Author Contributions
Conceptualization, C.M. and B.H.; Data curation, M.M. (Michael Moritz), M.M. (Marius Muenk) and M.M. (Meike Müller); Formal analysis, M.M. (Michael Moritz) and J.S.; Funding acquisition, C.M. and B.H.; Investigation, M.M. (Michael Moritz), M.M. (Marius Muenk) and M.M. (Meike Müller); Methodology, B.H.; Project administration, C.M. and B.H.; Resources, B.H.; Software, J.S.; Supervision, C.M. and B.H.; Visualization, J.S.; Writing—original draft preparation, M.M. (Michael Moritz), J.S. and B.H.; Writing—Review and Editing, J.S., B.H. and C.M. All authors have read and agreed to the published version of the manuscript.
Figure 1.
Optical coherence tomography (OCT) retinal layer annotation from a high-resolution OCT image of a control patient.
Figure 1.
Optical coherence tomography (OCT) retinal layer annotation from a high-resolution OCT image of a control patient.
Figure 2.
Alignment of the Bruch’s membrane opening base scan to the Fovea–Bruch’s membrane opening (BMO) centerline using the anatomical positioning system (APS). The 24 green scan lines are automatically generated by the Glaucoma Module Premium Edition (GMPE, Heidelberg Engineering GmbH, Heidelberg, Germany) and detect 48 Bruch membrane opening (BMO) positions along the optic nerve head, determining the BMO-based disc margins (red dots). The blue line shows the axis between the fovea and the centre of the BMO. The green arrow shows which scan line is currently selected (courtesy of Heidelberg Engineering GmbH, Heidelberg, Germany).
Figure 2.
Alignment of the Bruch’s membrane opening base scan to the Fovea–Bruch’s membrane opening (BMO) centerline using the anatomical positioning system (APS). The 24 green scan lines are automatically generated by the Glaucoma Module Premium Edition (GMPE, Heidelberg Engineering GmbH, Heidelberg, Germany) and detect 48 Bruch membrane opening (BMO) positions along the optic nerve head, determining the BMO-based disc margins (red dots). The blue line shows the axis between the fovea and the centre of the BMO. The green arrow shows which scan line is currently selected (courtesy of Heidelberg Engineering GmbH, Heidelberg, Germany).
Figure 3.
Analysis of peripapillary vessel density with anatomical boundary of Bruch’s membrane opening by the Erlangen Angio Tool (version 2.0 coded in Matlab (The MathWorks, Inc., Natick, MA, USA, R2017b)). S1 represents the superior sector, S2 the nasal sector, S3 the inferior sector, and S4 the temporal sector.
Figure 3.
Analysis of peripapillary vessel density with anatomical boundary of Bruch’s membrane opening by the Erlangen Angio Tool (version 2.0 coded in Matlab (The MathWorks, Inc., Natick, MA, USA, R2017b)). S1 represents the superior sector, S2 the nasal sector, S3 the inferior sector, and S4 the temporal sector.
