1. Introduction
Sutureless aortic bioprosthetic valves have emerged as an important innovation in cardiac surgery, aiming to simplify implantation and reduce operative times while maintaining the hemodynamic advantages of conventional bioprostheses [
1]. Over the past decade, these devices have become an established option for surgical aortic valve replacement (SAVR), particularly in elderly or high-risk patients with severe aortic stenosis. The Perceval valve was the first sutureless prosthesis to gain widespread clinical use and remains the most extensively studied device in this category [
1].
The concept of a sutureless valve seeks to bridge the gap between conventional SAVR and transcatheter aortic valve implantation (TAVI). By eliminating or minimizing the need for annular sutures, sutureless valves allow for shorter cardiopulmonary bypass (CPB) and aortic cross-clamp times, potentially reducing perioperative morbidity and facilitating minimally invasive surgical approaches [
2]. This technical evolution is especially advantageous in elderly patients, those with multiple comorbidities, or in redo surgeries where annular calcification and tissue fragility make traditional suturing challenging [
1,
2,
3].
The Perceval valve consists of a self-expanding stent frame onto which bovine pericardial leaflets are mounted [
2]. Its design allows radial anchoring within the native aortic annulus after decalcification, and its self-expanding properties ensure adequate sealing and positioning without the need for sutures. The device is available in multiple sizes to accommodate a wide range of annular diameters and is deployed under direct visualization using a simple release mechanism. Clinical studies and registry data have demonstrated favourable outcomes with the Perceval valve, including low transvalvular gradients, large effective orifice areas, and acceptable rates of structural valve deterioration over mid-term follow-up [
1,
2,
3].
Despite these advantages, the increasing adoption of sutureless valves has raised new clinical and pathophysiological questions. Among them, prosthetic valve endocarditis (PVE) represents one of the most serious and potentially devastating complications [
1]. Its diagnosis and management may pose unique challenges due to the prosthesis’ metallic frame, lack of a sewing ring, and its radiological characteristics on imaging modalities such as echocardiography. Endocarditis on a Perceval valve can be particularly challenging to diagnose due to acoustic shadowing and artifacts on echocardiography, which may obscure vegetations or abscesses.
The mechanisms that predispose to infection in sutureless valves may differ in part from those of traditional prostheses. Moreover, because these valves are often implanted in patients with advanced age and multiple comorbidities, the host-related risk of infection remains significant [
4].
Given these complexities, a comprehensive understanding of the epidemiology, microbiology, imaging features, and surgical management of endocarditis on Perceval valves is essential. This review aims to summarize current knowledge regarding the design and technical aspects of sutureless valves, the epidemiology and microbiology of PVE in this context, the diagnostic approach with advanced imaging, and the therapeutic strategies available, and to identify areas for future research.
2. Design, Indications and Surgical Characteristics of the Perceval Sutureless Valve
2.1. Design
The Perceval sutureless valve is composed of two main components.
First, the tissue component, made of bovine pericardium, undergoes phospholipid reduction using ethanol and 1,2-octanediol, followed by taurine treatment to neutralize free aldehyde residues, and is finally stabilized in a buffered glutaraldehyde solution [
5].
Second, the metallic component is a self-expanding nitinol stent coated with Carbofilm to enhance biocompatibility. This structure both supports the prosthesis (the tissue component) and secures it in position. The stent anchors the valve through two cylindrical segments: a proximal segment positioned at the patient’s aortic annulus and a distal segment fitting into the sinotubular junction. These two rings are connected by three straight posts with mid-portion tapering and by six sinusoidal posts designed to conform to the sinuses of Valsalva. The stent structure also renders the prosthesis radiopaque [
5].
The tissue component is sutured to the metallic scaffold at the straight posts and covers the lower ring. The nadir of each leaflet corresponds to the midpoint of the lower ring, whereas the free margin aligns with the upper portion of the tapered posts [
5].
2.2. Indications
As with all bioprosthetic valves, its primary indication—according to international convention—is in patients over 60 years of age [
6], as well as those with contraindications to anticoagulation or with reduced life expectancy due to comorbidities.
The prosthesis is designed for AVR in cases of aortic valve disease—stenosis or mixed lesions—where annular rigidity facilitates prosthesis anchoring, like TAVI systems. However, indications have progressively expanded, and Perceval implantation has been reported in pure aortic regurgitation [
7] and in cases of failed homografts or degenerated bioprostheses [
8].
Certain features make it particularly advantageous in specific situations:
Minimally invasive surgery [
9,
10,
11]: Because the prosthesis can be folded, implantation through smaller incisions is easier, and visualization of correct positioning is improved both in conventional and minimally invasive approaches, whether via ministernotomy or right lateral minithoracotomy.
Patients with small annuli [
12]: Since the inner diameter of the prosthesis is only 1 mm smaller than that of the stent frame, the effective orifice area is larger compared with sutured bioprostheses, reducing patient–prosthesis mismatch and often avoiding more complex aortic root-enlargement procedures.
Patients requiring reduced cross-clamp and cardiopulmonary bypass times: Multiple studies [
2] show that Perceval™ implantation results in shorter ischemic and bypass times compared with sutured prostheses, making it especially valuable in high-risk patients or in those undergoing concomitant procedures, where reduced times help decrease morbidity and mortality [
13,
14].
Certain contraindications listed in the technical specifications should also be considered, although many have become relative over time. A classic contraindication is a bicuspid aortic valve; however, several published series have reported successful Perceval implantation in this setting [
15,
16]. We consider it truly contraindicated only in bicuspid valves with two sinuses of Valsalva. Another contraindication is the performance of concomitant procedures such as multiple valve repair/replacement or coronary revascularization. In multiple valve procedures, Perceval should be implanted last to avoid valve displacement; in the case of proximal coronary anastomoses, these should be performed during cardiac arrest before aortic unclamping [
17]. A further contraindication is a dilated ascending aorta when the annulus-to-sinotubular junction ratio exceeds 1.3, as this compromises stent support at the sinotubular level [
5]; however, this can be addressed through concomitant ascending aortic replacement. Finally, metal allergy to the materials composing the stent represents an absolute contraindication to Perceval implantation [
5].
2.3. Surgical Technical Characteristics
Perceval implantation differs from sutured prosthesis placement in several specific aspects [
18,
19]:
Aortotomy: A transverse aortotomy—rather than a classical italic-S incision—should be performed 3 cm above the valvular plane to avoid interference with the stent during closure. The periaortic fat band serves as the reference landmark.
Annular decalcification: Decalcification must ensure a homogeneous surface for prosthesis seating, removing irregularities at the annular, subannular, and supra-annular levels, but without excessive aggressiveness to avoid compromising annular integrity.
Sizing: The sizing technique has evolved to reduce the initially high pacemaker implantation rates. Currently, prosthesis size should correspond to the largest size that allows a snug passage of the white-barrel sizer through the native annulus. Undersizing may lead to paravalvular leak or migration, whereas oversizing increases the risk of conduction disturbances requiring pacemaker implantation and produces higher postoperative gradients.
Implantation: Three guiding sutures are placed at the nadirs of the sinuses at 120º intervals. The sutures should enter 2 mm below and exit 2 mm above the annular plane, ensuring proper seating of the prosthesis. After threading the guiding sutures through the prosthesis eyelets, deployment must be performed perpendicular to the annulus while maintaining even tension across the three sutures. Although manual steady traction is standard, the use of snuggers during release (Snugger method) has also been described [
20].
Post-dilatation: Once correct positioning is verified, the prosthesis is dilated at 4 atmospheres for 30 s while irrigating with warm saline.
Aortotomy closure: Care must be taken to avoid inadvertently entangling the stent frame in the closure sutures.
Postoperative handling: Gentle manipulation of the heart is essential to prevent prosthesis dislocation or migration. Intraoperative echocardiographic assessment is mandatory to confirm proper function.
3. Epidemiology
The incidence of PVE in patients who have undergone AVR with the Perceval valve has been reported in meta-analyses, clinical trials and observational cohorts. Reported incidence rates range between 0.4% and 0.6% per patient-year. Reported cumulative incidence across studies ranges from 0.8% to 6.6% (
Table 1) [
21,
22,
23,
24,
25]. This variability reflects substantial differences in study design—particularly the scarcity of prospective trials—heterogeneous follow-up durations—mean follow-up from 0.9 to 7.03 years [
21,
26]—differences in baseline characteristics of included populations and, possibly, temporal trends reflecting increasing operator experience and shifts in PVE epidemiology.
