From Plastics to Prognosis: ANO4 Susceptibility Links Phthalate Exposure to Prostate Cancer Progression
Abstract
1. Introduction
2. Patient and Methods
2.1. Transcriptomic Analysis of MEHP Exposure in Human Prostate Epithelial Cells
2.2. Study Population and Clinical Data Collection
2.3. Single-Nucleotide Polymorphism (SNP) Selection and Genotyping
2.4. Bioinformatic Analyses of ANO4 Expression and Function
2.5. Statistical Analyses
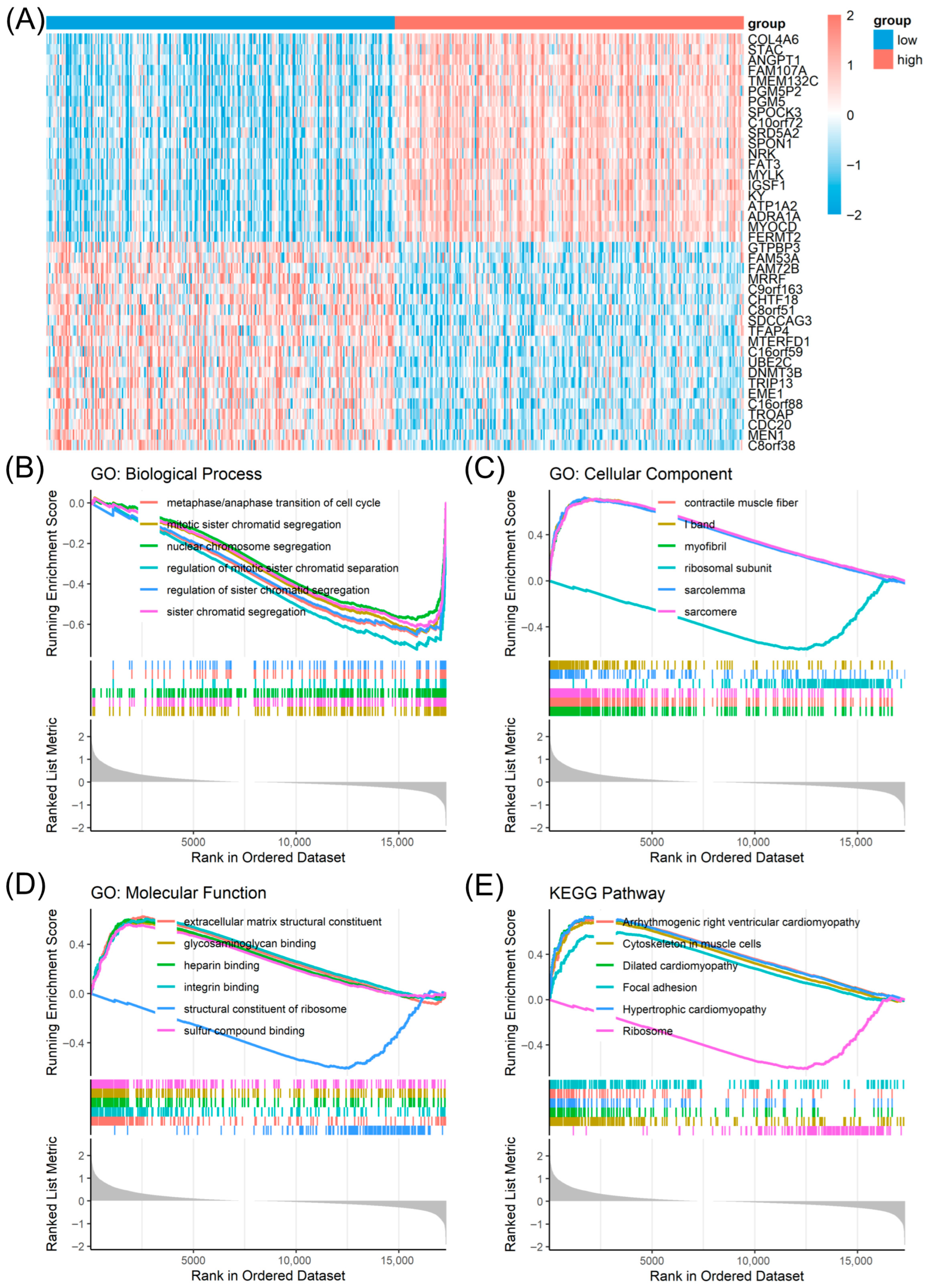
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Jaeger, R.J.; Rubin, R.J. Extraction, localization, and metabolism of di-2-ethylhexyl phthalate from PVC plastic medical devices. Environ. Health Perspect. 1973, 3, 95–102. [Google Scholar] [CrossRef]
- Ashaari, S.; Jamialahmadi, T.; Davies, N.M.; Almahmeed, W.; Sahebkar, A. Di (2-ethyl hexyl) phthalate and its metabolite-induced metabolic syndrome: A review of molecular mechanisms. Drug Chem. Toxicol. 2025, 48, 325–343. [Google Scholar] [CrossRef]
- Li, X.; Xiao, C.; Liu, J.; Wei, N.; Song, J.; Yuan, J.; Liu, L.; Song, R.; Yi, W.; Pan, R.; et al. Association of Di(2-ethylhexyl) Phthalate Exposure with Reproductive Hormones in the General Population and the Susceptible Population: A Systematic Review and Meta-Analysis. Environ. Health 2024, 2, 750–765. [Google Scholar] [CrossRef]
- Zheng, W.C.; Xu, W.T.; Chen, C.R.; Lin, F.; Chen, S.H.; Li, X.D.; Sun, X.L.; Zheng, Q.S.; Wei, Y.; Xue, X.Y.; et al. Integrative analysis of DEHP exposure and prostate cancer: Mechanistic insights and predictive modeling. Environ. Pollut. 2026, 392, 127628. [Google Scholar] [CrossRef]
- Martinez-Razo, L.D.; Rivero-Segura, N.A.; Almeida-Aguirre, E.K.P.; Mancilla-Herrera, I.; Rincon-Heredia, R.; Martinez-Ibarra, A.; Cerbon, M. Mono(2-ethylhexyl) Phthalate Disrupts Mitochondrial Function, Dynamics and Biogenesis in Human Trophoblast Cells at Human Exposure Range Concentrations. Toxics 2025, 13, 770. [Google Scholar] [CrossRef]
- Rusyn, I.; Corton, J.C. Mechanistic considerations for human relevance of cancer hazard of di(2-ethylhexyl) phthalate. Mutat. Res. 2012, 750, 141–158. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.H.; Chen, J.; Wang, Y.; Xia, B.; Wang, R.; Zhao, Y.; Wang, Q.X.; Song, Q.; Yao, S.H.; Zhang, Y.H.; et al. Effect of Mono-2-ethyhexyl Phthalate on DNA Methylation in Human Prostate Cancer LNCaP Cells. Biomed. Environ. Sci. 2017, 30, 641–648. [Google Scholar] [CrossRef] [PubMed]
