Plasma RNA-Based Dual Screening for Early/Extreme Spontaneous Preterm Birth and Early Onset Preeclampsia to Enable Prevention
Abstract
1. Background/Objectives
2. Precision Medicine for Complications of Pregnancy
2.1. Materials and Methods
2.1.1. Clinical Methods
2.1.2. Laboratory Methods
RNA Extraction
qRT-PCR Assays
2.1.3. Preamplification and qPCR
2.1.4. Statistics
2.2. Results
2.2.1. External Validation Studies
EOP with Delivery at ≤33 Wks
sPTB < 33 Wks
2.3. Explaining the Dual Efficacy of FutureBIRTHR to Predict Both sPTB and EOP
2.4. Why Has No Other PCF RNA Test Been Externally Validated for Any Disease or Complication Repeatedly Despite Extensive Effort?
3. Discussion
- All CL measurements guiding therapy should be transvaginal and performed at 18–22 wks’ gestation at the anatomic scan.
- A midtrimester CL of ≤25 mm is considered short in women with a singleton pregnancy but no prior sPTB, and those with a CL of ≤20 mm should be prescribed vaginal progesterone.
- In such women with a CL of 10–25 mm, cerclage should not be performed absent cervical dilation, nor should a pessary be prescribed.
- Serial vaginal CL (every 1–4 wks’) at 16 0/7–24 0/7 wks’ gestation.
- Offer vaginal progesterone for asymptomatic women with a prior sPTB.
- Consider cerclage with a CL of <25 mm (vs. progesterone if not already on it).
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Callaghan, W.M.; MacDorman, M.F.; Shapiro-Mendoza, C.K.; Barfield, W.D. Explaining the recent decrease in US infant mortality rate, 2007–2013. Am. J. Obstet. Gynecol. 2017, 216, 73.e1–73.e8. [Google Scholar] [CrossRef]
- Ohuma, E.O.; Moller, A.-B.; Bradley, E.; Chakwera, S.; Hussain-Alkhateeb, L.; Lewin, A.; Okwaraji, Y.B.; Mahanani, R.T.; Johansson, E.W.; Lavin, T. National, regional, and global estimates of preterm birth in 2020, with trends from 2010: A systematic analysis. Lancet 2023, 402, 1261–1271. [Google Scholar] [CrossRef]
- Osterman, M.J.K.; Hamilton, B.E.; Martin, J.A.; Driscoll, A.K.; Valenzuela, C.P. Births: Final Data for 2023; National Vital Statistics Reports; National Center for Health Statistics: Hyattsville, MD, USA, 2025; Volume 74. [Google Scholar] [CrossRef]
- American College of Obstetricians and Gynecologists’ Committee on Practice Bulletins—Obstetrics (ACOG). Prediction and prevention of spontaneous preterm birth. Obstet. Gynecol. 2021, 138, e61–e95. [Google Scholar] [CrossRef]
- Romero, R.; Meyyazhagan, A.; Hassan, S.S.; Creasy, G.W.; Conde-Agudelo, A. Vaginal Progesterone to Prevent Spontaneous Preterm Birth in Women With a Sonographic Short Cervix: The Story of the PREGNANT Trial. Clin. Obstet. Gynecol. 2024, 67, 433–457. [Google Scholar] [CrossRef] [PubMed]
- Burchard, J.; Polpitiya, A.D.; Fox, A.C.; Randolph, T.L.; Fleischer, T.C.; Dufford, M.T.; Garite, T.J.; Critchfield, G.C.; Boniface, J.J.; Saade, G.R.; et al. Clinical Validation of a Proteomic Biomarker Threshold for Increased Risk of Spontaneous Preterm Birth and Associated Clinical Outcomes: A Replication Study. J. Clin. Med. 2021, 10, 5088. [Google Scholar] [CrossRef] [PubMed]
