Transmission Electron Microscopy Corneal Ultrastructure Study in Hematocornea of Corneal Transplant Graft
Abstract
1. Introduction
1.1. Deep Anterior Lamellar Keratoplasty (DALK)
1.2. DALK and Hematocornea
1.3. Previous Transmission Electron Microscopy (TEM) Studies in Hematocornea
1.4. The Corneal Ultrastructure
2. Materials and Methods
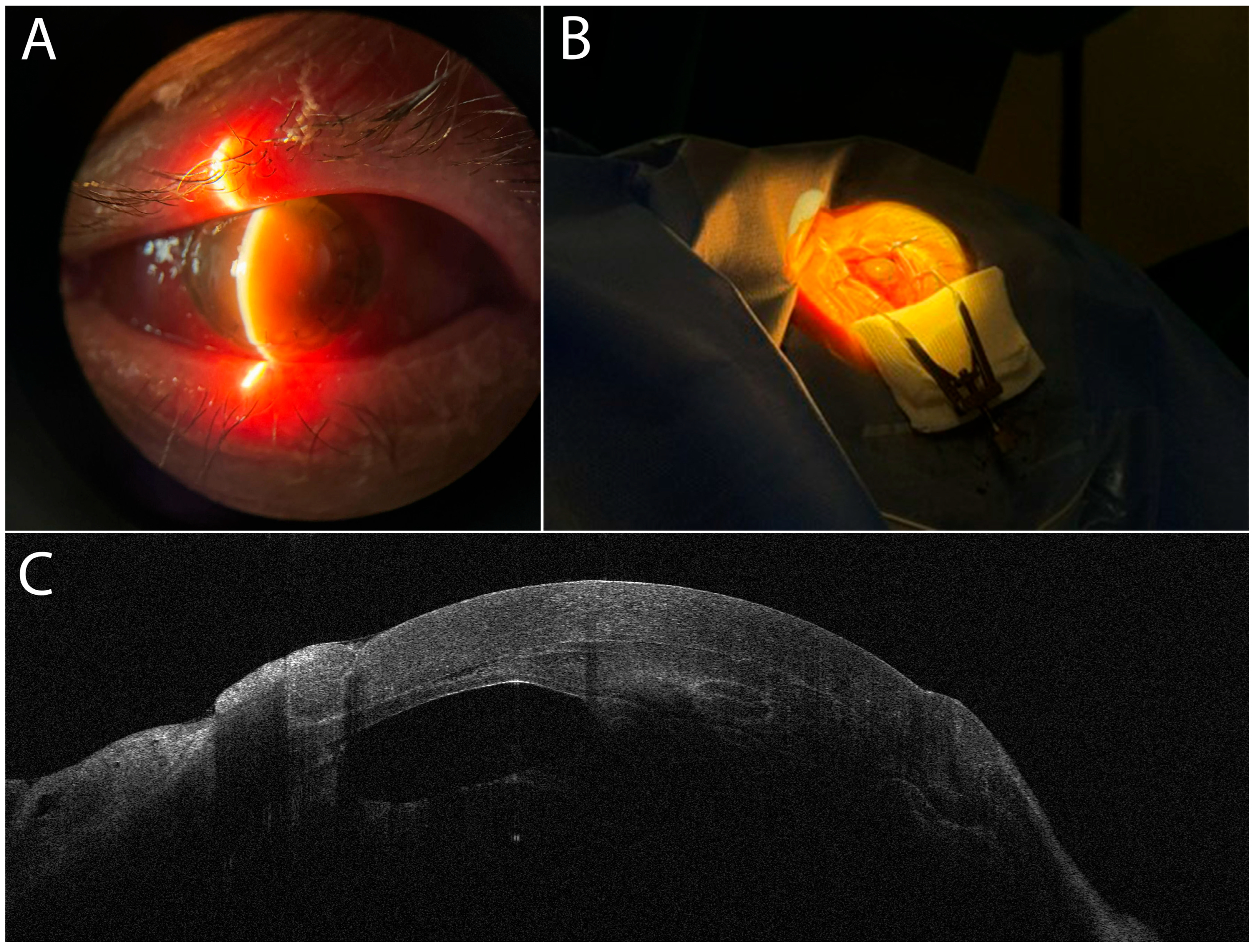
2.1. Clinical Context of Case Presentation
2.2. Sample Preparation and Analysis
3. Results
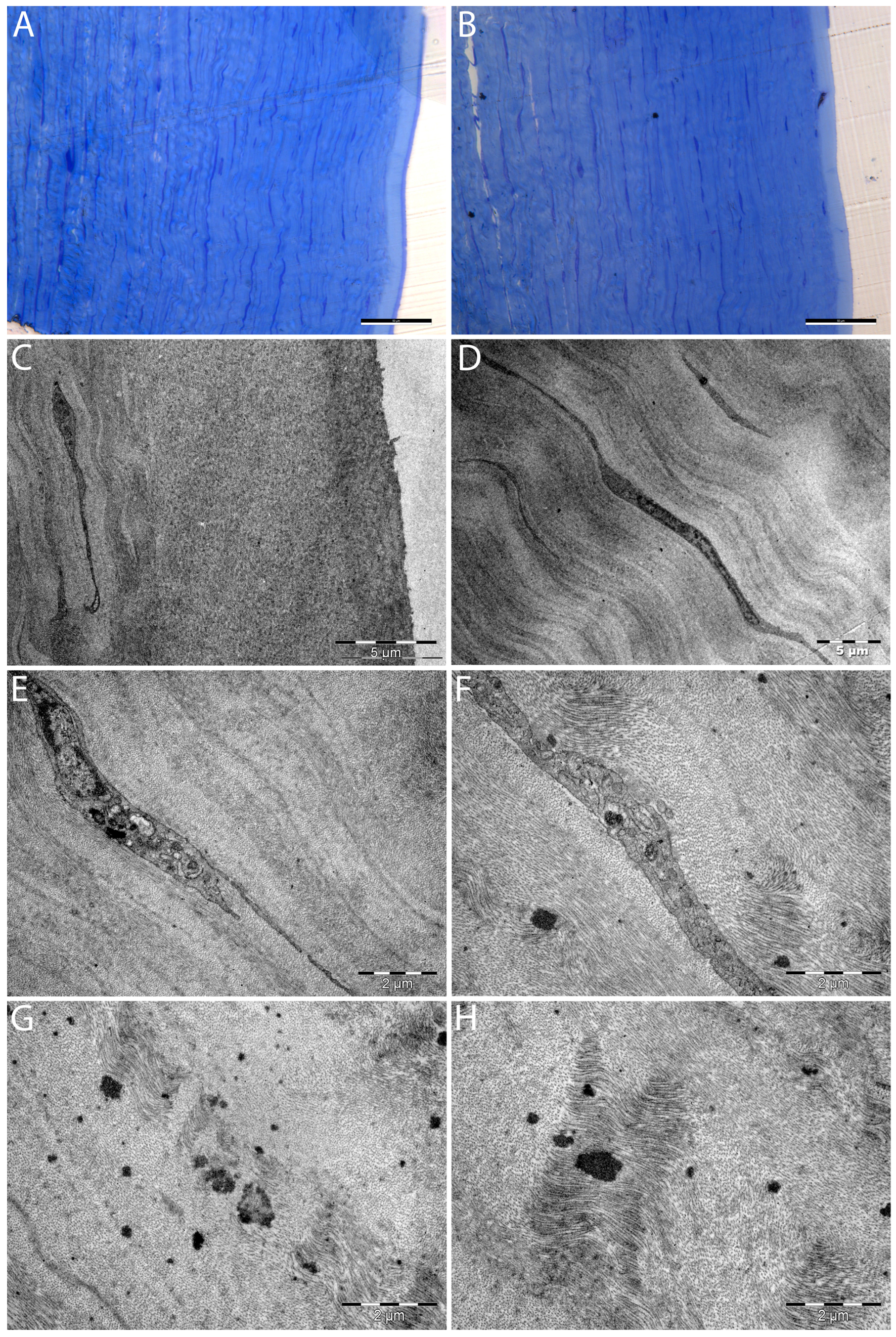
3.1. Corneal Ultrastructure in the Hematocornea DALK Transplant Graft (TEM)
3.2. Cellular Response to Presence of Electron-Dense Hem Material
4. Discussion
4.1. Common Findings with McDonnel et al. [10]
4.2. Common Findings with Yoshimura M. et al. [12]
4.3. Differences from Yoshimura M. et al. [12] and McDonnel et al. [10]
4.4. Further Research on Hemoglobin Clearance and Cellular Adaptation
4.5. Two Cellular Aspects in Hematocornea: Adaptation and Degradation
4.5.1. Cellular Adaptation
4.5.2. Cellular Degradation
4.5.3. Cellular Analysis and Study Limitations
4.6. Alteration of the Collagen Lamellae
4.7. Loss of the DALK Graft
4.8. Clinical Context of the Case
4.8.1. Preoperative Surgical Risk Assessment
4.8.2. Surgical and Postoperative Case Particularities
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| DALK | Deep anterior lamellar keratoplasty |
| TEM | Transmission electron microscopy |
| AS-OCT | Anterior pole optical coherence tomography |
| AC | Anterior chamber |
| mm | Millimeter |
| µm | Micron |
| M | Molar |
| °C | Degree Celsius |
| IVCM | In vivo confocal microscopy |
| LSECs | Liver sinusoidal cells |
| HbS | Hemoglobin S (from sickle cell disease) |
References
- Shams, M.; Sharifi, A.; Akbari, Z.; Maghsoudlou, A.; Reza Tajali, M. Penetrating Keratoplasty versus Deep Anterior Lamellar Keratoplasty for Keratoconus: A Systematic Review and Meta-analysis. J. Ophthalmic Vis. Res. 2022, 17, 89–107. [Google Scholar] [CrossRef]