- Wu, P.H.; Chen, S.C.; Chien, C.J.; Lin, J.; Lee, H.Y.; Lin, Y.T.; Weng, T.C.; Hsu, P.C.; Wu, M.T.; Yu, S.H. Epigenetic signatures of phthalate exposure and potential risks: A DNA methylation analysis using Infinium MethylationEPIC BeadChip. Environ. Epigenetics 2025, 11, dvaf020. [Google Scholar] [CrossRef] [PubMed]
- Wu, A.H.; Franke, A.A.; Wilkens, L.R.; Tseng, C.; Conroy, S.M.; Li, Y.; Polfus, L.M.; De Rouen, M.; Caberto, C.; Haiman, C.; et al. Urinary phthalate exposures and risk of breast cancer: The Multiethnic Cohort study. Breast Cancer Res. 2021, 23, 44. [Google Scholar] [CrossRef]
- Zhou, F.; Guo, C.; Wang, L.; Zhang, G.; Wang, J.; Chen, W.; Cui, K.; Tan, Y.; Zhou, Z. Mono-(2-ethylhexyl) Phthalate (MEHP)-Induced Telomere Structure and Function Disorder Mediates Cell Cycle Dysregulation and Apoptosis via c-Myc and Its Upstream Transcription Factors in a Mouse Spermatogonia-Derived (GC-1) Cell Line. Toxics 2023, 11, 448. [Google Scholar] [CrossRef]
- Dhaini, H.R.; Daher, Z. Genetic polymorphisms of PPAR genes and human cancers: Evidence for gene-environment interactions. J. Environ. Sci. Health C Environ. Carcinog. Ecotoxicol. Rev. 2019, 37, 146–179. [Google Scholar] [CrossRef] [PubMed]
- Ke, C.C.; Chen, L.C.; Yu, C.C.; Cheng, W.C.; Huang, C.Y.; Lin, V.C.; Lu, T.L.; Huang, S.P.; Bao, B.Y. Genetic Analysis Reveals a Significant Contribution of CES1 to Prostate Cancer Progression in Taiwanese Men. Cancers 2020, 12, 1346. [Google Scholar] [CrossRef]
- Liu, J.C.; Shen, W.C.; Shih, T.C.; Tsai, C.W.; Chang, W.S.; Cho, Y.; Tsai, C.H.; Bau, D.T. The current progress and future prospects of personalized radiogenomic cancer study. Biomedicine 2015, 5, 2. [Google Scholar] [CrossRef]
- Subramanian, A.; Tamayo, P.; Mootha, V.K.; Mukherjee, S.; Ebert, B.L.; Gillette, M.A.; Paulovich, A.; Pomeroy, S.L.; Golub, T.R.; Lander, E.S.; et al. Gene set enrichment analysis: A knowledge-based approach for interpreting genome-wide expression profiles. Proc. Natl. Acad. Sci. USA 2005, 102, 15545–15550. [Google Scholar] [CrossRef]