- Branch, D.W.; VanBuren, J.M.; Porter, T.F.; Holmgren, C.; Holubkov, R.; Page, K.; Burchard, J.; Lam, G.K.; Esplin, M.S. Prediction and Prevention of Preterm Birth: A Prospective, Randomized Intervention Trial. Am. J. Perinatol. 2021, 40, 1071–1080. [Google Scholar] [CrossRef] [PubMed]
- Hoffman, M.K.; Kitto, C.; Zhang, Z.; Shi, J.; Walker, M.G.; Shahbaba, B.; Ruhstaller, K. Neonatal Outcomes after Maternal Biomarker-Guided Preterm Birth Intervention: The AVERT PRETERM Trial. Diagnostics 2024, 14, 1462. [Google Scholar] [CrossRef]
- Cuenca-Gómez, D.; De Paco Matallana, C.; Rolle, V.; Mendoza, M.; Valiño, N.; Revello, R.; Adiego, B.; Casanova, M.C.; Molina, F.S.; Delgado, J.L.; et al. Comparison of different methods of first-trimester screening for preterm pre-eclampsia: Cohort study. Ultrasound Obstet. Gynecol. 2024, 64, 57–64. [Google Scholar] [CrossRef]
- Martins, J.G.; Miller, E.; Aboukhater, D.; Bittner, M.; Rolnik, D.L.; Kawakita, T. Performance of a first-trimester combined screening for preterm preeclampsia in the United States population using the Fetal Medicine Foundation competing risks model. Am. J. Obstet. Gynecol. MFM 2025, 7, 101803. [Google Scholar] [CrossRef]
- Carlson, S.E.; Gajewski, B.J.; Valentine, C.J.; Sands, S.A.; Brown, A.R.; Kerling, E.H.; Crawford, S.A.; Buhimschi, C.S.; Weiner, C.; Cackovic, M.; et al. Early and late preterm birth rates in participants adherent to randomly assigned high dose docosahexaenoic acid (DHA) supplementation in pregnancy. Clin. Nutr. 2023, 42, 235–243. [Google Scholar] [CrossRef]
- Weiner, C.P.; Dong, Y.; Zhou, H.; Cuckle, H.S.; Ramsey, R.; Egerman, R.; Buhimschi, I.; Buhimschi, C. Early pregnancy prediction of spontaneous preterm birth before 32 completed weeks of pregnancy using plasma RNA: Transcriptome discovery and initial validation of an RNA panel of markers. BJOG 2021, 128, 1870–1880. [Google Scholar] [CrossRef] [PubMed]
- Weiner, C.P.; Cuckle, H.; Weiss, M.L.; Buhimschi, I.A.; Dong, Y.; Zhou, H.; Ramsey, R.; Egerman, R.; Buhimschi, C.S. Evaluation of a Maternal Plasma RNA Panel Predicting Spontaneous Preterm Birth and Its Expansion to the Prediction of Preeclampsia. Diagnostics 2022, 12, 1327. [Google Scholar] [CrossRef] [PubMed]
- Weiner, C.P.; Zhou, H.; Cuckle, H.; Syngelaki, A.; Nicolaides, K.H.; Weiss, M.L.; Dong, Y. Maternal Plasma RNA in First Trimester Nullipara for the Prediction of Spontaneous Preterm Birth ≤ 32 Weeks: Validation Study. Biomedicines 2023, 11, 1149. [Google Scholar] [CrossRef] [PubMed]
- Weiner, C.P.; Cuckle, H.S.; Carlson, S.E.; Bernstein, I. Plasma Cell Free NAMPT Expression at 12–20 weeks in Women Destined for Early Onset Preeclampsia (EOP) with Delivery < 34 weeks. Reprod. Sci. 2025, 32, 1. [Google Scholar]
- Weiner, C.P.; Mason, C.W.; Dong, Y.; Buhimschi, I.A.; Swaan, P.W.; Buhimschi, C.S. Human effector/initiator gene sets that regulate myometrial contractility during term and preterm labor. Am. J. Obstet. Gynecol. 2010, 202, 474.e1–474.20. [Google Scholar] [CrossRef]
