If You Care About Autonomic Modulation—Do Not Let Seizure Seizure
Abstract
1. Introduction
2. Materials and Methods
2.1. Patient Selection
2.2. Clinical and Epilepsy Parameters
2.3. Parameters of Autonomic Cardiovascular Modulation
2.4. Statistical Analysis
- Disease localization, i.e., right- or left hemispheric TLE
- Seizure frequency < 1 vs. >1/month, and disease duration < 10 vs. >10 years
- Number of ASM
- Age, i.e., individuals younger than 30 years and those older than 30 years
3. Results
3.1. Baseline Characteristics
3.2. Association Between Autonomic Modulation and Hemispheric Localization of TLE
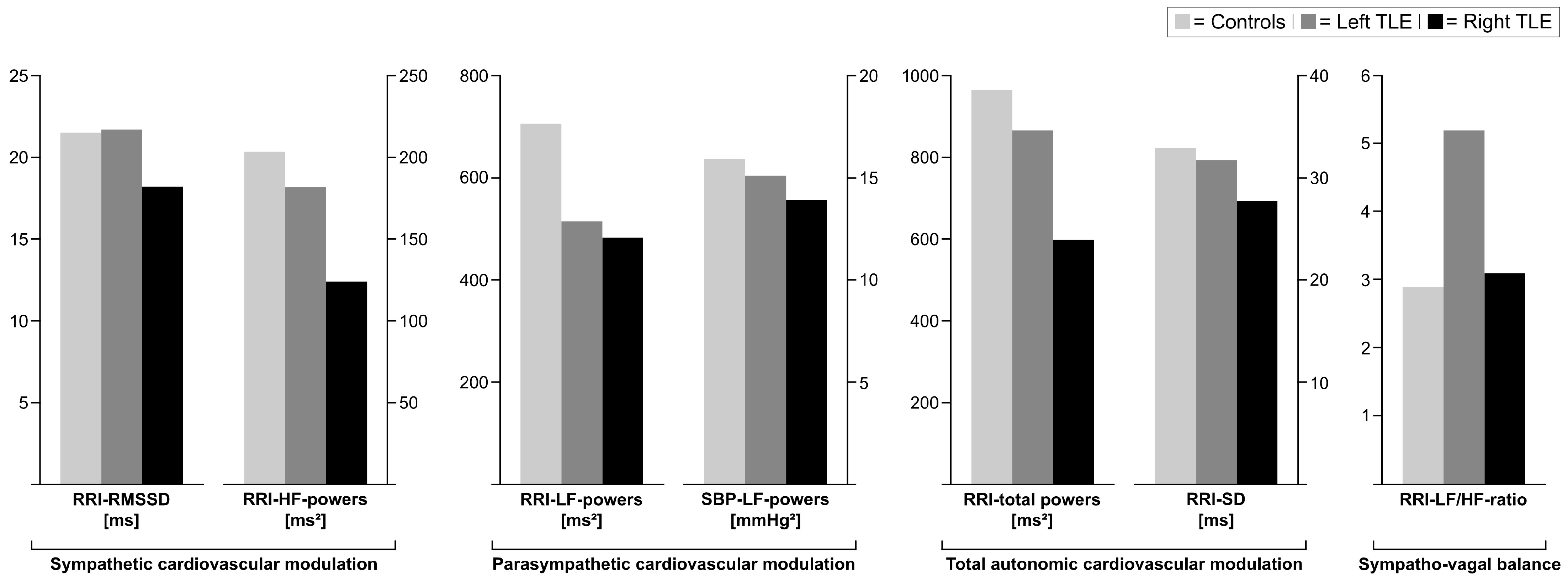
3.3. Association Between Autonomic Modulation, Disease Duration, and Seizure Frequency
3.4. Association Between Autonomic Modulation and Antiseizure Medication
3.5. Association Between Autonomic Modulation and Age
4. Discussion
4.1. Summary of Key Findings
- A trend toward lower values of sympathetic modulation in patients with right TLE suggests that the localization of the epileptic focus influences autonomic cardiovascular modulation, with structural alterations in sympathetic regulation predominantly mediated by the right hemisphere.
- Given the comparable alterations in autonomic cardiovascular modulation observed across age-matched subgroups, the differences between epilepsy patients and healthy controls cannot be explained solely by physiological, age-associated changes.
- A decrease in parasympathetic modulation with increasing seizure frequency underscores the importance of sufficient seizure control in order to prevent autonomic complications. The absence of significant associations between disease duration and alterations in autonomic modulation suggests that epilepsy exerts an early and clinically relevant effect on the autonomic nervous system.
- Although the interpretability is limited by the small sample size, the findings indicate that antiseizure medication alone does not account for the observed autonomic dysregulation in epilepsy patients. Comparable alterations in autonomic modulation were observed both in a patient without antiepileptic medication and in patients undergoing polytherapy with at least three different agents.