- Bao, B.Y.; Pao, J.B.; Huang, C.N.; Pu, Y.S.; Chang, T.Y.; Lan, Y.H.; Lu, T.L.; Lee, H.Z.; Juang, S.H.; Chen, L.M.; et al. Polymorphisms inside microRNAs and microRNA target sites predict clinical outcomes in prostate cancer patients receiving androgen-deprivation therapy. Clin. Cancer Res. 2011, 17, 928–936. [Google Scholar] [CrossRef] [PubMed]
- Ross, R.W.; Oh, W.K.; Xie, W.; Pomerantz, M.; Nakabayashi, M.; Sartor, O.; Taplin, M.E.; Regan, M.M.; Kantoff, P.W.; Freedman, M. Inherited variation in the androgen pathway is associated with the efficacy of androgen-deprivation therapy in men with prostate cancer. J. Clin. Oncol. 2008, 26, 842–847. [Google Scholar] [CrossRef] [PubMed]
- Chang, H.H.; Lee, C.H.; Chen, Y.T.; Huang, C.Y.; Yu, C.C.; Lin, V.C.; Geng, J.H.; Lu, T.L.; Huang, S.P.; Bao, B.Y. Genetic Analysis Reveals the Prognostic Significance of the DNA Mismatch Repair Gene MSH2 in Advanced Prostate Cancer. Cancers 2022, 14, 223. [Google Scholar] [CrossRef]
- Barrett, J.C.; Fry, B.; Maller, J.; Daly, M.J. Haploview: Analysis and visualization of LD and haplotype maps. Bioinformatics 2005, 21, 263–265. [Google Scholar] [CrossRef]
- Li, R.; Zhu, J.; Zhong, W.-D.; Jia, Z. PCaDB—A comprehensive and interactive database for transcriptomes from prostate cancer population cohorts. bioRxiv 2022. [Google Scholar] [CrossRef]
- Park, S.J.; Yoon, B.H.; Kim, S.K.; Kim, S.Y. GENT2: An updated gene expression database for normal and tumor tissues. BMC Med. Genom. 2019, 12, 101. [Google Scholar] [CrossRef]
- Yao, P.L.; Lin, Y.C.; Richburg, J.H. Mono-(2-ethylhexyl) phthalate (MEHP) promotes invasion and migration of human testicular embryonal carcinoma cells. Biol. Reprod. 2012, 86, 160–161. [Google Scholar] [CrossRef]
- Fan, P.; Li, Z.; Zuo, C.; Fang, M. Promotion effects of mono-2-ethyhexyl phthalate (MEHP) on migration and invasion of human melanoma cells via activation of TGF-β signals. Cell Biochem. Funct. 2020, 38, 38–46. [Google Scholar] [CrossRef]
- Wang, Z.; Shao, M.; Liu, Y. Promotion of Wilms’ tumor cells migration and invasion by mono-2-ethyhexyl phthalate (MEHP) via activation of NF-κB signals. Chem. Biol. Interact. 2017, 270, 1–8. [Google Scholar] [CrossRef]
