Safety of a Tailored Gadolinium-Based Contrast Agent Protocol Considering Excretion Pathways in Patients with Renal Impairment
Abstract
1. Introduction
2. Materials and Methods
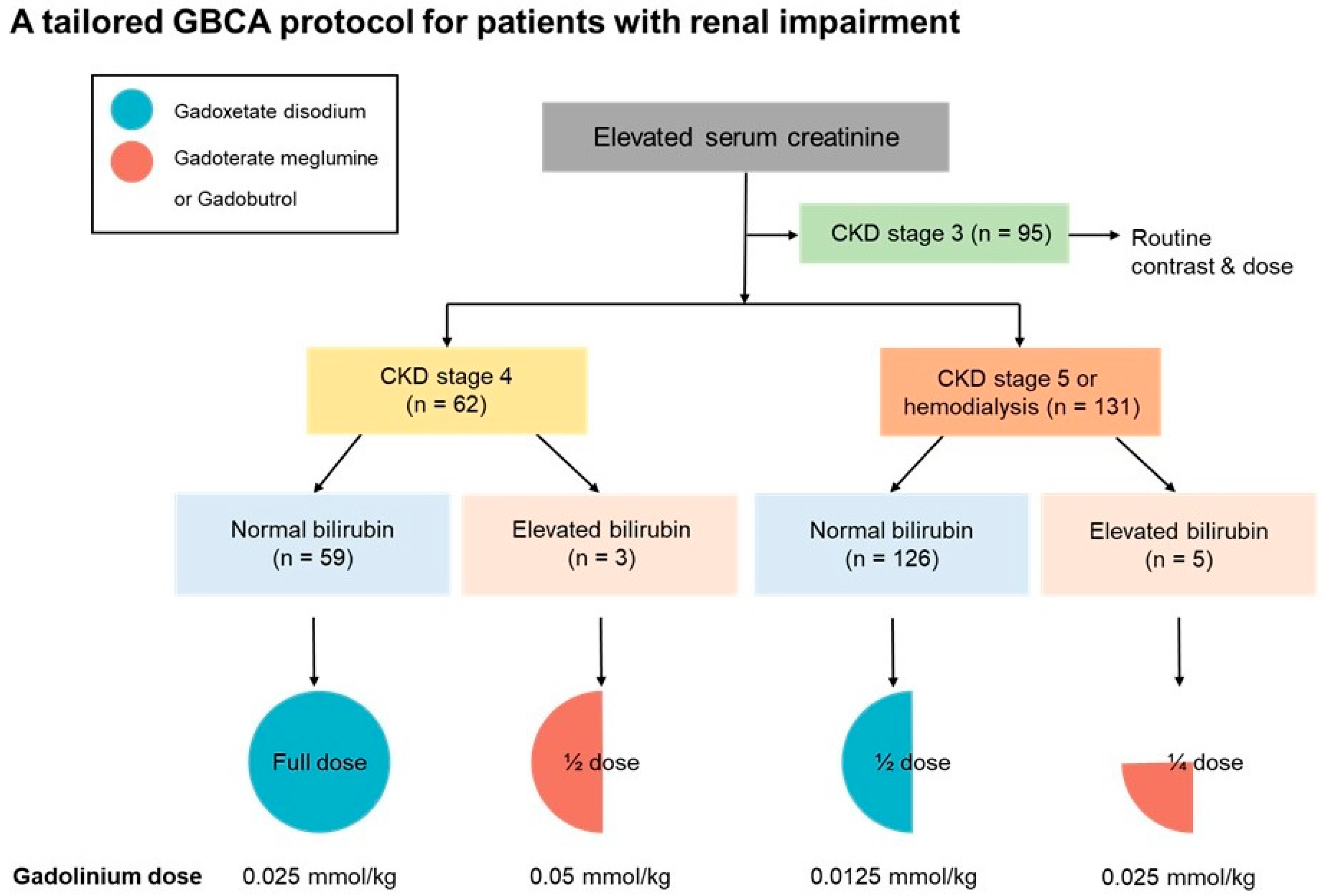
2.1. Patients
2.2. Clinical Outcomes
2.3. Image Quality Analysis for Non-Hepatobiliary MRI Examinations
2.4. Statistical Analysis
3. Results
3.1. Patients
3.2. Nephrogenic Systemic Fibrosis (NSF)
3.3. Gadolinium Deposition in Brain Tissue
3.4. Image Quality Analysis for Non-Hepatobiliary MRI Examinations
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| GBCA | Gadolinium-based contrast agent |
| NSF | Nephrogenic systemic fibrosis |
| ACR | American college of radiology |
| CKD | Chronic kidney disease |
| eGFR | Estimated glomerular filtration rate |
| CI | Confidence interval |
| ESUR | European society of urogenital radiology |
References
- Matsumura, T.; Hayakawa, M.; Shimada, F.; Yabuki, M.; Dohanish, S.; Palkowitsch, P.; Yoshikawa, K. Safety of gadopentetate dimeglumine after 120 million administrations over 25 years of clinical use. Magn. Reson. Med. Sci. 2013, 12, 297–304. [Google Scholar] [CrossRef]
- Tedeschi, E.; Caranci, F.; Giordano, F.; Angelini, V.; Cocozza, S.; Brunetti, A. Gadolinium retention in the body: What we know and what we can do. Radiol. Med. 2017, 122, 589–600. [Google Scholar] [CrossRef]
- Weinreb, J.C.; Rodby, R.A.; Yee, J.; Wang, C.L.; Fine, D.; McDonald, R.J.; Perazella, M.A.; Dillman, J.R.; Davenport, M.S. Use of intravenous gadolinium-based contrast media in patients with kidney disease: Consensus statements from the American College of Radiology and the National Kidney Foundation. Radiology 2021, 298, 28–35. [Google Scholar] [CrossRef]
- Starekova, J.; Bruce, R.J.; Sadowski, E.A.; Reeder, S.B. No cases of nephrogenic systemic fibrosis after administration of gadoxetic acid. Radiology 2020, 297, 556–562. [Google Scholar] [CrossRef]
