64Cu-DOTATATE-PET/CT in Neuroborreliosis Shows Increased Tracer Uptake in Dorsal Root and Paravertebral Ganglia
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design and Population
2.2. Ethics
2.3. Clinical Data
2.4. 64Cu-DOTATATE PET/CT
2.5. MRI of the Brain and Spinal Cord
2.6. Interpretation of Scans
2.7. Endpoint
2.8. Statistical Analysis
3. Results
3.1. Baseline Characteristics
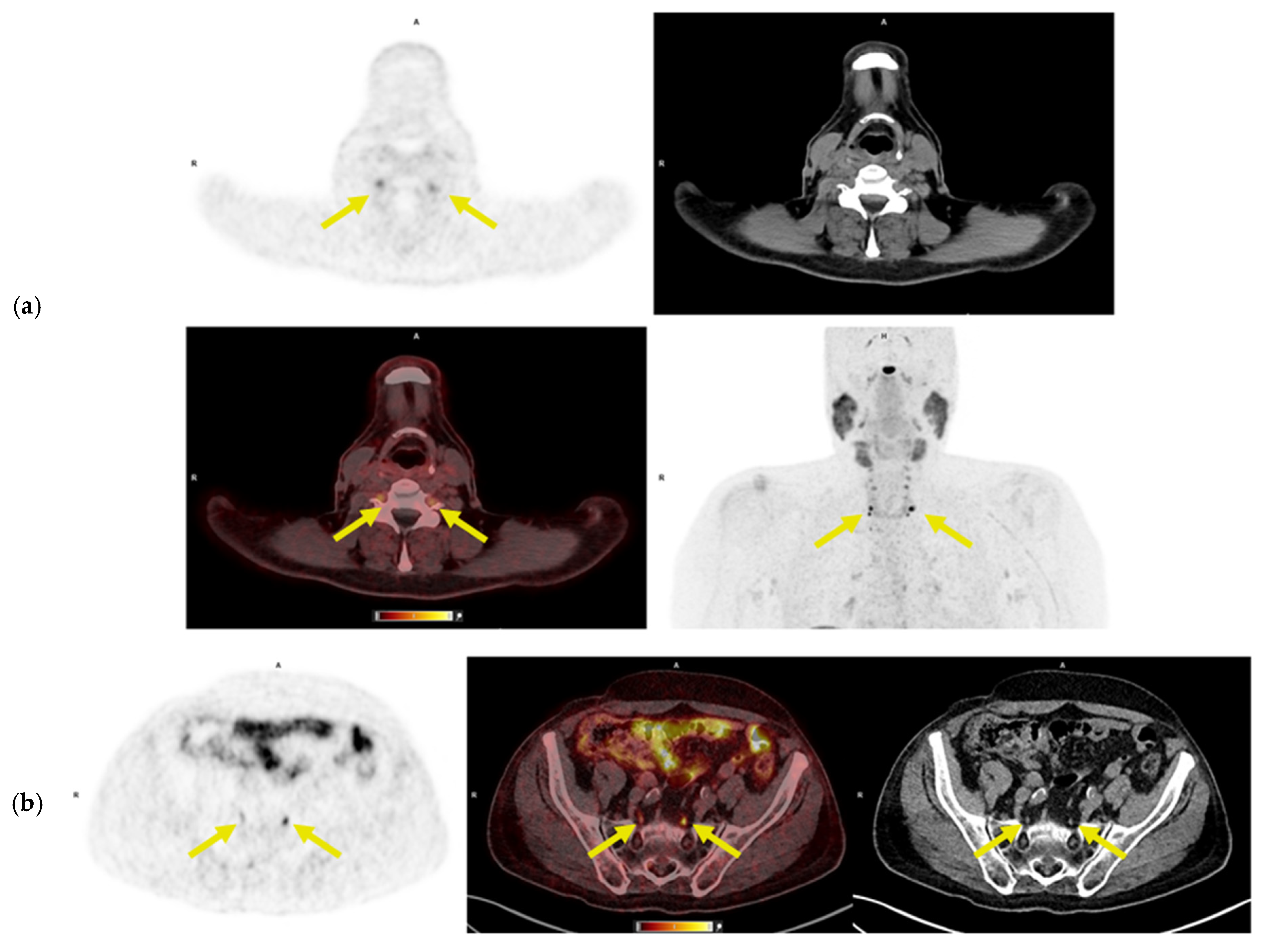
3.2. 64Cu-DOTATATE PET
3.3. MRI
3.4. Quality of Life
4. Discussion
Strength and Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| B. burgdorferi | Borrelia burgdorferi |
| CNS | Central nervous system |
| CSF | Cerebrospinal fluid |
| CT | Computed tomography |
| EFNSs | European Federation of Neurological Societies |
| IQR | Interquartile range |
| LAFOV | Long axial field of view |
| MBq | Megabecquerel |
| MFI-20 | Multidimensional Fatigue Inventory-20 |
| MIP | Maximum intensity projection |
| MoCA | Montreal Cognitive Assessment |
| MRI | Magnetic resonance imaging |
| NB | Neuroborreliosis |
| NET | Neuroendocrine tumor |
| PET | Positron emission tomography |
| PSF | Point spread function |
| REDCap | Research Electronic Data Capture |
| SF-36 | Short Form (36) Health Survey |
| SSTR2 | Somatostatin receptor subtype 2 |
| SUVmax | Maximum standardized uptake value |
| SUVmean | Mean standardized uptake value |
References
- Mygland, A.; Ljostad, U.; Fingerle, V.; Rupprecht, T.; Schmutzhard, E.; Steiner, I. EFNS guidelines on the diagnosis and management of European Lyme neuroborreliosis. Eur. J. Neurol. 2010, 17, 8-e4. [Google Scholar] [CrossRef] [PubMed]
- Halperin, J.J.; Eikeland, R.; Branda, J.A.; Dersch, R. Lyme neuroborreliosis: Known knowns, known unknowns. Brain 2022, 145, 2635–2647. [Google Scholar] [CrossRef] [PubMed]
- Hansen, K.; Lebech, A.M. The clinical and epidemiological profile of Lyme neuroborreliosis in Denmark 1985–1990. A prospective study of 187 patients with Borrelia burgdorferi specific intrathecal antibody production. Brain 1992, 115, 399–423. [Google Scholar] [CrossRef] [PubMed]
- Solheim, A.M.; Skarstein, I.; Quarsten, H.; Lorentzen, Å.R.; Berg-Hansen, P.; Eikeland, R.; Reiso, H.; Mygland, Å.; Ljøstad, U. Clinical and laboratory characteristics during a 1-year follow-up in European Lyme neuroborreliosis: A prospective cohort study. Eur. J. Neurol. 2024, 31, e16487. [Google Scholar] [CrossRef]
- Knudtzen, F.C.; Andersen, N.S.; Jensen, T.G.; Skarphédinsson, S. Characteristics and Clinical Outcome of Lyme Neuroborreliosis in a High Endemic Area, 1995–2014: A Retrospective Cohort Study in Denmark. Clin. Infect. Dis. 2017, 65, 1489–1495. [Google Scholar] [CrossRef] [PubMed]