4.2. Influence of TLE Localization on Autonomic Cardiovascular Modulation
4.3. Influence of (Patient) Age on Autonomic Cardiovascular Modulation
4.4. Influence of Seizure Frequency and Disease Duration on Autonomic Cardiovascular Modulation
4.5. Influence of Antiseizure Medication on Autonomic Cardiovascular Modulation
4.6. SUDEP
4.7. Limitations
4.8. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| ASM | antiseizure medication |
| TLE | temporal lobe epilepsy |
| RRI | R-R-interval |
| BP | blood pressure |
| CV | coefficient of variation |
| RMSSD | root mean square of successive differences |
| LF | low frequency |
| HF | high frequency |
| SBP | systolic blood pressure |
| SD | standard deviation |
| SeLEAS | self-limited epilepsy with autonomic seizures |
| SUDEP | sudden unexpected death in epilepsy |
| EEG | electroencephalography |
| rTLE | right temporal lobe epilepsy |
| lTLE | left temporal lobe epilepsy |
| DBP | diastolic blood pressure |
| Ct | controls |
| HRV | heart rate variability |
References
- Beghi, E. The Epidemiology of Epilepsy. Neuroepidemiology 2020, 54, 185–191. [Google Scholar] [CrossRef]
- Thijs, R.D.; Surges, R.; O’Brien, T.J.; Sander, J.W. Epilepsy in adults. Lancet 2019, 393, 689–701. [Google Scholar] [CrossRef] [PubMed]
- Akamatsu, N. Progress and Challenges in Epilepsy Treatment. Brain Nerve. 2024, 76, 917–921. (In Japanese) [Google Scholar] [CrossRef] [PubMed]
- Kanner, A.M.; Bicchi, M.M. Antiseizure Medications for Adults with Epilepsy: A Review. JAMA 2022, 327, 1269–1281. [Google Scholar] [CrossRef] [PubMed]
- Tomson, T.; Zelano, J.; Dang, Y.L.; Perucca, P. The pharmacological treatment of epilepsy in adults. Epileptic Disord. 2023, 25, 649–669. [Google Scholar] [CrossRef]
- Zaccara, G.; Lattanzi, S.; Brigo, F. Cardiac adverse effects of antiseizure medications. Expert Opin. Drug Saf. 2022, 21, 641–652. [Google Scholar] [CrossRef]
- D’Agnano, D.; Cernigliaro, F.; Ferretti, A.; Lo Cascio, S.; Correnti, E.; Terrin, G.; Santangelo, A.; Bellone, G.; Raieli, V.; Sciruicchio, V.; et al. The Role of the Autonomic Nervous System in Epilepsy and Migraine: A Narrative Review. J. Integr. Neurosci. 2024, 23, 128. [Google Scholar] [CrossRef]
- Barot, N.; Nei, M. Autonomic aspects of sudden unexpected death in epilepsy (SUDEP). Clin. Auton. Res. 2019, 29, 151–160. [Google Scholar] [CrossRef] [PubMed]
- Hilz, M.J.; Dütsch, M.; Perrine, K.; Nelson, P.K.; Rauhut, U.; Devinsky, O. Hemispheric influence on autonomic modulation and baroreflex sensitivity. Ann. Neurol. 2001, 49, 575–584. [Google Scholar] [CrossRef]
- Ghchime, R.; Benjelloun, H.; Kiai, H.; Belaidi, H.; Lahjouji, F.; Ouazzani, R. Cerebral Hemispheric Lateralization Associated with Hippocampal Sclerosis May Affect Interictal Cardiovascular Autonomic Functions in Temporal Lobe Epilepsy. Epilepsy Res. Treat. 2016, 2016, 7417540. [Google Scholar] [CrossRef]
- Choudhary, N.; Deepak, K.K.; Chandra, P.S.; Bhatia, S.; Sagar, R.; Jaryal, A.K.; Pandey, R.M.; Tripathi, M. Comparison of Autonomic Function before and after Surgical Intervention in Patients with Temporal Lobe Epilepsy. J. Epilepsy Res. 2017, 7, 89–98. [Google Scholar] [CrossRef] [PubMed]
- Dono, F.; Evangelista, G.; Frazzini, V.; Vollono, C.; Carrarini, C.; Russo, M.; Ferrante, C.; Di Stefano, V.; Marchionno, L.P.; De Angelis, M.V.; et al. Interictal Heart Rate Variability Analysis Reveals Lateralization of Cardiac Autonomic Control in Temporal Lobe Epilepsy. Front. Neurol. 2020, 11, 842. [Google Scholar] [CrossRef] [PubMed]