- Vornic, I.; Buciu, V.; Furau, C.G.; Zara, F.; Novacescu, D.; Barb, A.C.; Cumpanas, A.A.; Latcu, S.C.; Sas, I.; Serban, D.; et al. The Interplay of Molecular Factors and Morphology in Human Placental Development and Implantation. Biomedicines 2024, 12, 2908. [Google Scholar] [CrossRef]
- Moret, I.; Sánchez-Izquierdo, D.; Iborra, M.; Tortosa, L.; Navarro-Puche, A.; Nos, P.; Cervera, J.; Beltrán, B. Assessing an improved protocol for plasma microRNA extraction. PLoS ONE 2013, 8, e82753. [Google Scholar] [CrossRef]
- Meerson, A.; Ploug, T. Assessment of six commercial plasma small RNA isolation kits using qRT-PCR and electrophoretic separation: Higher recovery of microRNA following ultracentrifugation. Biol. Methods Protoc. 2016, 1, bpw003. [Google Scholar] [CrossRef]
- Yamamoto, K.; Chiba, M. Examination and comparison of the RNA extraction methods using mouse serum. Biomed. Rep. 2024, 20, 51. [Google Scholar] [CrossRef]
- Max, K.E.A.; Bertram, K.; Akat, K.M.; Bogardus, K.A.; Li, J.; Morozov, P.; Ben-Dov, I.Z.; Li, X.; Weiss, Z.R.; Azizian, A.; et al. Human plasma and serum extracellular small RNA reference profiles and their clinical utility. Proc. Natl. Acad. Sci. USA 2018, 115, E5334–E5343. [Google Scholar] [CrossRef]
- Weiner, C.P.; Weiss, M.L.; Zhou, H.; Syngelaki, A.; Nicolaides, K.H.; Dong, Y. Detection of Embryonic Trisomy 21 in the First Trimester Using Maternal Plasma Cell-Free RNA. Diagnostics 2022, 12, 1410. [Google Scholar] [CrossRef] [PubMed]
- Richter, L.L.; Shen, Y.; Lisonkova, S.; Bone, J.N.; Albert, A.; Ho, M.S.P.; Kieran, E.; Chan, E.S.; Mammen, C.; Lam, C.; et al. Preterm birth and risk of health service use, morbidity, and medication needs at 5 years. JAMA Pediatr. 2025, 179, 1172–1182. [Google Scholar] [CrossRef] [PubMed]
- Lo, K.W.; Lo, Y.M.; Leung, S.F.; Tsang, Y.S.; Chan, L.Y.; Johnson, P.J.; Hjelm, N.M.; Lee, C.J.; Huang, D.P. Analysis of cell-free Epstein-Barr virus associated RNA in the plasma of patients with nasopharyngeal carcinoma. Clin. Chem. 1999, 45, 1292–1294. [Google Scholar] [CrossRef] [PubMed]
- Poon, L.L.; Leung, T.N.; Lau, T.K.; Lo, Y.M. Presence of fetal RNA in maternal plasma. Clin. Chem. 2000, 46, 1832–1834. [Google Scholar] [CrossRef]
- Rasmussen, M.; Reddy, M.; Nolan, R.; Camunas-Soler, J.; Khodursky, A.; Scheller, N.M.; Cantonwine, D.E.; Engelbrechtsen, L.; Mi, J.D.; Dutta, A.; et al. RNA profiles reveal signatures of future health and disease in pregnancy. Nature 2022, 601, 422–427. [Google Scholar] [CrossRef]
- Moufarrej, M.N.; Vorperian, S.K.; Wong, R.J.; Campos, A.A.; Quaintance, C.C.; Sit, R.V.; Tan, M.; Detweiler, A.M.; Mekonen, H.; Neff, N.F.; et al. Early prediction of preeclampsia in pregnancy with cell-free RNA. Nature 2022, 602, 689–694. [Google Scholar] [CrossRef]
- Elovitz, M.A.; Gee, E.P.S.; Delaney-Busch, N.; Moe, A.B.; Reddy, M.; Khodursky, A.; La, J.; Abbas, I.; Mekaru, K.; Collins, H.; et al. Molecular subtyping of hypertensive disorders of pregnancy. Nat. Commun. 2025, 16, 2948. [Google Scholar] [CrossRef]
- March of Dimes. 2025. Available online: https://www.marchofdimes.org/peristats/data?reg=99&top=3&stop=61&lev=1&slev=4&obj=1&sreg=99 (accessed on 15 December 2025).