- Stupica, D.; Bajrović, F.F.; Blagus, R.; Cerar Kišek, T.; Collinet-Adler, S.; Lah, A.; Levstek, E.; Ružić-Sabljić, E. Clinical manifestations and long-term outcome of early Lyme neuroborreliosis according to the European Federation of Neurological Societies diagnostic criteria (definite versus possible) in central Europe. A retrospective cohort study. Eur. J. Neurol. 2021, 28, 3155–3166. [Google Scholar] [CrossRef]
- Nordberg, C.L.; Bodilsen, J.; Knudtzen, F.C.; Storgaard, M.; Brandt, C.; Wiese, L.; Hansen, B.R.; Andersen, Å.B.; Nielsen, H.; Lebech, A.M. Lyme neuroborreliosis in adults: A nationwide prospective cohort study. Ticks Tick. Borne Dis. 2020, 11, 101411. [Google Scholar] [CrossRef] [PubMed]
- Krüger, H.; Reuss, K.; Pulz, M.; Rohrbach, E.; Pflughaupt, K.W.; Martin, R.; Mertens, H.G. Meningoradiculitis and encephalomyelitis due to Borrelia burgdorferi: A follow-up study of 72 patients over 27 years. J. Neurol. 1989, 236, 322–328. [Google Scholar] [CrossRef] [PubMed]
- Vrijmoeth, H.D.; Ursinus, J.; Harms, M.G.; Tulen, A.D.; Baarsma, M.E.; Van De Schoor, F.R.; Gauw, S.A.; Zomer, T.P.; Vermeeren, Y.M.; Ferreira, J.A.; et al. Determinants of persistent symptoms after treatment for Lyme borreliosis: A prospective observational cohort study. eBioMedicine 2023, 98, 104825. [Google Scholar] [CrossRef] [PubMed]
- Carlsen, E.A.; Johnbeck, C.B.; Binderup, T.; Loft, M.; Pfeifer, A.; Mortensen, J.; Oturai, P.; Loft, A.; Berthelsen, A.K.; Langer, S.W.; et al. 64Cu-DOTATATE PET/CT and Prediction of Overall and Progression-Free Survival in Patients with Neuroendocrine Neoplasms. J. Nucl. Med. 2020, 61, 1491–1497. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Hadji-Turdeghal, K.; Fosbøl, M.; Hasbak, P.; Löfgren, J.; Bull Rasmussen, I.; Bundgaard, H.; Iversen, K.; Bruun, N.E.; Møller, C.H.; Tuxen, C.; et al. First-In-Human Study of [(64)Cu]Cu-DOTATATE PET/CT in Infective Endocarditis: A Prospective Head-to-Head Comparison With [(18)F]FDG. Circ. Cardiovasc. Imaging 2025, 18, e017156. [Google Scholar] [CrossRef] [PubMed]
- Jensen, J.K.; Madsen, J.S.; Jensen, M.E.K.; Kjaer, A.; Ripa, R.S. [64Cu]Cu-DOTATATE PET metrics in the investigation of atherosclerotic inflammation in humans. J. Nucl. Cardiol. 2023, 30, 986–1000. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Nobashi, T.; Nakamoto, Y.; Kubo, T.; Ishimori, T.; Handa, T.; Tanizawa, K.; Sano, K.; Mishima, M.; Togashi, K. The utility of PET/CT with 68Ga-DOTATOC in sarcoidosis: Comparison with 67Ga-scintigraphy. Ann. Nucl. Med. 2016, 30, 544–552. [Google Scholar] [CrossRef]
- Verhaegh, D.; Joosten, L.A.B.; Oosting, M. The role of host immune cells and Borrelia burgdorferi antigens in the etiology of Lyme disease. Eur. Cytokine Netw. 2017, 28, 70–84. [Google Scholar] [CrossRef] [PubMed]