- Kodavoor, S.K.; Rathi, N.; Dandapani, R. Complications in deep anterior lamellar keratoplasty—A retrospective interventional analysis in a large series. Indian J. Ophthalmol. 2022, 70, 3501–3507. [Google Scholar] [CrossRef]
- Kodavoor, S.K.; Rathi, N.; Dandapani, R. Complications in deep anterior lamellar keratoplasty—A retrospective cross sectional interventional analysis in a large series. Oman J. Ophthalmol. 2023, 16, 23–29. [Google Scholar] [CrossRef] [PubMed]
- Morgan, S.; Mukhija, R.; Nanavaty, M.A. Impact of Intraoperative Descemet Membrane Perforations on Deep Anterior Lamellar Keratoplasty Outcomes. J. Ophthalmol. 2025, 2025, 4101770. [Google Scholar] [CrossRef] [PubMed]
- Borderie, V.M.; Georgeon, C.; Sandali, O.; Bouheraoua, N. Long-term outcomes of deep anterior lamellar versus penetrating keratoplasty for keratoconus. Br. J. Ophthalmol. 2023, 108, 10–16. [Google Scholar] [CrossRef]
- Sarnicola, C.; Sarnicola, E.; Cheung, A.Y.; Sarnicola, V. Deep Anterior Lamellar Keratoplasty: Can All Ruptures Be Fixed? Cornea 2023, 42, 80–88. [Google Scholar] [CrossRef]
- Gheorghe, A.G.; Onofrei, A.G.; Arghirescu, A.M.; Coleașă, A.; Tiran, G.D.; Dinu, L.I.; Toader, E.V. Strategic Management of Descemet’s Membrane Perforation During DALK in Advanced Keratoconus. Rom. J. Ophthalmol. 2024, 68, 448–456. [Google Scholar] [CrossRef] [PubMed]
- Hayashi, T.; Siebelmann, S. Rescue Technique to Solve Postoperative Refractory Double Anterior Chamber in Deep Anterior Lamellar Keratoplasty. Cornea 2022, 41, 374–378. [Google Scholar] [CrossRef]
- Oganesyan, O.G.; Gusak, D.A.; Saakyan, S.V.; Amiryan, A.G.; Milash, S.V.; Oganesyan, C. Recurrence of hematocornea with intraocular neoplasm. Am. J. Ophthalmol. Case Rep. 2025, 39, 102372. [Google Scholar] [CrossRef]
- McDonnell, P.J.; Green, W.R.; Stevens, R.E.; Bargeron, C.B.; Riquelme, J.L. Blood staining of the cornea. Light microscopic and ultrastructural features. Ophthalmology 1985, 92, 1668–1674. [Google Scholar] [CrossRef]
- Gottsch, J.D.; Messmer, E.P.; McNair, D.S.; Font, R.L. Corneal blood staining. An animal model. Ophthalmology 1986, 93, 797–802. [Google Scholar] [CrossRef] [PubMed]