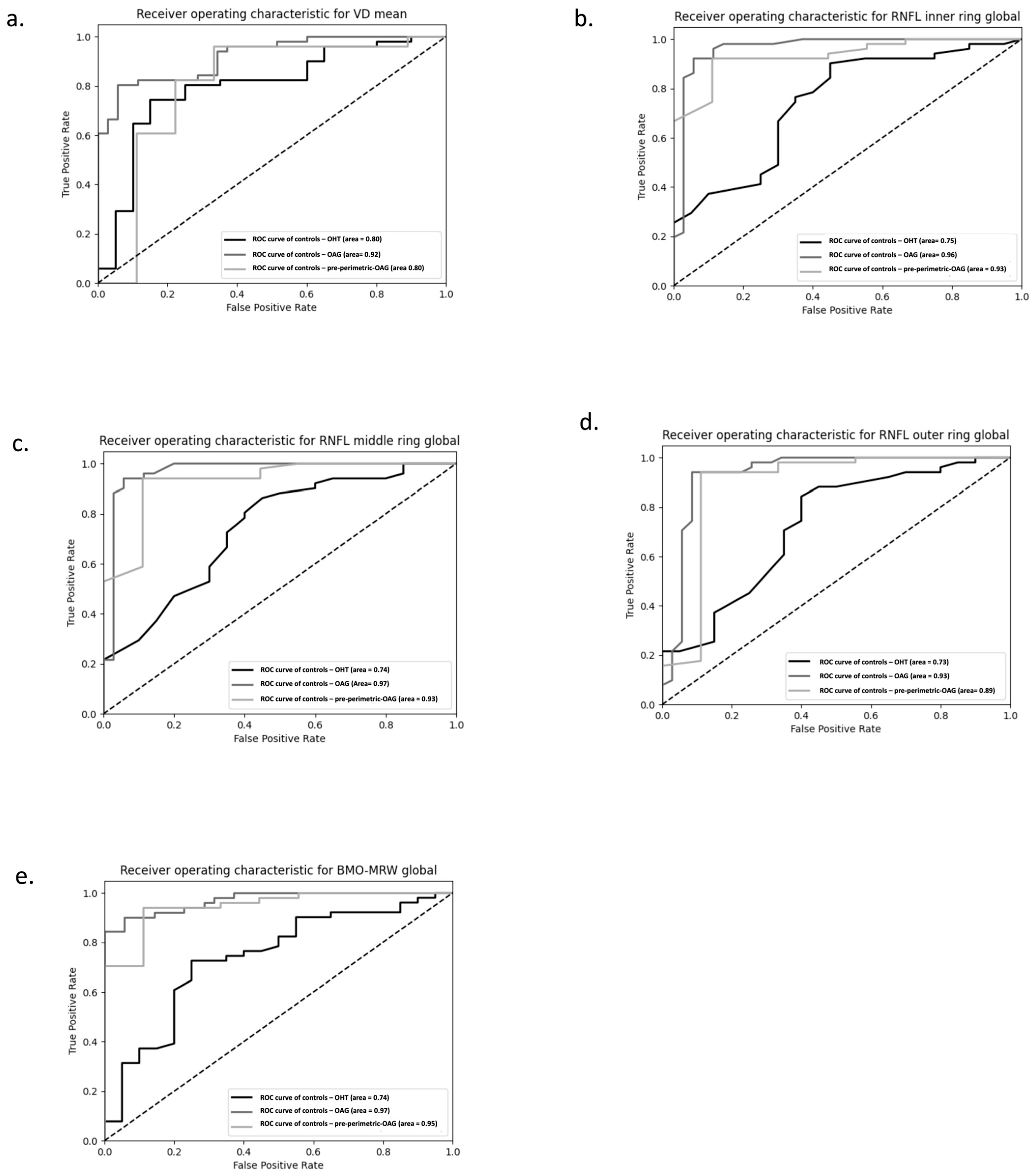
Figure 4.
Receiver operating curves (ROC) of (a) mean vessel density (VD), (b) inner ring of retinal nerve fiber layer (RNFL) thickness, (c) middle ring of RNFL thickness, (d) outer ring of RNFL thickness, and (e) the Bruch’s Membrane Opening–Minimum Rim Width (BMO-MRW) are used to differentiate between the individual patient groups (ocular hypertension (OHT), pre-perimetric open-angle glaucoma (pre-perimetric OAG), primary OAG (POAG), and secondary OAG (SOAG)) and the controls. The dashed line shows the random classifier (AUC = 0.5).
Figure 4.
Receiver operating curves (ROC) of (a) mean vessel density (VD), (b) inner ring of retinal nerve fiber layer (RNFL) thickness, (c) middle ring of RNFL thickness, (d) outer ring of RNFL thickness, and (e) the Bruch’s Membrane Opening–Minimum Rim Width (BMO-MRW) are used to differentiate between the individual patient groups (ocular hypertension (OHT), pre-perimetric open-angle glaucoma (pre-perimetric OAG), primary OAG (POAG), and secondary OAG (SOAG)) and the controls. The dashed line shows the random classifier (AUC = 0.5).
Table 1.
Demographic data of the study cohort: number of eyes, age and gender; ocular hypertension (OHT); pre-perimetric open-angle glaucoma (pre-perimetric OAG); primary OAG (POAG); secondary OAG (SOAG).
