Thermoacoustic Ultrasound Assessment of Liver Steatosis—A Novel Approach for MASLD Diagnosis
Abstract
1. Introduction
1.1. Clinical Background
1.2. MASLD Diagnosis
2. Materials and Methods
2.1. TAEUS® Liver System
2.2. Study Description
2.3. Data Analysis
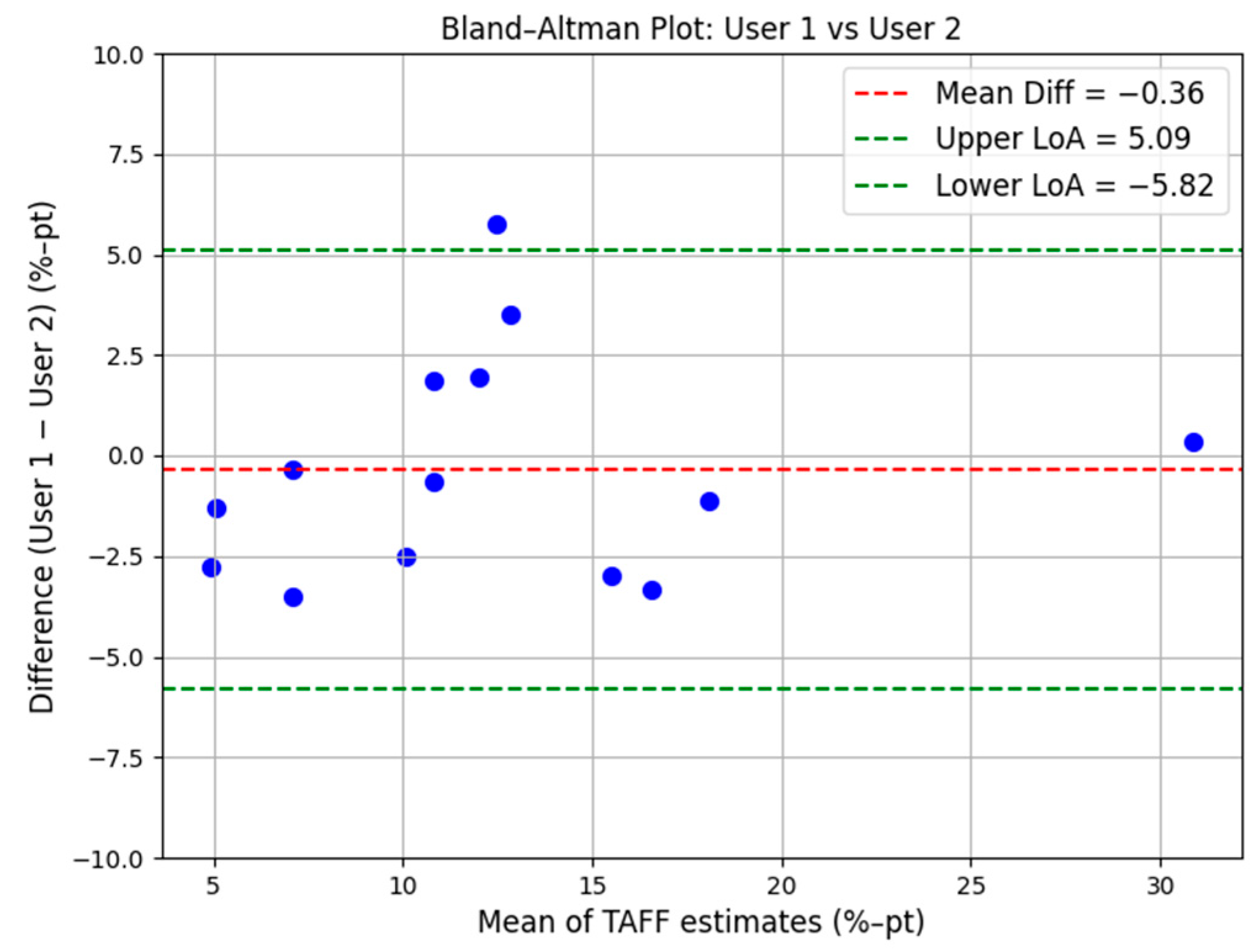
2.4. Measurement Reliability Analysis
3. Results
3.1. Study Subjects
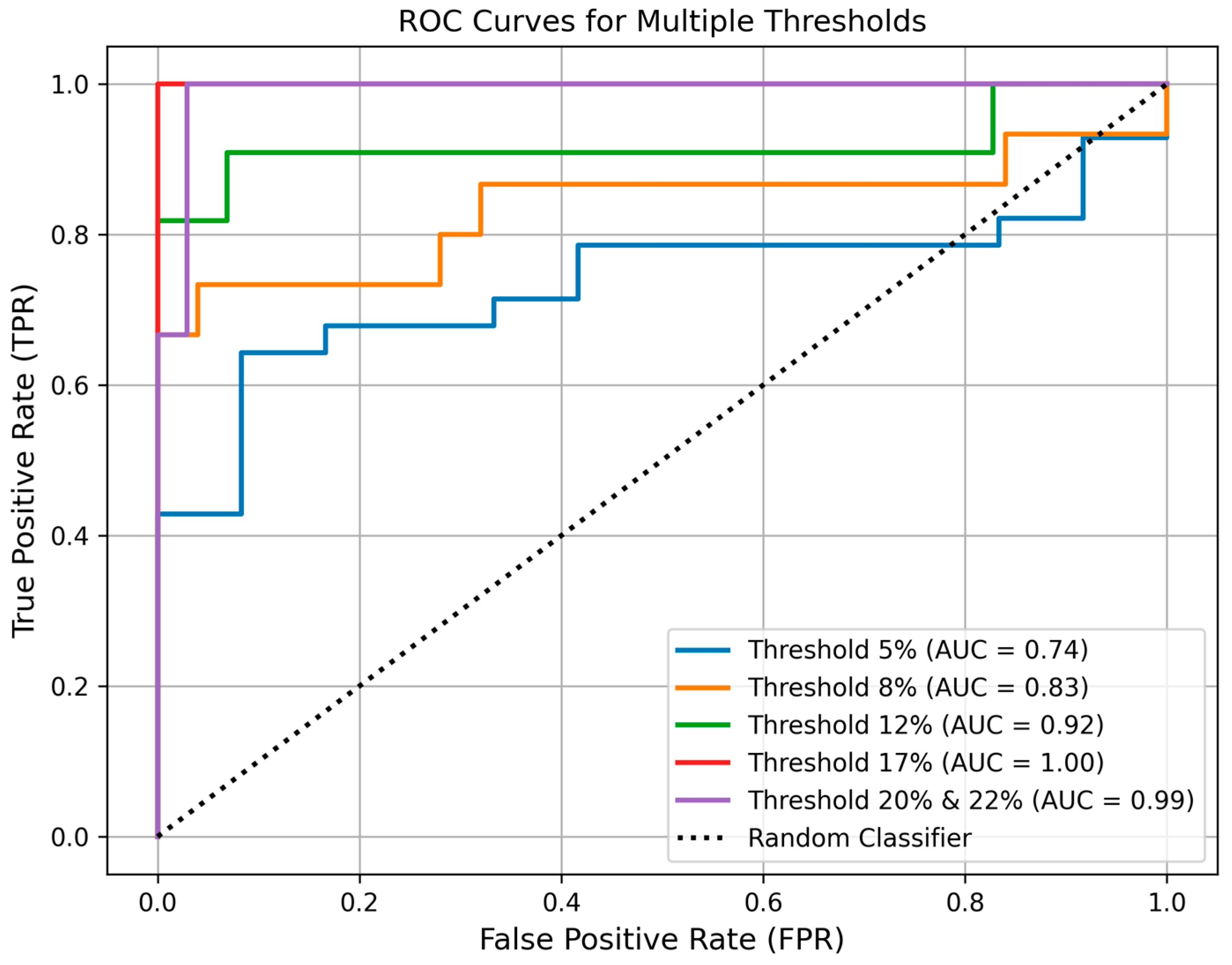
3.2. TAFF Performance
3.3. Relationship Between TAFF and Demographic Information
3.4. Measurement Reliability
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| AASLD | American Association for the Study of Liver Diseases |
| AC | Attenuation Coefficient |
| BMI | Body Mass Index |
| CAP | Controlled Attenuation Parameter |
| CDF | Cumulative Distribution Function |
| CVD | Cardio-Vascular Disease |
| EM | Electro-Magnetic |
| GLP-1 RA | Glucagon-Like Peptide-1 Receptor Agonists |
| HCC | Hepato-Cellular Carcinoma |
| ICC | Intraclass Correlation Coefficient |
| LFF | Liver Fat Fraction |
| LOA | Limits Of Agreement |
| MASH | Metabolic Dysfunction-Associated Steatohepatitis |
| MRI-PDFF | Magnetic Resonance Imaging-Proton Density Fat Fraction |
| MASLD | Metabolic Dysfunction-Associated Steatotic Liver Disease |
| MWA | Micro-Wave Ablation |
| NAFLD | Non-Alcoholic Fatty Liver Disease |
| NPV | Negative Predictive Value |
| QIBA | Quantitative Imaging Biomarkers Alliance |
| QUS | Quantitative Ultra-Sound |
| RF | Radio-Frequency |
| ROI | Region of Interest |
| RSNA | Radiological Society of North America |
| R&R | Repeatability and Reproducibility |
| SEM | Standard Error of Measurement |
| TA | Thermo-Acoustic |
| TAEUS | Thermo-Acoustic Enhanced Ultrasound System |
| TAFF | Thermo-Acoustic Fat Fraction |
| T2D | Type 2 Diabetes |
| US | United States |
| U/S | Ultra-Sound |
| UDFF | Ultrasound-Derived Fat Fraction |
| UGAP | Ultrasound-Guided Attenuation Parameter |
Appendix A
Detailed Description of TAFF Estimation
Appendix B
Raw Data
| Subject ID | TAFF Mean | MR PDFF Mean |
|---|---|---|
| END2501-001 | 4.76 | 1.5 |
| END2501-002 | 9.23 | 6.3 |
| END2501-003 | 2.97 | 12 |
| END2501-005 | 26.13 | 24.8 |
| END2501-006 | 3.02 | 2.9 |
| END2501-007 | 29.80 | 31.9 |
| END2501-009 | 15.94 | 24.9 |
| END2501-010 | 5.96 | 4.1 |
| END2501-011 | 7.19 | 6.9 |
| END2501-012 | 7.35 | 1.5 |
| END2501-014 | 10.94 | 15.7 |
| END2501-015 | 5.03 | 5.6 |
| END2501-016 | 3.63 | 0.9 |
| END2501-017 | 7.23 | 6.3 |
| END2501-018 | 14.13 | 12.5 |
| END2501-019 | 21.80 | 23.7 |
| END2501-020 | 2.18 | 1 |
| END2501-021 | 9.57 | 9.2 |
| END2501-022 | 2.15 | 7.9 |
| END2501-023 | 3.75 | 2.8 |
| END2501-024 | 25.50 | 25.2 |
| END2501-026 | 5.73 | 4.2 |
| END2501-028 | 5.61 | 4.2 |
| END2501-029 | 3.90 | 3.2 |
| END2501-030 | 2.43 | 5.9 |
| END2501-031 | 3.93 | 4.6 |
| END2501-033 | 18.41 | 24.3 |
| END2501-035 | 7.17 | 6.8 |
| END2501-037 | 6.73 | 5.9 |
| END2501-038 | 5.76 | 11.5 |
| END2501-039 | 4.06 | 7.8 |
| END2501-040 | 3.31 | 2.9 |
| END2501-041 | 20.05 | 17.6 |
| END2501-042 | 2.60 | 6.6 |
| END2501-043 | 6.40 | 11.4 |
| END2501-044 | 7.66 | 12.5 |
| END2501-045 | 3.06 | 5.1 |
| END2501-046 | 4.69 | 7.8 |
| END2501-047 | 0.75 | 11.1 |
| END2501-048 | 6.40 | 5.8 |
| Subject ID | User ID | Trial ID | TAFF |
|---|---|---|---|
| MCOM-021 | U1 | 1 | 3.76 |
| MCOM-021 | U2 | 1 | 3.84 |
| MCOM-021 | U1 | 2 | 5.1 |
| MCOM-021 | U2 | 2 | 7.6 |
| MCOM-022 | U1 | 1 | 12.28 |
| MCOM-022 | U2 | 1 | 11.86 |
| MCOM-022 | U1 | 2 | 11.18 |
| MCOM-022 | U2 | 2 | 7.92 |
| MCOM-023 | U1 | 1 | 8.1 |
| MCOM-023 | U2 | 1 | 12.48 |
| MCOM-023 | U1 | 2 | 9.58 |
| MCOM-023 | U2 | 2 | 10.22 |
| MCOM-024 | U1 | 1 | 3.58 |
| MCOM-024 | U2 | 1 | 5.64 |
| MCOM-024 | U1 | 2 | 3.5 |
| MCOM-024 | U2 | 2 | 7.02 |
| MCOM-025 | U1 | 1 | 15.36 |
| MCOM-025 | U2 | 1 | 8.16 |