- Gómez, T.; Castillo-Marco, N.; Igual, M.; Muñoz-Blat, I.; Gómez-Álvarez, C.; Bernat-González, N.; Gaspar-Doménech, A.; Ortiz-Domingo, E.; Vives, A.; Ortega-Sanchís, S.; et al. Maternal Plasma Cell-Free RNA as a Predictor of Early and Late-Onset Preeclampsia Throughout Pregnancy. Nat. Commun. 2025, 16, 1–14. [Google Scholar] [CrossRef]
- Esplin, M.S.; Elovitz, M.A.; Iams, J.D.; Parker, C.B.; Wapner, R.J.; Grobman, W.A.; Simhan, H.N.; Wing, D.A.; Haas, D.M.; Silver, R.M.; et al. Predictive accuracy of serial transvaginal cervical lengths and quantitative vaginal fetal fibronectin for spontaneous preterm birth among nulliparous women. JAMA 2017, 317, 1047–1056. [Google Scholar] [CrossRef]
- Wright, D.; Wright, A.; Nicolaides, K.H. The competing risk approach for prediction of preeclampsia. Am. J. Obstet. Gynecol. 2020, 223, 12–23.e7. [Google Scholar] [CrossRef]
- Bujold, E.; Roberge, S.; Nicolaides, K.H. Low-dose aspirin for prevention of adverse outcomes related to abnormal placentation. Prenat. Diagn 2014, 34, 642–648. [Google Scholar] [CrossRef]
- Olsen, S.F.; Hansen, H.S.; Sørensen, T.I.; Jensen, B.; Secher, N.J.; Sommer, S.; Knudsen, L.B. Intake of marine fat, rich in (n-3)-polyunsaturated fatty acids may increase birthweight by prolonging gestation. Lancet 1986, 2, 367–369. [Google Scholar] [CrossRef]
- Olsen, S.F.; Secher, N.J.; Tabor, A.; Weber, T.; Walker, J.J.; Gluud, C. Randomised clinical trials of fish oil supplementation in high-risk pregnancies. BJOG 2000, 107, 382–395. [Google Scholar] [CrossRef] [PubMed]
- Middleton, P.; Gomersall, J.C.; Gould, J.F.; Shepherd, E.; Olsen, S.F.; Makrides, M. Omega-3 fatty acid addition during pregnancy. Cochrane Database Syst. Rev. 2018, 11, CD003402. [Google Scholar] [CrossRef] [PubMed]
- Carlson, S.E.; Gajewski, B.J.; Valentine, C.J.; Kerling, E.H.; Weiner, C.P.; Cackovic, M.; Buhimschi, C.S.; Rogers, L.K.; Sands, S.A.; Brown, A.R.; et al. Higher dose docosahexaenoic acid supplementation during pregnancy and early preterm birth: A randomized, double-blind, adaptive-design superiority trial. EClinMed 2021, 36, 100905. [Google Scholar] [CrossRef]
- Makrides, M.; Best, K.; Yelland, L.; McPhee, A.; Zhou, S.; Quinlivan, J.; Dodd, J.; Atkinson, E.; Safa, H.; van Dam, J.; et al. A randomized trial of prenatal n-3 fatty acid supplementation and preterm delivery. N. Engl. J. Med. 2019, 381, 1035–1045. [Google Scholar] [CrossRef]