- Yang, W.; Tan, W.; Zheng, J.; Zhang, B.; Li, H.; Li, X. MEHP promotes the proliferation of cervical cancer via GPER mediated activation of Akt. Eur. J. Pharmacol. 2018, 824, 11–16. [Google Scholar] [CrossRef]
- Liu, B.; Wu, Q.; Xuan, Z.; Zheng, Z.; Du, Y.; Sui, X.; Wu, H.; Zhang, Z.; Zhang, Z.; Zhong, M.; et al. Mechanisms Involved in Focal Adhesion Signaling Regulating Tumor Anoikis Resistance. Cancer Sci. 2025, 116, 2640–2648. [Google Scholar] [CrossRef]
- Maniero, C.; Scudieri, P.; Haris Shaikh, L.; Zhao, W.; Gurnell, M.; Galietta, L.J.V.; Brown, M.J. ANO4 (Anoctamin 4) Is a Novel Marker of Zona Glomerulosa That Regulates Stimulated Aldosterone Secretion. Hypertension 2019, 74, 1152–1159. [Google Scholar] [CrossRef] [PubMed]
- Leitzke, S.; Seidel, J.; Ahrens, B.; Schreiber, R.; Kunzelmann, K.; Sperrhacke, M.; Bhakdi, S.; Reiss, K. Influence of Anoctamin-4 and -9 on ADAM10 and ADAM17 Sheddase Function. Membranes 2022, 12, 123. [Google Scholar] [CrossRef] [PubMed]
- Al Sharie, A.H.; Al Zu’bi, Y.O.; El-Elimat, T.; Al-Kammash, K.; Abu Lil, A.; Isawi, I.H.; Al Sharie, S.; Abu Mousa, B.M.; Al Malkawi, A.A.; Alali, F.Q. ANO4 Expression Is a Potential Prognostic Biomarker in Non-Metastasized Clear Cell Renal Cell Carcinoma. J. Pers. Med. 2023, 13, 295. [Google Scholar] [CrossRef]
- Yu, C.C.; Chen, L.C.; Huang, C.Y.; Lin, V.C.; Lu, T.L.; Lee, C.H.; Huang, S.P.; Bao, B.Y. Genetic association analysis identifies a role for ANO5 in prostate cancer progression. Cancer Med. 2020, 9, 2372–2378. [Google Scholar] [CrossRef] [PubMed]
- Cavalu, S.; Abdelhamid, A.M.; Saber, S.; Elmorsy, E.A.; Hamad, R.S.; Abdel-Reheim, M.A.; Yahya, G.; Salama, M.M. Cell cycle machinery in oncology: A comprehensive review of therapeutic targets. FASEB J. 2024, 38, e23734. [Google Scholar] [CrossRef]
- Hwang, S.P.; Denicourt, C. The impact of ribosome biogenesis in cancer: From proliferation to metastasis. NAR Cancer 2024, 6, zcae017. [Google Scholar] [CrossRef]
