Comparison of Manual Multi-ROI, Single-Location, and Volumetric Fat Fraction Measurements for Hepatic Steatosis Using MRI
Abstract
1. Introduction
2. Materials and Methods
2.1. Patient Selection
2.2. Magnetic Resonance Imaging Protocol
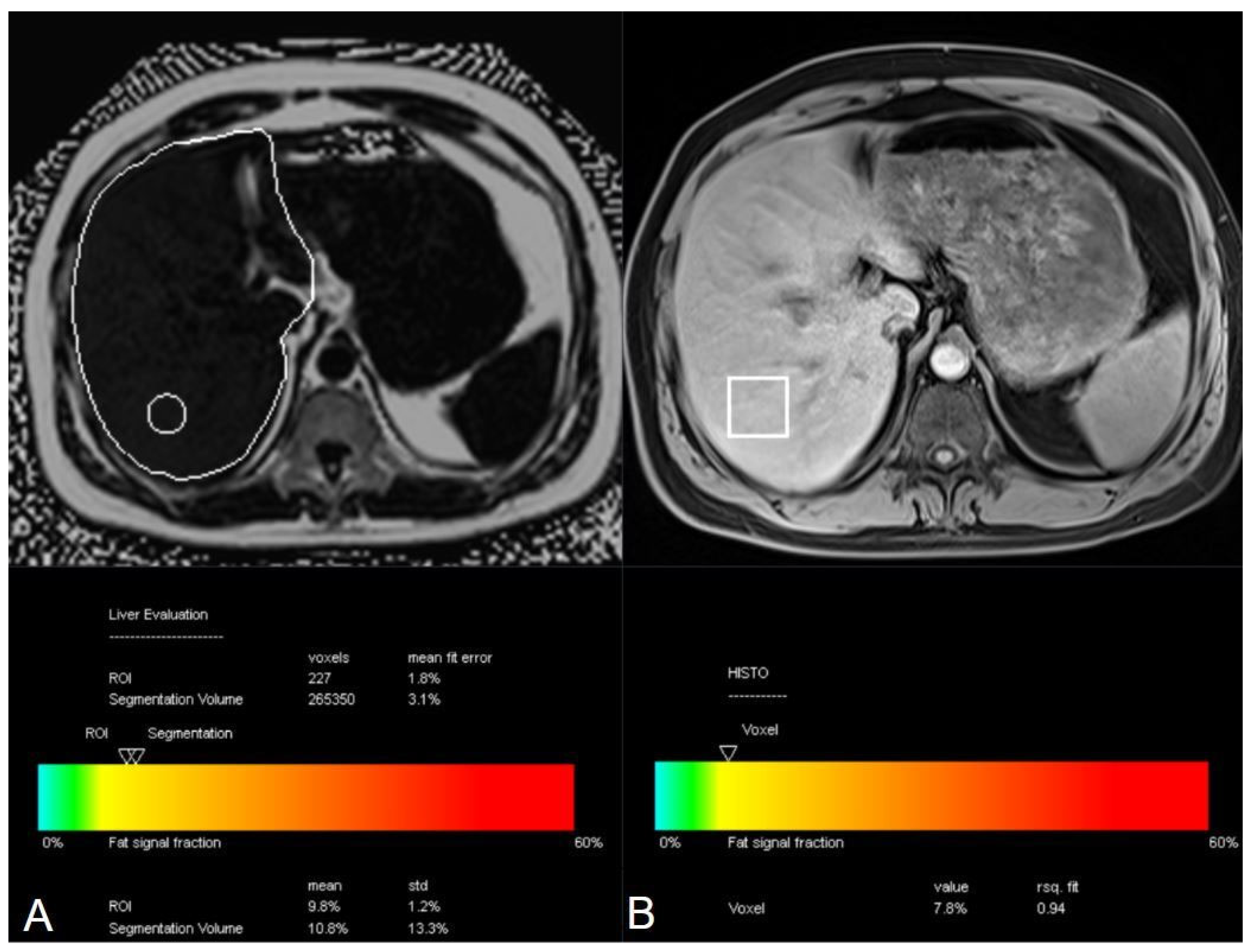
2.3. Image Analysis and Fat Fraction Measurements
2.4. Ethical Approval and Funding
2.5. Statistical Analysis
3. Results
3.1. Study Population Characteristics
3.2. Comparison of Fat Fraction Measurements Across Methods
3.3. Agreement in Steatosis Grading
3.4. Correlation Between the Techniques
3.5. Bland–Altman Agreement Analysis
3.6. Interobserver and Intraobserver Reliability
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| BMI | Body Mass Index |
| CI | Confidence Interval |
| ICC | Intraclass Correlation Coefficient |
| IQR | Interquartile Range |
| κ (Kappa) | Cohen’s Kappa Coefficient |
| LoA | Limits of Agreement |
| MASLD | Metabolic Dysfunction-Associated Steatotic Liver Disease |
| MRI | Magnetic Resonance Imaging |
| MRI-PDFF | Magnetic Resonance Imaging–Proton Density Fat Fraction |
| MRS | Magnetic Resonance Spectroscopy |
| PDFF | Proton Density Fat Fraction |
| QDixon | Quantitative Dixon |
| ROI | Region of Interest |
| S0–S3 | Steatosis Grades 0 to 3 |
| STEAM | Stimulated Echo Acquisition Mode |
| STROBE | Strengthening the Reporting of Observational Studies in Epidemiology |
| TE | Echo Time |
| TR | Repetition Time |
References
- European Association for the Study of the Liver (EASL); European Association for the Study of Diabetes (EASD); European Association for the Study of Obesity (EASO). EASL-EASD-EASO Clinical Practice Guidelines on the management of metabolic dysfunction-associated steatotic liver disease (MASLD). Obes. Facts 2024, 17, 374–444. [Google Scholar] [CrossRef]
- Eslam, M.; Newsome, P.N.; Sarin, S.K.; Anstee, Q.M.; Targher, G.; Romero-Gomez, M.; Zelber-Sagi, S.; Wong, V.W.-S.; Dufour, J.-F.; Schattenberg, J.M. A new definition for metabolic dysfunction-associated fatty liver disease: An international expert consensus statement. J. Hepatol. 2020, 73, 202–209. [Google Scholar] [CrossRef]
- Kanwal, F.; Neuschwander-Tetri, B.A.; Loomba, R.; Rinella, M.E. Metabolic dysfunction–associated steatotic liver disease: Update and impact of new nomenclature on the American Association for the Study of Liver Diseases practice guidance on nonalcoholic fatty liver disease. Hepatology 2024, 79, 1212–1219. [Google Scholar] [CrossRef]
- Loomba, R.; Friedman, S.L.; Shulman, G.I. Mechanisms and disease consequences of nonalcoholic fatty liver disease. Cell 2021, 184, 2537–2564. [Google Scholar] [CrossRef]
- Arab, J.P.; Arrese, M.; Trauner, M. Recent insights into the pathogenesis of nonalcoholic fatty liver disease. Annu. Rev. Pathol. Mech. Dis. 2018, 13, 321–350. [Google Scholar] [CrossRef] [PubMed]