- Simmonds, L.A.; Sullivan, T.R.; Skubisz, M.; Middleton, P.F.; Best, K.P.; Yelland, L.N.; Quinlivan, J.; Zhou, S.J.; Liu, G.; McPhee, A.J.; et al. Omega-3 fatty acid supplementation in pregnancy-baseline omega-3 status and early preterm birth: Exploratory analysis of a randomised controlled trial. BJOG 2020, 127, 975–981. [Google Scholar] [CrossRef]
- Gustafson, K.M.; Christifano, D.N.; Hoyer, D.; Schmidt, A.; Carlson, S.E.; Colombo, J.; Mathis, N.B.; Sands, S.A.; Chollet-Hinton, L.; Brown, A.R.; et al. Prenatal docosahexaenoic acid effect on maternal-infant equilibrium and fetal neurodevelopment: A randomized clinical trial. Pediatr. Res. 2022, 92, 255–264. [Google Scholar] [CrossRef]
- Christifano, D.N.; Crawford, S.A.; Lee, G.; Brown, A.R.; Camargo, J.T.; Kerling, E.H.; Gajewski, B.J.; Valentine, C.J.; Gustafson, K.M.; DeFranco, E.A.; et al. Docosahexaenoic acid (DHA) intake estimated from a 7-question survey identifies pregnancies most likely to benefit from high-dose DHA supplementation. Clin. Nutr. ESPEN 2023, 53, 93–99. [Google Scholar] [CrossRef]
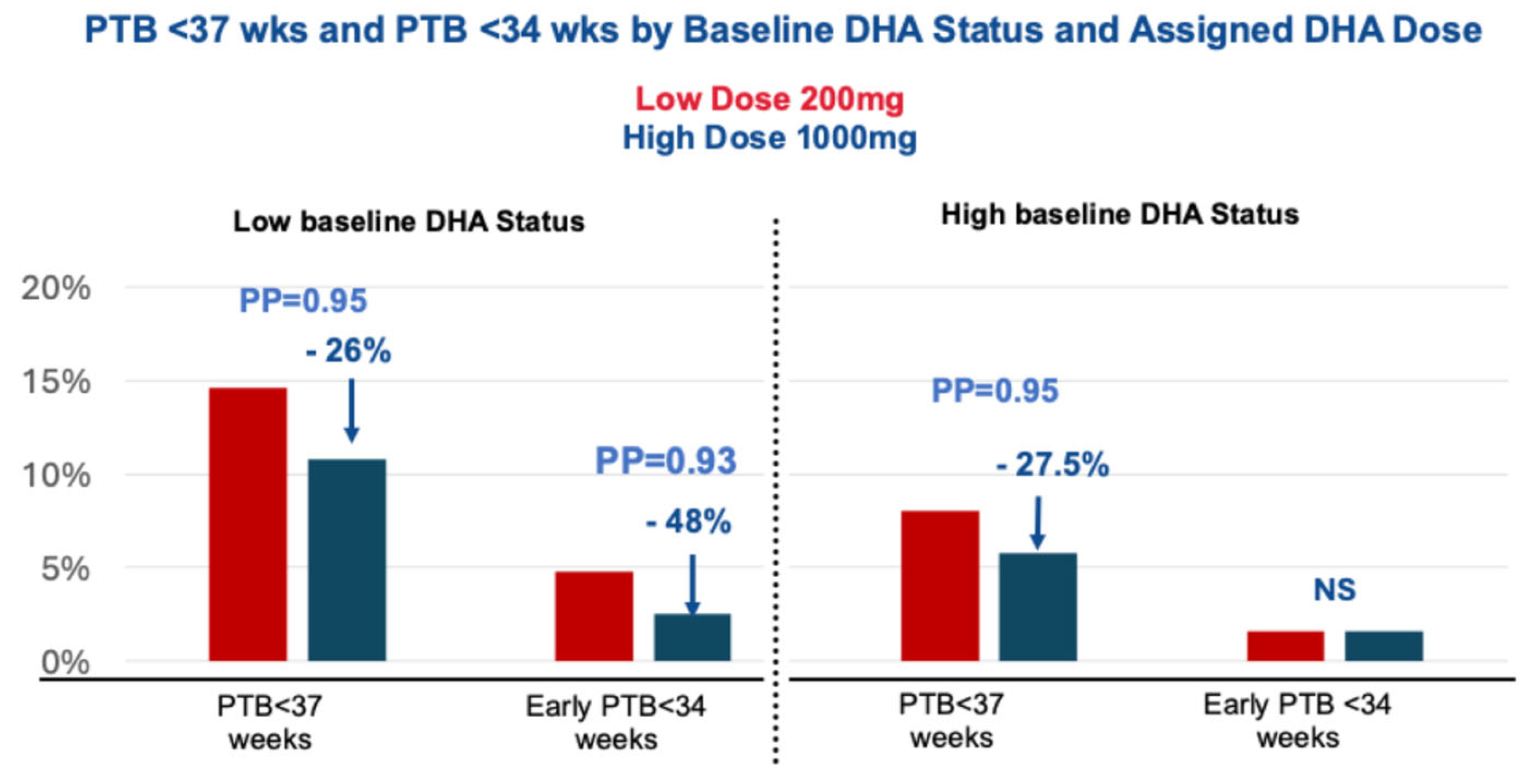
- Cetin, I.; Carlson, S.E.; Burden, C.; da Fonseca, E.B.; di Renzo, G.C.; Hadjipanayis, A.; Harris, W.S.; Kumar, K.R.; Olsen, S.F.; Mader, S.; et al. Omega-3 fatty acid supply in pregnancy for risk reduction of preterm and early preterm birth. Am. J. Obstet. Gynecol. MFM 2024, 6, 101251. [Google Scholar] [CrossRef]
- SMFM Publications Committee. SMFM Consult Series #70: Management of short cervix in individuals without a history of spontaneous preterm birth. Am. J. Obstet. Gynecol. 2024, 231, B2–B13. [Google Scholar] [CrossRef]
- Best, K.P.; Gibson, R.A.; Makrides, M. ISSFAL statement number 7—Omega-3 fatty acids during pregnancy to reduce preterm birth. Prostaglandins Leukot Essent Fat. Acids 2022, 186, 102495. [Google Scholar] [CrossRef]
- Rolnik, D.L.; Wright, D.; Poon, L.C.; O’Gorman, N.; Syngelaki, A.; de Paco Matallana, C.; Akolekar, R.; Cicero, S.; Janga, D.; Singh, M.; et al. Aspirin versus placebo in pregnancies at high risk for preterm preeclampsia. N. Engl. J. Med. 2017, 377, 613–622. [Google Scholar] [CrossRef] [PubMed]
- O’Gorman, N.; Wright, D.; Poon, L.C.; Rolnik, D.L.; Syngelaki, A.; Wright, A.; Akolekar, R.; Cicero, S.; Janga, D.E.; Jani, J.; et al. Accuracy of competing-risks model in screening for pre-eclampsia by maternal factors and biomarkers at 11-13 weeks’ gestation. Ultrasound Obstet. Gynecol. 2017, 49, 751–755. [Google Scholar] [CrossRef] [PubMed]
