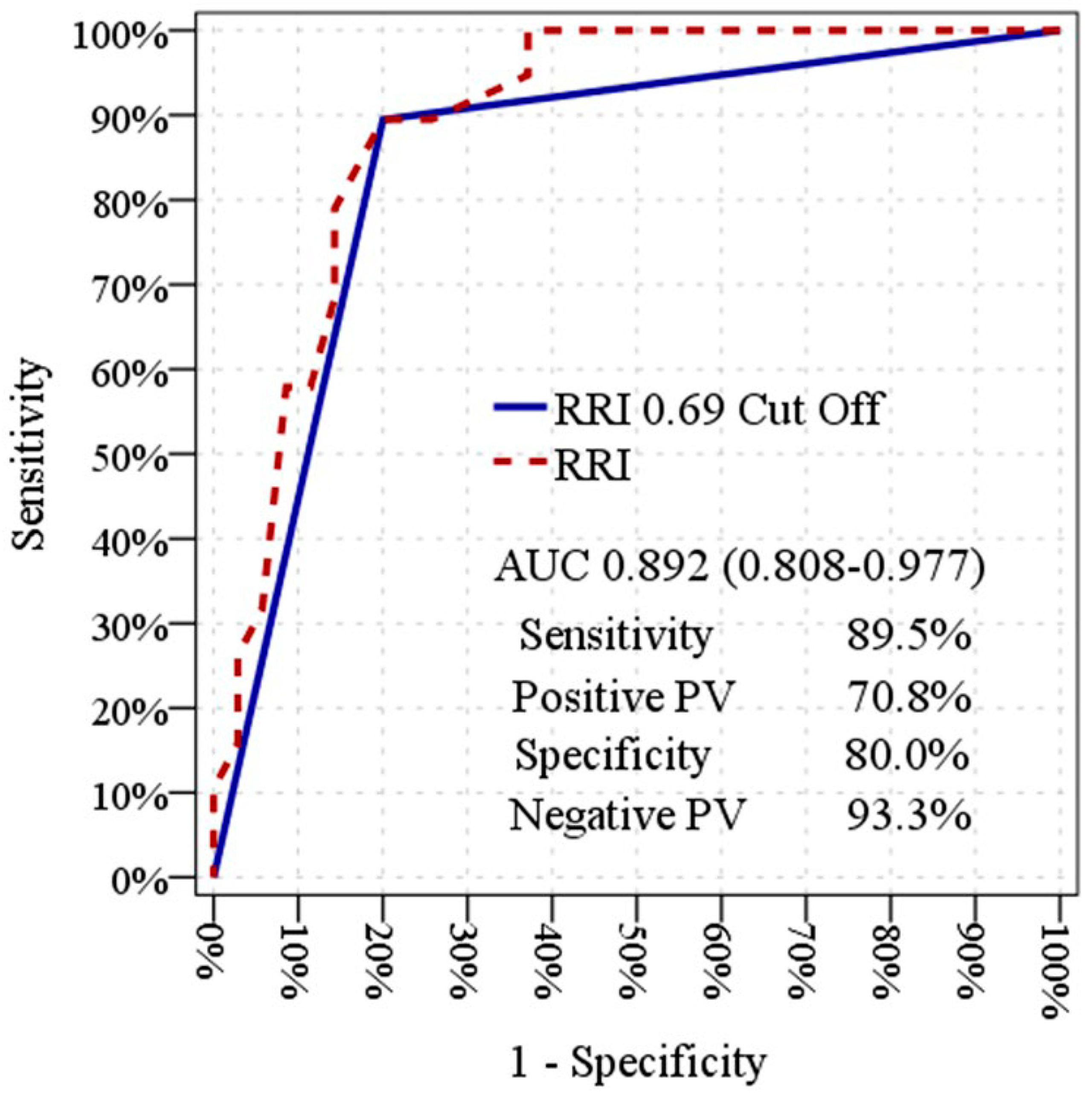
Statistical analyses were performed using SPSS Statistics version 28.0 for Windows (IBM Corp., Armonk, NY, USA). Continuous variables were assessed for normality using both the Kolmogorov–Smirnov and Shapiro–Wilk tests. Descriptive statistics were reported as mean ± standard deviation for normally distributed continuous variables, median (interquartile range) for non-normally distributed continuous variables, and n (%) for categorical variables. Because the SPDUS is a four-grade ordinal score, it was summarized as the median (interquartile range) and analyzed using nonparametric methods appropriate for ordinal data. Comparisons between independent groups were performed using the independent-samples t-test for normally distributed continuous variables and the Mann–Whitney U test for non-normally distributed variables. Categorical variables were compared using the Chi-square test or Fisher’s exact test, as appropriate. ROC curve analysis was used to describe the ability of baseline renal perfusion parameters to distinguish transient from non-transient kidney injury within the study cohort. Because SPDUS0 is a four-grade ordinal score, ROC/AUC analysis for this variable was interpreted within a nonparametric, rank-based framework rather than as an interval-level continuous measure. Accordingly, the AUC for SPDUS0 was considered a rank-based summary of its discriminatory ability. The 95% confidence intervals for the AUCs were calculated using DeLong’s nonparametric method. The area under the curve (AUC) was reported as a measure of separation rather than as a means of determining definitive diagnostic cut-off values. To explore the association between early renal perfusion parameters and SA-AKI, logistic regression analyses were performed. Given the study’s physiological focus and limited sample size, multivariable models were intentionally restricted to a small set of clinically relevant covariates to reduce the risk of overfitting. Accordingly, the study was not powered for formal multivariable predictive modeling, and these analyses were considered exploratory and hypothesis-generating. Given the exploratory nature of the between-group comparisons, no formal multiplicity correction was performed. Accordingly, the results, especially borderline p-values, should be interpreted cautiously in view of the potential inflation of type I error. All statistical tests were two-sided, and p-values < 0.05 were considered statistically significant.
4. Discussion
This prospective ED-based study showed that early renal microcirculatory perfusion patterns differed between patients who subsequently followed transient and non-transient SA-AKI trajectories. Rather than establishing a predictive or prognostic model, the present study was designed to characterize early intrarenal perfusion differences during the ED phase of sepsis, when conventional biochemical markers may still be insufficient to reflect evolving renal dysfunction.