- Woitzik, P.; Linder, S. Molecular Mechanisms of Borrelia burgdorferi Phagocytosis and Intracellular Processing by Human Macrophages. Biology 2021, 10, 567. [Google Scholar] [CrossRef]
- Skovsbo Clausen, A.; Ørbæk, M.; Renee Pedersen, R.; Oestrup Jensen, P.; Lebech, A.M.; Kjaer, A. 64Cu-DOTATATE Positron Emission Tomography (PET) of Borrelia Burgdorferi Infection: In Vivo Imaging of Macrophages in Experimental Model of Lyme Arthritis. Diagnostics 2020, 10, 790. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Ørbæk, M.; Bodilsen, J.; Gynthersen, R.M.M.; Shekhrajka, N.; Nordberg, C.L.; Larsen, L.; Storgaard, M.; Brandt, C.; Wiese, L.; Hansen, B.R.; et al. CT and MR neuroimaging findings in patients with Lyme neuroborreliosis: A national prospective cohort study. J. Neurol. Sci. 2020, 419, 117176. [Google Scholar] [CrossRef] [PubMed]
- Lindland, E.S.; Solheim, A.M.; Andreassen, S.; Quist-Paulsen, E.; Eikeland, R.; Ljostad, U.; Mygland, A.; Elsais, A.; Nygaard, G.O.; Lorentzen, A.R.; et al. Imaging in Lyme neuroborreliosis. Insights Imaging 2018, 9, 833–844. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Loft, M.; Carlsen, E.A.; Johnbeck, C.B.; Johannesen, H.H.; Binderup, T.; Pfeifer, A.; Mortensen, J.; Oturai, P.; Loft, A.; Berthelsen, A.K.; et al. 64Cu-DOTATATE PET in Patients with Neuroendocrine Neoplasms: Prospective, Head-to-Head Comparison of Imaging at 1 Hour and 3 Hours After Injection. J. Nucl. Med. 2021, 62, 73–80. [Google Scholar] [CrossRef] [PubMed]
- Newberg, A.; Hassan, A.; Alavi, A. Cerebral metabolic changes associated with Lyme disease. Nucl. Med. Commun. 2002, 23, 773–777. [Google Scholar] [CrossRef] [PubMed]
- Plotkin, M.; Hautzel, H.; Krause, B.J.; Mohr, S.; Langen, K.J.; Muller, H.W. Fluorine-18-labeled fluorodeoxyglucose-positron emission tomography studies of acute brainstem Lyme neuroborreliosis. J. Neurosurg. 2005, 102, 927–929. [Google Scholar] [CrossRef] [PubMed]
- Ørbæk, M.; Klausen, C.; Lebech, A.M.; Mens, H. Lyme Neuroborreliosis in a Patient with Breast Cancer: MRI and PET/CT Findings. Diagnostics 2020, 10, 36. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Glaudemans, A.W.; Slart, R.H.; van Dijl, J.M.; van Oosten, M.; van Dam, G.M. Molecular imaging of infectious and inflammatory diseases: A terra incognita. J. Nucl. Med. 2015, 56, 659–661. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Coughlin, J.M.; Yang, T.; Rebman, A.W.; Bechtold, K.T.; Du, Y.; Mathews, W.B.; Lesniak, W.G.; Mihm, E.A.; Frey, S.M.; Marshall, E.S.; et al. Imaging glial activation in patients with post-treatment Lyme disease symptoms: A pilot study using [11C]DPA-713 PET. J. Neuroinflammation 2018, 15, 346. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Kortela, E.; Kanerva, M.J.; Puustinen, J.; Hurme, S.; Airas, L.; Lauhio, A.; Hohenthal, U.; Jalava-Karvinen, P.; Nieminen, T.; Finnilä, T.; et al. Oral Doxycycline Compared to Intravenous Ceftriaxone in the Treatment of Lyme Neuroborreliosis: A Multicenter, Equivalence, Randomized, Open-label Trial. Clin. Infect. Dis. 2021, 72, 1323–1331. [Google Scholar] [CrossRef]
- Ørbæk, M.; Gynthersen, R.M.M.; Mens, H.; Brandt, C.; Stenør, C.; Wiese, L.; Andersen, Å.B.; Møller, H.J.; Lebech, A.M. Cerebrospinal fluid levels of the macrophage-specific biomarker sCD163 are diagnostic for Lyme neuroborreliosis: An observational cohort study. Clin. Chim. Acta 2023, 543, 117299. [Google Scholar] [CrossRef] [PubMed]
- Brem, C.E.; Goldberg, L.J. Early Erythema Migrans: Do Not Count on Plasma Cells. Am. J. Dermatopathol. 2022, 44, e23–e25. [Google Scholar] [CrossRef] [PubMed]
- Snik, M.E.; Stouthamer, N.; Hovius, J.W.; van Gool, M.M.J. Bridging the gap: Insights in the immunopathology of Lyme borreliosis. Eur. J. Immunol. 2024, 54, e2451063. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Ramesh, G.; Borda, J.T.; Gill, A.; Ribka, E.P.; Morici, L.A.; Mottram, P.; Martin, D.S.; Jacobs, M.B.; Didier, P.J.; Philipp, M.T. Possible role of glial cells in the onset and progression of Lyme neuroborreliosis. J. Neuroinflammation 2009, 6, 23. [Google Scholar] [CrossRef]
- Roberts, E.D.; Bohm, R.P., Jr.; Lowrie, R.C., Jr.; Habicht, G.; Katona, L.; Piesman, J.; Philipp, M.T. Pathogenesis of Lyme neuroborreliosis in the rhesus monkey: The early disseminated and chronic phases of disease in the peripheral nervous system. J. Infect. Dis. 1998, 178, 722–732. [Google Scholar] [CrossRef] [PubMed]
- Bär, K.J.; Schurigt, U.; Scholze, A.; Segond Von Banchet, G.; Stopfel, N.; Bräuer, R.; Halbhuber, K.J.; Schaible, H.G. The expression and localization of somatostatin receptors in dorsal root ganglion neurons of normal and monoarthritic rats. Neuroscience 2004, 127, 197–206. [Google Scholar] [CrossRef] [PubMed]
- Berg, Z.; Koppula, B.R. 68Ga-DOTATATE Uptake by Cervicothoracic (Stellate) Ganglia. Clin. Nucl. Med. 2019, 44, 810–811. [Google Scholar] [CrossRef] [PubMed]
- Mingels, C.; Chung, K.J.; Pantel, A.R.; Rominger, A.; Alberts, I.; Spencer, B.A.; Nardo, L.; Pyka, T. Total-Body PET/CT: Challenges and Opportunities. Semin. Nucl. Med. 2025, 55, 21–30. [Google Scholar] [CrossRef] [PubMed]
- Urso, L.; Frantellizzi, V.; Vincentis, G.D.; Schillaci, O.; Filippi, L.; Evangelista, L. Clinical applications of long axial field-of-view PET/CT scanners in oncology. Clin. Transl. Imaging 2023, 11, 365–380. [Google Scholar] [CrossRef]