- Yoshimura, M.; Sameshima, M.; Fujita, S.; Ohba, N. Blood staining of the cornea in Hansen’s disease. A light- and electron-microscopic study. Ophthalmologica 1980, 181, 314–319. [Google Scholar] [CrossRef] [PubMed]
- Messmer, E.P.; Gottsch, J.; Font, R.L. Blood staining of the cornea: A histopathologic analysis of 16 cases. Cornea 1984, 3, 205–212. [Google Scholar] [PubMed]
- Meek, K.M.; Knupp, C.; Lewis, P.N.; Morgan, S.R.; Hayes, S. Structural control of corneal transparency, refractive power and dynamics. Eye 2025, 39, 644–650. [Google Scholar] [CrossRef]
- Meek, K.M.; Quantock, A.J. The use of X-Ray scattering techniques to determine corneal ultrastructure. Prog. Retin Eye Res. 2001, 20, 95–137. [Google Scholar] [CrossRef] [PubMed]
- Fullwood, N.J.; Meek, K.M. A synchroton X-ray study of the changes occurring in the corneal stroma during processing for electron microscopy. J. Microsc. 1993, 169, 53–60. [Google Scholar] [CrossRef] [PubMed]
- Akhtar, S. Effect of processing methods for transmission electron microscopy on corneal collagen fibrils diameter and spacing. Microsc. Res. Tech. 2012, 75, 1420–1424. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.S.; Wang, H.; White, J.B.R.; Degtjarik, O.; Huynh, C.; Brannstrom, K.; Horn, M.T.; Muench, S.P.; Somers, W.S.; Chaparro-Riggers, J.; et al. Structural elucidation of the haptoglobin-hemoglobin clearance mechanism by macrophage scavenger receptor CD163. PLoS Biol. 2025, 23, e3003264. [Google Scholar] [CrossRef]
- Schaer, D.J.; Alayash, A.I. Clearance and control mechanisms of hemoglobin from cradle to grave. Antioxid. Redox Signal. 2010, 12, 181–184. [Google Scholar] [CrossRef]
- Zurawska, G.; Sas, Z.; Jończy, A.; Mahadeva, R.; Slusarczyk, P.; Chwałek, M.; Seehofer, D.; Damm, G.; Mazgaj, R.; Skórzyński, M.; et al. Liver sinusoidal endothelial cells constitute a major route for hemoglobin clearance. EMBO Rep. 2026, 27, 598–628. [Google Scholar] [CrossRef]
