Ultrasound Versus Fluoroscopy for Cervical Medial Branch Injections at C3–C6: A Single-Center Retrospective Cohort Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design and Setting
2.2. Participants
- (i)
- Clinically suspected cervical facet-related pain characterized by segmental paravertebral pain, exacerbation with extension/rotation, and tenderness on facet provocation tests;
- (ii)
- Radiological findings consistent with facet arthropathy or spondylosis at the C3–C6 levels;
- (iii)
- Prior administration of US- or FL-guided medial branch block (MBB) and/or radiofrequency ablation (RFA).
2.3. Interventions and Procedures
2.3.1. Ultrasound (US)-Guided MBB/RFA
2.3.2. Fluoroscopy (FL)-Guided MBB/RFA
2.4. Common Procedure Elements
2.5. Sample Size and Power Analysis
2.6. Outcome Measures
- Technical success, defined as real-time confirmation of correct needle placement and independent verification by a blinded algology expert.
- Procedure time (minutes).
- Pain intensity was assessed using a 10 cm Visual Analog Scale (VAS; 0 = no pain, 10 = worst imaginable pain) at baseline immediately prior to the procedure and at each follow-up visit. Pain intensity (VAS, 0–10) at 1, 3, 6, and 12 months. Responders were defined as demonstrating ≥2-point improvement.
- Functional outcomes, assessed using the NDI (0–50).
- Complications graded according to CIRSE classification.
- Radiation exposure in the FL group (fluoroscopy time, DAP, and air kerma).
- Sedation dosage and steroid use.
- MCID-based NI analyses at 6 and 12 months (margin: 1.5 points for VAS; 7 points for NDI) were applied in a post hoc framework.