Table 1.
Demographic data of the study cohort: number of eyes, age and gender; ocular hypertension (OHT); pre-perimetric open-angle glaucoma (pre-perimetric OAG); primary OAG (POAG); secondary OAG (SOAG).
| | | Number of Eyes | Age (Years) | Gender (Female/Male) |
|---|
| OHT | | 35 | 61 ± 15 | 17/18 |
| Pre-perimetric OAG | 16 | 66 ± 13 | 7/9 |
| OAG | POAG | 37 | 69 ± 8 | 23/14 |
| SOAG | 27 | 68 ± 13 | 12/15 |
| Controls | 65 | 41 ± 20 | 42/23 |
Table 2.
BMO-MRW morphometric data: Mean and standard deviation for controls, ocular hypertension (OHT), pre-perimetric open-angle glaucoma (pre-perimetric OAG), primary open-angle glaucoma (POAG) and secondary open-angle glaucoma (SOAG).
Table 2.
BMO-MRW morphometric data: Mean and standard deviation for controls, ocular hypertension (OHT), pre-perimetric open-angle glaucoma (pre-perimetric OAG), primary open-angle glaucoma (POAG) and secondary open-angle glaucoma (SOAG).
| | | Controls | OHT | Pre-Perimetric OAG | POAG | SOAG |
|---|
| BMO-MRW | Average ± SD in µm | 343.44 ± 78.57 | 278.16 ± 70.42 | 207.13 ± 47.65 | 173.43 ± 52.89 | 191. 13 ± 72.56 |
Table 3.
Significance of differences between BMO-MRW values in pairwise comparisons between subgroups: the first model compared controls with ocular hypertension (OHT), open-angle glaucoma (OAG), and pre-perimetric open-angle glaucoma (pre-perimetric-OAG); the second model examined pairwise comparisons for controls, OHT, pre-perimetric OAG, as well as a subanalysis of OAG, including primary open-angle glaucoma (POAG) and secondary open-angle glaucoma (SOAG).
Table 3.
Significance of differences between BMO-MRW values in pairwise comparisons between subgroups: the first model compared controls with ocular hypertension (OHT), open-angle glaucoma (OAG), and pre-perimetric open-angle glaucoma (pre-perimetric-OAG); the second model examined pairwise comparisons for controls, OHT, pre-perimetric OAG, as well as a subanalysis of OAG, including primary open-angle glaucoma (POAG) and secondary open-angle glaucoma (SOAG).
| | Model 1 | Model 2 |
|---|
| Control vs. OHT | p > 0.05 | p = 1.000 |
| Control vs. OAG | p < 0.0001 | N/A |
| Control vs. Pre-perimetric OAG | p = 0.005 | p = 0.008 |
| Control vs. POAG | N/A | p < 0.0001 |
| Control vs. SOAG | N/A | p < 0.0001 |
| OHT vs. OAG | p = 0.000 | N/A |
| OHT vs. Pre-perimetric OAG | p = 0.049 | p = 0.085 |
| OHT vs. POAG | N/A | p < 0.0001 |
| OHT vs. SOAG | N/A | p = 0.001 |
Table 4.
Retinal nerve fiber layer thickness (RNFL): Mean and standard deviation for controls, ocular hypertension (OHT), pre-perimetric open-angle glaucoma (pre-perimetric OAG), primary open-angle glaucoma (POAG), and secondary open-angle glaucoma (SOAG).
Table 4.
Retinal nerve fiber layer thickness (RNFL): Mean and standard deviation for controls, ocular hypertension (OHT), pre-perimetric open-angle glaucoma (pre-perimetric OAG), primary open-angle glaucoma (POAG), and secondary open-angle glaucoma (SOAG).
| | | | Controls | OHT | Pre-Perimetric OAG | POAG | SOAG |
|---|
| RNFL thickness | Inner Ring | Average ± SD in µm | 99.21 ± 9.74 | 87.78 ± 13.66 | 87.73 ± 13.73 | 65.73 ± 10.35 | 69.29 ± 16.13 |
| Middle Ring | 85.33 ± 7.99 | 75.14 ± 11.76 | 68.07 ± 11.64 | 58.84 ± 9.94 | 60.42 ± 13.08 |
| Outer Ring | 74.53 ± 6.67 | 66.47 ± 9.74 | 60.08 ± 10.28 | 58.29 ± 13.77 | 55.39 ± 11.59 |
Table 5.