- Taylor, R.S.; Taylor, R.J.; Bayliss, S.; Hagström, H.; Nasr, P.; Schattenberg, J.M.; Ishigami, M.; Toyoda, H.; Wong, V.W.-S.; Peleg, N. Association between fibrosis stage and outcomes of patients with nonalcoholic fatty liver disease: A systematic review and meta-analysis. Gastroenterology 2020, 158, 1611–1625.e1612. [Google Scholar] [CrossRef] [PubMed]
- Allen, A.M.; Therneau, T.M.; Larson, J.J.; Coward, A.; Somers, V.K.; Kamath, P.S. Nonalcoholic fatty liver disease incidence and impact on metabolic burden and death: A 20 year-community study. Hepatology 2018, 67, 1726–1736. [Google Scholar] [CrossRef] [PubMed]
- Caussy, C.; Reeder, S.B.; Sirlin, C.B.; Loomba, R. Noninvasive, quantitative assessment of liver fat by MRI-PDFF as an endpoint in NASH trials. Hepatology 2018, 68, 763–772. [Google Scholar] [CrossRef]
- Dag, N.; Kutlu, R. A to Z of Imaging Modalities for the Diagnosis and Quantification of Hepatic Steatosis. J. Inonu Liver Transplant. Inst. 2025, 3, 71–77. [Google Scholar] [CrossRef]
- Dag, N.; Sarici, B.; Igci, G.; Yagin, F.H.; Yilmaz, S.; Kutlu, R. Diagnostic Performance of Ultrasound-Based Liver Fat Quantification with Reference to Magnetic Resonance Imaging Proton Density Fat Fraction and Histology. J. Clin. Ultrasound 2025, 53, 1265–1271. [Google Scholar] [CrossRef]
- Tang, A.; Tan, J.; Sun, M.; Hamilton, G.; Bydder, M.; Wolfson, T.; Gamst, A.C.; Middleton, M.; Brunt, E.M.; Loomba, R. Nonalcoholic fatty liver disease: MR imaging of liver proton density fat fraction to assess hepatic steatosis. Radiology 2013, 267, 422–431. [Google Scholar] [CrossRef]
- Kukuk, G.M.; Hittatiya, K.; Sprinkart, A.M.; Eggers, H.; Gieseke, J.; Block, W.; Moeller, P.; Willinek, W.A.; Spengler, U.; Trebicka, J. Comparison between modified Dixon MRI techniques, MR spectroscopic relaxometry, and different histologic quantification methods in the assessment of hepatic steatosis. Eur. Radiol. 2015, 25, 2869–2879. [Google Scholar] [CrossRef] [PubMed]
- Idilman, I.S.; Aniktar, H.; Idilman, R.; Kabacam, G.; Savas, B.; Elhan, A.; Celik, A.; Bahar, K.; Karcaaltincaba, M. Hepatic steatosis: Quantification by proton density fat fraction with MR imaging versus liver biopsy. Radiology 2013, 267, 767–775. [Google Scholar] [CrossRef] [PubMed]
- Azizi, N.; Naghibi, H.; Shakiba, M.; Morsali, M.; Zarei, D.; Abbastabar, H.; Ghanaati, H. Evaluation of MRI proton density fat fraction in hepatic steatosis: A systematic review and meta-analysis. Eur. Radiol. 2025, 35, 1794–1807. [Google Scholar] [CrossRef] [PubMed]
- Gu, J.; Liu, S.; Du, S.; Zhang, Q.; Xiao, J.; Dong, Q.; Xin, Y. Diagnostic value of MRI-PDFF for hepatic steatosis in patients with non-alcoholic fatty liver disease: A meta-analysis. Eur. Radiol. 2019, 29, 3564–3573. [Google Scholar] [CrossRef]