| MCOM-025 | U1 | 2 | 15.34 |
| MCOM-025 | U2 | 2 | 11 |
| MCOM-026 | U1 | 1 | 7.16 |
| MCOM-026 | U2 | 1 | 7.16 |
| MCOM-026 | U1 | 2 | 6.68 |
| MCOM-026 | U2 | 2 | 7.36 |
| MCOM-027 | U1 | 1 | 20.34 |
| MCOM-027 | U2 | 1 | 13.32 |
| MCOM-027 | U1 | 2 | 9.42 |
| MCOM-027 | U2 | 2 | 23.12 |
| MCOM-028 | U1 | 1 | 20.02 |
| MCOM-028 | U2 | 1 | 11.44 |
| MCOM-028 | U1 | 2 | 7.98 |
| MCOM-028 | U2 | 2 | 22.5 |
| MCOM-029 | U1 | 1 | 5.52 |
| MCOM-029 | U2 | 1 | 7.3 |
| MCOM-029 | U1 | 2 | 5.16 |
| MCOM-029 | U2 | 2 | 10.36 |
| MCOM-030 | U1 | 1 | 10.46 |
| MCOM-030 | U2 | 1 | 12.34 |
| MCOM-030 | U1 | 2 | 10.48 |
| MCOM-030 | U2 | 2 | 9.92 |
| MCOM-031 | U1 | 1 | 15.94 |
| MCOM-031 | U2 | 1 | 9.4 |
| MCOM-031 | U1 | 2 | 13.22 |
| MCOM-031 | U2 | 2 | 12.76 |
| MCOM-032 | U1 | 1 | 13.96 |
| MCOM-032 | U2 | 1 | 8.7 |
| MCOM-032 | U1 | 2 | 12 |
| MCOM-032 | U2 | 2 | 13.36 |
| MCOM-033 | U1 | 1 | 20.88 |
| MCOM-033 | U2 | 1 | 18.02 |
| MCOM-033 | U1 | 2 | 14.14 |
| MCOM-033 | U2 | 2 | 19.26 |
| MCOM-034 | U1 | 1 | 31.6 |
| MCOM-034 | U2 | 1 | 26.96 |
| MCOM-034 | U1 | 2 | 30.46 |
| MCOM-034 | U2 | 2 | 34.4 |
Appendix C
Example of a Beamformed Thermoacoustic Image

References
- Koo, D.J.; Lee, W.Y. The crosstalk between insulin resistance and nonalcoholic fatty liver disease/metabolic dysfunction-associated fatty liver disease: A culprit or a consequence? Cardiovasc. Prev. Pharmacother. 2022, 4, 132–141. [Google Scholar] [CrossRef]
- Vesković, M.; Šutulović, N.; Hrnčić, D.; Stanojlović, O.; Macut, D.; Mladenović, D. The interconnection between hepatic insulin resistance and metabolic dysfunction-associated steatotic liver disease-the transition from an adipocentric to liver-centric approach. Curr. Issues Mol. Biol. 2023, 45, 9084–9102. [Google Scholar] [CrossRef]
- Diehl, A.M. Tumor necrosis factor and its potential role in insulin resistance and nonalcoholic fatty liver disease. Clin. Liver Dis. 2004, 8, 619–638. [Google Scholar] [CrossRef]
- Meex, R.C.R.; Watt, M.J. Hepatokines: Linking nonalcoholic fatty liver disease and insulin resistance. Nat. Rev. Endocrinol. 2017, 13, 509–520. [Google Scholar] [CrossRef]
- Stefan, N.; Schick, F.; Birkenfeld, A.L.; Häring, H.U.; White, M.F. The role of hepatokines in NAFLD. Cell Metab. 2023, 35, 236–252. [Google Scholar] [CrossRef]
- Zhao, Y.C.; Zhao, G.J.; Chen, Z.; She, Z.G.; Cai, J.; Li, H. Nonalcoholic fatty liver disease. Hypertension 2020, 75, 275–284. [Google Scholar] [CrossRef] [PubMed]
- Miao, X.; Alidadipour, A.; Saed, V.; Sayyadi, F.; Jadidi, Y.; Davoudi, M.; Amraee, F.; Jadidi, N.; Afrisham, R. Hepatokines: Unveiling the molecular and cellular mechanisms connecting hepatic tissue to insulin resistance and inflammation. Acta Diabetol. 2024, 61, 1339–1361. [Google Scholar] [CrossRef] [PubMed]
- Alam, S.; Mustafa, G.; Alam, M.; Ahmad, N. Insulin resistance in development and progression of nonalcoholic fatty liver disease. World J. Gastrointest. Pathophysiol. 2016, 7, 211–217. [Google Scholar] [CrossRef]
- Masarone, M.; Rosato, V.; Dallio, M.; Gravina, A.G.; Aglitti, A.; Loguercio, C.; Federico, A.; Persico, M. Role of oxidative stress in pathophysiology of nonalcoholic fatty liver disease. Oxid. Med. Cell. Longev. 2018, 2018, 9547613. [Google Scholar] [CrossRef]
- Gonzalez, A.; Huerta-Salgado, C.; Orozco-Aguilar, J.; Aguirre, F.; Tacchi, F.; Simon, F.; Cabello-Verrugio, C. Role of oxidative stress in hepatic and extrahepatic dysfunctions during nonalcoholic fatty liver disease (NAFLD). Oxid. Med. Cell. Longev. 2020, 2020, 1617805. [Google Scholar] [CrossRef] [PubMed]
- Petrescu, M.; Vlaicu, S.I.; Ciumărnean, L.; Milaciu, M.V.; Mărginean, C.; Florea, M.; Vesa, Ș.C.; Popa, M. Chronic inflammation-A link between nonalcoholic fatty liver disease (NAFLD) and dysfunctional adipose tissue. Medicina 2022, 58, 641. [Google Scholar] [CrossRef] [PubMed]