- Heye, A.K.; Culling, R.D.; Valdés Hernández Mdel, C.; Thrippleton, M.J.; Wardlaw, J.M. Assessment of blood-brain barrier disruption using dynamic contrast-enhanced MRI. A systematic review. Neuroimage Clin. 2014, 6, 262–274. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Jimenez-Andrade, J.M.; Herrera, M.B.; Ghilardi, J.R.; Vardanyan, M.; Melemedjian, O.K.; Mantyh, P.W. Vascularization of the dorsal root ganglia and peripheral nerve of the mouse: Implications for chemical-induced peripheral sensory neuropathies. Mol. Pain 2008, 4, 10. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Halperin, J.J. Facial nerve palsy associated with lyme disease. Muscle Nerve Off. J. Am. Assoc. Electrodiagn. Med. 2003, 28, 516–517. [Google Scholar] [CrossRef]
- Aslam, B.; Nisar, M.A.; Khurshid, M.; Farooq Salamat, M.K. Immune escape strategies of Borrelia burgdorferi. Future Microbiol. 2017, 12, 1219–1237. [Google Scholar] [CrossRef] [PubMed]
- Ogrinc, K.; Kastrin, A.; Lotrič-Furlan, S.; Bogovič, P.; Rojko, T.; Maraspin, V.; Ružić-Sabljić, E.; Strle, K.; Strle, F. Colocalization of Radicular Pain and Erythema Migrans in Patients With Bannwarth Syndrome Suggests a Direct Spread of Borrelia Into the Central Nervous System. Clin. Infect. Dis. 2022, 75, 81–87. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Hakami, M.A.; Khan, F.R.; Abdulaziz, O.; Alshaghdali, K.; Hazazi, A.; Aleissi, A.F.; Abalkhail, A.; Alotaibi, B.S.; Alhazmi, A.Y.M.; Kukreti, N.; et al. Varicella-zoster virus-related neurological complications: From infection to immunomodulatory therapies. Rev. Med. Virol. 2024, 34, e2554. [Google Scholar] [CrossRef] [PubMed]
- Ramesh, G.; Didier, P.J.; England, J.D.; Santana-Gould, L.; Doyle-Meyers, L.A.; Martin, D.S.; Jacobs, M.B.; Philipp, M.T. Inflammation in the pathogenesis of lyme neuroborreliosis. Am. J. Pathol. 2015, 185, 1344–1360. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Cadavid, D.; O’Neill, T.; Schaefer, H.; Pachner, A.R. Localization of Borrelia burgdorferi in the nervous system and other organs in a nonhuman primate model of lyme disease. Lab. Investig. 2000, 80, 1043–1054. [Google Scholar] [CrossRef] [PubMed]
- Foret, J.; Paren, A.J.; Zayet, S.; Chirouze, C.; Gendrin, V.; Bouiller, K.; Klopfenstein, T. Residual Symptoms and Quality of Life After Treated Lyme Neuroborreliosis: Case-Control Study (QoLYME). Open Forum Infect. Dis. 2025, 12, ofaf042. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Andreassen, S.; Lindland, E.M.S.; Beyer, M.K.; Solheim, A.M.; Ljøstad, U.; Mygland, Å.; Lorentzen, Å.R.; Reiso, H.; Bjuland, K.J.; Pripp, A.H.; et al. Assessment of cognitive function, structural brain changes and fatigue 6 months after treatment of neuroborreliosis. J. Neurol. 2023, 270, 1430–1438. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]