- Pal-Ghosh, S.; Datta-Majumdar, H.; Datta, S.; Dimri, S.; Hally, J.; Wehmeyer, H.; Chen, Z.; Watsky, M.; Ma, J.X.; Liang, W.; et al. Corneal epithelial cells upregulate macropinocytosis to engulf metabolically active axonal mitochondria released by injured axons. Ocul. Surf. 2025, 37, 173–188. [Google Scholar] [CrossRef]
- Peng, H.; Park, J.K.; Lavker, R.M. Eyeing autophagy and macropinocytosis in the corneal/limbal epithelia. Autophagy 2017, 13, 975–977. [Google Scholar] [CrossRef]
- Poole, K.; Iyer, K.S.; Schmidtke, D.W.; Petroll, W.M.; Varner, V.D. Corneal Keratocytes, Fibroblasts, and Myofibroblasts Exhibit Distinct Transcriptional Profiles In Vitro. Investig. Ophthalmol. Vis. Sci. 2025, 66, 28. [Google Scholar] [CrossRef]
- Acosta, A.C.; Joud, H.; Sun, M.; Avila, M.Y.; Margo, C.E.; Espana, E.M. Keratocyte-Derived Myofibroblasts: Functional Differences with Their Fibroblast Precursors. Investig. Ophthalmol. Vis. Sci. 2023, 64, 9. [Google Scholar] [CrossRef]
- Sato, T.; Sugioka, K.; Kodama-Takahashi, A.; Murakami, J.; Saito, A.; Mishima, H.; Nishida, T.; Kusaka, S. Stimulation of Phagocytic Activity in Cultured Human Corneal Fibroblasts by Plasminogen. Investig. Ophthalmol. Vis. Sci. 2019, 60, 4205–4214. [Google Scholar] [CrossRef]
- Klintworth, G.K. Experimental studies on the phagocytic capability of the corneal fibroblast. Am. J. Pathol. 1969, 55, 283–294. [Google Scholar]
- Lande, M.A.; Birk, D.E.; Nagpal, M.L.; Rader, R.L. Phagocytic properties of human keratocyte cultures. Investig. Ophthalmol. Vis. Sci. 1981, 20, 481–489. [Google Scholar]
- Tripathi, R.C.; Tripathi, B.J. Human trabecular endothelium, corneal endothelium, keratocytes, and scleral fibroblasts in primary cell culture. A comparative study of growth characteristics, morphology, and phagocytic activity by light and scanning electron microscopy. Exp. Eye Res. 1982, 35, 611–624. [Google Scholar] [CrossRef]
- Chakravarti, S.; Wu, F.; Vij, N.; Roberts, L.; Joyce, S. Microarray studies reveal macrophage-like function of stromal keratocytes in the cornea. Investig. Ophthalmol. Vis. Sci. 2004, 45, 3475–3484. [Google Scholar] [CrossRef][Green Version]