- Targher, G.; Byrne, C.; Tilg, H. NAFLD and increased risk of cardiovascular disease: Clinical associations, pathophysiological mechanisms and pharmacological implications. Gut 2020, 69, 1691–1705. [Google Scholar] [CrossRef] [PubMed]
- Mantovani, A.; Morieri, M.L.; Aldigeri, R.; Palmisano, L.; Masulli, M.; Bonomo, K.; Baroni, M.G.; Cossu, E.; Cimini, F.A.; Cavallo, G.; et al. MASLD, hepatic steatosis and fibrosis are associated with the prevalence of chronic kidney disease and retinopathy in adults with type 1 diabetes mellitus. Diabetes Metab. 2024, 50, 101497. [Google Scholar] [CrossRef]
- Mikkelsen, A.C.D.; Kjærgaard, K.; Schapira, A.H.V.; Mookerjee, R.P.; Thomsen, K.L. The liver-brain axis in metabolic dysfunction-associated steatotic liver disease. Lancet Gastroenterol. Hepatol. 2025, 10, 248–258. [Google Scholar] [CrossRef] [PubMed]
- Riazi, K.; Azhari, H.; Charette, J.H.; Underwood, F.E.; King, J.A.; Afshar, E.E.; Swain, M.G.; Congly, S.E.; Kaplan, G.G.; Shaheen, A.-A. The prevalence and incidence of NAFLD worldwide: A systematic review and meta-analysis. Lancet Gastroenterol. Hepatol. 2022, 7, 851–861. [Google Scholar] [CrossRef]
- ElSayed, N.A.; McCoy, R.G.; Aleppo, G.; Bajaj, M.; Balapattabi, K.; Beverly, E.A.; Briggs Early, K.; Bruemmer, D.; Cusi, K.; Echouffo-Tcheugui, J.B.; et al. 4. Comprehensive Medical Evaluation and Assessment of Comorbidities: Standards of Care in Diabetes—2025. Diabetes Care 2025, 48, S59–S85. [Google Scholar]
- Younossi, Z.M.; Koenig, A.B.; Abdelatif, D.; Fazel, Y.; Henry, L.; Wymer, M. Global epidemiology of nonalcoholic fatty liver disease-Meta-analytic assessment of prevalence, incidence, and outcomes. Hepatology 2016, 64, 73–84. [Google Scholar] [CrossRef]
- Le, M.H.; Yeo, Y.H.; Li, X.; Li, J.; Zou, B.; Wu, Y.; Ye, Q.; Huang, D.Q.; Zhao, C.; Zhang, J.; et al. 2019 global NAFLD prevalence: A systematic review and meta-analysis. Clin. Gastroenterol. Hepatol. 2022, 20, 2809–2817.e28. [Google Scholar] [CrossRef]
- Younossi, Z.M.; Paik, J.M.; Henry, L.; Yang, J.; Fernandes, G.; Stepanova, M.; Nader, F. The Growing Economic and Clinical Burden of Nonalcoholic Steatohepatitis (NASH) in the United States. J. Clin. Exp. Hepatol. 2023, 13, 454–467. [Google Scholar] [CrossRef]
- Zhang, C.; Yang, M. Current Options and Future Directions for NAFLD and NASH Treatment. Int. J. Mol. Sci. 2021, 22, 7571. [Google Scholar] [CrossRef]
- Mahathi, B.I.; Srikanth, R. Non-Alcoholic Fatty Liver Disease: An Emerging Cardiovascular Risk Enhancer. 2022. Available online: https://www.acc.org/latest-in-cardiology/articles/2022/07/13/14/58/non-alchoholic-fatty-liver-disease (accessed on 12 December 2025).
- Oh, Y.H.; Jeong, S.; Park, S.J.; Ahn, J.C.; Park, S.M. Reversal of nonalcoholic fatty liver disease reduces the risk of cardiovascular disease among Korean. Medicine 2023, 102, e35804. [Google Scholar] [CrossRef] [PubMed]
- Tsamos, G.; Vasdeki, D.; Koufakis, T.; Michou, V.; Makedou, K.; Tzimagiorgis, G. Therapeutic potentials of reducing liver fat in non-alcoholic fatty liver disease: Close association with type 2 diabetes. Metabolites 2023, 13, 517. [Google Scholar] [CrossRef]
- Laursen, T.L.; Hagemann, C.A.; Wei, C.; Kazankov, K.; Thomsen, K.L.; Knop, F.K.; Grønbæk, H. Bariatric surgery in patients with non-alcoholic fatty liver disease-from pathophysiology to clinical effects. World J. Hepatol. 2019, 11, 138–149. [Google Scholar] [CrossRef]
- Biobaku, F.; Ghanim, H.; Monte, S.V.; Caruana, J.A.; Dandona, P. Bariatric surgery: Remission of inflammation, cardiometabolic benefits, and common adverse effects. J. Endocr. Soc. 2020, 4, bvaa049. [Google Scholar] [CrossRef] [PubMed]