- Riishede, I.; Ekelund, C.K.; Sperling, L.; Overgaard, M.; Knudsen, C.S.; Clausen, T.D.; Pihl, K.; Zingenberg, H.J.; Wright, A.; Wright, D.; et al. Screening for pre-eclampsia with competing-risks model using placental growth factor measurement in blood samples collected before 11 weeks’ gestation. Ultrasound Obstet. Gynecol. 2024, 63, 342–349. [Google Scholar] [CrossRef]
- Chaemsaithong, P.; Pooh, R.K.; Zheng, M.; Ma, R.; Chaiyasit, N.; Tokunaka, M.; Shaw, S.W.; Seshadri, S.; Choolani, M.; Wataganara, T.; et al. Prospective evaluation of screening performance of first-trimester prediction models for preterm preeclampsia in an Asian population. Am. J. Obstet. Gynecol. 2019, 221, 650.e1–650.e16. [Google Scholar] [CrossRef]
- Tiruneh, S.A.; Rolnik, D.L.; Selvaratnam, R.; da Silva Costa, F.; McLennan, A.; Hyett, J.; Teede, H.; Enticott, J. External validation of the Fetal Medicine Foundation model for preterm pre-eclampsia prediction at 11-14 weeks in an Australian population. Acta Obstet. Gynecol. Scand. 2025, 104, 1774–1782. [Google Scholar] [CrossRef]
- Wright, D.; Rolnik, D.L.; Syngelaki, A.; de Paco Matallana, C.; Machuca, M.; de Alvarado, M.; Mastrodima, S.; Tan, M.Y.; Shearing, S.; Persico, N.; et al. Aspirin for Evidence-Based Preeclampsia Prevention trial: Effect of aspirin on length of stay in the neonatal intensive care unit. Am. J. Obstet. Gynecol. 2018, 218, 612.e1–612.e6. [Google Scholar] [CrossRef]
- Poon, L.C.; Rolnik, D.L.; Tan, M.Y.; Delgado, J.L.; Tsokaki, T.; Akolekar, R.; Singh, M.; Andrade, W.; Efeturk, T.; Jani, J.C.; et al. ASPRE trial: Incidence of preterm pre-eclampsia in patients fulfilling ACOG and NICE criteria according to risk by FMF algorithm. Ultrasound Obstet. Gynecol. 2018, 51, 738–742. [Google Scholar] [CrossRef]
- Tan, M.Y.; Syngelaki, A.; Poon, L.C.; Rolnik, D.L.; O’Gorman, N.; Delgado, J.L.; Akolekar, R.; Konstantinidou, L.; Tsavdaridou, M.; Galeva, S.; et al. Screening for pre-eclampsia by maternal factors and biomarkers at 11-13 weeks’ gestation. Ultrasound Obstet. Gynecol. 2018, 52, 186–195. [Google Scholar] [CrossRef]
- Mills, J.L.; DerSimonian, R.; Raymond, E.; Morrow, J.D.; Roberts, L.J., 2nd; Clemens, J.D.; Hauth, J.C.; Catalano, P.; Sibai, B.; Curet, L.B.; et al. Prostacyclin and thromboxane changes predating clinical onset of preeclampsia: A multicenter prospective study. JAMA 1999, 28, 356–362. [Google Scholar] [CrossRef]