- Guglielmo, F.F.; Barr, R.G.; Yokoo, T.; Ferraioli, G.; Lee, J.T.; Dillman, J.R.; Horowitz, J.M.; Jhaveri, K.S.; Miller, F.H.; Modi, R.Y. Liver fibrosis, fat, and iron evaluation with MRI and fibrosis and fat evaluation with US: A practical guide for radiologists. Radiographics 2023, 43, e220181. [Google Scholar] [CrossRef]
- Vandenbroucke, J.P.; Von Elm, E.; Altman, D.G.; Gøtzsche, P.C.; Mulrow, C.D.; Pocock, S.J.; Poole, C.; Schlesselman, J.J.; Egger, M.; Initiative, S. Strengthening the Reporting of Observational Studies in Epidemiology (STROBE): Explanation and elaboration. Int. J. Surg. 2014, 12, 1500–1524. [Google Scholar] [CrossRef]
- Kang, G.H.; Cruite, I.; Shiehmorteza, M.; Wolfson, T.; Gamst, A.C.; Hamilton, G.; Bydder, M.; Middleton, M.S.; Sirlin, C.B. Reproducibility of MRI-determined proton density fat fraction across two different MR scanner platforms. J. Magn. Reson. Imaging 2011, 34, 928–934. [Google Scholar] [CrossRef]
- Bonekamp, S.; Tang, A.; Mashhood, A.; Wolfson, T.; Changchien, C.; Middleton, M.S.; Clark, L.; Gamst, A.; Loomba, R.; Sirlin, C.B. Spatial distribution of MRI-determined hepatic proton density fat fraction in adults with nonalcoholic fatty liver disease. J. Magn. Reson. Imaging 2014, 39, 1525–1532. [Google Scholar] [CrossRef]
- Runge, J.H.; Smits, L.P.; Verheij, J.; Depla, A.; Kuiken, S.D.; Baak, B.C.; Nederveen, A.J.; Beuers, U.; Stoker, J. MR spectroscopy–derived proton density fat fraction is superior to controlled attenuation parameter for detecting and grading hepatic steatosis. Radiology 2018, 286, 547–556. [Google Scholar] [CrossRef]
- Kim, J.W.; Lee, C.H.; Yang, Z.; Kim, B.-H.; Lee, Y.-S.; Kim, K.A. The spectrum of magnetic resonance imaging proton density fat fraction (MRI-PDFF), magnetic resonance spectroscopy (MRS), and two different histopathologic methods (artificial intelligence vs. pathologist) in quantifying hepatic steatosis. Quant. Imaging Med. Surg. 2022, 12, 5251. [Google Scholar] [CrossRef] [PubMed]
- Martí-Aguado, D.; Jiménez-Pastor, A.; Alberich-Bayarri, Á.; Rodríguez-Ortega, A.; Alfaro-Cervello, C.; Mestre-Alagarda, C.; Bauza, M.; Gallén-Peris, A.; Valero-Pérez, E.; Ballester, M.P. Automated whole-liver MRI segmentation to assess steatosis and iron quantification in chronic liver disease. Radiology 2022, 302, 345–354. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.-H.; Zhao, Y.; Tian, S.-F.; Xie, L.-H.; Chen, L.-H.; Chen, A.-L.; Wang, N.; Song, Q.-W.; Zhang, H.-N.; Xie, L.-Z. Hepatic fat quantification of magnetic resonance imaging whole-liver segmentation for assessing the severity of nonalcoholic fatty liver disease: Comparison with a region of interest sampling method. Quant. Imaging Med. Surg. 2021, 11, 2933. [Google Scholar] [CrossRef] [PubMed]