Significance of RNFL thickness values for inner, middle, and outer rings in pairwise comparisons of subgroups. Model 1 compared controls, ocular hypertension (OHT), open-angle glaucoma (OAG), and pre-perimetric open-angle glaucoma (pre-perimetric OAG) pairwise. Model 2 additionally examined the subanalysis of OAG, which is divided into primary open-angle glaucoma (POAG) and secondary open-angle glaucoma (SOAG).
Table 5.
Significance of RNFL thickness values for inner, middle, and outer rings in pairwise comparisons of subgroups. Model 1 compared controls, ocular hypertension (OHT), open-angle glaucoma (OAG), and pre-perimetric open-angle glaucoma (pre-perimetric OAG) pairwise. Model 2 additionally examined the subanalysis of OAG, which is divided into primary open-angle glaucoma (POAG) and secondary open-angle glaucoma (SOAG).
| | Model 1 | Model 2 |
|---|
| Inner | Middle | Outer | Inner | Middle | Outer |
|---|
| Control vs. OHT | p = 1.000 | p = 0.705 | p = 1.000 | p = 1.000 | p = 1.000 | p = 1.000 |
| Control vs. OAG | p < 0.0001 | p < 0.0001 | p < 0.0001 | N/A |
| Control vs. Pre-perimetric OAG | p < 0.0001 | p < 0.0001 | p = 0.001 | p = 0.001 | p < 0.0001 | p = 0.002 |
| Control vs. POAG | N/A | p < 0.0001 | p < 0.0001 | p < 0.0001 |
| Control vs. SOAG | N/A | p < 0.0001 | p < 0.0001 | p < 0.0001 |
| OHT vs. OAG | p < 0.0001 | p < 0.0001 | p < 0.0001 | N/A |
| OHT vs. Pre-perimetric OAG | p = 0.013 | p = 0.007 | p = 0.027 | p = 0.021 | p = 0.011 | p = 0.046 |
| OHT vs. POAG | N/A | p < 0.0001 | p < 0.0001 | p < 0.0001 |
| OHT vs. SOAG | N/A | p < 0.0001 | p < 0.0001 | p < 0.0001 |
Table 6.
Mean defect: Mean and standard deviation for ocular hypertension (OHT), pre-perimetric open-angle glaucoma (pre-perimetric OAG), primary open-angle glaucoma (POAG), and secondary open-angle glaucoma (SOAG).
Table 6.
Mean defect: Mean and standard deviation for ocular hypertension (OHT), pre-perimetric open-angle glaucoma (pre-perimetric OAG), primary open-angle glaucoma (POAG), and secondary open-angle glaucoma (SOAG).
| | | OHT | Pre-Perimetric-OAG | POAG | SOAG |
|---|
| Visual field mean deviation | Average ± SD in dB | 2.83 ± 3.34 | 2.95 ± 2.26 | 8.43 ± 5.47 | 6.45 ± 4.55 |
Table 7.
Spherical equivalent: Mean and standard deviation for controls, ocular hypertension (OHT), pre-perimetric open-angle glaucoma (pre-perimetric OAG), primary open-angle glaucoma (POAG), and secondary open-angle glaucoma (SOAG).
Table 7.
Spherical equivalent: Mean and standard deviation for controls, ocular hypertension (OHT), pre-perimetric open-angle glaucoma (pre-perimetric OAG), primary open-angle glaucoma (POAG), and secondary open-angle glaucoma (SOAG).
| | | Controls | OHT | Pre-Perimetric OAG | POAG | SOAG |
|---|
| Spherical equivalent | Average ± SD | −0.5 ± 2 | −0.7 ± 3 | −1.1 ± 3 | −0.8 ± 2 | −1.1 ± 3 |
Table 8.
APSified-BMO-based peripapillary vessel density (VD): Mean and standard deviation for controls, ocular hypertension (OHT), pre-perimetric open-angle glaucoma (pre-perimetric OAG), primary open-angle glaucoma (POAG), and secondary open-angle glaucoma (SOAG).