A 2024 meta-analysis including retrospective and prospective studies evaluating up to 5-year outcomes of AVR with Perceval valves with or without concomitant procedures reported freedom from infective endocarditis (IE) ranging from 90.7% to 99% [
27]. Another meta-analysis of mid-term outcomes found a PVE incidence of 1.6% after a mean follow-up of 4.1 years, with 1.1% of patients requiring Perceval explantation due to IE [
28]. Combined data from three multicenter prospective clinical trials—Pilot, Pivotal and CAVALIER—showed a PVE incidence of 1.9% [
29], with similar rates reported among patients who underwent AVR with concomitant procedures [
17]. Comparable rates of late PVE were observed in a prospective cohort undergoing minimally invasive AVR [
11]. In the context of reoperative AVR, a prospective cohort study reported an incidence rate of 0.4% per patient-year [
30]. Additionally, a registry-based study found no cases of very early IE (within the first month of AVR) in patients with bicuspid aortic valves treated with sutureless AVR [
16].
A detailed description of patient characteristics in Perceval Valve PVE (PPVE) is lacking. A retrospective cohort with detailed reporting noted a mean age of 72 (standard deviation 7.22), a predominance of male patients (85.7%) and a mean EuroSCORE II score of 12.34% (standard deviation 7.4), partially driven by frequent comorbidities such as cardiovascular risk factors (with arterial hypertension and dyslipidemia occurring in 85% of patients), peripheral arterial disease and chronic renal insufficiency [
1]. Case reports similarly describe patients in their eighth decade of life affected by chronic cardiovascular comorbidity [
31,
32].
Although patient profiles are available, no study has identified specific risk factors for PPVE, whether attributable to patient comorbidity, procedural features, or periprocedural events. The risk of IE recurrence after IE requiring AVR with Perceval valves has not been reported.
PPVE remains the most frequent reason for Perceval valve replacement [
28,
33,
34]. Reported operability of PPVE patients ranges from 79% to 100%, mirroring the proportion of those who finally underwent surgical treatment for PPVE, possibly due to the high frequency of indications for surgery [
1,
35]. However, even if IE undoubtedly contributes to the elevated operative risk, no study has defined the determinants for inoperability in PPVE.
The timing of PPVE remains poorly defined due to the short and heterogeneous follow-up across available cohorts and the limited number of events. A predominance of early PVE (within the first year after AVR) over very early or late PVE (beyond the first year) has been reported in clinical trials [
29] and in both prospective observational [
11] and retrospective cohorts [
1]. In a single-arm prospective trial, the overall risk of PPVE was 1.4%, with most cases (8 out of 9) occurring between the first and the sixth postoperative months [
24]. A theoretically relevant distinction between PPVE and PVE involving conventional bioprosthetic valves is the potential for delayed diagnosis due to the absence of perivalvular leak in PPVE, potentially related to the self-expanding radial forces of the Perceval valve [
31,
32]. However, no study has directly compared the time distribution of PPVE with that of PVE affecting conventional bioprostheses.
No direct comparative studies evaluating the incidence or clinical and radiological manifestations of PPVE relative to those of PVE affecting conventional aortic bioprotheses exist. Available evidence is indirect and sparse. A case report of PPVE complicated with aortic abscess and aortic root extension describes a pattern of infection extension—particularly, a tendency to extend toward the left ventricular outflow tract—but these observations are still insufficient to define an epidemiological pattern [
31].
Table 1.
Relevant observational studies and clinical trials on the epidemiology of infective endocarditis following Perceval valve implantation.
Table 1.
Relevant observational studies and clinical trials on the epidemiology of infective endocarditis following Perceval valve implantation.
| First Author | Study Design | Study Period | Perceval
Population (n) | Perceval IE Cases (n) | Perceval IE Incidence Rate (Per 100 Patient-Years) | Perceval IE Cumulative Incidence (%) | Follow-Up Mean or Median Duration | Perceval IE Mortality (%) |
|---|
| Berastegui [1] | Retrospective cohort study; single-center | 2015–2020 | 670 | 14 | NR | 2.1 | 23 months | 14 (in-hospital), 41 (during follow-up) |
| Berastegui [33] | Prospective cohort study; multicentric | 2013–2016 | 448 | 2 | NR | 0.45 | 1 year | NR |
| Di Bacco [25] | Retrospective cohort study; multicentric | 2012–2015 | 518 | 8 | 0.6 | NR | 3.1 years | NR |
| Dokollari [22] | Systematic review and meta-analysis | - | 12,714 | NR | NR | 1.6–6.6 | - | NR |
| Flameng [35] | Retrospective cohort study; single-center | 2007–2009 | 32 | 1 | NR | 3.13 | 15.8 months | NR |
| Glauber [11] | Prospective cohort study; multicentric | 2011–2018 | 480 | 4 | 0.4 | NR | 2.4 years | NR |
| Jolliffe [28] | Systematic review and meta-analysis | - | 3196 | 50 | NR | 1.6 | - | NR |
| Lamberigts [26] | Retrospective cohort study; single-center | 2007–2019 | 784 | 13 | 0.46 | 1.7 | 7.03 years | NR |
| Rubino [21] | Retrospective cohort study; multicentric | 2007–2013 | 314 | NR | NR | 0.80 | 0.9 years | NR |
| Suri [23] | Single-arm clinical trial; multicentric | 2013–2025 | 300 | 5 | NR | 1.7 | 1 year | NR |
4. Microbiology
Information regarding the association between specific microorganisms and IE in the Perceval prosthesis is scarce. Most available data are derived from systematic reviews evaluating mid- and long-term outcomes of the Perceval valve and other sutureless bioprosthetic valves. Overall, it seems that the microbiological profile is similar to that reported for other biological prostheses, with streptococci, enterococci, and coagulase-negative staphylococci being the most frequently isolated organisms. The few series focusing only on Perceval endocarditis include fewer than 15 patients and provide limited microbiological data [
27,
28,
31,
34,
36]. The incidence of both early and late PVE in the Perceval prosthesis was similar to that reported in the literature for SAVR [
28], with cases of IE occurring from a few months up to 3 years after implantation [
37,
38]. There is just one study that focuses on the microbiological data for Perceval IE, which found that the most frequently isolated microorganism was
Staphylococcus epidermidis (7 out of 14 patients), and most cases (78.6%) were early PVE with an interval of more than 3 months after surgery [
1]. Another work based on a case report describes a case of IE caused by methicillin-resistant
Staphylococcus aureus 23 months after implantation [
31].
5. Clinical Presentation and Diagnosis
The clinical behaviour of PVE in sutureless devices, specifically the Perceval prosthesis, is fundamentally dictated by its unique anchoring mechanism. This design necessitates a specific pathophysiological understanding when infection occurs.
Current clinical evidence identifies a significant incidence of early-onset PVE, typically detected from the first month onwards following implantation. These cases are frequently associated with perioperative bacteraemia or nosocomial pathogens acquired during the initial surgical hospitalization. Conversely, late-onset PVE—defined as occurring more than one year after the procedure—follows a more indolent, subacute course. This late-stage infection often involves community-acquired pathogens and mimics the clinical progression observed in traditional stented bioprosthetic valves [
34,
39,
40].
5.1. Clinical Presentation and Symptomatology
Fever remains the most consistent clinical hallmark of PVE, documented in over 80% of the patient population. However, a high degree of clinical suspicion is required, as the fever may be absent or suppressed. This is particularly relevant in the Perceval recipient cohort, which often comprises elderly patients with multiple comorbidities or those receiving chronic anti-inflammatory medications.
Beyond pyrexia, the clinical spectrum encompasses non-specific constitutional symptoms such as generalized malaise, fatigue, and anorexia. In more virulent or aggressive presentations, patients may rapidly progress to septic syndrome characterized by hemodynamic instability and a significant rise in acute-phase reactants. The presence of these biomarkers, while non-specific, is a critical component of the diagnostic workup when evaluated alongside the patient’s surgical history [
4].
5.2. Mechanical Failure and Hemodynamic Dysfunction
A distinctive and critical feature of endocarditis in the Perceval valve is the early compromise of the periprosthetic seal. The infection typically colonizes the interface between the flexible nitinol frame and the native aortic annulus. As the infection progresses, it undermines the radial force and prosthetic apposition, leading to rapidly progressive paravalvular leaks.