- Pang, X.; He, X.; Qiu, Z.; Zhang, H.; Xie, R.; Liu, Z.; Gu, Y.; Zhao, N.; Xiang, Q.; Cui, Y. Targeting integrin pathways: Mechanisms and advances in therapy. Signal Transduct. Target. Ther. 2023, 8, 1. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Zhang, X.; Ben, T.; Li, M.; Jin, Y.; Wang, T.; Song, Y. Focal adhesion in the tumour metastasis: From molecular mechanisms to therapeutic targets. Biomark. Res. 2025, 13, 38. [Google Scholar] [CrossRef] [PubMed]
- Hong, S.Y.; Shih, Y.P.; Sun, P.; Hsieh, W.J.; Lin, W.C.; Lo, S.H. Down-regulation of tensin2 enhances tumorigenicity and is associated with a variety of cancers. Oncotarget 2016, 7, 38143–38153. [Google Scholar] [CrossRef] [PubMed]
- Ramirez, N.E.; Zhang, Z.; Madamanchi, A.; Boyd, K.L.; O’Rear, L.D.; Nashabi, A.; Li, Z.; Dupont, W.D.; Zijlstra, A.; Zutter, M.M. The α2β1 integrin is a metastasis suppressor in mouse models and human cancer. J. Clin. Investig. 2011, 121, 226–237. [Google Scholar] [CrossRef]
- Shih, Y.P.; Takada, Y.; Lo, S.H. Silencing of DLC1 upregulates PAI-1 expression and reduces migration in normal prostate cells. Mol. Cancer Res. 2012, 10, 34–39. [Google Scholar] [CrossRef]




Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Chang, C.-F.; Huang, S.-P.; Chen, Y.-T.; Chen, L.-C.; Huang, C.-Y.; Yu, C.-C.; Lin, V.C.; Lu, T.-L.; Bao, B.-Y. From Plastics to Prognosis: ANO4 Susceptibility Links Phthalate Exposure to Prostate Cancer Progression. Diagnostics 2026, 16, 794. https://doi.org/10.3390/diagnostics16050794
Chang C-F, Huang S-P, Chen Y-T, Chen L-C, Huang C-Y, Yu C-C, Lin VC, Lu T-L, Bao B-Y. From Plastics to Prognosis: ANO4 Susceptibility Links Phthalate Exposure to Prostate Cancer Progression. Diagnostics. 2026; 16(5):794. https://doi.org/10.3390/diagnostics16050794
Chicago/Turabian StyleChang, Chi-Fen, Shu-Pin Huang, Yei-Tsung Chen, Lih-Chyang Chen, Chao-Yuan Huang, Chia-Cheng Yu, Victor C. Lin, Te-Ling Lu, and Bo-Ying Bao. 2026. "From Plastics to Prognosis: ANO4 Susceptibility Links Phthalate Exposure to Prostate Cancer Progression" Diagnostics 16, no. 5: 794. https://doi.org/10.3390/diagnostics16050794
APA StyleChang, C.-F., Huang, S.-P., Chen, Y.-T., Chen, L.-C., Huang, C.-Y., Yu, C.-C., Lin, V. C., Lu, T.-L., & Bao, B.-Y. (2026). From Plastics to Prognosis: ANO4 Susceptibility Links Phthalate Exposure to Prostate Cancer Progression. Diagnostics, 16(5), 794. https://doi.org/10.3390/diagnostics16050794