- Alió Del Barrio, J.L.; Eldanasoury, A.M.; Arbelaez, J.; Faini, S.; Versaci, F. Artificial Neural Network for Automated Keratoconus Detection Using a Combined Placido Disc and Anterior Segment Optical Coherence Tomography Topographer. Transl. Vis. Sci. Technol. 2024, 13, 13. [Google Scholar] [CrossRef]
- Yu, A.C.; Sollazzo, A.; Bovone, C.; Busin, M. Large-diameter deep anterior lamellar keratoplasty: A narrative review. Taiwan J. Ophthalmol. 2024, 14, 27–33. [Google Scholar] [CrossRef]
- Janiszewska-Bil, D.; Kijonka, M.; Kokot-Lesiuk, J.; Derhartunian, V.; Lyssek-Boroń, A.; Dobrowolski, D.; Wylęgała, E.; Grabarek, B.O.; Krysik, K. Enhancing Safety in Refractive Surgery: A Pilot Evaluation of In Vivo Confocal Microscopy. J. Clin. Med. 2025, 14, 7714. [Google Scholar] [CrossRef]
- Gheorghe, A.G.; Arghirescu, A.M.; Marinescu, M.C.; Onofrei, A.G.; Pop, D.M.; Voinea, L.M.; Ciuluvică, R.C. In Vivo Confocal Microscopy and Anterior Segment Optical Coherence Tomography in Optimizing Diagnosis and Therapeutic Management in Fungal Keratitis: Case Reports and Literature Review. J. Clin. Med. 2025, 14, 8066. [Google Scholar] [CrossRef]
- Yeh, S.I.; Liu, T.S.; Ho, C.C.; Cheng, H.C. In vivo confocal microscopy of combined pre-descemet membrane corneal dystrophy and fuchs endothelial dystrophy. Cornea 2011, 30, 222–224. [Google Scholar] [CrossRef]
- Scorcia, V.; Giannaccare, G.; Pellegrini, M.; Camposampiero, D.; Ponzin, D.; Yu, A.C.; Busin, M. Stromal peeling for deep anterior lamellar keratoplasty in a post-penetrating keratoplasty eye with hematocornea. Am. J. Ophthalmol. Case Rep. 2023, 29, 101808. [Google Scholar] [CrossRef]
- Garner, A. Factors Involved in Post-traumatic Ocular Neovascularization. In Wundheilung des Auges und Ihre Komplikationen/Wound Healing of the Eye and Its Complications; Symposien der Deutschen Ophthalmologischen Gesellschaft; J.F. Bergmann-Verlag: Munich, Germany, 1980. [Google Scholar] [CrossRef]



| Protocol | Specifications |
|---|---|
| Sample fixation using glutaraldehyde 4% | 4–12 h |
| Resample with sodium cacodylate 0.1 M 1 | 4 °C, 2 × 10 min |