From a clinical perspective, this mechanical failure manifests as acute or subacute heart failure. The loss of anchorage results in sudden valvular dysfunction or massive regurgitation. The detection of a de novo cardiac murmur in a patient with a sutureless valve is a clinical emergency that necessitates immediate imaging to exclude prosthetic dehiscence. Furthermore, while classic peripheral stigmata—such as Janeway lesions or splinter hemorrhages—are statistically less common in PVE, their presence provides definitive evidence of systemic involvement [
1,
37].
5.3. Systemic Embolism and Perivalvular Complications
Systemic embolic events are reported in 20% to 30% of cases and usually involve the central nervous system, the spleen, and the kidneys. In many instances, an embolic stroke or acute abdominal pain serves as the initial clinical manifestation of the underlying endocarditis.
The proximity of the nitinol frame to the conduction system also increases the risk of serious local complications. The formation of perivalvular abscesses and pseudoaneurysms is a frequent and severe progression of the disease. These invasive processes often manifest as new-onset conduction disturbances, such as high-grade atrioventricular blocks. Such findings indicate deep tissue destruction and often suggest the involvement of the left ventricular outflow tract, carrying a poor prognosis without urgent surgical intervention [
41].
5.4. The Multimodal Diagnostic Paradigm
The diagnosis of IE in the Perceval valve must be managed through a multimodal framework established by the 2023 Duke-ISCVID Criteria and the 2023 ESC Guidelines. Central to this approach is the “Endocarditis Team,” a multidisciplinary group tasked with synthesizing complex clinical, microbiological, and imaging data.
Microbiological identification remains a cornerstone of the diagnosis. It is mandatory to obtain at least three sets of blood cultures prior to commencing antimicrobial therapy. The identification of typical microorganisms, such as
Staphylococcus or
Enterococcus species, provides the microbiological major criterion required for a definitive diagnosis [
42].
6. Imaging Diagnosis of Infective Endocarditis in the Perceval Prosthesis
The specific characteristics of these sutureless prostheses suggest that a multimodality imaging (MMI) approach to achieving a definitive diagnosis of infection in Perceval valves may be required more often than in other surgically (sutured) implanted prostheses, and may be more comparable to TAVI-IE.
6.1. Transthoracic Echocardiography (TTE) and Transesophageal Echocardiography (TEE)
Transthoracic echocardiography (TTE) and transesophageal echocardiography (TEE) are the first-line imaging modalities for suspected PVE in sutureless Perceval prosthesis patients, but diagnosis in this context is challenging.
There are no studies specifically addressing the limitations of echocardiography in Perceval PVE. In similar scenarios, such as transcatheter aortic valve implantation (TAVI), the combined sensitivity of TTE and TEE for the diagnosis of TAVI-IE is about 67%, compared with 73% in patients with surgically implanted prosthetic valves [
43].
The design of the Perceval valve creates specific diagnostic pitfalls on TEE imaging (similar to those observed with TAVI prostheses) and differs from the diagnostic findings in other forms of PVE.
Some of these findings are specifically detailed below:
- -
Vegetations: Mobile, irregular echo-bright masses on the prosthetic leaflets or at the sewing ring/interface with the aorta. The metal stent creates shadows and artifacts that can obscure vegetations. Conversely, valve “struts” and folds of pericardial leaflet tissue can be mistaken for vegetations.
- -
Leaflet thickening and obstructive patterns: Thickened bioprosthetic leaflets and increased transvalvular Doppler gradients or reduced valve opening (possibly due to vegetative infiltration or thrombus) can be observed. These changes, while nonspecific, may raise suspicion in the appropriate clinical context.
- -
Paravalvular complications/abscesses: A thickened, non-homogeneous perivalvular area with echolucent zones. In the Perceval valve, it often manifests as a space between the stent and the aortic wall. In some series, periannular thickening (<10 mm) on TEE is not diagnostic of abscess, and periannular extension is clearly larger when cardiac computed tomography is performed [
1].
- -
Perivalvular leak/aortic regurgitation: Due to the special design of the Perceval prosthesis, which anchors the valve to the aortic annulus by means of the radial force of the nitinol stent alone, aortic regurgitation due to perivalvular leak is not present in the early stages of infection. The lack of periprosthetic leak could delay the diagnosis, especially if the images of periannular involvement are not diagnostic of abscess in TEE [
1]. In advanced stages of the infection, anatomical dehiscence with stent “rocking” and secondary severe paravalvular aortic regurgitation may be observed, even on TTE.
Given the limitations of echocardiography in this context, a multi-modality approach with advanced imaging techniques such as cardiac CT and 18F-fluorodeoxyglucose positron emission tomography/computed tomography ([18F]FDG-PET/CT) is recommended. These modalities can facilitate early diagnosis when there is high clinical suspicion, allowing better evaluation of periannular anatomical involvement, even when TEE is negative or non-conclusive or in patients with contraindications to TEE evaluation.
6.2. Cardiac CT and [18F] FDG PET/CT in the Diagnosis of Perceval Valve Endocarditis
A multimodality work-up may include cardiac computed tomography (CT) and 18F-fluorodeoxyglucose positron emission tomography/computed tomography ([18F]FDG-PET/CT), which have consolidated their role in IE. Nevertheless, there are no specific recommendations in the guidelines regarding the use of cardiac CT or [18F]FDG-PET/CT depending on the subtype of prosthetic valve. Accordingly, the work-up in suspected Perceval valve infection should follow the general indications for prosthetic valve endocarditis (PVE) [
4,
42]. Moreover, the diagnostic accuracy of CT and/or PET/CT has been evaluated in the setting of suspected PVE in general series in which the specific prosthetic valve models were not well reported and may or may not have included sutureless valves. Therefore, until specific data become available, it is reasonable to extrapolate to Perceval valves the general diagnostic performance and application of the general guideline-based indications established for PVE.
6.2.1. CARDIAC CT
Cardiac CT provides high spatial resolution for the anatomical and functional assessment of prosthetic heart valves and has proven to be useful in the diagnosis of infective endocarditis and the detection of its complications [
44]. In particular, cardiac CT has shown higher sensitivity for the detection of periprosthetic complications, such as abscesses and pseudoaneurysms, when compared to transesophageal echocardiography (TEE) (87% for CT vs. 69% for TEE) [
45]. This advantage is especially useful in the suspicion of endocarditis involving Perceval prostheses, as these valves frequently develop periprosthetic abscesses, sometimes even in the absence of clearly identifiable vegetations. On CT, abscesses appear as periprosthetic soft-tissue thickening with variable extent. If disruption of the aortic wall or other structures occurs, cardiac CT also allows the detection of pseudoaneurysms. Although CT is not superior to echocardiography, it can also identify vegetations, which may be located on the leaflets or at different sites along the stent in this type of device. Vegetations may not be easily visualized by echocardiography due to metallic shadowing and artifacts. In addition, CT provides valuable information for surgical planning, including non-invasive assessment of coronary anatomy and atherosclerotic disease if patients require cardiac surgery.
6.2.2. [18F]FDG-PET/CT
[18F]FDG-PET/CT is a hybrid imaging technique that has demonstrated significant diagnostic utility in PVE, allowing the reclassification of a large proportion of cases that initially remain categorized as doubtful or “possible” after clinical and echocardiographic evaluation [
46]. The added value of PET/CT in PVE has been confirmed across patient series with mixed prosthetic valve types, or even in cohorts not including sutureless models [
47]. In general, a focal, multifocal, or heterogeneous FDG uptake pattern, visualized on the valve stent, is highly suggestive of infection. FDG activity extending into the periaortic space may suggest a possible periprosthetic abscess. When PET/CT is performed as a cardiac-dedicated examination, including cardiac CT within a single acquisition, it provides not only metabolic information but also allows the detection of the associated anatomic lesions (vegetations and periprosthetic complications), thereby significantly improving diagnostic yield.
Furthermore, both CT and [18F]FDG-PET/CT allow extracardiac evaluation for the detection of distant lesions, which are common in the setting of endocarditis. These findings may also fulfill minor criteria useful for reclassification of patients with suspected infective endocarditis and can contribute meaningfully to overall patient management [
47].
PET/CT can also identify the portal of entry of the infection of certain microorganisms, such as colonic lesions, which is particularly relevant in infective endocarditis, as eradication of the infectious source may prevent relapse and/or reinfections [
47]. Furthermore, when endocarditis is ruled out, PET/CT can suggest an alternative infectious or noninfectious diagnosis. Another important advantage of PET/CT is that, when endocarditis is excluded, it can suggest alternative infectious or noninfectious diagnoses.