- Vilar-Gomez, E.; Martinez-Perez, Y.; Calzadilla-Bertot, L.; Torres-Gonzalez, A.; Gra-Oramas, B.; Gonzalez-Fabian, L.; Friedman, S.L.; Diago, M.; Romero-Gomez, M. Weight loss through lifestyle modification significantly reduces features of nonalcoholic steatohepatitis. Gastroenterology 2015, 149, 367–378.e5; quiz e14-5. [Google Scholar] [CrossRef]
- Kotsiliti, E. Drug approvals in gastroenterology and hepatology in 2024. Nat. Rev. Gastroenterol. Hepatol. 2025, 22, 83. [Google Scholar] [CrossRef]
- Sanyal, A.J.; Newsome, P.N.; Kliers, I.; Østergaard, L.H.; Long, M.T.; Kjær, M.S.; Cali, A.M.G.; Bugianesi, E.; Rinella, M.E.; Roden, M.; et al. Phase 3 trial of semaglutide in metabolic dysfunction-associated steatohepatitis. N. Engl. J. Med. 2025, 392, 2089–2099. [Google Scholar] [CrossRef]
- McEwan, P.; Bøg, M.; Faurby, M.; Foos, V.; Lingvay, I.; Lübker, C.; Miller, R.; Toliver, J.; Yeates, F.; Lincoff, A.M. EE497 the cost-effectiveness of semaglutide 2.4MG in patients with overweight/obesity and cardiovascular disease from the select trial. Value Health 2024, 27, S148. [Google Scholar] [CrossRef]
- Bacha, Z.; Javed, J.; Sheraz, M.; Sikandar, M.; Zakir, M.; Ali, M.A.; Khan, M.; Iqbal, A.; Rehman, A.; Alam, U.; et al. Efficacy and safety of semaglutide on cardiovascular outcomes in patients with type 2 diabetes: A systematic review and meta-analysis of randomized controlled trials. Cardiol. Rev. 2025. [Google Scholar] [CrossRef]
- Kishimori, T.; Kato, T.; Wada, A.; Tani, A.; Yamaji, R.; Koike, J.; Iwasaki, Y.; Matsumoto, T.; Yagi, T.; Okada, M. Cardiovascular outcomes and safety of semaglutide in non-overweight populations with type 2 diabetes: A comparison with dipeptidyl peptidase 4 inhibitors. Eur. Heart J. Qual. Care Clin. Outcomes 2025, 11, 1319–1328. [Google Scholar] [CrossRef]
- Sumida, Y.; Nakajima, A.; Itoh, Y. Limitations of liver biopsy and non-invasive diagnostic tests for the diagnosis of nonalcoholic fatty liver disease/nonalcoholic steatohepatitis. World J. Gastroenterol. 2014, 20, 475–485. [Google Scholar] [CrossRef]
- Heyens, L.J.M.; Busschots, D.; Koek, G.H.; Robaeys, G.; Francque, S. Liver fibrosis in non-alcoholic fatty liver disease: From liver biopsy to non-invasive biomarkers in diagnosis and treatment. Front. Med. 2021, 8, 615978. [Google Scholar] [CrossRef] [PubMed]
- Caussy, C.; Reeder, S.B.; Sirlin, C.B.; Loomba, R. Noninvasive, quantitative assessment of liver fat by MRI-PDFF as an endpoint in NASH trials. Hepatology 2018, 68, 763–772. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.X.; Feng, Y.P.; You, C.L.; Zhang, L.Y. The diagnostic value of MRI-PDFF in hepatic steatosis of patients with metabolic dysfunction-associated steatotic liver disease: A systematic review and meta-analysis. BMC Gastroenterol. 2025, 25, 451. [Google Scholar] [CrossRef]
- Mikolasevic, I.; Orlic, L.; Franjic, N.; Hauser, G.; Stimac, D.; Milic, S. Transient elastography (FibroScan(®)) with controlled attenuation parameter in the assessment of liver steatosis and fibrosis in patients with nonalcoholic fatty liver disease-Where do we stand? World J. Gastroenterol. 2016, 22, 7236–7251. [Google Scholar] [CrossRef] [PubMed]
- Malandris, K.; Korakas, E.; Sarakapina, A.; Kalopitas, G.; Iatridi, F.; Liakos, A.; Bekiari, E.; Giouleme, O.; Tzatzagou, G.; Karagiannis, T.; et al. Accuracy of controlled attenuation parameter for liver steatosis in high-risk patients for MASLD using MRI-proton density fat fraction as reference standard. Dig. Dis. Sci. 2025, 70, 814–824. [Google Scholar] [CrossRef]
- Martinou, E.; Pericleous, M.; Stefanova, I.; Kaur, V.; Angelidi, A.M. Diagnostic modalities of non-Alcoholic Fatty Liver Disease: From biochemical biomarkers to multi-omics non-invasive approaches. Diagnostics 2022, 12, 407. [Google Scholar] [CrossRef]