The principal finding of this study is that baseline renal perfusion parameters were different in patients with transient and non-transient SA-AKI trajectories. This observation supports the concept that renal microcirculatory abnormalities are already detectable early in sepsis, during the ED phase, before prolonged ICU exposure or advanced organ support. In this context, the observed Doppler differences should be interpreted as early physiological pattern differences associated with renal recovery trajectories rather than as evidence of a direct causal or prognostic relationship [
2,
3,
4,
5]. Importantly, these differences were not confined to the admission measurements. At the fourth hour of ED stay, the non-transient SA-AKI group continued to exhibit lower SPDUS and higher RRI values, suggesting that the observed differences in perfusion patterns were sustained during short-term follow-up. However, because fourth-hour measurements may also reflect the effects of early therapeutic interventions and evolving hemodynamic changes, these findings should be interpreted as supportive rather than definitive evidence of persistent perfusion abnormalities.
The RRI reflects both renal and systemic hemodynamic influences on intrarenal vascular resistance, whereas SPDUS provides a semiquantitative assessment of cortical and medullary perfusion [
14,
15,
16]. When interpreted together, these measures may provide a more comprehensive physiological description of early renal perfusion status than either parameter alone. Recent reviews have highlighted the growing role of POCUS in evaluating renal hemodynamics and AKI in critically ill patients, supporting the view that Doppler-based renal perfusion assessment may provide clinically relevant bedside physiological information [
11]. In this broader context, renal Doppler ultrasound may be clinically useful not only for early monitoring of renal microcirculatory alterations but also for bedside hemodynamic assessment during the initial phase of sepsis. Moreover, integration of renal perfusion findings with other POCUS-based assessments commonly used in septic shock, including cardiac, lung, and venous congestion evaluations, may provide a more comprehensive physiological picture and improve early risk stratification. In line with this physiological interpretation, baseline MAP was significantly lower in the non-transient SA-AKI group. This suggests that the observed differences in baseline the RRI and SPDUS may reflect not only intrarenal microcirculatory alterations but also broader systemic hemodynamic impairment during the early phase of sepsis. However, in the present study, these parameters should be understood as descriptive markers of renal microcirculatory perfusion patterns within this cohort, rather than as validated diagnostic thresholds or prognostic tools for clinical decision-making [
22,
23].