2.7. Data Collection
2.8. Statistical Analysis
3. Results
3.1. Baseline Characteristics
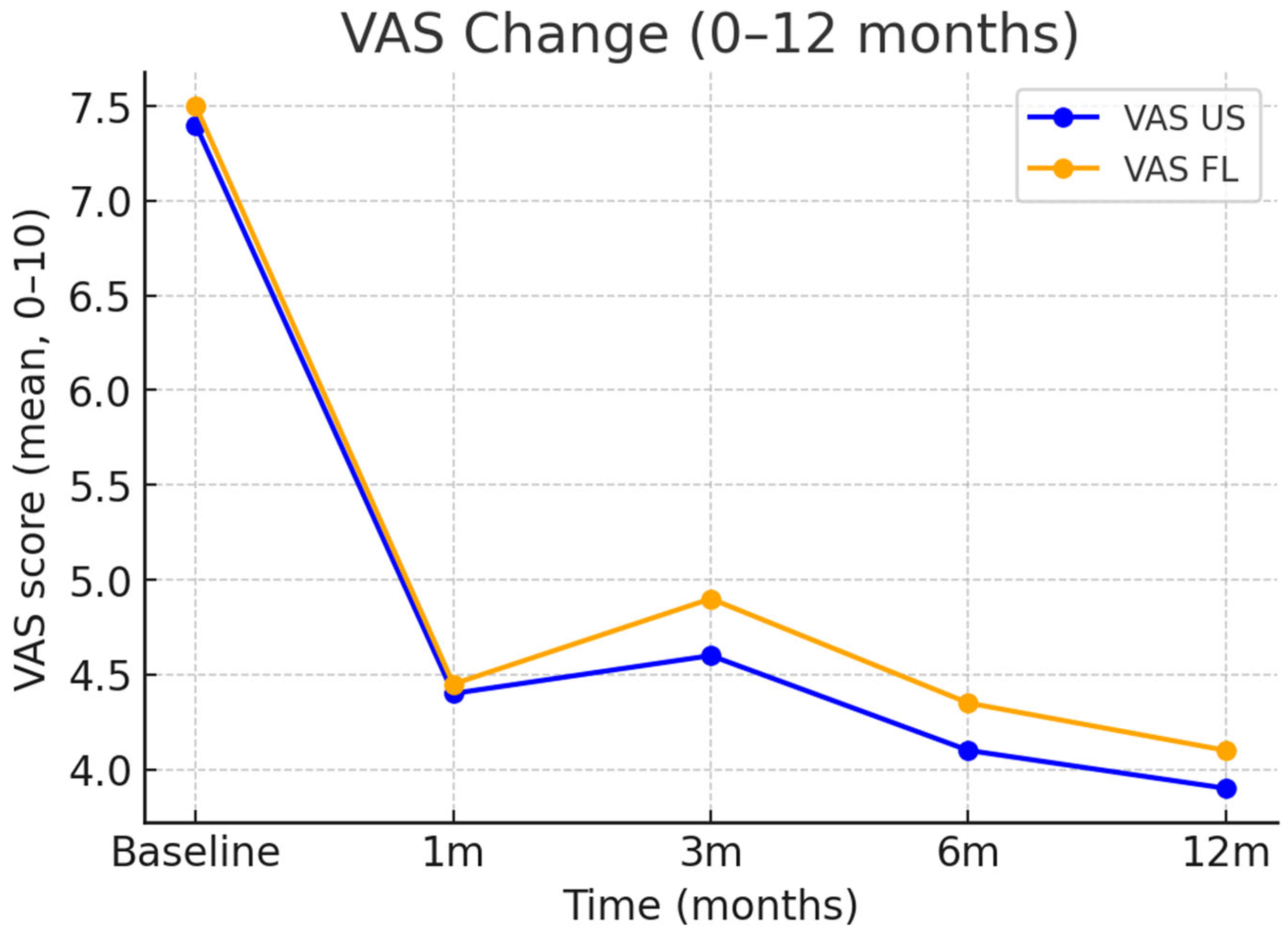
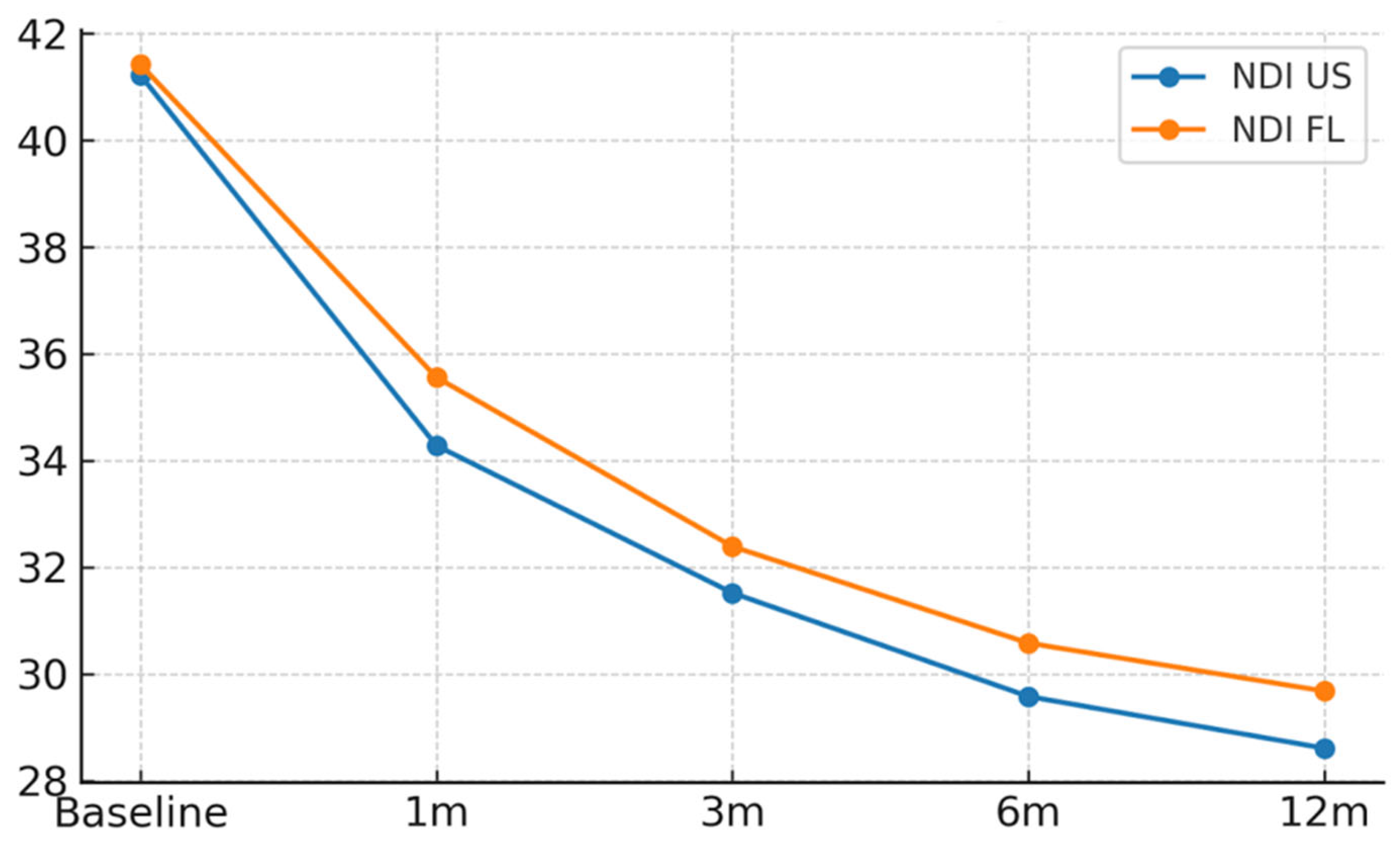
3.2. Pain and Functional Outcomes
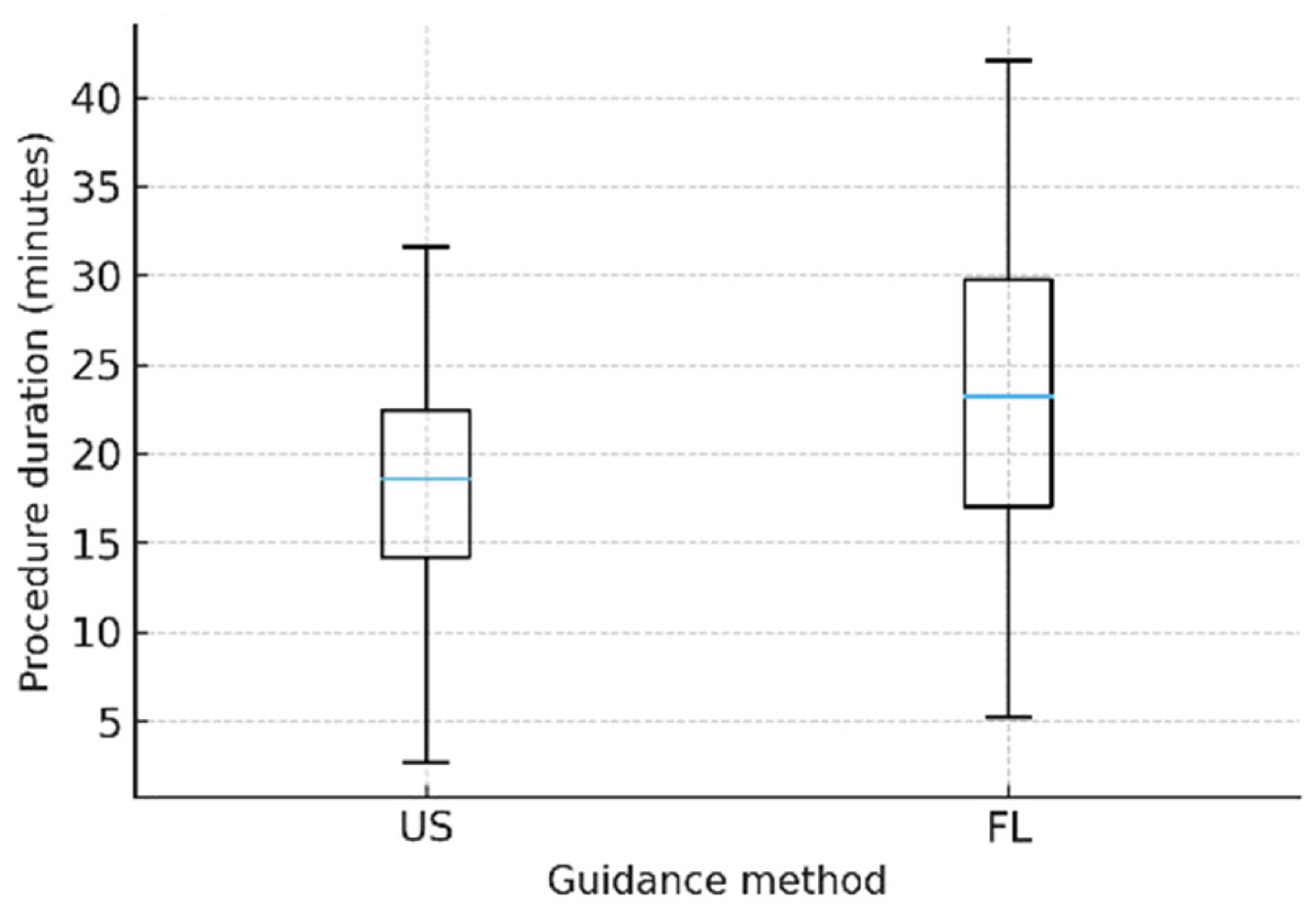
3.3. Secondary Endpoint: Procedure Time
3.4. Primary Endpoint: Technical Success
3.5. Exploratory NI Analyses of Clinical Outcomes
3.6. Sedation and Steroid Use
3.7. Safety Outcomes
3.8. Radiation Exposure (FL Group)
3.9. Subgroup Analyses
4. Discussion
5. Limitations
6. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| US | Ultrasound |
| FL | Fluoroscopy |
| NI | Non-Inferiority |
| VAS | Visual Analog Scale |
| NDI | Neck Disability Index |
| RFA | Radiofrequency Ablation |
| MCID | Minimum Clinically Important Difference |
| IPTW | Inverse Probability of Treatment Weighting |
| SMD | Standardized Mean Difference |
| CIRSE | Cardiovascular and Interventional Radiological Society of Europe (complication classification) |
| ALARA | As Low As Reasonably Achievable (radiation safety principle) |
| ROC | Receiver Operating Characteristic |
| AUC | Area Under the Curve |
| HRQoL | Health-Related Quality of Life |
| BMI | Body Mass Index |
| DAP | Dose–Area Product |
| GEE | Generalized Estimating Equations |
References
- Wu, A.-M.; Cross, M.; Elliott, J.M.; Culbreth, G.T.; Haile, L.M.; Steinmetz, J.D.; Hagins, H.; Kopec, J.A.; Brooks, P.M.; Woolf, A.D.; et al. Global, regional, and national burden of neck pain, 1990–2020, with projections to 2050: A systematic analysis for the Global Burden of Disease Study 2021. Lancet Rheumatol. 2024, 6, e142–e155. [Google Scholar] [CrossRef] [PubMed]