Figure 1 presents three representative cases assessed by echocardiography and PET/CT.
Since Perceval valve implantation is usually selected for patients at moderate to high surgical risk, particularly elderly patients [
48], some individuals with Perceval valve endocarditis may be considered poor candidates for cardiac surgery and managed with long-term antibiotic therapy. In this context, PET/CT findings can support clinical decision-making and may be used to monitor the response to antimicrobial treatment [
48].
Figure 2 highlights the importance of a comprehensive CT assessment, whereas
Figure 3 presents a case of endocarditis with spondylodiscitis, illustrating the value of PET/CT in a patient with inconclusive echocardiographic findings.
Based on the information above, a specific diagnostic approach is recommended for patients with a Perceval valve and suspected IE (
Figure 4).
7. Treatment
7.1. Antimicrobial Treatment
Evidence specifically addressing antimicrobial treatment for infective endocarditis involving the Perceval valve is limited, and no valve-specific therapeutic recommendations are currently available. Consequently, antibiotic management follows the general principles applied to patients with prosthetic valve infective endocarditis [
4].
Empirical therapy should therefore be guided by the timing of infection onset. In early prosthetic valve endocarditis (<12 months after valve implantation), microorganisms typically associated with healthcare exposure, particularly coagulase-negative Staphylococcus, are predominant. In contrast, in late prosthetic valve endocarditis (≥12 months after implantation), the microbiological spectrum more closely resembles that of native valve endocarditis [
49]. Once the causative pathogen has been identified, antimicrobial therapy should be tailored accordingly. In the absence of complications, the recommended duration of antibiotic treatment is at least 6 weeks [
1].
Although no alternative antimicrobial regimens can be recommended specifically for Perceval valve endocarditis, several aspects of this patient population warrant particular attention in clinical practice. First, this sutureless bioprosthetic valve, owing to its simplified implantation technique, is preferentially used in older patients and in those with significant comorbidities [
1,
37], who are more susceptible to antibiotic-related adverse events, including nephrotoxicity, fluid overload, and pharmacological interactions. As a result, careful individualized risk–benefit assessment is essential when selecting antimicrobial therapy. The use of agents such as aminoglycosides or rifamycins requires careful assessment and close monitoring, given their potential for severe toxicity in this patient population. Whenever feasible, antimicrobial regimens should be selected with the goal of preserving efficacy while minimizing treatment-related toxicity.
Second, owing to the valve’s anchoring and deployment mechanism, there is evidence of a higher incidence of perivalvular involvement [
1], including abscess formation [
31]. As a result, surgical management represents a cornerstone of treatment in these cases. In patients who are not candidates for repeat surgical intervention, standard-duration antibiotic therapy is often insufficient, and prolonged or suppressive antimicrobial treatment should be considered to achieve infection control, with high rates of failure and high mortality associated [
50].
7.2. Surgical Indications in Perceval Prosthesis Infective Endocarditis
Surgical indications for infective endocarditis (IE) affecting a Perceval sutureless aortic prosthesis follow the principles applied to PVE, as outlined in the 2023 ESC guidelines [
4]. Even so, the Perceval does not behave like a conventional sutured bioprosthesis when infection involves the annulus. As a result, patients often reach surgical thresholds earlier, making prompt Heart Team assessment essential.
The classic surgical triggers in PVE (acute prosthetic dysfunction with heart failure, persistent infection, perivalvular complications and significant embolic risk) apply fully to Perceval infections. These indications are supported by recent literature analysing sutureless prostheses and Perceval-specific series [
51,
52]. In practice, the mechanical vulnerability of this design means that these criteria tend to be met sooner when annular or perivalvular involvement develops.
Prosthesis instability is one of the most decisive reasons to proceed early. Because fixation depends exclusively on radial force, even small areas of annular compromise (such as an early abscess, localized necrosis or a phlegmon) can destabilize the prosthesis and lead to partial or complete dehiscence [
1]. This differs from sutured valves, where isolated infected stitches may still allow temporary stability. In Perceval prostheses, loss of support can be abrupt, and imaging signs of instability (rocking, displacement or new paravalvular leakage) should be considered urgent indications for surgery, even in the absence of advanced heart failure.
Acute leaflet dysfunction is another clear trigger for intervention. Infectious destruction, obstructive vegetations or sudden severe regurgitation cannot be managed conservatively in a sutureless system. Repair is not feasible, and significant dysfunction nearly always leads to explantation. Surgical reintervention may be complex due to inflamed or friable annular tissue, as reported in several case series [
53]. Early surgery therefore helps prevent further damage and reduces operative difficulty.
Perivalvular complications—abscesses, pseudoaneurysms and fistulas—are also strong indications for early intervention. The lack of a sewing ring offers minimal resistance to the outward spread of infection, which may accelerate the formation of invasive lesions [
1,
29]. When such complications are detected or suspected, early surgery is advisable, regardless of temporary clinical stability.
Vegetations ≥ 10–15 mm or those showing marked mobility carry a high embolic risk and remain a well-established reason for surgery. Although comparative imaging data are limited, the open-frame design of the Perceval may allow greater mobility of attached vegetations. Observational studies and expert reviews support early intervention in this setting [
29,
54].
Microbiology significantly influences timing.
Staphylococcus aureus, fungi and non-HACEK Gram-negative organisms often produce rapid tissue destruction. Given the limited mechanical tolerance of the Perceval prosthesis, these pathogens frequently lead to earlier surgical indication and make prolonged medical therapy alone unreliable [
4,
55].
The potential role of valve-in-valve transcatheter aortic valve implantation (TAVI) in the setting of active infective endocarditis remains unclear, as evidence is scarce and limited to anecdotal reports. To date, published data consist only of isolated case reports involving conventional bioprosthetic valves, including a recent single case [
55], and no studies have specifically addressed its use in Perceval sutureless prostheses. In light of this limited evidence, no therapeutic recommendation can be drawn. That said, in exceptional circumstances—particularly in patients considered unsuitable for surgery—valve-in-valve TAVI could be contemplated as a rescue approach after adequate infection control and thorough multidisciplinary Heart Team discussion. Further clinical experience and dedicated studies are needed to better define its potential role in this context.
7.3. Technical Surgical Considerations for Endocarditis on a Perceval Prosthesis
Large series describing Perceval explantation are lacking, and most of the recommended surgical maneuvers have been extrapolated from the extraction of self-expanding transcatheter valves due to their structural similarities [
56].
When facing a Perceval prosthesis that must be explanted—whether due to endocarditis or structural degeneration—preoperative imaging is essential to evaluate the degree of endothelialization of the stent frame. However, unlike in transcatheter prostheses, these findings do not guide the height of the aortotomy or the cannulation strategy. In general, explantation of a Perceval prosthesis is performed through a median sternotomy with central cannulation. The site of the aortotomy is usually determined by the location of the previous one.
Once the prosthesis is exposed, two main strategies have been described for its removal. In both techniques, the critical step is the accurate identification of the dissection plane between the prosthesis and the aortic intima [
57]. After locating this plane, extraction may proceed using one of the following methods:
7.3.1. X-Shaped Maneuver [32]
This is the technique traditionally used when the prosthesis must be repositioned during the initial implant. Using two forceps placed 180° apart on the upper crown of the stent, opposing traction is applied to create an “X-shaped” motion that detaches the stent posts from the aortic wall, facilitating prosthesis removal. This maneuver is particularly useful in recent implants or in cases with minimal adherence of the stent to the aorta and aortic annulus.
7.3.2. “Lasso” Technique [58]
Originally described for the extraction of self-expanding transcatheter valves, this technique can be adapted, with minor modifications, for Perceval prostheses. Once the dissection plane and the upper crown of the prosthesis are identified, a circumferential suture is passed around the crown and secured with a tourniquet, creating centripetal traction that partially collapses the prosthesis. This controlled folding facilitates traction, mobilization, and meticulous dissection of the prosthesis, ultimately enabling safe removal.
In both techniques, irrigating the prosthesis with cold saline has been reported as beneficial, as it induces contraction of the nitinol stent [
59], aiding dissection and reducing the risk of structural complications.
Following prosthesis explantation, the surgical approach depends on the extent of structural damage caused by endocarditis—such as abscesses, pseudoaneurysms, or fistulous tracts—as well as by the extraction maneuvers. The surgical strategy may range from simple aortic valve replacement with a new prosthesis to replacement combined with annular or root repair procedures, or even complete aortic root replacement. The need for associated or more complex procedures arises in up to 50% of cases when removing self-expanding prostheses [
59].