- Ansakorpi, H.; Korpelainen, J.T.; Suominen, K.; Tolonen, U.; Myllylä, V.V.; Isojärvi, J.I. Interictal cardiovascular autonomic responses in patients with temporal lobe epilepsy. Epilepsia 2000, 41, 42–47. [Google Scholar] [CrossRef] [PubMed]
- Goit, R.K.; Jha, S.K.; Pant, B.N. Alteration of cardiac autonomic function in patients with newly diagnosed epilepsy. Physiol. Rep. 2016, 4, e12826. [Google Scholar] [CrossRef][Green Version]
- Ansakorpi, H.; Korpelainen, J.T.; Huikuri, H.V.; Tolonen, U.; Myllylä, V.V.; Isojärvi, J.I. Heart rate dynamics in refractory and well controlled temporal lobe epilepsy. J. Neurol. Neurosurg. Psychiatry 2002, 72, 26–30. [Google Scholar] [CrossRef]
- Dütsch, M.; Hilz, M.J.; Devinsky, O. Impaired baroreflex function in temporal lobe epilepsy. J. Neurol. 2006, 253, 1300–1308. [Google Scholar] [CrossRef]
- Hennessy, M.J.; Tighe, M.G.; Binnie, C.D.; Nashef, L. Sudden withdrawal of carbamazepine increases cardiac sympathetic activity in sleep. Neurology 2001, 57, 1650–1654. [Google Scholar] [CrossRef]
- Leosuthamas, D.; Limotai, C.; Unwanatham, N.; Rattanasiri, S. Is anti-seizure medication the culprit of SUDEP? Neurol. Sci. 2023, 44, 3659–3668. [Google Scholar] [CrossRef]
- Sathyaprabha, T.N.; Koot, L.A.M.; Hermans, B.H.M.; Adoor, M.; Sinha, S.; Kramer, B.W.; Raju, T.R.; Satishchandra, P.; Delhaas, T. Effects of Chronic Carbamazepine Treatment on the ECG in Patients with Focal Seizures. Clin. Drug Investig. 2018, 38, 845–851. [Google Scholar] [CrossRef]
- Dono, F.; Evangelista, G.; Consoli, S.; Rodorigo, D.; Russo, M.; Carrarini, C.; Di Pietro, M.; De Angelis, M.V.; Faustino, M.; Anzellotti, F.; et al. Perampanel enhances the cardiovagal tone and heart rate variability (HRV) in patients with drug-resistant temporal lobe epilepsy. Seizure 2022, 99, 16–23. [Google Scholar] [CrossRef]
- Izzi, F.; Placidi, F.; Liguori, C.; Posca, I.; Lauretti, B.; Diomedi, M.; Pisani, A.; Mercuri, N.B.; Rocchi, C. Autonomic functions in focal epilepsy: A comparison between lacosamide and carbamazepine monotherapy. J. Neurol. Sci. 2020, 418, 117095. [Google Scholar] [CrossRef]
- Holtkamp, M.; May, T.W.; Berkenfeld, R.; Bien, C.G.; Coban, I.; Knake, S.; Michaelis, R.; Rémi, J.; Seeck, M.; Surges, R.; et al. First epileptic seizure and epilepsies in adulthood. Clin. Epileptol. 2024, 37, 87–107. [Google Scholar] [CrossRef] [PubMed]
- Task Force of the European Society of Cardiology and the North American Society of Pacing and Electrophysiology. Heart rate variability: Standards of measurement, physiological inter pretation and clinical use. Circulation 1996, 93, 1043–1065. [Google Scholar] [CrossRef]
- Hilz, M.J.; Dütsch, M. Quantitative studies of autonomic function. Muscle Nerve 2006, 33, 6–20. [Google Scholar] [CrossRef] [PubMed]
- Seifert, F.; Kallmünzer, B.; Gutjahr, I.; Breuer, L.; Winder, K.; Kaschka, I.; Kloska, S.; Doerfler, A.; Hilz, M.J.; Schwab, S.; et al. Neuroanatomical correlates of severe cardiac arrhythmias in acute ischemic stroke. J. Neurol. 2015, 262, 1182–1190. [Google Scholar] [CrossRef]
- Fontes, M.A.P.; Dos Santos Machado, L.R.; Viana, A.C.R.; Cruz, M.H.; Nogueira, Í.S.; Oliveira, M.G.L.; Neves, C.B.; Godoy, A.C.V.; Henderson, L.A.; Macefield, V.G. The insular cortex, autonomic asymmetry and cardiovascular control: Looking at the right side of stroke. Clin. Auton. Res. 2024, 34, 549–560. [Google Scholar] [CrossRef]
