A Complete Response to Immunotherapy in a Patient with Locally Advanced Squamous Cell Lung Cancer Harboring a Novel TMEM178B::BRAF Fusion: A Case Report
Abstract
1. Introduction
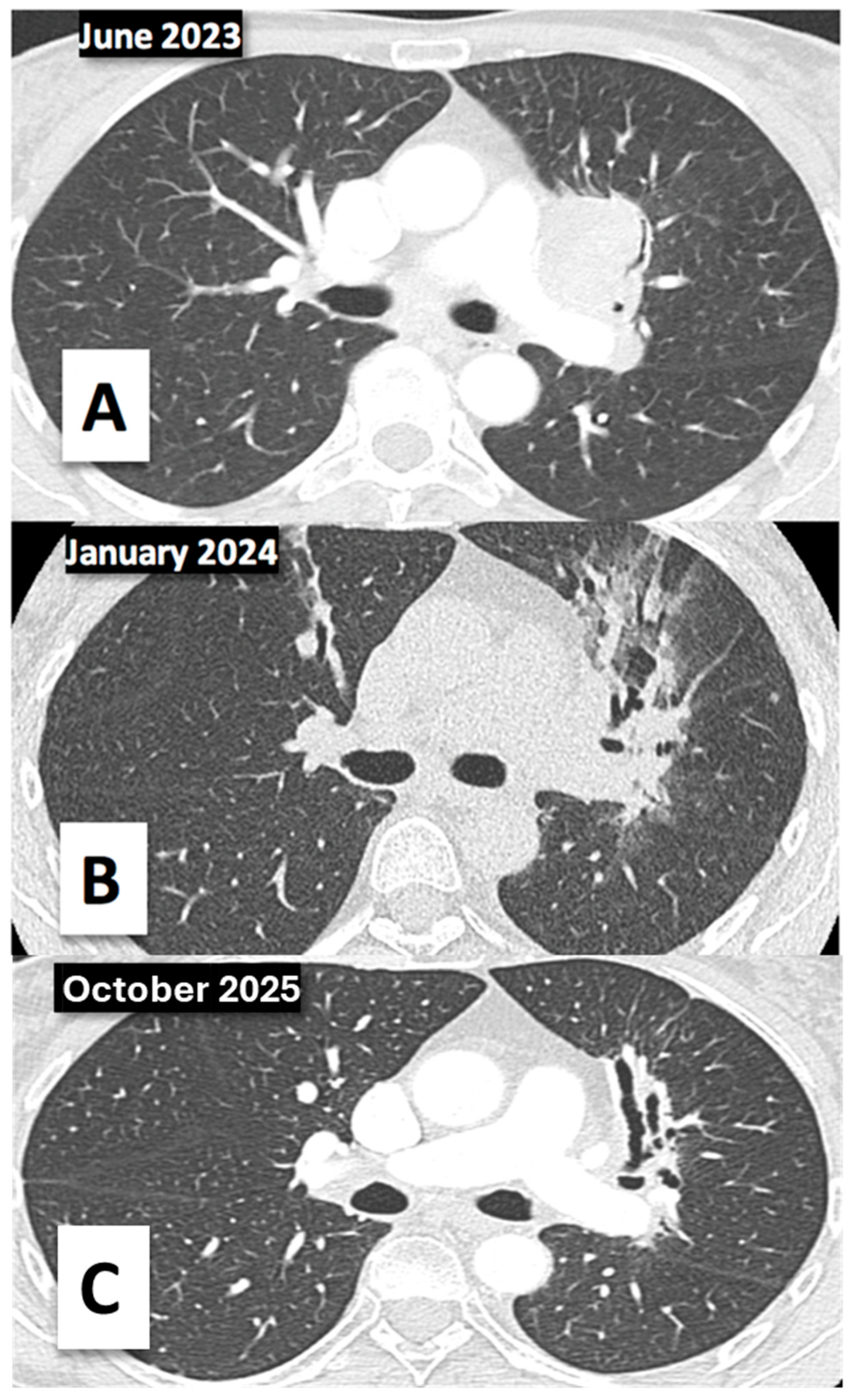
2. Case Report
3. Discussion
| Case | Date of Publication | Diagnosis | Age (yr)/Gender | BRAF Fusion | Targeted Therapy | Response |
|---|---|---|---|---|---|---|
| Castellano-Damaso et al. [21] | 23 May 2024 | Low-grade glioma | 18/male | KIAA1549-BRAF | Trametinib | PR |
| Zhou et al. [22] | 28 March 2024 | Tenosynovial giant cell tumor | 27/male | SLMAP-BRAF | Surgery + Chemotherapy | CR |
| Yasui et al. [23] | 23 February 2024 | Lung adenocarcinoma | 75/female | SLC44A1-BRAF | Trametinib | PR |
| Hirosi et al. [24] | 22 September 2023 | Melanoma | 71/female | BRAF-ZKSCAN5 | Dabrafenib/trametinib | PR |
| Heinrich et al. [25] | 26 June 2023 | Pancreatic cancer adenocarcinoma | ~ | TRIM24-BRAF | Chemotherapy + ICI | CR |
| Toshiro et al. [26] | 6 November 2022 | Melanoma | 73/female | BICD1-BRAF | ICI | PR |
| Kong et al. [27] | 30 January 2023 | Lung adenocarcinoma | 53/female | BTN2A1-BRAF | iEGFR | PR |
| Clarck et al. [28] | 21 May 2022 | Melanoma | 19/female | MYO5A-BRAF | ICI | PR |
| Yang et al. [29] | 19 August 2022 | Lung adenocarcinoma | 60/male | SDN1-BRAF | Trametinib | PR |
| Yun-Tse et al. [30] | 18 March 2022 | Lung adenocarcinoma | 67/male | BRAF-KIAA1549 MET amplification | Dabrafenib/trametinib/cacpmatinib | SD |
| Domen et al. [31] | 17 January 2022 | Myoepithelial carcinoma | 57/female | AGK-BRAF | Cobimetinib | PR |
| Cheng-You et al. [32] | 5 November 2021 | Lung adenocarcinoma | 66/male | LIMD1-BRAF | Trametinib | PR |
| Kervarrec et al. [33] | 1 October 2021 | Primary melanoma of the lung | 55/female | FNBP1-BRAF | ICI | SD |
| Chew et al. [34] | 15 April 2021 | Melanoma | 40/female | SKAP2-BRAF | Trametinib | PR |
| Hasegawa et al. [35] | 18 June 2021 | Rectal cancer | 40/female | EXOC4-BRAF | Chemotherapy + iEGFR | PR |
| Shao-Jie et al. [36] | 24 May 2020 | Malignant soft tissue tumor | 52/female | CDC42SE2-BRAF MET amplification | Crizotinib | SD |
| Isaacson et al. [37] | 25 February 2019 | Urothelial carcinoma | 69/male | NRF1-BRAF | Trametinib | PR |
| You-cai et al. [38] | 4 March 2019 | Lung adenocarcinoma | 60/male | TRIM24-BRAF | Vemurafenib | PR |
| Huat et al. [39] | 12 February 2018 | Cholangiocarcinoma | 58/male | YWHAZ-BRAF | Chemotherapy | PR |
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| NSCLC | Non-Small Cell Lung Cancer |