- Cao, D.; Yang, Y.; Li, M.; Liu, Y.; Yang, D.; Xu, H.; Lv, H.; Zhang, Z.; Zhang, P.; Jia, X. Quantitative Comparison of Liver Volume, Proton Density Fat Fraction, and Time Burden between Automatic Whole Liver Segmentation and Manual Sampling MRI Strategies for Diagnosing Metabolic Dysfunction-associated Steatotic Liver Disease in Obese Patients. Curr. Med. Imaging 2024, 20, e15734056282249. [Google Scholar] [CrossRef] [PubMed]
- Brancato, V.; Della Pepa, G.; Bozzetto, L.; Vitale, M.; Annuzzi, G.; Basso, L.; Cavaliere, C.; Salvatore, M.; Rivellese, A.A.; Monti, S. Evaluation of a whole-liver dixon-based MRI approach for quantification of liver fat in patients with type 2 diabetes treated with two isocaloric different diets. Diagnostics 2022, 12, 514. [Google Scholar] [CrossRef]
- Campo, C.A.; Hernando, D.; Schubert, T.; Bookwalter, C.A.; Pay, A.J.V.; Reeder, S.B. Standardized approach for ROI-based measurements of proton density fat fraction and R2* in the liver. Am. J. Roentgenol. 2017, 209, 592–603. [Google Scholar] [CrossRef]
- Li, Y.-W.; Jiao, Y.; Chen, N.; Gao, Q.; Chen, Y.-K.; Zhang, Y.-F.; Wen, Q.-P.; Zhang, Z.-M. How to select the quantitative magnetic resonance technique for subjects with fatty liver: A systematic review. World J. Clin. Cases 2022, 10, 8906. [Google Scholar] [CrossRef]
- Al-Huneidi, L.I.; Zhao, F.; Maas, R.; Ermans, S.J.; Runge, J.; Chen, X.; Ji, J.X.; Alic, L. Liver Fat Quantification and Steatosis Grading in Fatty Liver Disease by Magnetic Resonance Imaging: Systematic Review and Meta-Analysis. J. Gastroenterol. Hepatol. 2025, 40, 2808–2819. [Google Scholar] [CrossRef]
- Fortier, V.; Mohamed, A.; McNabb, E.; Dana, J.; Zakarian, R.; Levesque, I.R.; Reinhold, C. R2* Impact on Hepatic Fat Quantification with a Commercial Single Voxel Technique at 1.5 and 3.0 T. Can. Assoc. Radiol. J. 2024, 75, 838–846. [Google Scholar] [CrossRef]
- Hu, F.; Yang, R.; Huang, Z.; Wang, M.; Yuan, F.; Xia, C.; Wei, Y.; Song, B. 3D Multi-Echo Dixon technique for simultaneous assessment of liver steatosis and iron overload in patients with chronic liver diseases: A feasibility study. Quant. Imaging Med. Surg. 2019, 9, 1014. [Google Scholar] [CrossRef]
- Starekova, J.; Rutkowski, D.; Bae, W.C.; Do, H.; Madhuranthakam, A.J.; Malis, V.; Lin, S.Q.; Serai, S.; Yokoo, T.; Reeder, S.B. Multi-Center, Multi-Vendor Validation of Simultaneous MRI-Based Proton Density Fat Fraction and R2* Mapping Using a Combined Proton Density Fat Fraction-R2* Phantom. J. Magn. Reson. Imaging 2025, 62, 800–811. [Google Scholar] [CrossRef]
- Elfaal, M.; Supersad, A.; Ferguson, C.; Locas, S.; Manolea, F.; Wilson, M.P.; Sam, M.; Tu, W.; Low, G. Two-point Dixon and six-point Dixon magnetic resonance techniques in the detection, quantification and grading of hepatic steatosis. World J. Radiol. 2023, 15, 293. [Google Scholar] [CrossRef]