| Characteristics | All Participants (n = 20) | NB Patients 1 (n = 15) | Controls 2 (n = 5) |
|---|---|---|---|
| Age, years, median (IQR) | 56 (48–66) | 58 (51–65) | 53 (39–66) |
| Sex, female (%) | 7 (35) | 5 (33) | 2 (40) |
| BMI, median (IQR) | 23.9 (22.9–27.6) | 23.5 (22.0–26.0) | 28.7 (24.7–29.1) |
| Charlson Comorbidity Index, median (IQR) | 2 (1–3) | 2 (1–3) | 1 (0–2) |
| History of tick bite (yes, %) | 14 (70) | 10 (67) | 4 (80) |
| Symptom duration before lumbar puncture, n (%) | |||
| 0–14 days | 3 (15) | 1 (7) | 2 (40) |
| 15–44 days | 13 (65) | 11 (73) | 2 (40) |
| 45–179 days | 4 (20) | 3 (20) | 1 (20) |
| Days of antibiotic treatment before PET/CT, median (IQR) 3 | 9.5 (7–11) | 10 (6–11) | 8 (7–16) |
| Self-reported symptoms (yes, %) | |||
| Radiating pain | 10 (50) | 9 (60) | 1 (20) |
| Facial/eye nerve paralysis | 5 (25) | 5 (33) | 0 (0) |
| Limb paresis | 3 (15) | 3 (20) | 0 (0) |
| Facial pain/sensory changes | 3 (15) | 2 (13) | 1 (20) |
| Sensory disturbances | 10 (50) | 7 (47) | 3 (60) |
| Headache | 9 (45) | 7 (47) | 2 (40) |
| Fatigue | 17 (85) | 13 (87) | 4 (80) |
| Dizziness | 11 (55) | 8 (53) | 3 (60) |
| Muscle pain | 12 (60) | 10 (67) | 2 (40) |
| Joint pain | 8 (40) | 6 (40) | 2 (40) |
| Memory/concentration issues | 8 (40) | 7 (46) | 1 (20) |
| Altered bowel movement | 4 (20) | 3 (20) | 1 (20) |
| Changes in hearing or taste | 5 (25) | 4 (27) | 1 (20) |
| CSF analysis | |||
| Leucocytes, cells/µL, median (IQR) | 110 (14–185) | 155 (103–246) | 1 (1–4) |
| Mononuclear cells/µL, median (IQR) | 105 (15–188) | 155 (102–246) | 1 (1–5.5) |
| Polynuclear cells, cells/µL, median (IQR) | 2 (1–4) | 3 (2–5) | 1 (0.5–1) |
| CXCL13 > 500 pg/mL (yes, %) | 6 (30) | 6 (40) | 0 (0) |
| B. burgdorferi AI index (positive, %) | 10 (50) | 10 (67) | 0 (0) |
| Blood analysis | |||
| B. burgdorferi IgG (positive, %) missing | 11 (69) (4) | 9 (60) (4) | 2 (40) |
| Elevated infectious parameters (yes, %) missing | 5 (29) 2 | 4 (29) 1 | 1 (25) 1 |
| Leucocytes, ×109/L, median (IQR) | 7.1 (5.9–8.5) | 6.9 (5.8–8.5) | 7.2 (5.9–8.4) |
| C-reactive protein, mg/L, median (IQR) | 1.0 (1–5.6) | 2.0 (1–5.6) | 1 (1–4) |
| 64Cu-DOTATATE Uptake | Number of Patients | Main Symptoms | Number of Patients |
|---|---|---|---|
| Cervical ganglia | 1 | Headache | 1 |
| Stellate ganglia only | 4 | Ipsilateral facial palsy and ipsilateral radiating upper extremity pain | 1 |
| Meningeal symptoms | 1 | ||
| Ipsilateral facial palsy and contralateral radiating upper extremity pain | 1 | ||
| Bilateral radiating lower extremity pain | 1 | ||