- Safiri, S.; Kolahi, A.A.; Hoy, D.; Buchbinder, R.; Mansournia, M.A.; Bettampadi, D.; Ashrafi-Asgarabad, A.; Almasi-Hashiani, A.; Smith, E.; Sepidarkish, M.; et al. Global, regional, and national burden of neck pain in the general population, 1990–2017: Systematic analysis of the Global Burden of Disease Study 2017. BMJ 2020, 368, m791. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Falco, F.J.; Datta, S.; Manchikanti, L.; Sehgal, N.; Geffert, S.; Singh, V.; Smith, H.S.; Boswell, M.V. An updated review of the diagnostic utility of cervical facet joint injections. Pain Physician 2012, 15, E807–E838. [Google Scholar] [CrossRef] [PubMed]
- Manchikanti, L.; Kaye, A.D.; Boswell, M.V.; Bakshi, S.; Gharibo, C.G.; Grami, V.; Grider, J.S.; Gupta, S.; Jha, S.S.; Mann, D.P.; et al. A Systematic Review and Best Evidence Synthesis of the Effectiveness of Therapeutic Facet Joint Interventions in Managing Chronic Spinal Pain. Pain Physician 2015, 18, E535–E582. [Google Scholar] [CrossRef] [PubMed]
- Hurley, R.W.; Adams, M.C.B.; Barad, M.; Bhaskar, A.; Bhatia, A.; Chadwick, A.; Deer, T.R.; Hah, J.; Hooten, W.M.; Kissoon, N.R.; et al. Consensus practice guidelines on interventions for cervical spine (facet) joint pain from a multispecialty international working group. Reg. Anesth. Pain Med. 2022, 47, 3–59. [Google Scholar] [CrossRef]
- Moreno, B.; Barbosa, J. Ultrasound-Guided Procedures in the Cervical Spine. Cureus 2021, 13, e20361. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Manchikanti, L.; Kosanovic, R.; Pampati, V.; Sanapati, M.R.; Hirsch, J.A. Outcomes of Cervical Therapeutic Medial Branch Blocks and Radiofrequency Neurotomy: Clinical Outcomes and Cost Utility are Equivalent. Pain Physician 2022, 25, 35–47. [Google Scholar] [PubMed]