- Boettger, M.K.; Schulz, S.; Berger, S.; Tancer, M.; Yeragani, V.K.; Voss, A.; Bär, K.J. Influence of age on linear and nonlinear measures of autonomic cardiovascular modulation. Ann. Noninvasive Electrocardiol. 2010, 15, 165–174. [Google Scholar] [CrossRef]
- Athira, S.B.; Pal, P.; Nair, P.P.; Nanda, N.; Aghoram, R. Cardiovascular autonomic function and baroreflex sensitivity in drug-resistant temporal lobe epilepsy. Epilepsy Behav. 2023, 138, 109013. [Google Scholar] [CrossRef] [PubMed]
- Devinsky, O.; Hesdorffer, D.C.; Thurman, D.J.; Lhatoo, S.; Richerson, G. Sudden unexpected death in epilepsy: Epidemiology, mechanisms, and prevention. Lancet Neurol. 2016, 15, 1075–1088. [Google Scholar] [CrossRef]
- Sun, X.; Lv, Y.; Lin, J. The mechanism of sudden unexpected death in epilepsy: A mini review. Front. Neurol. 2023, 14, 1137182. [Google Scholar] [CrossRef]
- Lin, S.; Schwartz, T.H.; Pitt, G.S. Sudden unexpected death in epilepsy: Respiratory vs. cardiac contributions. Cardiovasc. Res. 2025, 120, 2385–2394. [Google Scholar] [CrossRef]
- Taiwo, R.O.; Goldberg, H.S.; Ilouz, N.; Singh, P.K.; Shekh-Ahmad, T.; Levite, M. Enigmatic intractable Epilepsy patients have antibodies that bind glutamate receptor peptides, kill neurons, damage the brain, and cause Generalized Tonic Clonic Seizures. J. Neural Transm. 2025, 132, 663–688. [Google Scholar] [CrossRef]
- Ryvlin, P.; Nashef, L.; Lhatoo, S.D.; Bateman, L.M.; Bird, J.; Bleasel, A.; Boon, P.; Crespel, A.; Dworetzky, B.A.; Høgenhaven, H.; et al. Incidence and mechanisms of cardiorespiratory arrests in epilepsy monitoring units (MORTEMUS): A retrospective study. Lancet Neurol. 2013, 12, 966–977. [Google Scholar] [CrossRef]
| Controls (n = 30) | All Patients with TLE (n = 31) | Patients with rTLE (n = 12) | Patients with lTLE (n = 19) | p-Values (rTLE vs. lTLE) | |
|---|---|---|---|---|---|
| Age [years], Median (IQR) | 38.0 (28.0–52.0) | 36.0 (27.0–46.0) | 37.5 (28.0–56.5) | 32.0 (25.0–43.0) | 0.22 |
| Age split | - | ||||
| <30 years, n (%) | 11 (35.5) | 3 (25.0) | 8 (42.1) | 0.45 | |
| <30 years, Median (IQR) | 25.0 (21.0–28.0) | 27.0 (-) | 23.5 (21.0–28.8) | 0.61 | |
| ≥30 years, n (%) | 20 (64.5) | 9 (75.0) | 11 (57.9) | 0.45 | |
| ≥30 years, Median (IQR) | 42.0 (36.3–52.0) | 51.0 (35.5–61.5) | 41.0 (37.0–46.0) | 0.52 | |
| Female sex, n (%) | 16 (53.3) | 16 (51.6) | 5 (41.7) | 11 (57.9) | 0.47 |
| Disease duration | - | ||||
| <10 years, n (%) | 13 (41.9) | 7 (58.3) | 6 (31.6) | 0.26 | |
| ≥10 years, n (%) | 18 (58.1) | 5 (41.7) | 13 (68.4) | 0.26 | |
| Seizure frequency | - | ||||
| daily, n (%) | 2 (6.5) | 1 (8.3) | 1 (5.3) | 1 | |
| weekly, n (%) | 3 (9.7) | 1 (8.3) | 2 (10.5) | 1 | |
| monthly, n (%) | 10 (32.3) | 4 (33.4) | 6 (31.6) | 1 | |
| yearly, n (%) | 7 (22.6) | 3 (25.0) | 4 (21.1) | 1 | |
| rarely/unknown, n (%) | 8 (25.8) | 3 (25.0) | 6 (31.6) | 1 | |
| Number of ASM | - | ||||
| none, n (%) | 1 (3.2) | 1 (8.3) | 0 (0.0) | 0.39 | |
| 1 ASM, n (%) | 6 (19.4) | 2 (16.7) | 4 (21.1) | 1 | |
| 2 ASM, n (%) | 16 (51.6) | 5 (41.7) | 11 (58.0) | 0.47 | |
| 3 ASM, n (%) | 8 (25.8) | 4 (33.3) | 4 (21.1) | 0.68 | |
| ASM premedication | - | ||||
| SCB, n (%) | 23 (74.2) | 9 (75.0) | 14 (73.7) | 1 | |
| Dual SCB therapy, n (%) | 8 (25.8) | 3 (25.0) | 5 (26.3) | 1 | |