| SCC | Squamous Cell Carcinoma |
| BRAF | V-Raf murine sarcoma viral oncogene homolog B1 |
| MAPK | Mitogen-Activated Protein Kinase |
| ERK | Extracellular Signal-Regulated Kinase |
| EGFR | Epidermal Growth Factor Receptor |
| ROS1 | ROS Proto-Oncogene 1 |
| NGS | Next-Generation Sequencing |
| CRT | Chemoradiotherapy |
| PD-1 | Programmed Cell Death Protein 1 |
| PD-L1 | Programmed Death-Ligand 1 |
| CT | Computed Tomography |
| PET-CT | Positron Emission Tomography–Computed Tomography |
| OPA | Oncomine Precision Assay |
| CTCAE | Common Terminology Criteria for Adverse Events |
| ECOG | Eastern Cooperative Oncology Group |
| TCGA | The Cancer Genome Atlas |
| MEK | Mitogen-Activated Protein Kinase Kinase |
| Gy | Gray |
| AUC | Area Under the Curve |
References
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J. Clin. 2021, 71, 209–249. [Google Scholar] [PubMed]
- Sun, S.; Schiller, J.H.; Gazdar, A.F. Lung cancer in never smokers—A different disease. Nat. Rev. Cancer 2007, 7, 778–790. [Google Scholar]
- Davies, H.; Bignell, G.R.; Cox, C.; Stephens, P.; Edkins, S.; Clegg, S.; Teague, J.; Woffendin, H.; Garnett, M.J.; Bottomley, W.; et al. Mutations of the BRAF gene in human cancer. Nature 2002, 417, 949–954. [Google Scholar] [CrossRef] [PubMed]
- Yan, N.; Guo, S.; Zhang, H.; Zhang, Z.; Shen, S.; Li, X. BRAF-Mutated Non-Small Cell Lung Cancer: Current Treatment Status and Future Perspective. Front. Oncol. 2022, 12, 863043. [Google Scholar] [CrossRef]
- Brustugun, O.T.; Khattak, A.M.; Trømborg, A.K.; Beigi, M.; Beiske, K.; Lund-Iversen, M.; Helland, Å. BRAF-mutations in non-small cell lung cancer. Lung Cancer 2014, 84, 36–38. [Google Scholar] [CrossRef]
- Kinno, T.; Tsuta, K.; Shiraishi, K.; Mizukami, T.; Suzuki, M.; Yoshida, A.; Suzuki, K.; Asamura, H.; Furuta, K.; Kohno, T.; et al. Clinicopathological features of nonsmall cell lung carcinomas with BRAF mutations. Ann. Oncol. 2014, 25, 138–142. [Google Scholar]
- Kris, M.G.; Johnson, B.E.; Kwiatkowski, D.J.; Iafrate, A.J.; Wistuba, I.I.; Aronson, S.L.; Engelman, J.A.; Shyr, Y.; Khuri, F.R.; Rudin, C.M.; et al. Identification of driver mutations in tumor specimens from 1000 patients with lung adenocarcinoma: The NCI’s Lung Cancer Mutation Consortium (LCMC). J. Clin. Oncol. 2011, 29, CRA7506. [Google Scholar]
- Adib, E.; Nassar, A.H.; Abou Alaiwi, S.; Groha, S.; Akl, E.W.; Sholl, L.M.; Michael, K.S.; Awad, M.M.; Jänne, P.A.; Gusev, A. Variation in targetable genomic alterations in non-small cell lung cancer by genetic ancestry, sex, smoking history, and histology. Genome Med. 2022, 14, 39. [Google Scholar] [CrossRef]