Table 8.
APSified-BMO-based peripapillary vessel density (VD): Mean and standard deviation for controls, ocular hypertension (OHT), pre-perimetric open-angle glaucoma (pre-perimetric OAG), primary open-angle glaucoma (POAG), and secondary open-angle glaucoma (SOAG).
| | | | Controls | OHT | Pre-Perimetric OAG | POAG | SOAG |
|---|
| APSified-BMO-based peripapillary VD | Sector 1 | Average ± SD | 37.58 ± 7.67 | 33.23 ± 6.94 | 34.61 ± 14.96 | 26.06 ± 7.76 | 25.34 ± 9.71 |
| Sector 2 | 42.32 ± 9.77 | 37.11 ± 7.35 | 38.51 ± 12.11 | 32.26 ± 9.13 | 31.21 ± 10.99 |
| Sector 3 | 39.99 ± 5.59 | 36.78 ± 7.01 | 36.91 ± 12.55 | 27.27 ± 7.29 | 27.09 ± 8.21 |
| Sector 4 | 42.19 ± 8.64 | 36.42 ± 10.57 | 38.31 ± 13.29 | 30.70 ± 10.68 | 28.49 ± 10.52 |
| Mean | 40.27 ± 6.76 | 35.89 ± 6.73 | 37.08 ± 12.47 | 29.07 ± 7.37 | 28.03 ± 8.66 |
Table 9.
Significance of APSified-BMO-based peripapillary vessel density (VD) values in pairwise comparisons of subgroups: model 1 compared controls, ocular hypertension (OHT), open-angle glaucoma (OAG), and pre-perimetric open-angle glaucoma (pre-perimetric OAG) pairwise; model 2 additionally examined the subanalysis of OAG, which is divided into primary open-angle glaucoma (POAG) and secondary open-angle glaucoma (SOAG).
Table 9.
Significance of APSified-BMO-based peripapillary vessel density (VD) values in pairwise comparisons of subgroups: model 1 compared controls, ocular hypertension (OHT), open-angle glaucoma (OAG), and pre-perimetric open-angle glaucoma (pre-perimetric OAG) pairwise; model 2 additionally examined the subanalysis of OAG, which is divided into primary open-angle glaucoma (POAG) and secondary open-angle glaucoma (SOAG).
| | Model 1 | Model 2 |
|---|
| Control vs. OHT | p = 1.000 | p = 1.000 |
| Control vs. OAG | p < 0.0001 | N/A |
| Control vs. Pre-perimetric-OAG | p = 1.000 | p = 1.000 |
| Control vs. POAG | N/A | p = 0.001 |
| Control vs. SOAG | N/A | p = 0.018 |
| OHT vs. OAG | p = 0.016 | N/A |
| OHT vs. Pre-perimetric-OAG | p = 1.000 | p = 1.000 |
| OHT vs. POAG | N/A | p = 0.036 |
| OHT vs. SOAG | N/A | p = 0.324 |
Table 10.
Results of the pairwise comparison of all individual sectoral APSified-BMO-based peripapillary vessel density significance values among all subgroups (controls, ocular hypertension (OHT), pre-perimetric open-angle glaucoma (pre-perimetric OAG), primary OAG (POAG), and secondary OAG (SOAG)) were combined, as they did not differ from each other.
Table 10.
Results of the pairwise comparison of all individual sectoral APSified-BMO-based peripapillary vessel density significance values among all subgroups (controls, ocular hypertension (OHT), pre-perimetric open-angle glaucoma (pre-perimetric OAG), primary OAG (POAG), and secondary OAG (SOAG)) were combined, as they did not differ from each other.
| | Control, OHT, Pre-Perimetric OAG, POAG, SOAG |
|---|
| Sector 1 vs. 2 | p < 0.0001 |
| Sector 1 vs. 3 | p = 0.098 |
| Sector 1 vs. 4 | p < 0.0001 |
| Sector 2 vs. 3 | p < 0.0001 |
| Sector 2 vs. 4 | p = 0.017 |
| Sector 3 vs. 4 | p = 0.241 |