- Narouze, S.N.; Benzon, H.T.; Provenzano, D.A.; Buvanendran, A.; De Andres, J.; Deer, T.R.; Rauck, R.; Huntoon, M.A. Interventional spine and pain procedures in patients on antiplatelet and anticoagulant medications: Guidelines from the American Society of Regional Anesthesia and Pain Medicine, the European Society of Regional Anaesthesia and Pain Therapy, the American Academy of Pain Medicine, the International Neuromodulation Society, the North American Neuromodulation Society, and the World Institute of Pain. Reg. Anesth. Pain Med. 2015, 40, 182–212. [Google Scholar] [CrossRef]
- Smith, J.; Finnoff, J.T.; Fitzgerald, J.; Minkowitz, G.; Narouze, S.N.; Shipp, T.; Sofferman, R.A.; Antonakis, J.; Baskin, H.J.; Brennan, F.H.; et al. AIUM Practice Parameter for the Performance of Selected Ultrasound-Guided Procedures. J. Ultrasound Med. 2016, 35, 1–40. [Google Scholar] [CrossRef]
- Klauser, A.S.; Peetrons, P. Developments in musculoskeletal ultrasound and clinical applications. Skelet. Radiol. 2010, 39, 1061–1071. [Google Scholar] [CrossRef] [PubMed]
- Tranquart, F.; Grenier, N.; Eder, V.; Pourcelot, L. Clinical use of ultrasound tissue harmonic imaging. Ultrasound Med. Biol. 1999, 25, 889–894. [Google Scholar] [CrossRef] [PubMed]
- International Council for Harmonisation (ICH). E9(R1) Addendum on Estimands and Sensitivity Analysis in Clinical Trials. 2019. Available online: https://database.ich.org/sites/default/files/E9-R1_Step4_Guideline_2019_1203.pdf (accessed on 10 February 2026).