- Tseng, T.Y.; Tseng, J.H.; Huang, B.S.; Lin, S.-Y.; Chen, C.-B.; Fang, Y.-W.; Lin, G.; Lai, Y.-C. Risk of nephrogenic systemic fibrosis in patients with impaired renal function undergoing fixed-dose gadoxetic acid–enhanced magnetic resonance imaging. Abdom. Radiol. 2021, 46, 3995–4001. [Google Scholar] [CrossRef]
- Gauthier, I.D.; Macleod, C.A.; Sathiadoss, P.; McGrath, T.A.; Nair, V.; Schieda, N. Risk of nephrogenic systemic fibrosis in oncology patients receiving gadoxetic acid and updated risk estimate in patients with moderate and severe renal impairment. Abdom. Radiol. 2022, 47, 1196–1201. [Google Scholar] [CrossRef]
- Choi, J.W.; Moon, W.J. Gadolinium deposition in the brain: Current updates. Korean J. Radiol. 2019, 20, 134–147. [Google Scholar] [CrossRef] [PubMed]
- Frydrychowicz, A.; Lubner, M.G.; Brown, J.J.; Merkle, E.M.; Nagle, S.K.; Rofsky, N.M.; Reeder, S.B. Hepatobiliary MR imaging with gadolinium-based contrast agents. J. Magn. Reson. Imaging 2012, 35, 492–511. [Google Scholar] [CrossRef] [PubMed]
- Mühler, A.; Heinzelmann, I.; Weinmann, H.J. Elimination of gadolinium-ethoxybenzyl-DTPA in a rat model of severely impaired liver and kidney excretory function: An experimental study in rats. Investig. Radiol. 1994, 29, 213–216. [Google Scholar] [CrossRef]
- Kirchin, M.A.; Lorusso, V.; Pirovano, G. Compensatory biliary and urinary excretion of gadobenate ion after administration of gadobenate dimeglumine (MultiHance®) in cases of impaired hepatic or renal function: A mechanism that may aid in the prevention of nephrogenic systemic fibrosis? Br. J. Radiol. 2015, 88, 20140526. [Google Scholar] [CrossRef] [PubMed]
- Gschwend, S.; Ebert, W.; Schultze-Mosgau, M.; Breuer, J. Pharmacokinetics and imaging properties of Gd-EOB-DTPA in patients with hepatic and renal impairment. Investig. Radiol. 2011, 46, 556–566. [Google Scholar] [CrossRef]
- American College of Radiology Committee on Drugs and Contrast Media. ACR Manual on Contrast Media. Available online: https://www.acr.org/Clinical-Resources/Contrast-Manual (accessed on 30 December 2025).