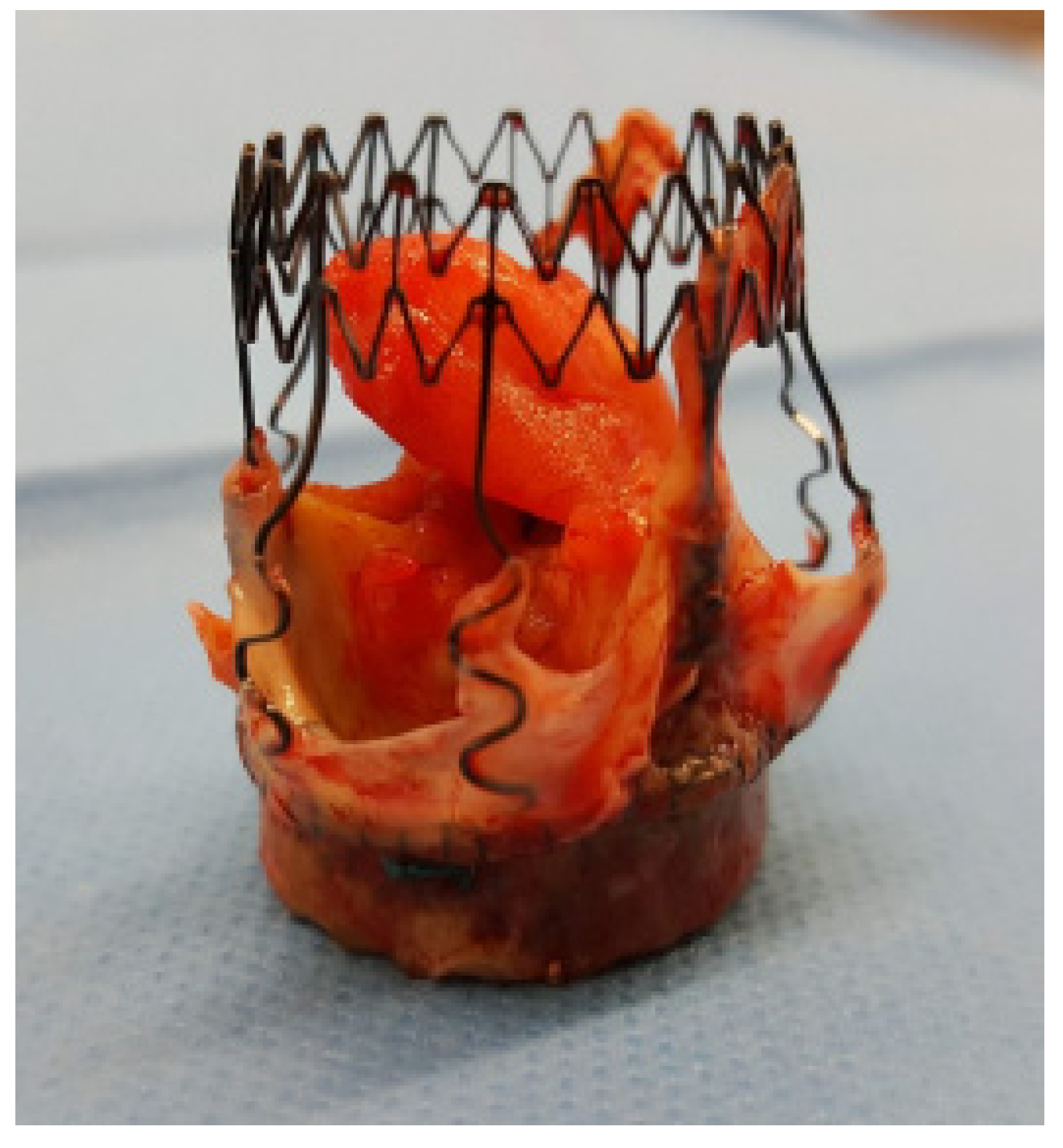
Figure 5 and
Figure 6 show an explanted Perceval valve and intraoperative findings of endocarditis involving a Perceval valve.
8. Prognosis
8.1. Comparative Durability
The clinical landscape of Sutureless Aortic Valve Replacement (Su-AVR) has been significantly shaped by the performance of the Perceval valve. Current evidence suggests that the incidence of PVE in these models is notably low. Long-term follow-up studies have reported freedom from PVE rates as high as 98% at the 8-year mark [
40]. However, a critical observation in longitudinal cohorts is that PVE has emerged as the primary indication for late valvular explant. This stands in stark contrast to conventional bioprostheses, as Structural Valve Deterioration (SVD) is remarkably rare—and in many series, practically non-existent—within this specific sutureless model [
37,
40]. Consequently, while the valve demonstrates exceptional mechanical durability, infection remains the most significant threat to its long-term viability.
8.2. Prognostic Determinants and Clinical Gravity
The prognosis for patients developing PPVE is intrinsically severe, mirroring the high-risk profile associated with any prosthetic heart valve infection. Contemporary registries indicate a hospital mortality rate ranging between 25% and 35% [
60,
61]. This elevated mortality is often a reflection of the patient population typically selected for sutureless valves—often older individuals with higher EuroSCORE II values and multiple comorbidities. Survival outcomes are drastically undermined by the rapid onset of intracardiac complications. Specifically, the development of perivalvular abscesses, acute heart failure, septic shock, or acute kidney injury serves as a primary driver of poor clinical outcomes.
8.3. Therapeutic Strategies: The Surgical Imperative
Perioperative mortality for redo interventions in prosthetic endocarditis remains high, often exceeding 25%. Despite this surgical risk, the data overwhelmingly support an aggressive surgical approach when indicated. Large-scale retrospective analyses have demonstrated a striking disparity in outcomes based on management strategy: patients treated with medical therapy alone faced a 34% mortality rate, compared to only 9% among those who underwent surgical intervention [
61]. These figures reinforce the critical role of timely surgery, despite the inherent technical complexity of explanting a nitinol-based frame and debriding the aortic root. For those who survive the acute phase, long-term survival rates are comparable to those seen in native valve endocarditis, though patients must be closely monitored for recurrent infection or residual hemodynamic dysfunction [
40,
61].
8.4. Technical Advantages in Redo Scenarios
Notably, the Perceval valve serves not only as a subject of PVE but also as a feasible therapeutic option for its management. In cases of active endocarditis necessitating re-replacement, the device has demonstrated clinical utility as a redo surgical option. Its sutureless design facilitates rapid deployment, which significantly reduces cardiopulmonary bypass and aortic cross-clamp durations—parameters that are critical for survival in hemodynamically unstable patients with high operative risk [
40].
Furthermore, its favourable hemodynamic performance and the absence of a conventional sewing ring may allow for improved adaptation to a friable or damaged aortic annulus.
9. Future Directions
Endocarditis is a constantly evolving disease, increasingly affecting complex patient populations, many of whom are elderly and have a high burden of comorbidities. At the same time, the incidence of infections involving prosthetic valves, aortic grafts, and cardiac implantable electronic devices continues to rise. As with many other cardiovascular conditions, this heterogeneity of clinical presentations underscores the need for an individualized approach. Despite this, epidemiological studies describing endocarditis according to the type of valve affected remain scarce. In this context, although Perceval prostheses are gaining acceptance and are increasingly used in surgical practice, there are still very few published case series reporting PVE.
Therefore, an individualized approach to PVE is warranted, supported by robust population-based incidence data and clinical registries that take into account the type of prosthesis, both those currently available and those that may become available in the future. Such registries should incorporate detailed clinical, microbiological, imaging, and prognostic information. Additionally, there is a critical need to determine the optimal duration of antimicrobial therapy for patients who have a surgical indication but cannot be operated on, particularly patients with paravalvular involvement. In this scenario, advanced imaging techniques, such as PET-CT, may play a key role in guiding clinical decisions, including the timing of antimicrobial withdrawal. A comprehensive analysis of these factors will help refine not only the diagnostic strategies but also the management and outcomes of these highly complex patients.
10. Conclusions
Sutureless aortic bioprostheses, and particularly the Perceval valve, represent a major step forward in the surgical treatment of aortic valve disease. However, as their use expands, clinicians are increasingly faced with complex postoperative complications such as prosthetic valve endocarditis.