- Piaggio, G.; Elbourne, D.R.; Pocock, S.J.; Evans, S.J.W.; Altman, D.G.; CONSORT Group. Reporting of noninferiority and equivalence randomized trials: Extension of the CONSORT 2010 statement. JAMA 2012, 308, 2594–2604. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, D.; Piché, F.; Mares, C.; Denis, I. Radiation exposure in fluoroscopy-guided spinal interventions: A prospective observational study of standard practice in a physiatry academic center. Interv. Pain Med. 2023, 2, 100273. [Google Scholar] [CrossRef] [PubMed]
- Hertault, A.; Maurel, B.; Midulla, M.; Bordier, C.; Desponds, L.; Kilani, M.S. Editor’s Choice—Minimizing radiation exposure during endovascular procedures: Basic knowledge, literature review, and reporting standards. Eur. J. Vasc. Endovasc. Surg. 2015, 50, 21–36. [Google Scholar] [CrossRef]
- von Elm, E.; Altman, D.G.; Egger, M.; Pocock, S.J.; Gøtzsche, P.C.; Vandenbroucke, J.P.; STROBE Initiative. The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement: Guidelines for reporting observational studies. Lancet 2007, 370, 1453–1457. [Google Scholar] [CrossRef]
- Filippiadis, D.K.; Binkert, C.; Pellerin, O.; Hoffmann, R.T.; Krajina, A.; Pereira, P.L. Cirse Quality Assurance Document and Standards for Classification of Complications: The Cirse Classification System. Cardiovasc. Interv. Radiol. 2017, 40, 1141–1146. [Google Scholar] [CrossRef]
- Farrar, J.T.; Young, J.P., Jr.; LaMoreaux, L.; Werth, J.L.; Poole, R.M. Clinical importance of changes in chronic pain intensity measured on an 11-point numerical pain rating scale. Pain 2001, 94, 149–158. [Google Scholar] [CrossRef]
- Dworkin, R.H.; Turk, D.C.; Wyrwich, K.W.; Beaton, D.; Cleeland, C.S.; Farrar, J.T.; Haythornthwaite, J.A.; Jensen, M.P.; Kerns, R.D.; Ader, D.N.; et al. Interpreting the clinical importance of treatment outcomes in chronic pain clinical trials: IMMPACT recommendations. J. Pain 2008, 9, 105–121. [Google Scholar] [CrossRef]