- Lauschke, V.M. Practice guidance documents for the diagnosis and management of non-alcoholic fatty liver disease-recent updates and open questions. Hepatobiliary Surg. Nutr. 2023, 12, 780–784. [Google Scholar] [CrossRef]
- Wear, K.A.; Han, A.; Rubin, J.M.; Gao, J.; Lavarello, R.; Cloutier, G.; Bamber, J.; Tuthill, T. US backscatter for liver fat quantification: An AIUM-RSNA QIBA pulse-echo quantitative Ultrasound initiative. Radiology 2022, 305, 526–537. [Google Scholar] [CrossRef]
- Park, J.; Lee, J.M.; Lee, G.; Jeon, S.K.; Joo, I. Quantitative evaluation of hepatic steatosis using advanced imaging techniques: Focusing on new quantitative ultrasound techniques. Korean J. Radiol. 2022, 23, 13–29. [Google Scholar] [CrossRef]
- Han, A. US attenuation coefficient for liver fat quantification: A need for standardization. Radiology 2024, 313, e242511. [Google Scholar] [CrossRef]
- Johansen, S.; Åberg, F.; Tsochatzis, E.A.; Krag, A. Screening for advanced steatotic liver disease. Lancet Gastroenterol. Hepatol. 2025, 10, 842–854. [Google Scholar] [CrossRef]
- Yang, Y.; Xia, Z.; Xia, H.; Li, Y.; Liu, G.; Xu, J. Applied current thermoacoustic imaging for biological tissues. Technol. Health Care 2020, 28, 57–65. [Google Scholar] [CrossRef] [PubMed]
- Gabriel, S.; Lau, R.W.; Gabriel, C. The dielectric properties of biological tissues: III. Parametric models for the dielectric spectrum of tissues. Phys. Med. Biol. 1996, 41, 2271–2293. [Google Scholar] [CrossRef] [PubMed]
- Gabriel, S.; Lau, R.W.; Gabriel, C. The dielectric properties of biological tissues: II. Measurements in the frequency range 10 Hz to 20 GHz. Phys. Med. Biol. 1996, 41, 2251–2269. [Google Scholar] [CrossRef]
- Gabriel, C.; Gabriel, S.; Corthout, E. The dielectric properties of biological tissues: I. Literature survey. Phys. Med. Biol. 1996, 41, 2231–2249. [Google Scholar] [CrossRef]
- Ku, G.; Wang, L.V. Scanning microwave-induced thermoacoustic tomography: Signal, resolution, and contrast. Med. Phys. 2001, 28, 4–10. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Huang, L.; Chi, Z.; Jiang, H. Integrated thermoacoustic and ultrasound imaging based on the combination of a hollow concave transducer array and a linear transducer array. Phys. Med. Biol. 2021, 66, 115011. [Google Scholar] [CrossRef]
- Roggenbuck, M.A.; Walker, R.D.; Catenacci, J.W.; Patch, S.K. Volumetric thermoacoustic imaging over large fields of view. Ultrason. Imaging 2013, 35, 57–67. [Google Scholar] [CrossRef]
- Razansky, D.; Kellnberger, S.; Ntziachristos, V. Near-field radiofrequency thermoacoustic tomography with impulse excitation: NRT. Med. Phys. 2010, 37, 4602–4607. [Google Scholar] [CrossRef]
- Liu, Q.; Liang, X.; Qi, W.; Gong, Y.; Jiang, H.; Xi, L. Biomedical microwave-induced thermoacoustic imaging. J. Innov. Opt. Health Sci. 2022, 15, 2230007. [Google Scholar] [CrossRef]
- Li, J.; Wu, Z.; Peng, C.; Song, L.; Luo, Y. Microwave-induced thermoacoustic imaging for the early detection of canine intracerebral hemorrhage. Front. Physiol. 2022, 13, 1067948. [Google Scholar] [CrossRef]
- Islam, T.; Avanaki, K. Thermoacoustic imaging: Principles and applications in preclinical research. In Biomaterials, Bioengineering and Sustainability; Springer Nature: Cham, Switzerland, 2025; pp. 365–397. [Google Scholar] [CrossRef]
- Song, L.; Peng, W.; Lu, Q.; Feng, L.; Yang, Z.; Huang, L.; Luo, Y. Thermoacoustic imaging using single-channel data acquisition system for non-invasive assessment of liver microwave ablation: A feasibility study. Photonics 2024, 11, 807. [Google Scholar] [CrossRef]
- Kruger, R.A.; Kopecky, K.K.; Aisen, A.M.; Reinecke, D.R.; Kruger, G.A.; Kiser, W.L., Jr. Thermoacoustic CT with radio waves: A medical imaging paradigm. Radiology 1999, 211, 275–278. [Google Scholar] [CrossRef]
- IEC 60601-2-33; Medical Electrical Equipment. International Electrotechnical Commission: Geneva, Switzerland, 2023.