Endocarditis involving a Perceval valve remains an uncommon but severe entity associated with high morbidity and mortality. Diagnostic accuracy continues to depend on a multimodality imaging approach, combining echocardiography and nuclear imaging techniques. Management requires a tailored strategy that balances antimicrobial therapy and timely surgical intervention. Surgical explantation of the Perceval prosthesis can be technically demanding due to its self-expanding metallic frame and potential adhesion to the aortic wall.
Future research should focus on refining perioperative infection prevention strategies, understanding the pathophysiological mechanisms of bacterial adherence specific to sutureless devices, and optimizing both medical and surgical management protocols.
In conclusion, while sutureless aortic bioprostheses have broadened the therapeutic options for aortic valve disease, vigilance for infectious complications remains crucial. The multidisciplinary collaboration among cardiologists, cardiac surgeons, infectious disease specialists, and imaging experts is fundamental to improving prognosis and ensuring the continued safe use of these innovative devices.
Author Contributions
Conceptualization, N.F.H., original draft preparation, all authors.; writing—review and editing, all authors; supervision, N.F.H. and P.R. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Data Availability Statement
The data supporting the findings of this study are not publicly available due to ethical and privacy restrictions.
Acknowledgments
Endocarditis Team, Hospital Universitari Vall d’Hebron, Barcelona, Spain. Lluís Admella, Montserrat Bach, Gemma Farga, Nuria Fernández-Hidalgo, Álvaro García, Víctor González, Olga Maisterra, Arwa Mehmood, María Nazarena Pizzi, Andrés Rodrigo Parra-Rojas, Xavier Peris, Antonia Pijuan, Pau Rello, Remedios Ríos, Albert Roque, Marco Tomasino, Nuria Vallejo-Camazón, Carlota Vigil.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- García, E.B.; Camazón, N.V.; Pruñonosa, L.M.; Carrasco, S.L.; Bayés-Genís, A.; Guijosa, C.M. En-docarditis on a Perceval S sutureless prosthesis. A new valve with a new form of clinical presentation. Rev. Esp. Cardiol. 2021, 74, 635–637. [Google Scholar] [CrossRef]
- Meco, M.; Montisci, A.; Miceli, A.; Panisi, P.; Donatelli, F.; Cirri, S.; Ferrarini, M.; Lio, A.; Glauber, M. Sutureless Perceval Aortic Valve Versus Conventional Stented Bioprostheses: Meta-Analysis of Postoperative and Midterm Results in Isolated Aortic Valve Replacement. J. Am. Heart Assoc. 2018, 7, e006091. [Google Scholar] [CrossRef] [PubMed]
- Fischlein, T.; Folliguet, T.; Meuris, B.; Shrestha, M.L.; Roselli, E.E.; McGlothlin, A.; Kappert, U.; Pfeiffer, S.; Corbi, P.; Lorusso, R.; et al. Sutureless versus conventional bioprostheses for aortic valve replacement in severe symptomatic aortic valve stenosis. J. Thorac. Cardiovasc. Surg. 2021, 161, 920–932. [Google Scholar] [CrossRef] [PubMed]
- Delgado, V.; Marsan, N.A.; de Waha, S.; Bonaros, N.; Brida, M.; Burri, H.; Caselli, S.; Doenst, T.; Ederhy, S.; Erba, P.A.; et al. ESC Guidelines for the management of endocarditis. Eur. Heart J. 2023, 44, 3948–4042. [Google Scholar] [CrossRef] [PubMed]
- Perceval Sutureless Aortic Heart Valve Instructions for Use. Available online: https://www.accessdata.fda.gov/cdrh_docs/pdf15/P150011d.pdf (accessed on 20 January 2026).
- Praz, F.; A Borger, M.; Lanz, J.; Marin-Cuartas, M.; Abreu, A.; Adamo, M.; Marsan, N.A.; Barili, F.; Bonaros, N.; Cosyns, B.; et al. ESC/EACTS Guidelines for the management of valvular heart disease. Eur. Heart J. 2025, 46, 4635–4736. [Google Scholar] [CrossRef] [PubMed]
- Zubarevich, A.; Rad, A.A.; Amanov, L.; Szczechowicz, M.; Osswald, A.; Torabi, S.; Schmack, B.; Ruhparwar, A.; Weymann, A. Sutureless aortic valve replacement in pure aortic regurgitation: Expanding the indications. J. Cardiothorac. Surg. 2022, 17, 198. [Google Scholar] [CrossRef]
- Folliguet, T.A.; Laborde, F. Sutureless Perceval aortic valve replacement in aortic homograft. Ann. Thorac. Surg. 2013, 96, 1866–1868. [Google Scholar] [CrossRef]
- Misfeld, M. Minimally invasive aortic valve replacement using the Perceval S sutureless valve. Cardiothorac. Surg. 2015, 4, 203–205–205. [Google Scholar] [CrossRef]
- Glauber, M.; Moten, S.C.; Quaini, E.; Solinas, M.; Folliguet, T.A.; Meuris, B.; Miceli, A.; Oberwalder, P.J.; Rambaldini, M.; Teoh, K.H.T.; et al. International Expert Consensus on Sutureless and Rapid Deployment Valves in Aortic Valve Replacement Using Minimally Invasive Approaches. Innov. Technol. Tech. Cardiothorac. Vasc. Surg. 2016, 11, 165–173. [Google Scholar] [CrossRef]
- Glauber, M.; Di Bacco, L.; Cuenca, J.; Di Bartolomeo, R.; Baghai, M.; Zakova, D.; Fischlein, T.; Troise, G.; Viganò, G.; Solinas, M. Minimally Invasive Aortic Valve Replacement with Sutureless Valves: Results from an International Prospective Registry. Innov. Technol. Tech. Cardiothorac. Vasc. Surg. 2020, 15, 120–130. [Google Scholar] [CrossRef]
- Fiore, A.; Gueldich, M.; Folliguet, T. Sutureless valves fit/perform well in a small aortic annulus. Ann. Cardiothorac. Surg. 2020, 9, 414–416. [Google Scholar] [CrossRef] [PubMed]
- Zubarevich, A.; Szczechowicz, M.; Zhigalov, K.; Osswald, A.; Eynde, J.V.D.; Rad, A.A.; Vardanyan, R.; Wendt, D.; Schmack, B.; Ruhparwar, A.; et al. Sutureless aortic valve replacement in multivalve procedures. J. Thorac. Dis. 2021, 13, 3392–3398. [Google Scholar] [CrossRef] [PubMed]
- Zubarevich, A.; Rad, A.A.; Szczechowicz, M.; Ruhparwar, A.; Weymann, A. Sutureless aortic valve replacement in high-risk patients with active infective endocarditis. J. Thorac. Dis. 2022, 14, 3178–3186. [Google Scholar] [CrossRef]
- Nguyen, A.; Fortin, W.; Mazine, A.; Bouchard, D.; Demers, P. Should Bicuspid Disease be a Contraindication? Can. J. Cardiol. 2014, 30, S238. [Google Scholar] [CrossRef]
- Miceli, A.; Berretta, P.; Fiore, A.; Andreas, M.; Solinas, M.; Santarpino, G.; Kappert, U.; Misfeld, M.; Savini, C.; Albertini, A.; et al. Sutureless and rapid deployment implantation in bicuspid aortic valve: Results from the sutureless and rapid-deployment aortic valve replacement international registry. Ann. Cardiothorac. Surg. 2020, 9, 298–304. [Google Scholar] [CrossRef]
- Shrestha, M.; Folliguet, T.A.; Pfeiffer, S.; Meuris, B.; Carrel, T.; Bechtel, M.; Flameng, W.J.; Fischlein, T.; Laborde, F.; Haverich, A. Aortic valve replacement and concomitant procedures with the perceval valve: Results of european trials. Ann. Thorac. Surg. 2014, 98, 1294–1300. [Google Scholar] [CrossRef]