- Austin, P.C. Balance diagnostics for comparing the distribution of baseline covariates between treatment groups in propensity-score matched samples. Stat. Med. 2009, 28, 3083–3107. [Google Scholar] [CrossRef]
- Finlayson, R.J.; Etheridge, J.-P.B.; Tiyaprasertkul, W.; Nelems, B.; Tran, D.Q. A randomized comparison between ultrasound- and fluoroscopy-guided C7 medial branch block. Reg. Anesth. Pain Med. 2015, 40, 52–57. [Google Scholar] [CrossRef]
- Park, K.D.; Lim, D.J.; Lee, W.Y.; Ahn, J.; Park, Y. Ultrasound versus fluoroscopy-guided cervical medial branch block for the treatment of chronic cervical facet joint pain: A retrospective comparative study. Skelet. Radiol. 2017, 46, 81–91. [Google Scholar] [CrossRef]
- Viderman, D.; Aubakirova, M.; Aryngazin, A.; Yessimova, D.; Kaldybayev, D.; Tankacheyev, R.; Abdildin, Y.G. Ultrasound-Guided vs. Fluoroscopy-Guided Interventions for Back Pain Management: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Diagnostics 2023, 13, 3474. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Finlayson, R.J.; Thonnagith, A.; Elgueta, M.F.; Perez, J.; Etheridge, J.P.B.; Tran, D.Q. Ultrasound-guided cervical medial branch radiofrequency neurotomy: Can multitined deployment cannulae be the solution? Reg. Anesth. Pain Med. 2017, 42, 45–51. [Google Scholar] [CrossRef] [PubMed]
- Dindo, D.; Demartines, N.; Clavien, P.A. Classification of surgical complications: A new proposal with evaluation in a cohort of 6336 patients and results of a survey. Ann. Surg. 2004, 240, 205–213. [Google Scholar] [CrossRef] [PubMed]
- Rauch, S.; Kasuya, Y.; Turan, A.; Neamtu, A.; Vinayakan, A.; Sessler, D.I. Ultrasound-guided lumbar medial branch block in obese patients: A fluoroscopically confirmed clinical feasibility study. Reg. Anesth. Pain Med. 2009, 34, 340–342. [Google Scholar] [CrossRef] [PubMed]
- Putzu, M.; Marchesini, M. Ultrasound block of the medial branch: Learning the technique using CUSUM curves. Anesth. Essays Res. 2022, 15, 385–390. [Google Scholar] [CrossRef] [PubMed]