| Levetiracetam, n (%) | 15 (58.4) | 5 (25.0) | 10 (52.6) | 0.72 | |
| Brivaracetam, n (%) | 2 (6.5) | 1 (8.3) | 1 (5.3) | 1 | |
| Valproate, n (%) | 2 (6.5) | 0 (0.0) | 2 (10.5) | 0.51 | |
| Topiramate, n (%) | 1 (3.2) | 0 (0.0) | 1 (5.3) | 1 | |
| Zonisamide, n (%) | 4 (12.9) | 3 (25.0) | 4 (21.1) | 1 | |
| Perampanel, n (%) | 1 (3.2) | 0 (0.0) | 1 (5.3) | 1 | |
| Pregabalin, n (%) | 1 (3.2) | 1 (8.3) | 0 (0.0) | 0.39 | |
| SCBs | - | ||||
| Lamotrigine, n (%) | 12 (38.7) | 4 (33.4) | 8 (42.1) | 0.72 | |
| Lacosamide, n (%) | 12 (38.7) | 5 (41.7) | 7 (36.8) | 1 | |
| Carbamazepine, n (%) | 1 (3.2) | 1 (8.3) | 0 (0.0) | 0.39 | |
| Oxcarbazepine, n (%) | 5 (16.1) | 1 (8.3) | 4 (21.1) | 0.63 | |
| Eslicarbazepine, n (%) | 4 (12.9) | 3 (25.0) | 1 (5.3) | 0.27 | |
| Drug-resistant epilepsy, n (%) | 21 (67.7) | 9 (75.0) | 12 (63.2) | 0.70 |
| Controls (n = 30) | Patients with rTLE (n = 12) | Patients with rTLE (n = 19) | p-Values (Ct vs. lTLE) | p-Values (Ct vs. lTLE) | p-Values (rTLE vs. lTLE) | |
|---|---|---|---|---|---|---|
| Biosignals, Median (IQR) | ||||||
| mean RRI [ms] | 754.1 (723.9–865.9) | 727.9 (647.2–847.2) | 787.4 (675.5–847.1) | 1.0 | 0.22 | 0.50 |
| mean SBP [ms] | 127.2 (122.0–139.3) | 120.8 (113.0–139.4) | 121.7 (112.6–130.7) | 0.05 | 0.33 | 0.63 |
| Parasympathetic modulation | ||||||
| RRI-RMSSD [ms] | 21.5 (17.2–35.6) | 18.2 (7.5–29.6) | 21.7 (11.4–29.3) | 0.31 | 0.15 | 0.72 |
| RRI-HF powers [ms2] | 203.4 (85.3–498.1) | 124.0 (29.0–450.5) | 181.7 (38.5–407.0) | 0.47 | 0.32 | 0.87 |
| Sympathetic modulation | ||||||
| RRI-LF powers [ms2] | 705.7 (362.2–2024.3) | 482.4 (98.4–837.0) | 514.9 (316.1–1409.4) | 0.23 | 0.07 | 0.47 |
| SBP-LF powers [mmHg2] | 15.9 (9.9–30.8) | 13.9 (6.2–26.2) | 15.1 (6.6–21.4) | 0.23 | 0.30 | 0.81 |
| Total autonomic modulation | ||||||
| RRI total powers [ms2] | 964.6 (437.3–2521.8) | 597.8 (145.8–1352.0) | 866.0 (360.5–1506.8) | 0.33 | 0.10 | 0.54 |
| RRI-SD [ms] | 32.9 (24.0–52.9) | 27.7 (13.3–36.6) | 31.7 (21.4–43.0) | 0.34 | 0.09 | 0.52 |
| RRI-CV [%] | 4.4 (3.2–6.7) | 3.3 (2.2–4.8) | 4.1 (2.7–5.1) | 0.24 | 0.11 | 0.52 |
| Sympatho-vagal balance | ||||||
| RRI-LF/HF-ratio | 2.9 (1.8–4.5) | 3.1 (1.5–7.5) | 5.2 (2.4–10.4) | 0.25 | 0.08 | 0.72 |
| (a) | ||||||||||||||||||
| Controls (n = 30) | Patients with rTLE <10 a (n = 7) | Patients with rTLE > 10 a (n = 5) | Patients with lTLE < 10 a (n = 6) | Patients with lTLE > 10 a (n = 13) | ||||||||||||||
| Biosignals, Median (IQR) | ||||||||||||||||||
| mean RRI [ms] | 754.1 (723.9–865.9) | 730.6 (573.7–856.3) | 725.2 (678.7–903.7) | 757.9 (669.4–842.3) | 793.4 (663.4–921.9) | |||||||||||||
| mean SBP [ms] | 127.2 (122.0–139.3) | 125.4 (104.4–144.8) | 115.8 (113.4–136.2) | 126.5 (119.4–130.2) | 118.8 (107.1–131.1) | |||||||||||||
| Parasympathetic modulation | ||||||||||||||||||
| RRI-RMSSD [ms] | 21.5 (17.2–35.6) | 15.6 (7.4–22.2) | 23.9 (6.8–33.5) | 17.1 (6.7–24.5) | 22.6 (11.6–30.8) | |||||||||||||
| RRI-HF powers [ms2] | 203.4 (85.3–498.1) | 63.1 (45.8–239.1) | 301.5 (16.9–665.6) | 94.3 (17.6–435.5) | 189.0 (41.5–390.4) | |||||||||||||
| Sympathetic modulation | ||||||||||||||||||
| RRI-LF powers [ms2] | 705.7 (362.2–2024.3) | 371.7 (129.0–872.1) | 673.6 (79.9–1635.6) | 644.3 (58.8–1582.8) | 514.9 (357.7–1356.1) | |||||||||||||
| SBP-LF powers [mmHg2] | 15.9 (9.9–30.8) | 12.8 (6.3–26.1) | 19.9 (4.7–42.2) | 9.6 (5.5–22.6) | 17.5 (6.8–21.1) | |||||||||||||
| Total autonomic modulation | ||||||||||||||||||