| Lumbosacral ganglia and stellate ganglia | 2 | Ipsilateral facial palsy, headache, and neck pain | 1 |
| Ipsilateral lower extremity pain | 1 | ||
| Both cervical and lumbosacral | 7 | Upper extremity pain and paresis | 1 |
| Ipsilateral facial palsy and upper extremity pain and paresis | 1 | ||
| Ipsilateral facial palsy and upper extremity paresis | 1 | ||
| Ipsilateral lower extremity pain and bilateral upper extremity sensory disturbances | 1 | ||
| Thoracic and bilateral radiating lower extremity pain | 1 | ||
| Ipsilateral facial palsy, thoracic, and bilateral radiating lower extremity pain | 1 | ||
| Ipsilateral facial palsy and radiating lower extremity pain | 1 | ||
| No enhancement | 1 | Ipsilateral facial palsy | 1 |
| Number of controls | Main symptoms | Number of controls | |
| Cervical ganglia | 1 | Erythema migrans and ipsilateral radiating neck pain | 1 |
| Stellate ganglia only | 1 | Bilateral facial sensory disturbances | 1 |
| Lumbosacral ganglia and stellate ganglia | 0 | NA | |
| Both cervical and lumbosacral | 0 | NA | |
| No enhancement | 3 | Headache and neck pain | 2 |
| Neck pain; sensory disturbances | 1 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Ørbæk, M.; Fosbøl, M.Ø.; Florescu, A.M.; Stenør, C.M.; Jepsen, M.P.G.; Carlsen, J.F.; Brandt, C.T.; Petersen, P.T.; Mens, H.; Andersen, Å.B.; et al. 64Cu-DOTATATE-PET/CT in Neuroborreliosis Shows Increased Tracer Uptake in Dorsal Root and Paravertebral Ganglia. Diagnostics 2026, 16, 561. https://doi.org/10.3390/diagnostics16040561
Ørbæk M, Fosbøl MØ, Florescu AM, Stenør CM, Jepsen MPG, Carlsen JF, Brandt CT, Petersen PT, Mens H, Andersen ÅB, et al. 64Cu-DOTATATE-PET/CT in Neuroborreliosis Shows Increased Tracer Uptake in Dorsal Root and Paravertebral Ganglia. Diagnostics. 2026; 16(4):561. https://doi.org/10.3390/diagnostics16040561
Chicago/Turabian StyleØrbæk, Mathilde, Marie Øbro Fosbøl, Anna Maria Florescu, Christian Midtgaard Stenør, Micha Phill Grønholm Jepsen, Jonathan Frederik Carlsen, Christian Thomas Brandt, Pelle Trier Petersen, Helene Mens, Åse Bengaard Andersen, and et al. 2026. "64Cu-DOTATATE-PET/CT in Neuroborreliosis Shows Increased Tracer Uptake in Dorsal Root and Paravertebral Ganglia" Diagnostics 16, no. 4: 561. https://doi.org/10.3390/diagnostics16040561
APA StyleØrbæk, M., Fosbøl, M. Ø., Florescu, A. M., Stenør, C. M., Jepsen, M. P. G., Carlsen, J. F., Brandt, C. T., Petersen, P. T., Mens, H., Andersen, Å. B., Andersen, F. L., Law, I., Loft, A., Kjaer, A., & Lebech, A.-M. (2026). 64Cu-DOTATATE-PET/CT in Neuroborreliosis Shows Increased Tracer Uptake in Dorsal Root and Paravertebral Ganglia. Diagnostics, 16(4), 561. https://doi.org/10.3390/diagnostics16040561