- Tekin, I.; Mirzai, H.; Ok, G.; Erbuyun, K.; Vatansever, D. A comparison of conventional and pulsed radiofrequency denervation in the treatment of chronic facet joint pain. Clin. J. Pain 2007, 23, 524–529. [Google Scholar] [CrossRef]
- Leveille, S.G.; Bean, J.; Ngo, L.; McMullen, W.; Guralnik, J.M. The pathway from musculoskeletal pain to mobility difficulty in older disabled women. Pain 2007, 128, 69–77. [Google Scholar] [CrossRef]
- Wong, M.J.; Rajarathinam, M. Ultrasound-guided axial facet joint interventions for chronic spinal pain: A narrative review. Can. J. Pain 2023, 7, 2193617. [Google Scholar] [CrossRef]

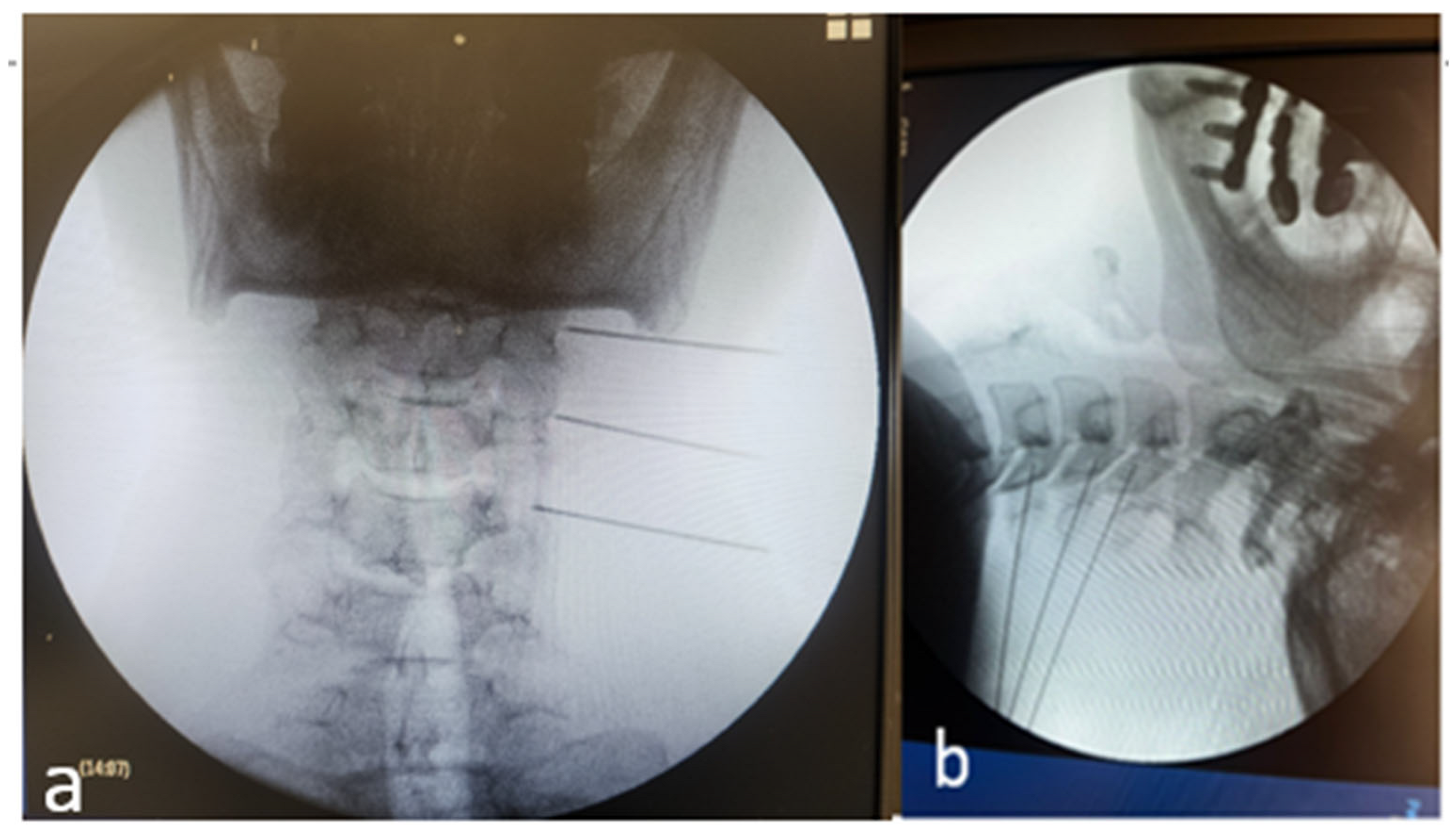





| Characteristic | US (n = 104) | FL (n = 120) |
|---|---|---|
| Age (years), mean ± SD | 57.3 ± 9.7 | 56.9 ± 10.4 |
| BMI (kg/m2), mean ± SD | 27.7 ± 4.3 | 27.0 ± 4.9 |
| Female, n (%) | 54 (51.9) | 66 (55.0) |
| Target level: C3, n (%) | 35 (33.7) | 43 (35.8) |
| Target level: C4, n (%) | 44 (42.3) | 56 (46.7) |
| Target level: C5, n (%) | 37 (35.6) | 47 (39.2) |
| Target level: C6, n (%) | 41 (39.4) | 41 (34.2) |
| Procedure: RFA, n (%) | 48 (46.2) | 50 (41.7) |
| Procedure: Diagnostic block, n (%) | 34 (32.7) | 44 (36.7) |
| Procedure: Therapeutic block, n (%) | 22 (21.2) | 26 (21.7) |
| Outcome | Timepoint | US (Mean) | FL (Mean) | Difference (US–FL) |
|---|---|---|---|---|
| VAS | Baseline | 7.31 | 7.47 | −0.16 |
| NDI | Baseline | 41.22 | 41.42 | −0.20 |
| VAS | 1 month | 4.40 | 4.45 | −0.05 |
| NDI | 1 month | 34.28 | 35.56 | −1.28 |
| VAS | 3 months | 4.62 | 4.87 | −0.25 |
| NDI | 3 months | 31.52 | 32.39 | −0.87 |
| VAS | 6 months | 4.09 | 4.35 | −0.26 |
| NDI | 6 months | 29.58 | 30.58 | −1.00 |
| VAS | 12 months | 3.82 | 4.09 | −0.27 |
| NDI | 12 months | 28.61 | 29.68 | −1.07 |
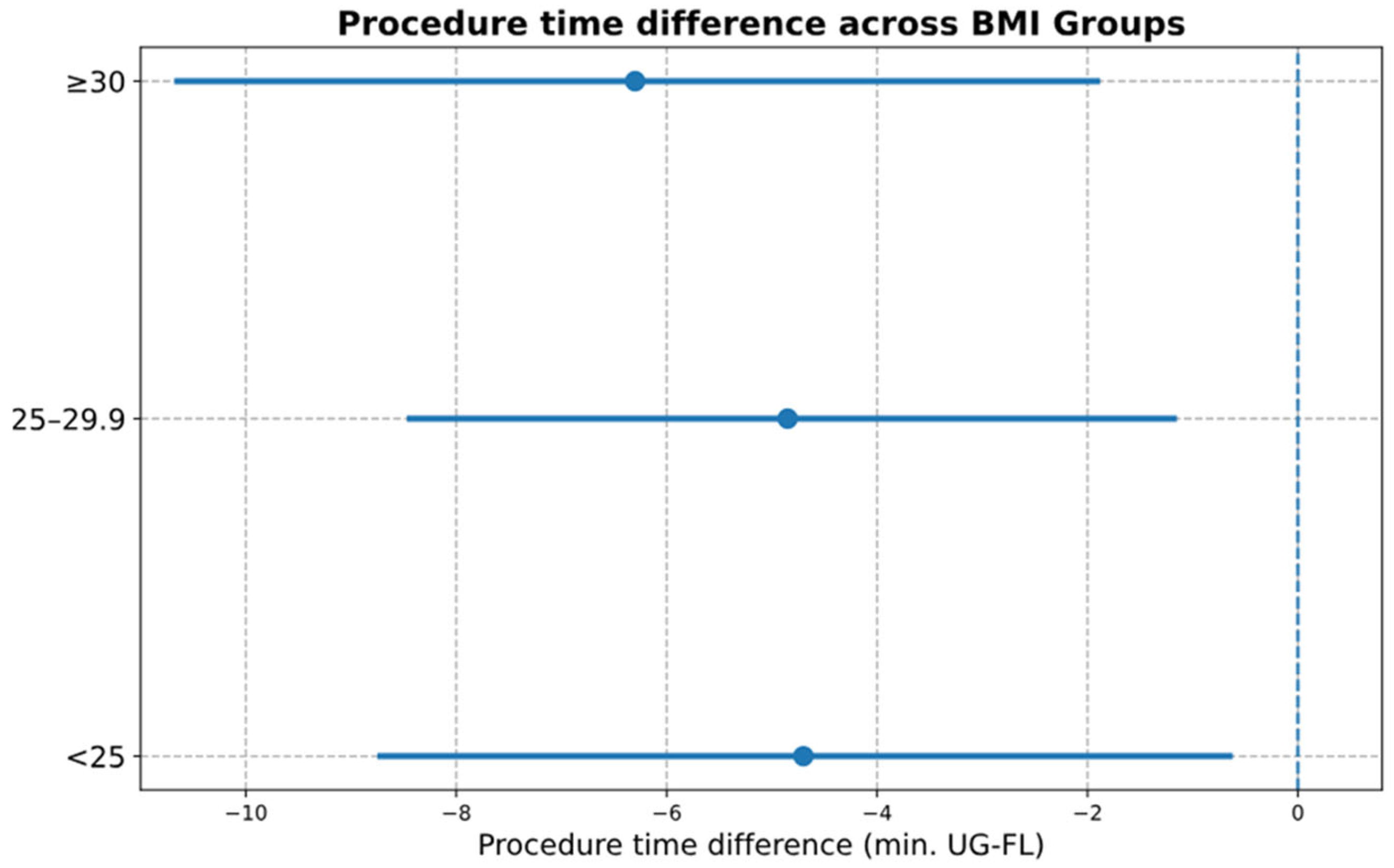
| Subgroup | US–FL Difference (min) | 95% CI | Interaction p | Comment |