- Li, Y.; Gu, J.; Xu, F.; Zhu, Q.; Ge, D.; Lu, C. Transcriptomic and functional network features of lung squamous cell carcinoma through integrative analysis of GEO and TCGA data. Sci. Rep. 2018, 8, 15834. [Google Scholar] [CrossRef]
- Riely, G.J.; Smit, E.F.; Ahn, M.J.; Felip, E.; Ramalingam, S.S.; Tsao, A.; Melissa Johnson, M.; Gelsomino, F.; Esper, R.; Nadal, E.; et al. Phase II, Open-Label Study of Encorafenib Plus Binimetinib in Patients with BRAFV600—Mutant Metastatic Non–Small-Cell Lung Cancer. J. Clin. Oncol. 2023, 41, 3700–3711. [Google Scholar]
- Ciampi, R.; Knauf, J.A.; Kerler, R.; Gandhi, M.; Zhu, Z.; Nikiforova, M.N.; Rabes, H.M.; Fagin, J.A.; Nikiforov, Y.E. Oncogenic AKAP9-BRAF fusion is a novel mechanism of MAPK pathway activation in thyroid cancer. J. Clin. Investig. 2005, 115, 94–101. [Google Scholar] [CrossRef]
- Ross, J.S.; Wang, K.; Chmielecki, J.; Gay, L.; Johnson, A.; Chudnovsky, J.; Yelensky, R.; Lipson, D.; Ali, S.M.; Elvin, J.A.; et al. The distribution of BRAF gene fusions in solid tumors and response to targeted therapy. Int. J. Cancer 2016, 138, 881–890. [Google Scholar] [CrossRef]
- Wiesweg, M.; Preuß, C.; Roeper, J.; Metzenmacher, M.; Eberhardt, W.; Stropiep, U.; Wedeken, K.; Reis, H.; Herold, T.; Darwiche, K.; et al. BRAF mutations and BRAF mutation functional class have no negative impact on the clinical outcome of advanced NSCLC and associate with susceptibility to immunotherapy. Eur. J. Cancer 2021, 149, 211–221. [Google Scholar] [CrossRef] [PubMed]
- Johnson, D.B.; Nebhan, C.A.; Noel, M.S. MEK inhibitors in non-V600 BRAF mutations and fusions. Oncotarget 2020, 11, 3900–3903. [Google Scholar] [CrossRef] [PubMed]
- Dagogo-Jack, I.; Piotrowska, Z.; Cobb, R.; Banwait, M.; Lennerz, J.K.; Hata, A.N.; Digumarthy, S.R.; Sequist, L.V. Response to the Combination of Osimertinib and Trametinib in a Patient with EGFR-Mutant NSCLC Harboring an Acquired BRAF Fusion. J. Thorac. Oncol. 2019, 14, e226–e228. [Google Scholar] [CrossRef]
- Zhang, C.; Zhang, C.; Lin, J.; Li, Z.; Wang, H. Patients with BRAF-Mutant NSCLC May Not Benefit From Immune Checkpoint Inhibitors: A Population-Based Study. JTO Clin. Res. Rep. 2020, 1, 100006. [Google Scholar] [PubMed]
- Guisier, F.; Dubos-Arvis, C.; Viñas, F.; Doubre, H.; Ricordel, C.; Ropert, S.; Janicot, H.; Bernardi, M.; Fournel, P.; Lamy, R.; et al. Efficacy and Safety of Anti–PD-1 Immunotherapy in Patients with Advanced NSCLC with BRAF, HER2, or MET Mutations or RET Translocation: GFPC 01-2018. J. Thorac. Oncol. 2020, 15, 628–636. [Google Scholar] [CrossRef]