- Kang, B.-k.; Kim, M.; Song, S.-Y.; Jun, D.W.; Jang, K. Feasibility of modified Dixon MRI techniques for hepatic fat quantification in hepatic disorders: Validation with MRS and histology. Br. J. Radiol. 2018, 91, 20170378. [Google Scholar] [CrossRef]







| Variable | Value (n = 490) |
|---|---|
| Age (years), mean ± SD (range) | 52.6 ± 15.4 (18–90) |
| Sex, n (%) | |
| –Female | 244 (49.8) |
| –Male | 246 (50.2) |
| Height (cm), mean ± SD (range) | 169.1 ± 7.2 (110–192) |
| Weight (kg), mean ± SD (range) | 75.5 ± 11.3 (45–130) |
| Body mass index (kg/m2), median (IQR) | 25.86 (24.22–28.67) |
| BMI categories, n (%) | |
| –Normal weight | 209 (42.7) |
| –Overweight | 207 (42.2) |
| –Obese | 74 (15.1) |
| Measurement Method | Median | IQR | Median Difference | p-Value |
|---|---|---|---|---|
| manual multi-ROI MRI-PDFF | 2.95 | 2.00–5.50 | – | – |
| single-ROI MRI-PDFF | 3.60 | 2.20–7.05 | +0.20 | <0.001 |
| volumetric MRI-PDFF | 6.00 | 4.10–9.63 | +1.90 | <0.001 |
| single-voxel MRS | 2.80 | 1.30–6.90 | −0.70 | <0.001 |
| Measurement Method | Exact Agreement (%) | Weighted κ (95% CI) | Predominant Direction of Disagreement | Non-Adjacent Class Shift (%) |
|---|---|---|---|---|
| single-ROI MRI-PDFF | 86.9 | 0.82 (excellent) | S0 → S1, S1 → S2 | <1% |
| volumetric MRI-PDFF | 76.1 | 0.68 (substantial) | S1 → S2, S2 → S3 (overestimation) | 3–5% |
| single-voxel MRS | 87.4 | 0.83 (excellent) | S0 → S1, S1 → S2 | <1% |
| Comparison | Spearman’s ρ | p-Value |
|---|---|---|
| manual multi-ROI MRI-PDFF vs. single-ROI MRI-PDFF | 0.72 | <0.001 |
| manual multi-ROI MRI-PDFF vs. volumetric MRI-PDFF | 0.74 | <0.001 |
| manual multi-ROI MRI-PDFF vs. single-voxel MRS | 0.75 | <0.001 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Dag, N.; Bilaloğlu, M.K.; Kutlu, R. Comparison of Manual Multi-ROI, Single-Location, and Volumetric Fat Fraction Measurements for Hepatic Steatosis Using MRI. Diagnostics 2026, 16, 716. https://doi.org/10.3390/diagnostics16050716
Dag N, Bilaloğlu MK, Kutlu R. Comparison of Manual Multi-ROI, Single-Location, and Volumetric Fat Fraction Measurements for Hepatic Steatosis Using MRI. Diagnostics. 2026; 16(5):716. https://doi.org/10.3390/diagnostics16050716
Chicago/Turabian StyleDag, Nurullah, Muhammed Kürşat Bilaloğlu, and Ramazan Kutlu. 2026. "Comparison of Manual Multi-ROI, Single-Location, and Volumetric Fat Fraction Measurements for Hepatic Steatosis Using MRI" Diagnostics 16, no. 5: 716. https://doi.org/10.3390/diagnostics16050716
APA StyleDag, N., Bilaloğlu, M. K., & Kutlu, R. (2026). Comparison of Manual Multi-ROI, Single-Location, and Volumetric Fat Fraction Measurements for Hepatic Steatosis Using MRI. Diagnostics, 16(5), 716. https://doi.org/10.3390/diagnostics16050716