- IEEE C95.1-2019; Standard for Safety Levels with Respect to Human Exposure to Electric, Magnetic, and Electromagnetic Fields, 0 Hz to 300 GHz. Institute of Electrical and Electronics Engineers (IEEE): New York, NY, USA, 2019.
- Xu, M.; Wang, L.V. Universal back-projection algorithm for photoacoustic computed tomography. Phys. Rev. E Stat. Nonlin. Soft Matter. Phys. 2005, 71, 016706. [Google Scholar] [CrossRef]
- Bonekamp, S.; Tang, A.; Mashhood, A.; Wolfson, T.; Changchien, C.; Middleton, M.S.; Clark, L.; Gamst, A.; Loomba, R.; Sirlin, C.B. Spatial distribution of MRI-Determined hepatic proton density fat fraction in adults with nonalcoholic fatty liver disease: Spatial Distribution of Liver Fat in NAFLD. J. Magn. Reson. Imaging 2014, 39, 1525–1532. [Google Scholar] [CrossRef] [PubMed]
- Martin Bland, J.; Altman, D. Statistical methods for assessing agreement between two methods of clinical measurement. Lancet 1986, 327, 307–310. [Google Scholar] [CrossRef]
- Deming, W.E. Statistical Adjustment of Data; John Wiley and Sons: Hoboken, NJ, USA, 1943; Volume 261, Available online: https://psycnet.apa.org/record/1944-00642-000 (accessed on 10 December 2025).
- Guidance, D. Guidance for Industry. Available online: https://pink.citeline.com/-/media/supporting-documents/pink-sheet/2022/12/p1222fda_2.pdf (accessed on 14 December 2025).
- Gassert, F.T.; Kufner, A.; Gassert, F.G.; Leonhardt, Y.; Kronthaler, S.; Schwaiger, B.J.; Boehm, C.; Makowski, M.R.; Kirschke, J.S.; Baum, T.; et al. MR-based proton density fat fraction (PDFF) of the vertebral bone marrow differentiates between patients with and without osteoporotic vertebral fractures. Osteoporos. Int. 2022, 33, 487–496. [Google Scholar] [CrossRef] [PubMed]
- Tang, A.; Tan, J.; Sun, M.; Hamilton, G.; Bydder, M.; Wolfson, T.; Gamst, A.C.; Middleton, M.; Brunt, E.M.; Loomba, R.; et al. Nonalcoholic fatty liver disease: MR imaging of liver proton density fat fraction to assess hepatic steatosis. Radiology 2013, 267, 422–431. [Google Scholar] [CrossRef]
- Park, S.; Kwon, J.H.; Kim, S.; Kang, J.H.; Chung, J.I.; Jang, J.K.; Jang, H.Y.; Shim, J.H.; Lee, S.S.; Kim, K.W.; et al. Cutoff values for diagnosing hepatic steatosis using contemporary MRI-proton density fat fraction measuring methods. Korean J. Radiol. 2022, 23, 1260–1268. [Google Scholar] [CrossRef] [PubMed]
- Kühn, J.P.; Hernando, D.; Muñoz del Rio, A.; Evert, M.; Kannengiesser, S.; Völzke, H.; Mensel, B.; Puls, R.; Hosten, N.; Reeder, S.B. Effect of multipeak spectral modeling of fat for liver iron and fat quantification: Correlation of biopsy with MR imaging results. Radiology 2012, 265, 133–142. [Google Scholar] [CrossRef]
- Szczepaniak, L.S.; Nurenberg, P.; Leonard, D.; Browning, J.D.; Reingold, J.S.; Grundy, S.; Hobbs, H.H.; Dobbins, R.L. Magnetic resonance spectroscopy to measure hepatic triglyceride content: Prevalence of hepatic steatosis in the general population. Am. J. Physiol. Endocrinol. Metab. 2005, 288, E462–E468. [Google Scholar] [CrossRef]
- Tang, A.; Desai, A.; Hamilton, G.; Wolfson, T.; Gamst, A.; Lam, J.; Clark, L.; Hooker, J.; Chavez, T.; Ang, B.D.; et al. Accuracy of MR imaging-estimated proton density fat fraction for classification of dichotomized histologic steatosis grades in nonalcoholic fatty liver disease. Radiology 2015, 274, 416–425. [Google Scholar] [CrossRef]
- Chen, V.L.; Morgan, T.R.; Rotman, Y.; Patton, H.M.; Cusi, K.; Kanwal, F.; Kim, W.R. Erratum: Resmetirom therapy for metabolic dysfunction-associated steatotic liver disease: October 2024 updates to AASLD Practice Guidance. Hepatology 2025, 81, E133. [Google Scholar] [CrossRef]
- Harrell FEJr Califf, R.M.; Pryor, D.B.; Lee, K.L.; Rosati, R.A. Evaluating the yield of medical tests. JAMA 1982, 247, 2543–2546. [Google Scholar] [CrossRef]
- Sun, J.Y.; Huang, W.J.; Hua, Y.; Qu, Q.; Cheng, C.; Liu, H.L.; Kong, X.Q.; Ma, Y.X.; Sun, W. Trends in general and abdominal obesity in US adults: Evidence from the National Health and Nutrition Examination Survey (2001–2018). Front. Public Health 2022, 10, 925293. [Google Scholar] [CrossRef] [PubMed]
- Han, A.; Zhang, Y.N.; Boehringer, A.S.; Montes, V.; Andre, M.P.; Erdman, J.W.; Loomba, R.; Valasek, M.A.; Sirlin, C.B.; O’Brien, W.D., Jr. Assessment of hepatic steatosis in nonalcoholic fatty liver disease by using quantitative US. Radiology 2020, 295, 106–113. [Google Scholar] [CrossRef]