These findings should also be interpreted in the broader context of early AKI risk stratification. In recent years, biomarker-based approaches, such as urinary [TIMP-2] × [IGFBP7], and functional markers, such as proenkephalin, have been investigated to identify patients at high risk of AKI during the early therapeutic window, particularly in perioperative and critical care settings [
24,
25]. In parallel, biomarker-guided implementation of supportive measures based on the KDIGO AKI care bundle has been explored as a strategy to reduce progression to clinically significant AKI [
26]. In this framework, RRI and SPDUS may occupy a complementary rather than competing role: unlike biochemical markers of kidney stress or dysfunction, they provide a bedside physiological snapshot of renal perfusion and microcirculatory status during the early phase of sepsis [
19]. It is plausible that combining perfusion-based ultrasonographic parameters with biochemical biomarkers could add value to early SA-AKI risk stratification, although this hypothesis requires validation in larger prospective studies.
Distinguishing transient from non-transient SA-AKI remains clinically relevant because persistent renal dysfunction has been associated with worse short- and long-term outcomes, including mortality and progression to chronic kidney disease [
27,
28,
29]. Although previous studies have reported mixed findings regarding the role of RRI in differentiating transient from persistent AKI [
30], the present results indicate that early Doppler-based perfusion patterns differ between these renal recovery trajectories. These findings support the possibility that the degree of early microcirculatory disturbance is associated with subsequent patterns of renal recovery, although no causal or predictive inferences can be drawn from the present data. In exploratory multivariable models, baseline RRI
0 and SPDUS
0 remained associated with non-transient SA-AKI after adjustment for qSOFA and MAP. However, given the limited sample size, these findings should be interpreted as hypothesis-generating rather than as evidence of a definitive independent prognostic effect.
The mortality rate observed in this cohort was high, particularly among patients with non-transient kidney injury. This finding should be interpreted in the context of greater sepsis severity in the non-transient SA-AKI group, as reflected by higher qSOFA and SAPS III scores, together with the advanced age of the cohort and the predominance of ICU admission. SA-AKI has been consistently associated with increased mortality, particularly in elderly patients with limited physiological reserve and multiple comorbidities [
2,
3,
31,
32]. Renal recovery itself is an important determinant of outcome, independent of the severity of initial sepsis, underscoring the clinical relevance of distinguishing between persistent and transient kidney injury. The relatively high mean SAPS III score in our cohort, despite a more moderate mean SOFA score, likely reflects the predominance of elderly individuals in the study population and the broader structure of SAPS III, which incorporates age, comorbidity burden, and baseline clinical characteristics in addition to acute physiological derangement.