| RRI total powers [ms2] | 964.6 (437.3–2521.8) | 556.7 (152.4–1111.2) | 1033.1 (96.8–2272.1) | 901.8 (76.4–1773.4) | 790.1 (454.9–1611.2) | |||||||||||||
| RRI-SD [ms] | 32.9 (24.0–52.9) | 26.2 (14.5–35.2) | 33.4 (11.6–49.2) | 33.0 (10.0–46.7) | 31.5 (23.7–41.3) | |||||||||||||
| RRI-CV [%] | 4.4 (3.2–6.7) | 3.2 (2.2–4.8) | 3.4 (1.7–6.7) | 4.5 (1.4–5.8) | 4.0 (2.8–5.5) | |||||||||||||
| Sympatho-vagal balance | ||||||||||||||||||
| RRI-LF/HF-ratio | 2.9 (1.8–4.5) | 3.8 (1.7–4.7) | 2.3 (1.7–3.9) | 2.3 (1.3–5.0) | 5.0 (1.6–10.6) | |||||||||||||
| p-Values (Ct vs. rTLE < 10 a) | p-Values (Ct vs. rTLE > 10 a) | p-Values (Ct vs. lTLE < 10 a) | p-Values (Ct vs. lTLE > 10 a) | p-Values (rTLE < 10 a vs. >10 a) | p-Values (lTLE < 10 a vs. >10 a) | |||||||||||||
| Biosignals, Median (IQR) | ||||||||||||||||||
| mean RRI [ms] | 0.28 | 0.45 | 0.64 | 0.71 | 0.69 | 0.54 | ||||||||||||
| mean SBP [ms] | 0.64 | 0.28 | 0.45 | 0.04 | 0.69 | 0.34 | ||||||||||||
| Parasympathetic modulation | ||||||||||||||||||
| RRI-RMSSD [ms] | 0.11 | 0.60 | 0.13 | 0.71 | 0.89 | 0.88 | ||||||||||||
| RRI-HF powers [ms2] | 0.19 | 0.93 | 0.22 | 0.87 | 0.89 | 0.96 | ||||||||||||
| Sympathetic modulation | ||||||||||||||||||
| RRI-LF powers [ms2] | 0.07 | 0.35 | 0.37 | 0.33 | 0.89 | 0.73 | ||||||||||||
| SBP-LF powers [mmHg2] | 0.26 | 0.78 | 0.14 | 0.54 | 0.78 | 0.66 | ||||||||||||
| Total autonomic modulation | ||||||||||||||||||
| RRI total powers [ms2] | 0.09 | 0.45 | 0.37 | 0.46 | 0.78 | 0.81 | ||||||||||||
| RRI-SD [ms] | 0.09 | 0.42 | 0.14 | 0.44 | 0.76 | 0.73 | ||||||||||||
| RRI-CV [%] | 0.19 | 0.28 | 0.42 | 0.32 | 0.76 | 0.66 | ||||||||||||
| Sympatho-vagal balance | ||||||||||||||||||
| RRI-LF/HF-ratio | 0.33 | 0.07 | 0.14 | 0.58 | 0.25 | 0.4 | ||||||||||||
| (b) | ||||||||||||||||||
| Controls (n = 30) | Patients with rTLE < 1/M (n = 6) | Patients with rTLE > 1/M (n = 6) | Patients with lTLE < 1/M (n = 9) | Patients with lTLE > 1/M (n = 10) | ||||||||||||||
| Biosignals, Median (IQR) | ||||||||||||||||||
| mean RRI [ms] | 754.1 (723.9–865.9) | 771.2 (566.8–891.1) | 717.2 (648.0–841.6) | 743.8 (607.9–841.3) | 801.2 (759.6–907.6) | |||||||||||||
| mean SBP [ms] | 127.2 (122.0–139.3) | 120.8 (113.0–131.6) | 127.9 (110.5–146.1) | 120.0 (107.1–130.1) | 124.9 (115.9–131.8) | |||||||||||||
| Parasympathetic modulation | ||||||||||||||||||
| RRI-RMSSD [ms] | 21.5 (17.2–35.6) | 14.3 (7.0–36.0) | 18.9 (9.8–25.8) | 23.3 (14.6–32.1) | 18.6 (7.1–25.6) | |||||||||||||
| RRI-HF powers [ms2] | 203.4 (85.3–498.1) | 122.7 (17.5–518.3) | 151.1 (40.2–415.8) | 224.1 (79.5–382.1) | 97.4 (28.7–444.3) | |||||||||||||
| Sympathetic modulation | ||||||||||||||||||
| RRI-LF powers [ms2] | 705.7 (362.2–2024.3) | 2230.0 (80.3–1878.2) | 633.3 (215.5–766.7) | 530.2 (378.5–1030.3) | 514.9 (162.0–1704.2) | |||||||||||||
| SBP-LF powers [mmHg2] | 15.9 (9.9–30.8) | 8.4 (5.0–33.5) | 17.5 (11.2–26.8) | 7.4 (5.7–15.3) | 19.8 (16.3–23.0) | |||||||||||||
| Total autonomic modulation | ||||||||||||||||||
| RRI total powers [ms2] | 964.6 (437.3–2521.8) | 350.1 (101.6–2396.5) | 836.0 (268.7–1191.5) | 828.0 (531.4–1324.5) | 937.7 (192.7–1985.2) | |||||||||||||
| RRI-SD [ms] | 32.9 (24.0–52.9) | 21.8 (11.8–58.4) | 29.8 (17.6–35.7) | 32.9 (25.1–38.0) | 31.7 (15.7–48.6) | |||||||||||||
| RRI-CV [%] | 4.4 (3.2–6.7) | 2.7 (1.7–7.5) | 4.0 (2.3–4.7) | 4.1 (2.7–5.2) | 4.3 (2.8–5.8) | |||||||||||||