- Glauber, M.; Miceli, A.; di Bacco, L. Sutureless and rapid deployment valves: Implantation technique from A to Z-the Perceval valve. Ann. Cardiothorac. Surg. 2020, 9, 330–340. [Google Scholar] [CrossRef]
- Vasanthan, V.; Kent, W.; Gregory, A.; Maitland, A.; Cutrara, C.; Bouchard, D.; Asch, F.; Adams, C. Perceval Valve Implantation: Technical Details and Echocardiographic Assessment. Ann. Thorac. Surg. 2019, 107, e223–e225. [Google Scholar] [CrossRef]
- Mashhour, A.; Zhigalov, K.; Szczechowicz, M.; Mkalaluh, S.; Easo, J.; Eichstaedt, H.; Borodin, D.; Ennker, J.; Weymann, A. Snugger method—The Oldenburg modi-fication of perceval implantation technique. World J. Cardiol. 2018, 10, 119–122. [Google Scholar] [CrossRef]
- Rubino, A.S.; Santarpino, G.; De Praetere, H.; Kasama, K.; Dalén, M.; Sartipy, U.; Lahtinen, J.; Heikkinen, J.; Deste, W.; Pollari, F.; et al. Early and intermediate outcome after aortic valve replacement with a sutureless bioprosthesis: Results of a multicenter study. J. Thorac. Cardiovasc. Surg. 2014, 148, 865–871. [Google Scholar] [CrossRef]
- Dokollari, A.; Torregrossa, G.; Sicouri, S.; Veshti, A.; Margaryan, R.; Cameli, M.; Mandoli, G.E.; Maccherini, M.; Montesi, G.; Cabrucci, F.; et al. Pearls, pitfalls, and surgical indications of the Intuity TM heart valve: A rapid deployment bioprosthesis. A systematic review of the literature. J. Card. Surg. 2022, 37, 5411–5417. [Google Scholar] [CrossRef] [PubMed]
- Suri, R.M.; Javadikasgari, H.; Heimansohn, D.A.; Weissman, N.J.; Ailawadi, G.; Ad, N.; Aldea, G.S.; Thourani, V.H.; Szeto, W.Y.; Michler, R.E.; et al. Prospective US investigational device exemption trial of a sutureless aortic bioprosthesis: One-year outcomes. J. Thorac. Cardiovasc. Surg. 2019, 157, 1773–1782.e3. [Google Scholar] [CrossRef] [PubMed]
- Fischlein, T.; Meuris, B.; Hakim-Meibodi, K.; Misfeld, M.; Carrel, T.; Zembala, M.; Gaggianesi, S.; Madonna, F.; Laborde, F.; Asch, F.; et al. The sutureless aortic valve at 1 year: A large multicenter cohort study. J. Thorac. Cardiovasc. Surg. 2016, 151, 1617–1626.e4. [Google Scholar] [CrossRef] [PubMed]
- Di Bacco, L.; Rosati, F.; Folliguet, T.; Petruccelli, R.D.; Concistrè, G.; Santarpino, G.; Di Bartolomeo, R.; Bisleri, G.; Fischlein, T.J.; Muneretto, C. Sutureless aortic valves in elderly patients with aortic stenosis and intermediate-risk profile. J. Cardiovasc. Med. 2021, 22, 297–304. [Google Scholar] [CrossRef]
- Lamberigts, M.; Szecel, D.; Rega, F.; Verbrugghe, P.; Dubois, C.; Meuris, B. Sutureless aortic valves in isolated and combined pro-cedures: Thirteen years of experience in 784 patients. J. Thorac. Cardiovasc. Surg. 2024, 167, 1724–1732.e1. [Google Scholar] [CrossRef]
- Chiariello, G.A.; Di Mauro, M.; Villa, E.; Koulouroudias, M.; Bruno, P.; Mazza, A.; Pasquini, A.; D’avino, S.; De Angelis, G.; Corigliano, K.; et al. Sutureless Bioprostheses for Aortic Valve Replacement: An Updated Systematic Review with Long-Term Results. J. Clin. Med. 2024, 13, 6829. [Google Scholar] [CrossRef]
- Jolliffe, J.; Moten, S.; Tripathy, A.; Skillington, P.; Tatoulis, J.; Muneretto, C.; Di Bacco, L.; Galvao, H.B.F.; Goldblatt, J. Perceval valve intermediate outcomes: A systematic review and meta-analysis at 5-year follow-up. J. Cardiothorac. Surg. 2023, 18, 129. [Google Scholar] [CrossRef]
- Shrestha, M.; Fischlein, T.; Meuris, B.; Flameng, W.; Carrel, T.; Madonna, F.; Misfeld, M.; Folliguet, T.; Haverich, A.; Laborde, F. European multicentre experience with the su-tureless Perceval valve: Clinical and haemodynamic outcomes up to 5 years in over 700 patients. Eur. J. Cardio-Thoracic Surg. 2016, 49, 234–241. [Google Scholar] [CrossRef]
- Glauber, M.; Kent, W.D.; Asimakopoulos, G.; Troise, G.; Padrò, J.M.; Royse, A.; Marnette, J.-M.; Noirhomme, P.; Baghai, M.; Lewis, M.; et al. Sutureless Valve in Repeated Aortic Valve Replacement: Results from an International Prospective Registry. Innov. Technol. Tech. Cardiothorac. Vasc. Surg. 2021, 16, 273–279. [Google Scholar] [CrossRef]
- Fujimoto, R.; Hirao, S.; Komiya, T. Infective endocarditis with perivalvular abscess following sutureless valve implantation, successfully treated with aortic root reconstruction and biological Bentall procedure. J. Cardiothorac. Surg. 2025, 20, 279. [Google Scholar] [CrossRef]
- Di Bacco, L.; Pfeiffer, S.; Fischlein, T.J.M.; Santarpino, G. Rapid Explantation of Rapid-Deployment Sutureless Valve in Case of Acute Endocarditis How to Remove Safely Sutureless Perceval S Prostheses. Innov. Technol. Tech. Cardiothorac. Vasc. Surg. 2017, 12, 483–485. [Google Scholar] [CrossRef]
- Muneretto, C.; Di Bacco, L.; Pollari, F.; Baudo, M.; Solinas, M.; D’alonzo, M.; Di Eusanio, M.; Rosati, F.; Folliguet, T.; Fischlein, T. Sutureless versus transcatheter valves in patients with aortic stenosis at intermediate risk: A multi-institutional European study. Surgery 2023, 174, 1153–1160. [Google Scholar] [CrossRef] [PubMed]
- García, E.B.; Rosell, M.L.C.; Cid, F.E.; Dominguez, E.S.; Barrera, R.R.; Sbraga, F.; Puente, J.G.; Diez, E.R.; Caballero, G.P.C.; Munguira, J.B.; et al. Perceval Less Invasive Aortic Replacement Register: Multicentric Spanish experience with the Perceval S bioprosthesis in moderate-high-risk aortic surgery. Interact. Cardiovasc. Thorac. Surg. 2018, 26, 596–601. [Google Scholar] [CrossRef] [PubMed]
- Flameng, W.; Herregods, M.-C.; Hermans, H.; Van der Mieren, G.; Vercalsteren, M.; Poortmans, G.; Van Hemelrijck, J.; Meuris, B. Effect of sutureless im-plantation of the Perceval S aortic valve bioprosthesis on intraoperative and early postoperative outcomes. J. Thorac. Cardiovasc. Surg. 2011, 142, 1453–1457. [Google Scholar] [CrossRef] [PubMed]
- Deblier, I.; De Bock, D.; Rodrigus, I.; Mistiaen, W. The Use of Perceval Valves in Older Patients: A Systematic Review. Rev. Cardiovasc. Med. 2025, 26, 39463. [Google Scholar] [CrossRef]
- Szecel, D.; Eurlings, R.; Rega, F.; Verbrugghe, P.; Meuris, B. Perceval Sutureless Aortic Valve Implantation: Midterm Outcomes. Ann. Thorac. Surg. 2021, 111, 1331–1337. [Google Scholar] [CrossRef]
- García, E.B.; Rosell, M.L.C.; Da Silva, P.A.; Gamarra, S.B.; Ramis, L.D.; Gallego, C.F.; Sistach, E.F.; Almill, I.J.; Ayerbe, J.L.; Ferrer, B.R.; et al. Prótesis sin sutura Perceval S en la cirugía valvular aórtica de alto riesgo. Una herramienta fundamental para el cirujanoSutureless Perceval S (Livanova, Saluggia, Italia) aortic valve in high risk aortic patients. A key tool for the cardiac surgeon. Cirugia Cardiovasc. 2017, 24, 267–273. [Google Scholar] [CrossRef]