- Gatto, M.; Esposito, M.; Morelli, M.; De Rose, S.; Gizurarson, S.; Meiri, H.; Mandalà, M. Placental Protein 13: Vasomodulatory Effects on Human Uterine Arteries and Potential Implications for Preeclampsia. Int. J. Mol. Sci. 2024, 25, 7522. [Google Scholar] [CrossRef]
- Trilla, C.; Platero, J.; Mora, J.; Nan, M.N.; Medina, C.; Alejos, O.; Parra, J.; Llurba, E. Role of aspirin therapy in modulating uterine artery resistance and placental growth between first and second trimesters of pregnancy. Ultrasound Obs. Gynecol. 2025, 66, 641–648. [Google Scholar] [CrossRef]
- Sibai, B.M.; Caritis, S.N.; Thom, E.; Klebanoff, M.; McNellis, D.; Rocco, L.; Paul, R.H.; Romero, R.; Witter, F.; Rosen, M.; et al. Prevention of preeclampsia with low-dose aspirin in healthy, nulliparous pregnant women. N. Engl. J. Med. 1993, 329, 1213–1218. [Google Scholar] [CrossRef]
- Roberge, S.; Bujold, E.; Nicolaides, K.H. Meta-analysis on the effect of aspirin use for prevention of preeclampsia on placental abruption and antepartum hemorrhage. Am. J. Obstet. Gynecol. 2018, 218, 483–489. [Google Scholar] [CrossRef]
- Roberge, S.; Bujold, E.; Nicolaides, K.H. Aspirin for the prevention of preterm and term preeclampsia: Systematic review and metaanalysis. Am. J. Obstet. Gynecol. 2018, 218, 287–293. [Google Scholar] [CrossRef]
- Lee, R.H.; Weiner, C.P. Robust cost efficacy of a novel, validated screening test at 12–20 Weeks gestation for the prediction of preterm birth (PTB) at or before 32 Weeks in singletons. Gynecol. Obstet. Clin. Med. 2021, 1, 107–111. [Google Scholar] [CrossRef]


| Outcome | Model Variables | AUC [95% CI] | DR (%) |
|---|---|---|---|
| EOP with delivery at ≤33 wks | NAMPT, MAP | 0.89 [0.81–0.97] | 89 |
| Outcome | Model Variables | AUC [95% CI] | DR (%) |
|---|---|---|---|
| EOP with delivery at ≤33 wks | NAMPT, APOA1, parity, MAP | 0.96 [0.92–0.996] | 100 |
| All preeclampsia | NAMPT, APOA1, parity, MAP | 0.82 [0.72–0.91] | 71 |
| Marker | Cases | Controls | Values ≥ Median | p-Value | |
|---|---|---|---|---|---|
| Cases | Controls | ||||
| PSME2 | 20 | 20 | 19 (95%) | 1 (5%) | <0.0001 |
| NAMPT | 13 | 11 | 12 (92%) | 0 (0%) | <0.0005 |
| APOA4 | 18 | 20 | 18 (100%) | 1 (5%) | <0.0001 |
| LET-7g | 20 | 20 | 19 (95%) | 1 (5%) | <0.0001 |
| Outcome | Model Variables | AUC [95% CI] | DR (%) |