| Post-fixation with osmium tetroxide 0.1 M in sodium cacodylate 0.2 M | 4 °C, 30 min |
| Sample suspension in osmium tetroxide 0.1 M | 4 °C, 2 × 10 min |
| Dehydration in ethylic alcohol 30° | 4 °C, 15 min |
| Dehydration in ethylic alcohol 50° | 4 °C, 15 min |
| Dehydration in ethylic alcohol 70° | 4 °C, 15 min |
| Dehydration in ethylic alcohol 90° | 4 °C, 15 min |
| Dehydration in ethylic alcohol 96° | 4 °C, 15 min |
| Dehydration in ethylic alcohol 100° | Room temperature, 2 × 15 min |
| Propylene oxide | Room temperature, 3 × 10 min |
| Bath I: propylene oxide/epoxy resin (2/1) | Room temperature, 2 h |
| Bath II: propylene oxide/epoxy resin (1/2) | Room temperature, overnight |
| Bath III: epoxy resin (Agar 100) | Room temperature, 3 h |
| Epoxy resin polymerization | 60 °C, 48 h |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Curcă, P.F.; Macovei, L.; Mușat, O.; Zemba, M.; Dinu, V.; Gherghiceanu, M.; Tătaru, C.I.; Tătaru, C.P. Transmission Electron Microscopy Corneal Ultrastructure Study in Hematocornea of Corneal Transplant Graft. Diagnostics 2026, 16, 890. https://doi.org/10.3390/diagnostics16060890
Curcă PF, Macovei L, Mușat O, Zemba M, Dinu V, Gherghiceanu M, Tătaru CI, Tătaru CP. Transmission Electron Microscopy Corneal Ultrastructure Study in Hematocornea of Corneal Transplant Graft. Diagnostics. 2026; 16(6):890. https://doi.org/10.3390/diagnostics16060890
Chicago/Turabian StyleCurcă, Paul Filip, Laura Macovei, Ovidiu Mușat, Mihail Zemba, Valentin Dinu, Mihaela Gherghiceanu, Cătălina Ioana Tătaru, and Călin Petru Tătaru. 2026. "Transmission Electron Microscopy Corneal Ultrastructure Study in Hematocornea of Corneal Transplant Graft" Diagnostics 16, no. 6: 890. https://doi.org/10.3390/diagnostics16060890
APA StyleCurcă, P. F., Macovei, L., Mușat, O., Zemba, M., Dinu, V., Gherghiceanu, M., Tătaru, C. I., & Tătaru, C. P. (2026). Transmission Electron Microscopy Corneal Ultrastructure Study in Hematocornea of Corneal Transplant Graft. Diagnostics, 16(6), 890. https://doi.org/10.3390/diagnostics16060890