- Wang, L.; Chen, A.; She, X.; Xiang, J.; Tang, J. VPS41-BRAF, a novel BRAF fusion gene identified in a lung adenocarcinoma patient by next-generation sequencing. Lung Cancer 2020, 146, 380–381. [Google Scholar]
- Chen, J.; Jiang, C.C.; Jin, L.; Zhang, X.D. Regulation of PD-L1: A novel role of pro-survival signalling in cancer. Ann. Oncol. 2016, 27, 409–416. [Google Scholar]
- Atefi, M.; Avramis, E.; Lassen, A.; Wong, D.J.L.; Robert, L.; Foulad, D.; Cerniglia, M.; Titz, B.; Chodon, T.; Graeber, T.G.; et al. Effects of MAPK and PI3K Pathways on PD-L1 Expression in Melanoma. Clin. Cancer Res. 2014, 20, 3446–3457. [Google Scholar] [CrossRef]
- Castellano-Damaso, S.; Vazquez-Gomez, F.; Moreno-Carrasco, J.L.; Arce, B.; Borrego, P.; Lassaletta, A. Continuous response despite reduced dose of trametinib as single agent in an adolescent with a relapsed disseminated pediatric low-grade glioma KIAA1549-BRAF fusion positive: A case report and review of the literature. Front. Oncol. 2024, 14, 1381354. [Google Scholar] [CrossRef]
- Zhou, P.; Liu, W.; Zheng, J.; Zhang, H.; Luo, J. Case report: Primary sarcoma of the mandible with a novel SLMAP-BRAF fusion. Front. Oncol. 2024, 14, 1369046. [Google Scholar] [CrossRef]
- Yasui, S.; Honda, T.; Onishi, I.; Ikeda, S.; Miyazaki, Y. Effective Treatment of Lung Adenocarcinoma with a Novel SLC44A1-BRAF Fusion Using Pembrolizumab Followed by Trametinib: A Case Report. Cureus 2024, 16, e54739. [Google Scholar] [PubMed]
- Kato, H.; Kano, S.; Yasui, Y.; Nojiri, Y.; Yoshimitsu, M.; Nakamura, M.; Morita, A. Effectiveness Treatment of a BRAF-ZKSCAN5 Fusion Gene Melanoma Case with Dabrafenib/Trametinib. Case Rep. Oncol. 2023, 16, 1007–1012. [Google Scholar] [CrossRef]
- Heinrich, K.; Fischer, L.E.; De Toni, E.N.; Markwardt, D.; Roessler, D.; Beyer, G.; Günther, M.; Ormanns, S.; Klauschen, F.; Kunz, W.G.; et al. Case of a Patient with Pancreatic Cancer with Sporadic Microsatellite Instability Associated with a BRAF Fusion Achieving Excellent Response to Immunotherapy. JCO Precis. Oncol. 2023, 7, e2200650. [Google Scholar] [CrossRef]
- Takai, T.; Sudo, T.; Kajimoto, K.; Goto, K. Reconsideration of the pathogenic significance of kinase gene fusion in melanocytic tumors based on a case of melanoma with non-in-frame BICD1::BRAF fusion. J. Cutan. Pathol. 2023, 50, 185–187. [Google Scholar] [CrossRef]