| Sympatho-vagal balance | ||||||||||||||||||
| RRI-LF/HF-ratio | 2.9 (1.8–4.5) | 3.5 (1.5–6.2) | 2.9 (1.8–3.9) | 2.9 (1.2–5.2) | 5.3 (1.6–14.5) | |||||||||||||
| p-Values (Ct vs. rTLE < 1/M) | p-Values (Ct vs. rTLE > 1/M) | p-Values (rTLE < 1 vs. >1/M) | p-Values (Ct vs. lTLE < 1/M) | p-Values (Ct vs. lTLE > 1/M) | p-Values (lTLE < 1 vs. >1/M) | p-Values (<1/M r vs. lTLE) | p-Values (>1/M r vs. lTLE) | |||||||||||
| Biosignals, Median (IQR) | ||||||||||||||||||
| mean RRI [ms] | 0.58 | 0.19 | 0.87 | 0.33 | 0.32 | 0.87 | 0.81 | 0.19 | ||||||||||
| mean SBP [ms] | 0.16 | 0.93 | 0.75 | 0.06 | 0.24 | 0.75 | 0.64 | 0.66 | ||||||||||
| Parasympathetic modulation | ||||||||||||||||||
| RRI-RMSSD [ms] | 0.22 | 0.33 | 0.87 | 0.90 | 0.13 | 0.17 | 0.52 | 0.56 | ||||||||||
| RRI-HF powers [ms2] | 0.40 | 0.50 | 0.63 | 0.90 | 0.30 | 0.51 | 0.66 | 0.72 | ||||||||||
| Sympathetic modulation | ||||||||||||||||||
| RRI-LF powers [ms2] | 0.11 | 0.24 | 0.52 | 0.32 | 0.39 | 0.87 | 0.45 | 0.72 | ||||||||||
| SBP-LF powers [mmHg2] | 0.19 | 0.87 | 0.34 | 0.02 | 0.57 | 0.05 | 1 | 0.81 | ||||||||||
| Total autonomic modulation | ||||||||||||||||||
| RRI total powers [ms2] | 0.14 | 0.29 | 0.52 | 0.42 | 0.46 | 1 | 0.45 | 0.81 | ||||||||||
| RRI-SD [ms] | 0.17 | 0.22 | 0.75 | 0.49 | 0.41 | 0.74 | 0.59 | 0.81 | ||||||||||
| RRI-CV [%] | 0.20 | 0.25 | 0.52 | 0.27 | 0.46 | 0.74 | 0.59 | 0.41 | ||||||||||
| Sympatho-vagal balance | ||||||||||||||||||
| RRI-LF/HF-ratio | 0.22 | 0.15 | 0.75 | 0.08 | 1 | 0.22 | 0.59 | 0.41 | ||||||||||
| Patients with no ASM (n = 1) | Patients with 1 ASM (n = 6) | Patients with 2 ASM (n = 16) | Patients with 3 ASM (n = 8) | |
|---|---|---|---|---|
| Biosignals, Median (IQR) | ||||
| mean RRI [ms] | 725.2 | 734.4 (580.7–832.1) | 771.3 (714.6–831.7) | 843.9 (649.0–926.9) |
| mean SBP [ms] | 112.5 | 114.2 (103.9–120.3) | 126.7 (109.3–139.0) | 127.1 (118.0–132.8) |
| Parasympathetic modulation | ||||
| RRI-RMSSD [ms] | 7.7 | 16.7 (9.0–24.8) | 23.1 (8.6–33.5) | 17.1 (9.3–23.4) |
| RRI-HF powers [ms2] | 18.2 | 152.5 (34.8–470.0) | 187.0 (44.0–469.1) | 19.2 (11.7–41.2) |
| Sympathetic modulation | ||||
| RRI-LF powers [ms2] | 88.2 | 633.3 (250.1–967.6) | 575.2 (228.3–1913.7) | 466.8 (157.8–764.0) |
| SBP-LF powers [mmHg2] | 3.3 | 13.2 (6.1–35.3) | 18.0 (7.6–23.4) | 11.2 (6.2–21.0) |
| Total autonomic modulation | ||||
| RRI total powers [ms2] | 106.4 | 794. 6 (284.8–1616.7) | 824.5 (301.6–2.387.3) | 828.0 (191.1–1009.3) |
| RRI-SD [ms] | 12.1 | 30.4 (18.2–39.8) | 32.6 (19.0–56.6) | 30.8 (14.4–34.0) |
| RRI-CV [%] | 1.7 | 4.5 (2.1–5.6) | 4.2 (2.8–6.9) | 3.3 (2.3–4.7) |
| Sympatho-vagal balance | ||||
| RRI-LF/HF-ratio | 1.5 | 2.9 (1.7–4.6) | 3.9 (1.6–8.3) | 2.9 (2.0–5.4) |
| Controls < 30 a (n = 10) | Controls > 30 a (n = 20) | Pat. < 30 a with rTLE (n = 3) | Pat. > 30 a with rTLE (n = 9) | Pat. < 30 a with lTLE (n = 8) | Pat. > 30 a with lTLE (n = 11) | ||||||||
| Biosignals, Median (IQR) | |||||||||||||
| mean RRI [ms] | 767.3 (683.1–841.6) | 754.1 (728.7–927.0) | 709.2 (-) | 730.6 (647.9–881.2) | 790.4 (600.1–837.6) | 781.3 (743.8–893.3) | |||||||
| mean SBP [ms] | 124.4 (120.8–137.6) | 129.3 (122.2–140.0) | 114.4 (-) | 125.4 (114.2–137.4) | 125.6 (117.3–131.4) | 120.2 (107.9–128.1) | |||||||
| Parasympathetic modulation | |||||||||||||
| RRI-RMSSD [ms] | 25.3 (20.4–45.5) | 21.2 (14.7–29.5) | 35.3 (-) | 15.6 (7.4–23.1) | 21.8 (18.7–25.5) | 15.5 (7.7–32.1) | |||||||