|---|---|---|---|---|
| BMI < 25 | −4.7 | −8.8 to −0.6 | 0.851 | Exploratory; not significant |
| BMI 25–29.9 | −4.8 | −8.5 to −1.1 | - | - |
| BMI ≥ 30 | −6.3 | −10.7 to −1.9 | - | - |
| Female | −3.1 | −6.2 to 0.1 | 0.049 | Exploratory; not significant after adjustment |
| Male | −7.7 | −11.0 to −4.4 | - | - |
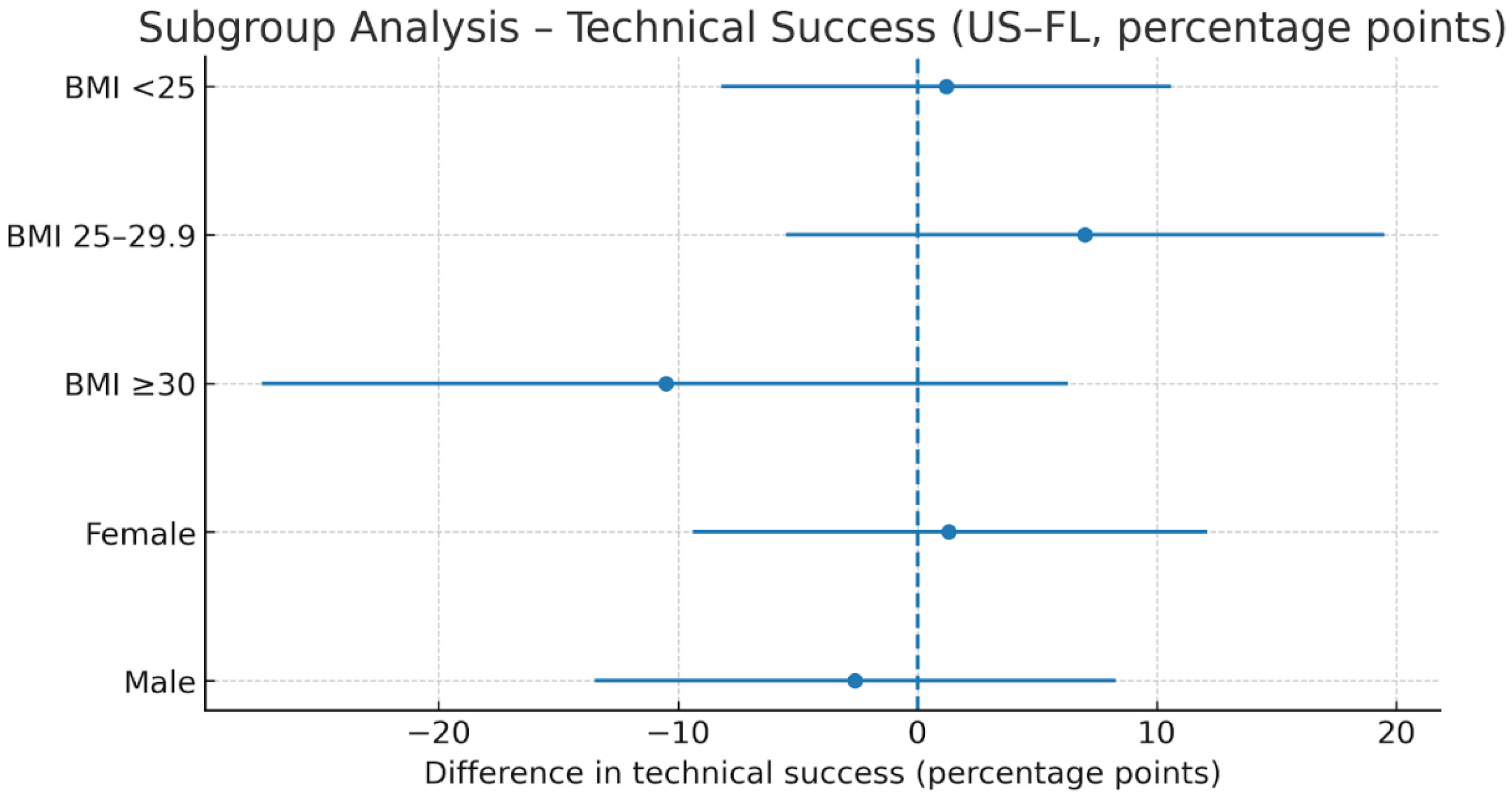
| Subgroup | US–FL Difference (%) | 95% CI | Interaction p | Comment |
|---|---|---|---|---|
| BMI < 25 | +1.2 | −8.2 to 10.6 | 0.267 | Exploratory, not significant |
| BMI 25–29.9 | +7.0 | −5.5 to 19.5 | - | - |
| BMI ≥ 30 | −10.5 | −27.4 to 6.3 | - | - |
| Female | +1.3 | −9.4 to 12.1 | 0.608 | Exploratory, not significant |
| Male | −2.6 | −13.5 to 8.3 | - | - |
| Metric | Mean ± SD | Median (IQR) |
|---|---|---|
| Fluoroscopy time (min) | 2.28 | 2.34 (1.66–2.94) |
| DAP (Gy·cm2) | 4.08 | 4.18 (2.83–5.48) |
| Air kerma (mGy) | 37.5 | 38.9 (25.5–47.2) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Kucukbingoz, C.; Yılmaz, A. Ultrasound Versus Fluoroscopy for Cervical Medial Branch Injections at C3–C6: A Single-Center Retrospective Cohort Study. Diagnostics 2026, 16, 592. https://doi.org/10.3390/diagnostics16040592
Kucukbingoz C, Yılmaz A. Ultrasound Versus Fluoroscopy for Cervical Medial Branch Injections at C3–C6: A Single-Center Retrospective Cohort Study. Diagnostics. 2026; 16(4):592. https://doi.org/10.3390/diagnostics16040592
Chicago/Turabian StyleKucukbingoz, Cagatay, and Ahmet Yılmaz. 2026. "Ultrasound Versus Fluoroscopy for Cervical Medial Branch Injections at C3–C6: A Single-Center Retrospective Cohort Study" Diagnostics 16, no. 4: 592. https://doi.org/10.3390/diagnostics16040592
APA StyleKucukbingoz, C., & Yılmaz, A. (2026). Ultrasound Versus Fluoroscopy for Cervical Medial Branch Injections at C3–C6: A Single-Center Retrospective Cohort Study. Diagnostics, 16(4), 592. https://doi.org/10.3390/diagnostics16040592