- Shao, C.X.; Ye, J.; Dong, Z.; Li, F.; Lin, Y.; Liao, B.; Feng, S.; Zhong, B. Steatosis grading consistency between controlled attenuation parameter and MRI-PDFF in monitoring metabolic associated fatty liver disease. Ther. Adv. Chronic Dis. 2021, 12, 20406223211033120. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Nie, M.; Zhou, H.; Mao, F.; Zhao, L.; Ding, J.; Jing, X. Head-to-head comparison of three different US-based quantitative parameters for hepatic steatosis assessment: A prospective study. Abdom. Radiol. 2024, 49, 2262–2271. [Google Scholar] [CrossRef]
- Gao, R.; Han, J.; Song, D.; Wang, P.; Chen, H.; Shen, H.; Li, J. Quantitative assessment of hepatic steatosis by ultrasound-guided attenuation parameter in patients with impaired glucose tolerance. Insights Imaging 2025, 16, 247. [Google Scholar] [CrossRef]
- De Robertis, R.; Spoto, F.; Autelitano, D.; Guagenti, D.; Olivieri, A.; Zanutto, P.; Incarbone, G.; D’Onofrio, M. Ultrasound-derived fat fraction for detection of hepatic steatosis and quantification of liver fat content. Radiol. Med. 2023, 128, 1174–1180. [Google Scholar] [CrossRef]
- Cao, Y.T.; Xiang, L.L.; Qi, F.; Zhang, Y.J.; Chen, Y.; Zhou, X.Q. Accuracy of controlled attenuation parameter (CAP) and liver stiffness measurement (LSM) for assessing steatosis and fibrosis in non-alcoholic fatty liver disease: A systematic review and meta-analysis. EClinicalMedicine 2022, 51, 101547. [Google Scholar] [CrossRef]
- Karlas, T.; Petroff, D.; Sasso, M.; Fan, J.G.; Mi, Y.Q.; de Ledinghen, V.; Kumar, M.; Lupsor-Platon, M.; Han, K.H.; Cardoso, A.C.; et al. Individual patient data meta-analysis of controlled attenuation parameter (CAP) technology for assessing steatosis. J. Hepatol. 2017, 66, 1022–1030. [Google Scholar] [CrossRef]
- Miura, D. Compatibility and bias between ultrasound-guided attenuation parameter and controlled attenuation parameter for ultrasound-based hepatic steatosis assessment. medRxiv 2025. [Google Scholar] [CrossRef]
- López-Mendez, I.; Romero-Flores, J.L.; Castro-Narro, G.; Uribe, M.; Juárez-Hernández, E. Factors associated with obtaining lower IQR-CAP values in the detection of hepatic steatosis by transient elastography. Ann. Hepatol. 2025, 30, 101762. [Google Scholar] [CrossRef]
- Heinitz, S.; Müller, J.; Jenderka, K.V.; Schlögl, H.; Stumvoll, M.; Blüher, M.; Blank, V.; Karlas, T. The application of high-performance ultrasound probes increases anatomic depiction in obese patients. Sci. Rep. 2023, 13, 16297. [Google Scholar] [CrossRef] [PubMed]
- Maar, M.; Lee, J.; Tardi, A.; Zheng, Y.Y.; Wong, C.; Gao, J. Inter-transducer variability of ultrasound image quality in obese adults: Qualitative and quantitative comparisons. Clin. Imaging 2022, 92, 63–71. [Google Scholar] [CrossRef] [PubMed]
- Bril, F.; Ortiz-Lopez, C.; Lomonaco, R.; Orsak, B.; Freckleton, M.; Chintapalli, K.; Hardies, J.; Lai, S.; Solano, F.; Tio, F.; et al. Clinical value of liver ultrasound for the diagnosis of nonalcoholic fatty liver disease in overweight and obese patients. Liver Int. 2015, 35, 2139–2146. [Google Scholar] [CrossRef]
- Ferraioli, G.; Raimondi, A.; Maiocchi, L.; De Silvestri, A.; Poma, G.; Kumar, V.; Barr, R.G. Liver fat quantification with ultrasound: Depth dependence of attenuation coefficient: Depth dependence of attenuation coefficient. J. Ultrasound Med. 2023, 42, 2247–2255. [Google Scholar] [CrossRef] [PubMed]
- Lin, S.C.; Heba, E.; Wolfson, T.; Ang, B.; Gamst, A.; Han, A.; Erdman, J.W., Jr.; O’Brien, W.D., Jr.; Andre, M.P.; Sirlin, C.B.; et al. Noninvasive diagnosis of nonalcoholic fatty liver disease and quantification of liver fat using a new quantitative ultrasound technique. Clin. Gastroenterol. Hepatol. 2015, 13, 1337–1345.e6. [Google Scholar] [CrossRef]
- Dillman, J.R.; Thapaliya, S.; Tkach, J.A.; Trout, A.T. Quantification of hepatic steatosis by ultrasound: Prospective comparison with MRI proton density fat fraction as reference standard. AJR Am. J. Roentgenol. 2022, 219, 784–791. [Google Scholar] [CrossRef]
- Ianni Filho, D.; Boin, I.d.F.S.F.; Yamanaka, A. Bioimpedance: New approach to non-invasive detection of liver fibrosis-a pilot study. Arq. Gastroenterol. 2018, 55, 2–6. [Google Scholar] [CrossRef][Green Version]
- Kyung, E.J.; Kim, H.B.; Hwang, E.S.; Lee, S.; Choi, B.K.; Kim, J.W.; Kim, H.J.; Lim, S.M.; Kwon, O.I.; Woo, E.J. Evaluation of hepatoprotective effect of curcumin on liver cirrhosis using a combination of biochemical analysis and magnetic resonance-based electrical conductivity imaging. Mediat. Inflamm. 2018, 2018, 5491797. [Google Scholar] [CrossRef]