| RRI-HF powers [ms2] | 323.7 (202.6–834.6) | 155.3 (40.1–277.3) | 500.2 (-) | 63.1 (20.8–270.3) | 314.8 (107.1–398.7) | 91.2 (34.5–464.8) | |||||||
| Sympathetic modulation | |||||||||||||
| RRI-LF powers [ms2] | 1620.8 (722.1–3979.9) | 21.2 (14.7–29.5) | 1657.8 (-) | 244.4 (85.7–702.6) | 663.3 (443.3–1285.9) | 424.9 (180.5–1999.0) | |||||||
| SBP-LF powers [mmHg2] | 15.7 (12.5–46.2) | 155.3 (40.1–277.3) | 26.2 (-) | 10.7 (5.8–23.0) | 17.5 (8.1–25.0) | 11.9 (6.6–20.2) | |||||||
| Total autonomic modulation | |||||||||||||
| RRI total powers [ms2] | 1835.6 (1039.7–5250.6) | 21.2 (14.7–29.5) | 2158.0 (-) | 307.5 (125.0–1072.2) | 996.5 (827.0–1393.1) | 588.4 (219.0–2463.7) | |||||||
| RRI-SD [ms] | 43.8 (34.0–77.1) | 155.3 (40.1–277.3) | 57.4 (-) | 19.2 (12.5–34.3) | 34.1 (30.4–40.9) | 26.3 (16.7–54.2) | |||||||
| RRI-CV [%] | 6.1 (4.9–8.3) | 3.7 (3.1–5.3) | 7.1 (-) | 2.4 (2.0–4.0) | 4.3 (3.4–5.1) | 3.5 (2.4–6.5) | |||||||
| Sympatho-vagal balance | |||||||||||||
| RRI-LF/HF-ratio | 4.7 (2.8–7.9) | 6.3 (1.8–10.5) | 2.3 (1.7–3.7) | 2.3 (1.7–3.7) | 4.7 (1.6–5.7) | 2.7 (1.3–13.8) | |||||||
| p-Values (Ct < 30 a vs. >30 a) | p-Values (Ct vs. rTLE < 30 a) | p-Values (Ct vs. rTLE > 30 a) | p-Values (Ct vs. lTLE < 30 a) | p-Values (Ct vs. lTLE > 30 a) | p-Values (r vs. lTLE < 30 a) | p-Values (r vs. lTLE > 30 a) | |||||||
| Biosignals, Median (IQR) | |||||||||||||
| mean RRI [ms] | 0.57 | 0.50 | 0.32 | 0.79 | 0.71 | 0.68 | 0.43 | ||||||
| mean SBP [ms] | 0.60 | 0.61 | 0.40 | 0.53 | 0.05 | 0.68 | 0.31 | ||||||
| Parasympathetic modulation | |||||||||||||
| RRI-RMSSD [ms] | 0.14 | 0.61 | 0.13 | 0.29 | 0.51 | 0.31 | 0.47 | ||||||
| RRI-HF powers [ms2] | 0.02 | 0.74 | 0.51 | 0.29 | 0.59 | 0.31 | 0.85 | ||||||
| Sympathetic modulation | |||||||||||||
| RRI-LF powers [ms2] | 0.01 | 0.61 | 0.07 | 0.03 | 0.64 | 0.22 | 0.34 | ||||||
| SBP-LF powers [mmHg2] | 0.57 | 0.61 | 0.17 | 0.79 | 0.23 | 0.41 | 0.91 | ||||||
| Total autonomic modulation | |||||||||||||
| RRI total powers [ms2] | 0.01 | 0.61 | 0.09 | 0.04 | 0.62 | 0.41 | 0.47 | ||||||
| RRI-SD [ms] | 0.01 | 0.87 | 0.08 | 0.08 | 0.65 | 0.31 | 0.47 | ||||||
| RRI-CV [%] | <0.01 | 0.87 | 0.08 | 0.03 | 0.48 | 0.10 | 0.31 | ||||||
| Sympatho-vagal balance | |||||||||||||
| RRI-LF/HF-ratio | 0.76 | 0.61 | 0.11 | 0.42 | 0.54 | 0.41 | 0.62 | ||||||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Borutta, M.C.; Royle, V.; Rothballer, C.; Kraemer, F.; Gollwitzer, S.; Hamer, H.; Schwab, S.; Koehn, J. If You Care About Autonomic Modulation—Do Not Let Seizure Seizure. Diagnostics 2026, 16, 698. https://doi.org/10.3390/diagnostics16050698
Borutta MC, Royle V, Rothballer C, Kraemer F, Gollwitzer S, Hamer H, Schwab S, Koehn J. If You Care About Autonomic Modulation—Do Not Let Seizure Seizure. Diagnostics. 2026; 16(5):698. https://doi.org/10.3390/diagnostics16050698
Chicago/Turabian StyleBorutta, Matthias C., Vayra Royle, Christina Rothballer, Florian Kraemer, Stephanie Gollwitzer, Hajo Hamer, Stefan Schwab, and Julia Koehn. 2026. "If You Care About Autonomic Modulation—Do Not Let Seizure Seizure" Diagnostics 16, no. 5: 698. https://doi.org/10.3390/diagnostics16050698
APA StyleBorutta, M. C., Royle, V., Rothballer, C., Kraemer, F., Gollwitzer, S., Hamer, H., Schwab, S., & Koehn, J. (2026). If You Care About Autonomic Modulation—Do Not Let Seizure Seizure. Diagnostics, 16(5), 698. https://doi.org/10.3390/diagnostics16050698