This study has several important limitations that should be acknowledged when interpreting the findings. First, this was a single-center study with a relatively small sample size, which inherently limits statistical power and external validity. Although the cohort size was sufficient to explore physiological associations, the study was not designed or powered to establish definitive prognostic thresholds or to support broad clinical decision-making. Accordingly, all discriminatory analyses should be interpreted as cohort-specific and hypothesis-generating rather than confirmatory. Although the observed AUC values suggest substantial separation within this cohort, they should not be interpreted as validated predictive thresholds, and external validation in larger multicenter cohorts is required before any broader clinical application can be considered. Second, the study population was highly selected, with extensive exclusion criteria to minimize confounding factors that could affect renal perfusion and Doppler-derived indices. While this approach allowed a more focused assessment of sepsis-related renal microcirculatory alterations, it limits generalizability to the broader, heterogeneous population of critically ill patients with sepsis, in whom comorbid conditions such as chronic kidney disease, hypertension, diabetes mellitus, and vasoactive medication use are common. Consequently, the findings may not be directly applicable to unselected ICU populations, in whom comorbidities and treatment-related confounders are more prevalent. At the same time, this design reflects the clinical reality that patients with sepsis are initially evaluated and managed in the ED, where early physiological alterations precede ICU admission and advanced organ support. From this perspective, the study specifically targets the early diagnostic window during which renal microcirculatory dysfunction may still be modifiable. In particular, the exclusion of diabetes mellitus, essential hypertension, and RAAS inhibitor use was intended to reduce chronic vascular and renal hemodynamic confounding that could influence Doppler-derived perfusion indices, but this came at the cost of reduced external validity. Because these conditions are highly prevalent among patients with sepsis in routine clinical practice, the present findings should be interpreted as applying primarily to a selected population rather than to unselected real-world sepsis cohorts. Available comorbidity data did not suggest that the non-transient group represented a broadly more multimorbid population; rather, between-group differences appeared to be more closely related to acute illness severity within this selected cohort. Third, ultrasonographic measurements were performed by a single experienced operator, and neither interobserver nor intraobserver variability was formally assessed. Although this strategy improved internal consistency and reduced technical variability, it introduces potential operator-dependent bias and limits reproducibility across centers and operators with different levels of ultrasound expertise. In addition, SPDUS is inherently subjective, and despite standardized acquisition protocols, grading variability cannot be fully excluded. Accordingly, the cohort-specific AUC values reported in this study should not be extrapolated to multi-operator settings or assumed to reflect SPDUS performance beyond this single-operator context. Fourth, renal perfusion parameters are influenced by systemic hemodynamics, vascular compliance, and age-related arterial stiffness. Although efforts were made to obtain measurements early, prior to major therapeutic interventions and before vasopressor exposure, residual confounding by factors such as baseline blood pressure, cardiac output, and evolving volume status cannot be excluded. Moreover, because baseline MAP was lower in the non-transient SA-AKI group, it is not possible to fully disentangle the contribution of systemic hemodynamic impairment from that of intrarenal microcirculatory alterations in the observed RRI and SPDUS differences. In addition, vasopressor exposure specifically at the time of fourth-hour ultrasonographic reassessment, as well as dose-related effects, was not recorded separately. Because both vasopressor therapy and fluid resuscitation may influence Doppler-derived renal indices, their potential influence on RRI4 cannot be completely excluded. Fifth, pre-admission baseline creatinine was unavailable for 17 of 54 patients (31.5%), which may have introduced misclassification in distinguishing transient from non-transient SA-AKI. In these patients, renal recovery had to be interpreted based on in-hospital creatinine trends and urine output rather than on a confirmed pre-morbid renal baseline. As the group assignment was based on the 72 h clinical course and creatinine trajectory rather than a standardized pre-morbid reference, misclassification of borderline cases cannot be completely excluded. In addition, although trajectory classification was based on 72 h renal recovery, detailed descriptors of creatinine dynamics, such as peak serum creatinine, rate of rise, and time-to-peak, were not reported separately. Therefore, heterogeneity in the magnitude and temporal pattern of kidney injury within each trajectory group may not have been fully captured, potentially introducing bias into the between-group comparisons. Because the transient versus non-transient SA-AKI classification was determined over a 72 h period that extended beyond the ED stay, variation in post-ED management may have influenced renal recovery trajectories. Although urine output was clinically followed and considered during classification of renal recovery, it was not systematically recorded in the final study dataset as a standalone, analyzable variable. Therefore, separate quantitative reporting of urine output findings was not possible. In addition, serum creatinine and urine output—used as reference standards—are imperfect surrogates for early kidney injury and may lag behind microcirculatory dysfunction, further complicating temporal interpretation. Finally, the predominance of elderly individuals in the study population introduces additional confounding due to age-associated vascular changes and reduced renal reserve. Given the strong associations among age, sepsis severity, and mortality, it is not possible to fully disentangle the independent contribution of renal perfusion parameters from the overall disease burden and host vulnerability. Multicenter studies that incorporate larger, more diverse populations and standardized hemodynamic adjustments are required to validate these findings and clarify their clinical relevance.