- Shi, Y.; Bai, X.; Yang, J.; Wu, X.; Wang, L. Optimized measurement methods and systems for the dielectric properties of active biological tissues in the 10Hz-100 MHz frequency range. Front. Physiol. 2025, 16, 1537537. [Google Scholar] [CrossRef] [PubMed]
- Reiter, R.; Freise, C.; Jöhrens, K.; Kamphues, C.; Seehofer, D.; Stockmann, M.; Somasundaram, R.; Asbach, P.; Braun, J.; Samani, A.; et al. Wideband MRE and static mechanical indentation of human liver specimen: Sensitivity of viscoelastic constants to the alteration of tissue structure in hepatic fibrosis. J. Biomech. 2014, 47, 1665–1674. [Google Scholar] [CrossRef] [PubMed]
- Yeh, W.C.; Li, P.C.; Jeng, Y.M.; Hsu, H.C.; Kuo, P.L.; Li, M.L.; Yang, P.M.; Lee, P.H. Elastic modulus measurements of human liver and correlation with pathology. Ultrasound Med. Biol. 2002, 28, 467–474. [Google Scholar] [CrossRef] [PubMed]





| Inclusion Criteria | Exclusion Criteria |
|---|---|
|
|
| Gender | N | Variable | Units | Mean | Std Dev | Minimum | Maximum |
|---|---|---|---|---|---|---|---|
| Female | 25 | Age | years | 54.2 | 12.0 | 34.0 | 74.0 |
| Height | cm | 165.7 | 7.6 | 153.0 | 184.0 | ||
| Weight | kg | 93.6 | 22.0 | 54.0 | 148.8 | ||
| BMI | kg/m2 | 34.0 | 7.1 | 21.0 | 47.0 | ||
| Hip circumference | cm | 120.3 | 16.7 | 84.0 | 150.0 | ||
| Waist circumference | cm | 113.4 | 16.8 | 77.0 | 148.0 | ||
| Waist-to-hip ratio | # | 0.95 | 0.09 | 0.79 | 1.17 | ||
| Male | 15 | Age | years | 49.2 | 10.3 | 29.0 | 65.0 |
| Height | cm | 176.5 | 8.0 | 157.0 | 186.0 | ||
| Weight | kg | 100.5 | 14.3 | 79.2 | 130.0 | ||
| BMI | kg/m2 | 32.2 | 3.7 | 24.8 | 37.6 | ||
| Hip circumference | cm | 112.0 | 6.1 | 103.0 | 123.0 | ||
| Waist circumference | cm | 112.1 | 13.8 | 83.0 | 134.0 | ||
| Waist-to-hip ratio | # | 1.01 | 0.09 | 0.81 | 1.13 | ||
| Both | 40 | Age | years | 52.3 | 11.5 | 29.0 | 74.0 |
| Height | cm | 169.8 | 9.3 | 153.0 | 186.0 | ||
| Weight | kg | 96.2 | 19.6 | 54.0 | 148.8 | ||
| BMI | kg/m2 | 33.3 | 6.0 | 21.0 | 47.0 | ||
| Hip circumference | cm | 117.1 | 14.2 | 84.0 | 150.0 | ||
| Waist circumference | cm | 112.9 | 15 | 77.0 | 148.0 | ||
| Waist-to-hip ratio | # | 0.97 | 0.09 | 0.79 | 1.17 |
| Threshold | 5% LFF: S0-S1 | 8% LFF: THrβ | 12% LFF: S1-S2 | 17% LFF: S1-S2 | 20% LFF: S2-S3 | 22% LFF: S2-S3 |
|---|---|---|---|---|---|---|
| PPV [%] | 75.9 (59.3, 90.6) | 68.8 (43.8, 90.9) | 100.0 (100.0, 100.0) | 100.0 (100.0, 100.0) | 80.0 (33.3, 100.0) | 100.0 (100.0, 100.0) |
| NPV [%] | 45.5 (16.7, 75.0) | 83.3 (66.7, 96.3) | 93.5 (83.3, 100.0) | 100.0 (100.0, 100.0) | 94.3 (85.7, 100.0) | 94.4 (86.1, 100.0) |
| Sensitivity [%] | 78.6 (62.1, 92.6) | 73.3 (50.0, 94.1) | 81.8 (54.55, 100.0) | 100.0 (100.0, 100.0) | 66.7 (20.0, 100.0) | 66.7 (20.0, 100.0) |
| Specificity [%] | 41.7 (12.5, 72.7) | 80.0 (63.0, 95.0) | 100.0 (100.0, 100.0) | 100.0 (100.0, 100.0) | 97.1 (90.3, 100.0) | 100.0 (100.0, 100.0) |
| Accuracy [%] | 67.5 (52.5, 82.5) | 77.5 (65.0, 90.0) | 95.0 (87.5, 100.0) | 100.0 (100.0, 100.0) | 92.5 (82.50, 100.0) | 95.0 (87.5, 100.0) |
| AUROC [#] | 0.74 (0.57, 0.88) | 0.83 (0.66, 0.98) | 0.92 (0.75, 1.00) | 1.0 (1.0,1.0) | 0.99 (0.96, 1.00) | 0.99 (0.96, 1.00) |
| Variable | Correlation | p-Value |
|---|---|---|
| Age | −0.080 | 0.2574 |
| Height | −0.158 | 0.3295 |
| Weight | −0.150 | 0.3563 |
| BMI | −0.056 | 0.7370 |
| Hip circumference | −0.052 | 0.7530 |
| Waist circumference | −0.1049 | 0.5252 |
| Waist/hip ratio | −0.085 | 0.6114 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Cho, J.H.; Bull, C.M.; Thornton, M.; Gao, J.; Rubin, J.M.; Steinberg, I. Thermoacoustic Ultrasound Assessment of Liver Steatosis—A Novel Approach for MASLD Diagnosis. Diagnostics 2026, 16, 804. https://doi.org/10.3390/diagnostics16050804
Cho JH, Bull CM, Thornton M, Gao J, Rubin JM, Steinberg I. Thermoacoustic Ultrasound Assessment of Liver Steatosis—A Novel Approach for MASLD Diagnosis. Diagnostics. 2026; 16(5):804. https://doi.org/10.3390/diagnostics16050804
Chicago/Turabian StyleCho, Jang Hwan, Christopher M. Bull, Michael Thornton, Jing Gao, Jonathan M. Rubin, and Idan Steinberg. 2026. "Thermoacoustic Ultrasound Assessment of Liver Steatosis—A Novel Approach for MASLD Diagnosis" Diagnostics 16, no. 5: 804. https://doi.org/10.3390/diagnostics16050804
APA StyleCho, J. H., Bull, C. M., Thornton, M., Gao, J., Rubin, J. M., & Steinberg, I. (2026). Thermoacoustic Ultrasound Assessment of Liver Steatosis—A Novel Approach for MASLD Diagnosis. Diagnostics, 16(5), 804. https://doi.org/10.3390/diagnostics16050804