- Kong, W.M.; Guo, Y.J.; Ma, J.; Shi, C. BTN2A1-BRAF fusion may be a novel mechanism of resistance to osimertinib in lung adenocarcinoma: A case report. Transl. Cancer Res. 2023, 12, 186–193. [Google Scholar] [CrossRef]
- Clark, H.E.; Huang, Y.Y.M.; Vance, G.H.; Alomari, A.K. Fatal melanoma with a novel MYO5A-BRAF fusion and small associated conventional nevus: A case report and review of literature. J. Cutan. Pathol. 2022, 49, 808–812. [Google Scholar] [CrossRef] [PubMed]
- Yu, Y.; Yu, M.; Li, Y.; Zhou, X.; Tian, T.; Du, Y.; Tu, Z.; Huang, M. Rapid response to monotherapy with MEK inhibitor trametinib for a lung adenocarcinoma patient harboring primary SDN1-BRAF fusion: A case report and literature review. Front. Oncol. 2022, 12, 945620. [Google Scholar] [CrossRef]
- Chou, Y.T.; Lin, C.C.; Lee, C.T.; Pavlick, D.C.; Su, P.L. Durable Response of Dabrafenib, Trametinib, and Capmatinib in an NSCLC Patient with Co-Existing BRAF-KIAA1549 Fusion and MET Amplification: A Case Report. Front. Oncol. 2022, 12, 838798. [Google Scholar] [CrossRef] [PubMed]
- Domen, A.; Van Paesschen, C.; Zwaenepoel, K.; Lambin, S.; Pauwels, P.; Rasschaert, M.; Segelov, E.; Peeters, M.; Prenen, H. Excellent Response to MEK Inhibition in an AGK-BRAF Gene Fusion Driven Carcinoma: Case Report and Literature Review. Anticancer Res. 2022, 42, 373–379. [Google Scholar]
- Wang, C.Y.; Hsia, J.Y.; Li, C.H.; Ho, C.C.; Chao, W.R.; Wu, M.F. Lung Adenocarcinoma with Primary LIMD1-BRAF Fusion Treated with MEK Inhibitor: A Case Report. Clin. Lung Cancer 2021, 22, e878–e880. [Google Scholar] [CrossRef]
- Kervarrec, T.; Jean-Jacques, B.; Pissaloux, D.; Tirode, F.; de la Fouchardière, A. FNBP1-BRAF fusion in a primary melanoma of the lung. Pathology 2021, 53, 785–788. [Google Scholar] [CrossRef]
- Chew, S.M.; Lucas, M.; Brady, M.; Kelly, C.M. SKAP2-BRAF fusion and response to an MEK inhibitor in a patient with metastatic melanoma resistant to immunotherapy. BMJ Case Rep. 2021, 14, e238494. [Google Scholar] [CrossRef]
- Hasegawa, H.; Miyo, M.; Mori, K.; Mano, M.; Ishida, H.; Mita, E. A Rare BRAF Fusion in Advanced Rectal Cancer Treated with Anti-Epidermal Growth Factor Receptor Therapy. Case Rep. Oncol. 2021, 14, 938–943. [Google Scholar] [CrossRef] [PubMed]
- Sheng, S.; Li, J.; Zou, Y.; Peng, X.; Wang, Q.; Fang, H.; Li, X.; Ding, Y.; Fan, Q.; Zhang, Z.; et al. A low-grade malignant soft tissue tumor with S100 and CD34 co-expression showing novel CDC42SE2-BRAF fusion with distinct features. Genes Chromosomes Cancer 2020, 59, 595–600. [Google Scholar]