|---|---|---|---|
| sPTB at <33 wks | PSME2, Let 7g, prior PTB, race | 0.83 [0.74–0.92] | 77 |
| sPTB at <37 wks | PSME2, Let 7g, prior PTB, race | 0.77 [0.70–0.83] | 73 |
| Outcome | Model Variables | AUC [95% CI] | DR (%) |
|---|---|---|---|
| sPTB at <33 wks | APOA1 + CRL, MA, MW, race, tobacco | 0.79 [0.66–0.91] | 79 |
| Test Name [References] | Patient Inclusions /Exclusions | Earliest Test Use (wks) | Screening Target and Validation | AUC | DR (%) |
|---|---|---|---|---|---|
| FutureBIRTH® [12,13,14] | None | 12 | sPTB at <33 wks (External validation) | 0.79–0.83 | 77–79 |
| Fetal fibronectin [31] | None | 16–22 | All PTB at <37 wks (External validation) | 0.51–0.52 | 3–15 |
| TV Cervical Length [31] | None | 16–22 | All PTB at <37 wks (External validation) | 0.53 | 4–8 |
| PreTRM® [6,7,8] | None | 19 | All PTB at <37 wks (Internal and external validations) | 0.68–0.75 PTB at <37 wks 0.76 PTB at <32 wks | 75 |
| FutureBIRTH® [13] | None | 12 | EOP delivery at ≤33 wks (External validation) | 0.89–0.96 | 89–100 |
| FMF Combined Test [32,33] | None | 11 | EOP with delivery at <33 wks’ gestation (External validation) | 0.80–0.93 | 90% |
| EncompassTM [26,27,28] |
| 18 | All preeclampsia (Internal validation) Preeclampsia with delivery at ≤35 wks (Internal validation) | 0.88 0.83 | 90 81% |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Weiner, C.P.; Carlson, S.E.; Meiri, H. Plasma RNA-Based Dual Screening for Early/Extreme Spontaneous Preterm Birth and Early Onset Preeclampsia to Enable Prevention. Diagnostics 2026, 16, 660. https://doi.org/10.3390/diagnostics16050660
Weiner CP, Carlson SE, Meiri H. Plasma RNA-Based Dual Screening for Early/Extreme Spontaneous Preterm Birth and Early Onset Preeclampsia to Enable Prevention. Diagnostics. 2026; 16(5):660. https://doi.org/10.3390/diagnostics16050660
Chicago/Turabian StyleWeiner, Carl P., Susan E. Carlson, and Hamutal Meiri. 2026. "Plasma RNA-Based Dual Screening for Early/Extreme Spontaneous Preterm Birth and Early Onset Preeclampsia to Enable Prevention" Diagnostics 16, no. 5: 660. https://doi.org/10.3390/diagnostics16050660
APA StyleWeiner, C. P., Carlson, S. E., & Meiri, H. (2026). Plasma RNA-Based Dual Screening for Early/Extreme Spontaneous Preterm Birth and Early Onset Preeclampsia to Enable Prevention. Diagnostics, 16(5), 660. https://doi.org/10.3390/diagnostics16050660