- Pollari, F.; Mamdooh, H.; Hitzl, W.; Grossmann, I.; Vogt, F.; Fischlein, T. Ten years’ experience with the sutureless aortic valve replacement: Incidence and predictors for survival and valve durability at follow-up. Eur. J. Cardio-Thoracic Surg. 2023, 63, ezac572. [Google Scholar] [CrossRef]
- Concistré, G.; Baghai, M.; Santarpino, G.; Royse, A.; Scherner, M.; Troise, G.; Glauber, M.; Solinas, M. Clinical and hemodynamic outcomes of the Perceval sutureless aortic valve from a real-world registry. Interdiscip. Cardiovasc. Thorac. Surg. 2023, 36, ivad103. [Google Scholar] [CrossRef]
- Li, M.; Kim, J.B.; Sastry, B.K.S.; Chen, M. Infective endocarditis. Lancet 2024, 404, 377–392. [Google Scholar] [CrossRef]
- Fowler, V.G.; Durack, D.T.; Selton-Suty, C.; Athan, E.; Bayer, A.S.; Chamis, A.L.; Dahl, A.; DiBernardo, L.; Durante-Mangoni, E.; Duval, X.; et al. The 2023 Duke-International Society for Cardiovascular Infectious Diseases Criteria for Infective Endocarditis: Updating the Modified Duke Criteria. Clin. Infect. Dis. 2023, 77, 518–526. [Google Scholar] [CrossRef] [PubMed]
- Mangner, N.; Panagides, V.; del Val, D.; Abdel-Wahab, M.; Crusius, L.; Durand, E.; Ihlemann, N.; Urena, M.; Pellegrini, C.; Giannini, F.; et al. Incidence, Clinical Characteristics, and Impact of Absent Echocardiographic Signs in Patients With Infective Endocarditis After Transcatheter Aortic Valve Implantation. Clin. Infect. Dis. 2023, 76, 1003–1012. [Google Scholar] [CrossRef] [PubMed]
- Bin Saeedan, M.; Wang, T.K.M.; Cremer, P.; Wahadat, A.R.; Budde, R.P.J.; Unai, S.; Pettersson, G.B.; Bolen, M.A. Role of Cardiac CT in Infective Endocarditis: Current Evidence, Opportunities, and Challenges. Radiol. Cardiothorac. Imaging 2021, 3, e200378. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, M.; Guittet, L.; Hamon, M.; Hamon, M. Comparative Value of Cardiac CT and Transesophageal Echocardiography in Infective Endocarditis: A Systematic Review and Meta-Analysis. Radiol. Cardiothorac. Imaging 2020, 2, e190189. [Google Scholar] [CrossRef]
- Wang, T.K.M.; Sánchez-Nadales, A.; Igbinomwanhia, E.; Cremer, P.; Griffin, B.; Xu, B. Diagnosis of Infective Endocarditis by Subtype Using 18F-Fluorodeoxyglucose Positron Emission Tomography/Computed Tomography: A Contemporary Meta-Analysis. Circ. Cardiovasc. Imaging 2020, 13, e010600. [Google Scholar] [CrossRef]
- Pizzi, M.N.; Roque, A.; Fernández-Hidalgo, N.; Cuéllar-Calabria, H.; Ferreira-González, I.; Gonzàlez-Alujas, M.T.; Oristrell, G.; Gracia-Sánchez, L.; González, J.J.; Rodríguez-Palomares, J.; et al. Improving the Diagnosis of Infective Endocarditis in Prosthetic Valves and Intracardiac Devices with 18F-Fluordeoxyglucose Positron Emission Tomography/Computed Tomography Angiography: Initial Results at an Infective Endocarditis Referral Center. Circulation 2015, 132, 1113–1126. [Google Scholar] [CrossRef]
- Roselló-Díez, E.; Cuerpo, G.; Estévez, F.; Muñoz-Guijosa, C.; Tauron, M.; Cuenca, J.J.; González-Pinto, Á.; Padró, J.M. Use of the Perceval Sutureless Valve in Active Prosthetic Aortic Valve Endocarditis. Ann. Thorac. Surg. 2018, 105, 1168–1174. [Google Scholar] [CrossRef]
- López, J.; Revilla, A.; Vilacosta, I.; Villacorta, E.; González-Juanatey, C.; Gómez, I.; Rollán, M.J.; Román, J.A.S. Definition, clinical profile, microbiological spectrum, and prognostic factors of early-onset prosthetic valve endocarditis. Eur. Heart J. 2007, 28, 760–765. [Google Scholar] [CrossRef]
- Camazon, N.V.; Mateu, L.; Cediel, G.; Escolà-Vergé, L.; Fernández-Hidalgo, N.; Ferrer, M.G.; Rodriguez, M.T.P.; Cuervo, G.; Aragón, R.N.; Llibre, C.; et al. Long-term antibiotic therapy in patients with surgery-indicated not undergoing surgery infective endocarditis. Cardiol. J. 2021, 28, 566–578. [Google Scholar] [CrossRef]
- Elgharably, H.; Unai, S.; Pettersson, G.B. Sutureless Prosthesis for Prosthetic Aortic Valve Endocarditis: Time to Put Brakes on a Speedy Bus? Ann. Thorac. Surg. 2024, 118, 519. [Google Scholar] [CrossRef]
- Elgharably, H.; Sarhan, H.; Poddi, S.; Fraser, T.; Xu, B.; Pettersson, G.B. Sutureless and Trans-catheter Valve Use in Aortic Valve Endocarditis: A Review. Heart Surg. Forum 2025, 28, E452–E461. [Google Scholar] [CrossRef]
- Baran, C.; Kayan, A.; Baran, C.S.; Karacuha, A.F.; Eryilmaz, S. Comparison of Sutureless and Sutured Aortic Valve Replacements in Patients with Redo Infective Endocarditis. Medicina 2024, 60, 2037. [Google Scholar] [CrossRef] [PubMed]
- Dokollari, A.; Ramlawi, B.; Torregrossa, G.; Sá, M.P.; Sicouri, S.; Prifti, E.; Gelsomino, S.; Bonacchi, M. Benefits and Pitfalls of the Perceval Sutureless Bio-prosthesis. Front. Cardiovasc. Med. 2022, 8, 789392. [Google Scholar] [CrossRef] [PubMed]
- Powell, R.; Pelletier, M.P.; Chu, M.W.A.; Bouchard, D.; Melvin, K.N.; Adams, C. The Perceval Sutureless Aortic Valve: Review of Outcomes, Complications, and Future Direction. Innov. Technol. Tech. Cardiothorac. Vasc. Surg. 2017, 12, 155–173. [Google Scholar] [CrossRef] [PubMed]
- Hernandez-Vaquero, D.; Pascual, I.; Diaz, R.; Avanzas, P.; Moris, C.; Silva, J. Surgical Explantation of a Transcatheter-Implanted Aortic Valve Prosthesis Is Feasible and Easy. Ann. Thorac. Surg. 2019, 108, e173–e174. [Google Scholar] [CrossRef] [PubMed]
- Kefer, J.; Vancraeynest, D.; Roelants, V.; Belkhir, L. Valve-in-valve transcatheter aortic valve implantation (TAVI): A new valuable approach to bioprosthetic infective endocarditis? Eur. Heart J. Case Rep. 2024, 8, ytae384. [Google Scholar] [CrossRef]
- Kim, Y.S.; Yoo, J.S. Easy surgical explantation technique for sutureless Perceval S prosthesis, ‘lasso technique’: A case report. J. Cardiothorac. Surg. 2023, 18, 64. [Google Scholar] [CrossRef]
- Explante Quirúrgico de TAVI: Enseñanzas Clave Para una Técnica Segura y Reproducible—Sociedad Española de Cirugía Car-diovascular y Endovascular. Available online: https://secce.es/explante-quirurgico-de-tavi-ensenanzas-clave-para-una-tecnica-segura-y-reproducible/ (accessed on 20 January 2026).
- Ramos-Martínez, A.; Domínguez, F.; Muñoz, P.; Marín, M.; Pedraz, Á.; Fariñas, M.C.; Tascón, V.; de Alarcón, A.; Rodríguez-García, R.; Miró, J.M.; et al. Clinical presentation, microbiology, and prognostic factors of prosthetic valve endocarditis. Lessons learned from a large prospective registry. PLoS ONE 2023, 18, e0290998. [Google Scholar] [CrossRef]
- Cuervo, G.; Quintana, E.; Regueiro, A.; Perissinotti, A.; Vidal, B.; Miro, J.M.; Baddour, L.M. The Clinical Challenge of Prosthetic Valve Endo-carditis. JACC 2024, 83, 1418–1430. [Google Scholar] [CrossRef]
| Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |