Integration of Photon-Counting CT into the Surgical Workflow of Complex Maxillofacial Reconstruction: A Pilot Feasibility Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design and Patient Selection
2.2. Study Endpoints
- (1)
- Successful transfer of PCCT data into the CAD/CAM software platforms (CMX Portal, version 2.6.1158, Medartis AG, Basel, Switzerland; or ProPlan CMF, version 5.7.8.025, Materialise NV, Leuven, Belgium);
- (2)
- Adequacy of tumor-related osseous delineation for surgical margin planning;
- (3)
- Reliability of donor-site vascular assessment within the same imaging session.
2.3. Imaging Protocol (Photon-Counting CT)
2.4. Image Reconstruction and Metal Artifact Reduction (MAR)
2.5. Virtual Surgical Planning (VSP) and CAD/CAM Integration
- Diagnostic Precision: The ability to delineate tumor margins and medullary invasion.
- Workflow Integration: The seamless transfer of PCCT datasets into the CAD/CAM planning software (CMX Portal, version 2.6.1158, Medartis AG, Basel, Switzerland; or ProPlan CMF, version 5.7.8.025, Materialise NV, Leuven, Belgium) for the fabrication of 3D-printed cutting guides and patient-specific implants (PSI).
- Intraoperative Correlation: The correlation between the PCCT-based virtual plan and the actual surgical findings.
2.6. Surgical Procedure
2.7. Statistical Analysis
3. Results
3.1. Diagnostic Performance and Surgical Decision-Making
3.2. Integration into Virtual Planning Workflow
3.3. Vascular Assessment and Donor Site Planning
3.4. Surgical and Oncologic Outcomes
3.5. Observations on Workflow Feasibility and Subjective Image Quality
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| CT | Computed Tomography |
| VSP | Virtual surgical planning |
| CAD/CAM | Computer-Aided Design and Computer-Aided Manufacturing |
| PCCT | Photon-Counting CT |
| ORN | Osteoradionecrosis |
| FFF | Fibula free flap |
| DCIA | Deep circumflex iliac artery |
| ALT | Anterolateral Thigh |
| SCIP | Superficial Circumflex Iliac Perforator |
| MRI | Magnetic Resonance Imaging |
| PET/CT | Positron Emission Tomography/Computed Tomography |
| PCD | Photon-Counting Detectors |
| EID | Energy Integrating Detectors |
| MAR | Metal artifact reduction |
| CdTe | Cadmium telluride |
| QIR | Quantum Iterative Reconstruction |
| VMI | Virtual monoenergetic images |
| DICOM | Digital Imaging and Communications in Medicine |
| PSI | Patient-specific implants |
| CNR | Contrast-To-Noise Ratio |
| SNR | Signal-to-noise ratio |
| OCSCC | Oral Cavity Squamous Cell Carcinoma |
| MEC | Mucoepidermoid carcinoma |
| Lt | Left |
| Rt | Right |
| A. poplitea | popliteal artery |
| ATA | anterior tibial artery |
| PTA | posterior tibial artery |
| TPT | tibioperoneal trunk |
| AF | fibular artery |
| CNN | convolutional neural network |
| MDCT | Multi-Detector CT |
| HR-pQCT | high-resolution peripheral quantitative computed tomography |
| iMAR | iterative metal artifact reduction |
| CBCT | Cone Beam CT |
| vBMD | volumetric bone mineral density |
| UHR | ultra-high-resolution |
References
- Likhterov, Ι.; Roche, A.; Urken, M. Contemporary Osseous Reconstruction of the Mandible and the Maxilla. Oral. Maxillofac. Surg. Clin. N. Am. 2019, 31, 101–116. [Google Scholar] [CrossRef]
- Pu, J.J.; Hakim, S.G.; Melville, J.C.; Su, Y.-X. Current Trends in the Reconstruction and Rehabilitation of Jaw following Ablative Surgery. Cancers 2022, 14, 3308. [Google Scholar] [CrossRef]
- Wilkman, T.; Husso, A.; Lassus, P. Clinical Comparison of Scapular, Fibular, and Iliac Crest Osseal Free Flaps in Maxillofacial Reconstructions. Scand. J. Surg. 2019, 108, 76–82. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Zhang, S.; Zou, J.; Jiang, T.; Chen, L.; Gong, Z. Reconstruction of Complex Oral and Maxillofacial Defects After Oral Cancer Resection With Individualized Anterolateral Thigh Multiple Paddle Flaps. J. Craniofac. Surg. 2025, 36, 241–246. [Google Scholar] [CrossRef]
- Zubler, C.; Lese, I.; Pastor, T.; Attinger, M.; Constantinescu, M.A.; Olariu, R. The osteocutaneous SCIP flap: A detailed description of the surgical technique and retrospective cohort study of consecutive cases in a tertiary European centre. J. Plast. Reconstr. Aesthet. Surg. 2023, 77, 21–30. [Google Scholar] [CrossRef] [PubMed]
- Yadav, S.K.; Shrestha, S. Microvascular Free Flaps in Oral and Maxillofacial Reconstruction following Cancer Ablation. J. Nepal Health Res. Counc. 2017, 15, 88–95. [Google Scholar] [CrossRef]
- Ritschl, L.M.; Mücke, T.; Hart, D.; Unterhuber, T.; Kehl, V.; Wolff, K.D.; Fichter, A.M. Retrospective analysis of complications in 190 mandibular resections and simultaneous reconstructions with free fibula flap, iliac crest flap or reconstruction plate: A comparative single centre study. Clin. Oral Investig. 2021, 25, 2905–2914. [Google Scholar] [CrossRef]
- Mashrah, M.A.; Aldhohrah, T.; Abdelrehem, A.; Sakran, K.A.; Ahmad, H.; Mahran, H.; Abu-Lohom, F.; Su, H.; Fang, Y.; Wang, L. Survival of vascularized osseous flaps in mandibular reconstruction: A network meta-analysis. PLoS ONE 2021, 16, e0257457. [Google Scholar] [CrossRef] [PubMed]
- Butler, D.P.; Dunne, J.A.; Wood, S.H.; Jallali, N. A Unifying Algorithm in Microvascular Reconstruction of Oral Cavity Defects Using the Trilaminar Concept. Plast. Reconstr. Surg. Glob. Open 2019, 7, e2267. [Google Scholar] [CrossRef]
- Cao, C.; Gan, X.; He, Y.; Su, Y.; Liu, Z.; Hu, X.; Peng, X. Diagnostic efficacy of PET-CT, CT, and MRI in preoperative assessment of mandibular invasion caused by head and neck cancer: A systematic review and meta-analysis. Oral. Oncol. 2021, 116, 105264. [Google Scholar] [CrossRef]
- Wolff, K.D.; Follmann, M.; Nast, A. The diagnosis and treatment of oral cavity cancer. Dtsches Arztebl. Int. 2012, 109, 829–835. [Google Scholar] [CrossRef]
- Brockenbrough, J.M.; Petruzzelli, G.J.; Lomasney, L. DentaScan as an accurate method of predicting mandibular invasion in patients with squamous cell carcinoma of the oral cavity. Arch. Otolaryngol. Head Neck Surg. 2003, 129, 113–117. [Google Scholar] [CrossRef]
- Rao, D.; Weyh, A.; Bunnell, A.; Hernandez, M. The Role of Imaging in Mandibular Reconstruction with Microvascular Surgery. Oral Maxillofac. Surg. Clin. N. Am. 2023, 35, 327–344. [Google Scholar] [CrossRef]
- Layer, Y.C.; Kravchenko, D.; Dell, T.; Kütting, D. [CT technology: Photon-counting detector computed tomography]. Radiologie 2023, 63, 497–506. [Google Scholar] [CrossRef]
- Gil, R.S.; Roig, A.M.; Obispo, C.A.; Morla, A.; Pagès, C.M.; Perez, J.L. Surgical planning and microvascular reconstruction of the mandible with a fibular flap using computer-aided design, rapid prototype modelling, and precontoured titanium reconstruction plates: A prospective study. Br. J. Oral Maxillofac. Surg. 2015, 53, 49–53. [Google Scholar] [CrossRef]
- Weitz, J.; Bauer, F.J.M.; Hapfelmeier, A.; Rohleder, N.H.; Wolff, K.D.; Kesting, M.R. Accuracy of mandibular reconstruction by three-dimensional guided vascularised fibular free flap after segmental mandibulectomy. Br. J. Oral Maxillofac. Surg. 2016, 54, 506–510. [Google Scholar] [CrossRef] [PubMed]
- Kaariainen, M.; Kuuskeri, M.; Gremoutis, G.; Kuokkanen, H.; Miettinen, A.; Laranne, J. Utilization of Three-Dimensional Computer-Aided Preoperative Virtual Planning and Manufacturing in Maxillary and Mandibular Reconstruction with a Microvascular Fibula Flap. J. Reconstr. Microsurg. 2016, 32, 137–141. [Google Scholar] [CrossRef] [PubMed]
- Muñoz, N.M.H.; Zapata, M.R.M.; Guerrero, D.D.S.; Ortega, E.F.M.; Felicita, K.L.L.; Cedillo, J.C.; Molina, N.S.O.; Guerrero, G.X.G. Orbital Defect Reconstruction using Customized Implants: Current Techniques and Long-Term Outcomes in Maxillofacial Surgery. Int. J. Med. Sci. Dent. Health 2024, 10, 74–85. [Google Scholar] [CrossRef]
- Greffier, J.; Viry, A.; Robert, A.; Khorsi, M.; Si-Mohamed, S. Photon-counting CT systems: A technical review of current clinical possibilities. Diagn. Interv. Imaging 2025, 106, 53–59. [Google Scholar] [CrossRef]
- Algin, O.; Tokgoz, N.; Cademartiri, F. Photon-counting computed tomography in radiology. Pol. J. Radiol. 2024, 89, e433–e442. [Google Scholar] [CrossRef] [PubMed]
- Bette, S.; Risch, F.; Becker, J.; Popp, D.; Decker, J.A.; Kaufmann, D.; Friedrich, L.; Scheurig-Münkler, C.; Schwarz, F.; Kröncke, T.J. Photon-counting detector CT—First experiences in the field of musculoskeletal radiology. Rofo 2025, 197, 34–43. [Google Scholar] [CrossRef]
- Gascho, D. Photon-counting CT for forensic death investigations-a glance into the future of virtual autopsy. Front. Radiol. 2024, 16, 1463236. [Google Scholar] [CrossRef]
- Chang, S.; Benson, J.C.; Lane, J.I.; Bruesewitz, M.R.; Swicklik, J.R.; Thorne, J.E.; Koons, E.K.; Carlson, M.L.; McCollough, C.H.; Leng, S. Ultra-High-Resolution Photon-Counting-Detector CT with a Dedicated Denoising Convolutional Neural Network for Enhanced Temporal Bone Imaging. AJNR Am. J. Neuroradiol. 2025, 46, 1188–1195. [Google Scholar] [CrossRef] [PubMed]
- Azari, F.; Uniyal, P.; Soete, J.; Coudyzer, W.; Wyers, C.E.; Quintiens, J.; van den Bergh, J.P.; van Lenthe, G.H. Accuracy of photon-counting computed tomography for the measurement of bone quality in the knee. Bone 2024, 117027. [Google Scholar] [CrossRef]
- Hermans, R.; Boomgaert, L.; Cockmartin, L.; Binst, J.; De Stefanis, R.; Bosmans, H. Photon-counting CT allows better visualization of temporal bone structures in comparison with current generation multi-detector CT. Insights Imaging 2023, 14, 112. [Google Scholar] [CrossRef] [PubMed]
- Quintiens, J.; Coudyzer, W.; Bevers, M.; Vereecke, E.; van den Bergh, J.P.; Manske, S.L.; van Lenthe, G.H. The quantification of bone mineral density using photon counting computed tomography and its implications for detecting bone remodeling. J. Bone Miner. Res. 2024, 39, 1774–1782. [Google Scholar] [CrossRef]
- Shi, G.; Sivakumar, A.; Abadi, E.; McCabe, C.; Quevedo-González, F.; Breighner, R.; Manske, S.L.; van Lenthe, G.H. Quantitative trabecular bone morphometry using photon-counting CT. In Medical Imaging 2024: Clinical and Biomedical Imaging; Gimi, B.S., Krol, A., Eds.; SPIE: San Diego, CA, USA, 2024; Volume 12930, pp. 16–19. [Google Scholar] [CrossRef]
- Vanden Broeke, L.; Grillon, M.; Yeung, A.W.K.; Wu, W.; Tanaka, R.; Vardhanabhuti, V. Feasibility of photon-counting spectral CT in dental applications-a comparative qualitative analysis. BDJ Open 2021, 7, 4. [Google Scholar] [CrossRef]
- Pallasch, F.B.; Rau, A.; Reisert, M.; Rau, S.; Diallo, T.; Stein, T.; Faby, S.; Bamberg, F.; Weiss, J. Impact of different metal artifact reduction techniques in photon-counting computed tomography head and neck scans in patients with dental hardware. Eur. Radiol. 2024, 34, 3742–3749. [Google Scholar] [CrossRef]
- Sawall, S.; Maier, J.; Sen, S.; Gehrig, H.; Kim, T.S.; Schlemmer, H.P.; Schönberg, S.O.; Kachelrieß, M.; Rütters, M. Dental imaging in clinical photon-counting CT at a quarter of DVT dose. J. Dent. 2024, 142, 104859. [Google Scholar] [CrossRef]
- Klintström, E.; Ly, A.; Sandborg, M.; Woisetschläger, M.; Tesselaar, E. Image quality of photon-counting detector CT for visualization of maxillofacial anatomy in comparison with energy-integrating detector CT and intraoperative C-arm CBCT. Eur. J. Radiol. 2024, 181, 111785. [Google Scholar] [CrossRef]
- Rak, K.; Spahn, B.; Müller-Graff, F.-T.; Engert, J.; Voelker, J.; Hackenberg, S.; Hagen, R.; Petritsch, B.; Grunz, J.-P.; Bley, T. The Photon-Counting CT Enters the Field of Cochlear Implantation: Comparison to Angiography DynaCT and Conventional Multislice CT. Otol. Neurotol. 2024, 45, 662–670. [Google Scholar] [CrossRef] [PubMed]
- Doyle, N.S.; Benson, J.C.; Carr, C.M.; Diehn, F.E.; Carlson, M.L.; Leng, S.; Lane, J.I. Photon Counting Versus Energy-integrated Detector CT in Detection of Superior Semicircular Canal Dehiscence. Clin. Neuroradiol. 2024, 34, 251–255. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, Y.; Higaki, F.; Sugaya, A.; Asano, Y.; Kojima, K.; Morimitsu, Y.; Akagi, N.; Itoh, T.; Matsui, Y.; Hiraki, T. Evaluation of the ear ossicles with photon-counting detector CT. Jpn J. Radiol. 2024, 42, 158–164. [Google Scholar] [CrossRef]
- Rajendran, K.; Voss, B.A.; Zhou, W.; Tao, S.; Delone, D.R.; Lane, J.I.; Weaver, J.M.; Carlson, M.L.; Fletcher, J.G.; McCollough, C.H.; et al. Dose Reduction for Sinus and Temporal Bone Imaging Using Photon-Counting Detector CT With an Additional Tin Filter. Investig. Radiol. 2020, 55, 91–100. [Google Scholar] [CrossRef] [PubMed]
- Khanungwanitkul, K.; Sliwicka, O.; Schwartz, F.R. Abdominal Applications of Photon-Counting CT. Br. J. Radiol. 2024, 98, 1854–1858. [Google Scholar] [CrossRef]
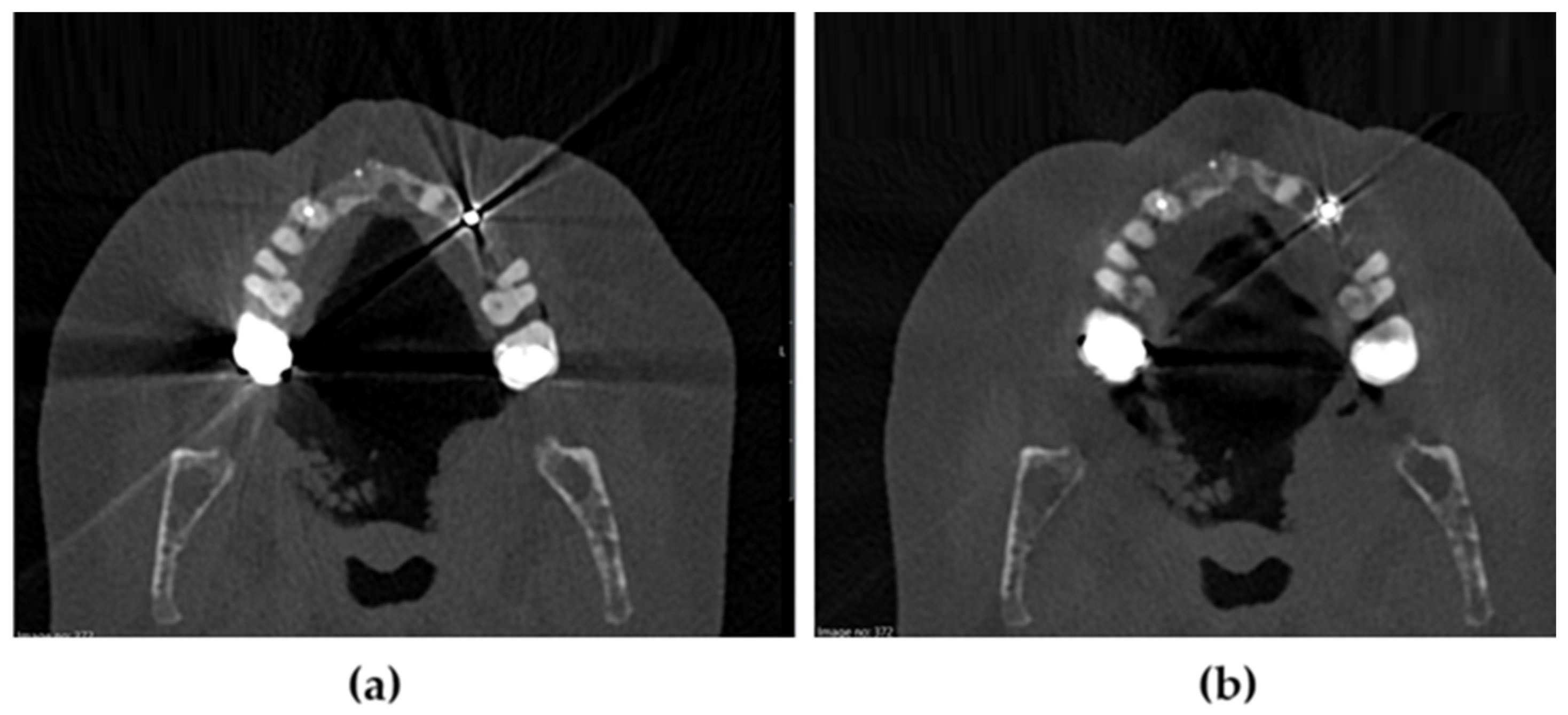
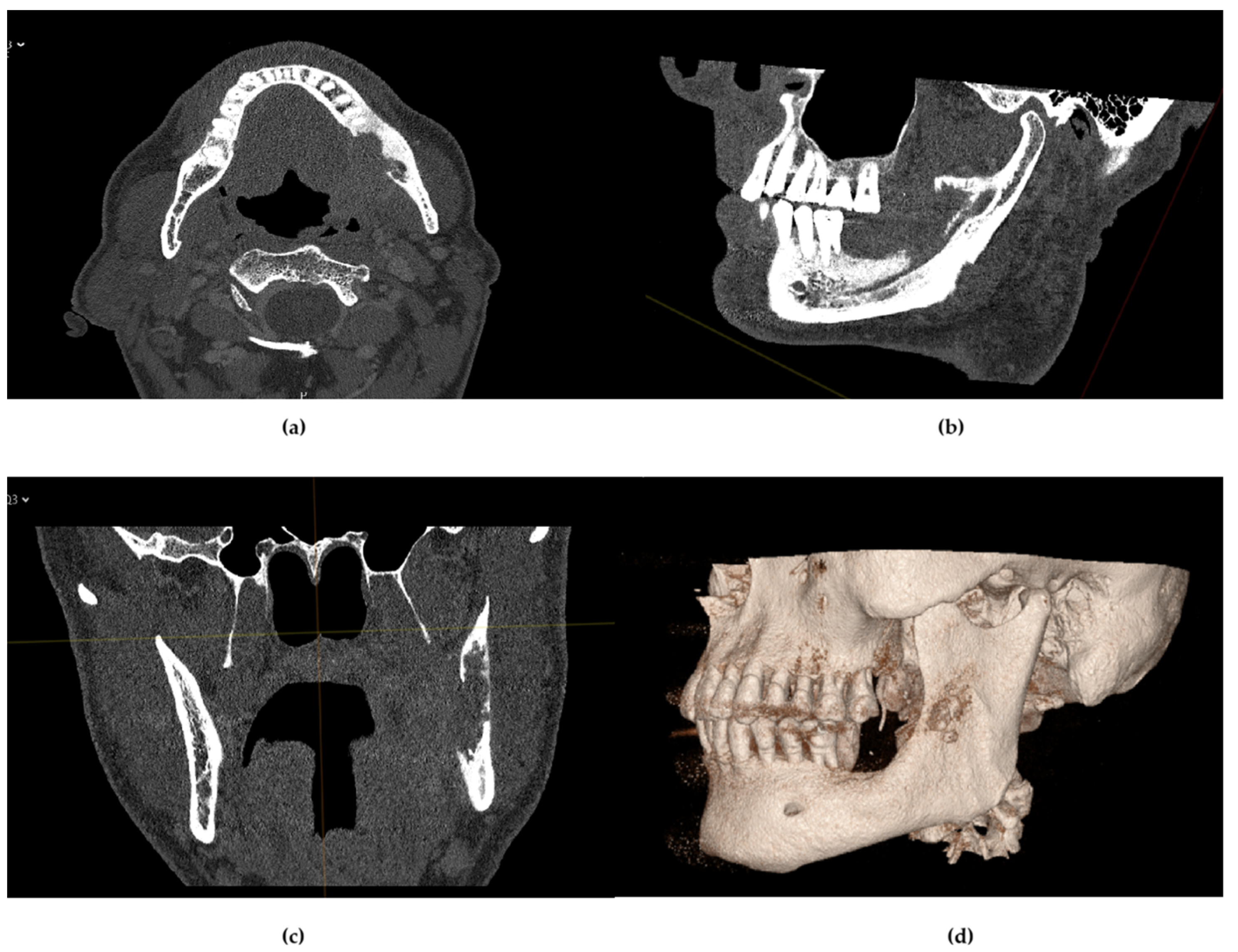


| Parameter | Value |
|---|---|
| Scanner | NAEOTOM Alpha (Siemens Healthineers, Forchheim, Germany) |
| Detector Type | Cadmium Telluride (CdTe) PCD |
| Tube Voltage | 120 kV |
| Tube Current Modulation | CARE Dose4D (automated) |
| Slice Thickness | 0.2–0.4 mm |
| Reconstruction Increment | 0.1–0.2 mm |
| Kernels | Br64 (bone), Br40 (soft tissue) |
| Contrast Agent | Iopromide (Ultravist 370; 100 mL @ 4 mL/s) |
| Saline Bolus | 30 mL post-contrast |
| Angiography Protocol | Biphasic (arterial + venous phase) |
| Case | Age/Sex | Diagnosis & Site | Reconstruction (Flap) | Reconstruction Goal | Outcome | Specific PCCT Contribution |
|---|---|---|---|---|---|---|
| 1 | 68/M | SCC, Floor of Mouth | FFF | Segmental Resection | R0/ Success | Detected medullary invasion (occult on MRI); Upstaged from cT3 to cT4a. |
| 2 | 59/F | SCC, Maxilla | FFF | Brown IIb Maxillectomy | R0/ Success | High-resolution delineation of residual bone for PSI fit. |
| 3 | 56/M | SCC, Retromolar | FFF | Hemimandibulectomy | R0/ Success | Defined cortical & medullary extension for safe margins. |
| 4 | 67/M | Recurrent SCC, Mandible | FFF (2-seg) | Subtotal Mandibulectomy | R0/ Success | Detected vessel occlusion (Rt leg); Guided switch to Lt leg. |
| 5 | 59/F | SCC, Floor of Mouth | FFF (2-seg) + ALT flap | Mandibulectomy | R0/ Success | Defined cortical & medullary extension for safe margins. |
| 6 | 77/F | SCC, Floor of Mouth & Mandible | FFF (2-seg) | Mandibulectomy | R0/ Success | Defined cortical & medullary extension for safe margins. |
| 7 | 65/M | Tumor persistence SCC, Mandible | FFF | Mandibulectomy | R0/ Success | High-resolution imaging enabled differentiation between postoperative changes and tumor persistence |
| 8 | 68/M | SCC, Maxilla | FFF | Brown IIb Maxillectomy | R0/ Success | High-resolution delineation of residual bone for PSI fit. |
| 9 | 49/M | Tumor persistence/progression of mucoepidermoid carcinoma (MEC), Floor of Mouth | FFF (2-seg) | Mandibulectomy | R0/ Success | Detected medullary invasion (occult on MRI); Upstaged from cT3 to cT4a. |
| 10 | 63/M | SCC, Maxilla | Osteocutaneous SCIP flap | Brown IIb Maxillectomy | R0/ Success | High-resolution delineation of residual bone for PSI fit. |
| Metric | Value/Observation |
|---|---|
| Number of patients | 10 |
| Indications | SCC (n = 9), MEC (n = 1) |
| Mean Age | 63.1 years (range 49–77) |
| Segmentation Method | Semi-automated + manual refinement |
| Segmentation/Planning Software | CMX Portal Medartis, ProPlan CMF |
| Virtual Surgical Planning Compatibility | 100% (no DICOM transfer failures) |
| Flap Type | FFF (n = 9), SCIP (n = 1) |
| Intraoperative Fit of Guides | Excellent; no modifications required |
| Resection Margins (oncologic cases) | R0 achieved in 10/10 |
| Flap Survival | 100% |
| Postoperative Complications | None |
| Follow-up Duration | 12–16 months |
| Local Recurrence | None |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Kalaitsidou, I.; Maissen, M.; Dammann, F.; Schedeit, C.; Toneatti, D.J.; Schaller, B. Integration of Photon-Counting CT into the Surgical Workflow of Complex Maxillofacial Reconstruction: A Pilot Feasibility Study. Diagnostics 2026, 16, 876. https://doi.org/10.3390/diagnostics16060876
Kalaitsidou I, Maissen M, Dammann F, Schedeit C, Toneatti DJ, Schaller B. Integration of Photon-Counting CT into the Surgical Workflow of Complex Maxillofacial Reconstruction: A Pilot Feasibility Study. Diagnostics. 2026; 16(6):876. https://doi.org/10.3390/diagnostics16060876
Chicago/Turabian StyleKalaitsidou, Ioanna, Matias Maissen, Florian Dammann, Christian Schedeit, Daniel Jan Toneatti, and Benoît Schaller. 2026. "Integration of Photon-Counting CT into the Surgical Workflow of Complex Maxillofacial Reconstruction: A Pilot Feasibility Study" Diagnostics 16, no. 6: 876. https://doi.org/10.3390/diagnostics16060876
APA StyleKalaitsidou, I., Maissen, M., Dammann, F., Schedeit, C., Toneatti, D. J., & Schaller, B. (2026). Integration of Photon-Counting CT into the Surgical Workflow of Complex Maxillofacial Reconstruction: A Pilot Feasibility Study. Diagnostics, 16(6), 876. https://doi.org/10.3390/diagnostics16060876