- Isaacson, A.L.; Guseva, N.V.; Bossler, A.D.; Ma, D. Urothelial carcinoma with an NRF1-BRAF rearrangement and response to targeted therapy. Cold Spring Harb. Mol. Case Stud. 2019, 5, a003848. [Google Scholar]
- Zhu, Y.C.; Wang, W.X.; Xu, C.W.; Zhuang, W.; Du, K.Q.; Chen, G.; Lv, T.F.; Song, Y. A Patient with Lung Adenocarcinoma with BRAF Gene Fusion and Response to Vemurafenib. Clin. Lung Cancer 2019, 20, e224–e228. [Google Scholar] [CrossRef] [PubMed]
- Lim, H.C.; Montesion, M.; Botton, T.; Collisson, E.A.; Umetsu, S.E.; Behr, S.C.; Gordan, J.D.; Stephens, P.J.; Kelley, R.K. Hybrid Capture-Based Tumor Sequencing and Copy Number Analysis to Confirm Origin of Metachronous Metastases in BRCA1-Mutant Cholangiocarcinoma Harboring a Novel YWHAZ-BRAF Fusion. Oncologist 2018, 23, 998. [Google Scholar] [CrossRef] [PubMed]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Redondo-González, J.C.; San Miguel, I.; Rodríguez-González, M.; Montero, J.C.; Sayagués, J.M.; Abad Hernández, M.; Fonseca Sánchez, E.; Del Barco-Morillo, E.; Olivares-Hernández, A. A Complete Response to Immunotherapy in a Patient with Locally Advanced Squamous Cell Lung Cancer Harboring a Novel TMEM178B::BRAF Fusion: A Case Report. Diagnostics 2026, 16, 909. https://doi.org/10.3390/diagnostics16060909
Redondo-González JC, San Miguel I, Rodríguez-González M, Montero JC, Sayagués JM, Abad Hernández M, Fonseca Sánchez E, Del Barco-Morillo E, Olivares-Hernández A. A Complete Response to Immunotherapy in a Patient with Locally Advanced Squamous Cell Lung Cancer Harboring a Novel TMEM178B::BRAF Fusion: A Case Report. Diagnostics. 2026; 16(6):909. https://doi.org/10.3390/diagnostics16060909
Chicago/Turabian StyleRedondo-González, Juan Carlos, Iñigo San Miguel, Marta Rodríguez-González, Juan Carlos Montero, José María Sayagués, Mar Abad Hernández, Emilio Fonseca Sánchez, Edel Del Barco-Morillo, and Alejandro Olivares-Hernández. 2026. "A Complete Response to Immunotherapy in a Patient with Locally Advanced Squamous Cell Lung Cancer Harboring a Novel TMEM178B::BRAF Fusion: A Case Report" Diagnostics 16, no. 6: 909. https://doi.org/10.3390/diagnostics16060909
APA StyleRedondo-González, J. C., San Miguel, I., Rodríguez-González, M., Montero, J. C., Sayagués, J. M., Abad Hernández, M., Fonseca Sánchez, E., Del Barco-Morillo, E., & Olivares-Hernández, A. (2026). A Complete Response to Immunotherapy in a Patient with Locally Advanced Squamous Cell Lung Cancer Harboring a Novel TMEM178B::BRAF Fusion: A Case Report. Diagnostics, 16(6), 909. https://doi.org/10.3390/diagnostics16060909