- Girardi, M.; Kay, J.; Elston, D.M.; LeBoit, P.E.; Abu-Alfa, A.; Cowper, S.E. Nephrogenic systemic fibrosis: Clinicopathological definition and workup recommendations. J. Am. Acad. Dermatol. 2011, 65, 1095–1106.e7. [Google Scholar] [CrossRef]
- Newcombe, R.G. Two-sided confidence intervals for the single proportion: Comparison of seven methods. Stat. Med. 1998, 17, 857–872. [Google Scholar] [CrossRef]
- Davies, J.; Siebenhandl-Wolff, P.; Tranquart, F.; Jones, P.; Evans, P. Gadolinium: Pharmacokinetics and toxicity in humans and laboratory animals following contrast agent administration. Arch. Toxicol. 2022, 96, 403–429. [Google Scholar] [CrossRef]
- Aime, S.; Caravan, P. Biodistribution of gadolinium-based contrast agents, including gadolinium deposition. J. Magn. Reson. Imaging 2009, 30, 1259–1267. [Google Scholar] [CrossRef]
- Perazella, M.A. Current status of gadolinium toxicity in patients with kidney disease. Clin. J. Am. Soc. Nephrol. 2009, 4, 461–469. [Google Scholar] [CrossRef]
- Lauenstein, T.; Ramirez-Garrido, F.; Kim, Y.H.; Rha, S.E.; Ricke, J.; Phongkitkarun, S.; Boettcher, J.; Gupta, R.T.; Korpraphong, P.; Tanomkiat, W.; et al. Nephrogenic systemic fibrosis risk after liver magnetic resonance imaging with gadoxetate disodium in patients with moderate to severe renal impairment: Results of a prospective, open-label, multicenter study. Investig. Radiol. 2015, 50, 416–422. [Google Scholar] [CrossRef] [PubMed]
- Schieda, N.; van der Pol, C.B.; Walker, D.; Tsampalieros, A.K.; Maralani, P.J.; Woo, S.; Davenport, M.S. Adverse events to the gadolinium-based contrast agent gadoxetic acid: Systematic review and meta-analysis. Radiology 2020, 297, 565–572. [Google Scholar] [CrossRef]
- Endrikat, J.S.; Dohanish, S.; Balzer, T.; Breuer, J.A. Safety of gadoxetate disodium: Results from the clinical phase II–III development program and postmarketing surveillance. J. Magn. Reson. Imaging 2015, 42, 634–643. [Google Scholar] [CrossRef] [PubMed]
- Song, K.D.; Kim, S.H.; Lee, J.; Kang, K.A.; Kim, J.; Yoo, H. Half-dose gadoxetic acid–enhanced liver magnetic resonance imaging in patients at risk for nephrogenic systemic fibrosis. Eur. J. Radiol. 2015, 84, 378–383. [Google Scholar] [CrossRef]
- Heinz-Peer, G.; Neruda, A.; Watschinger, B.; Vychytil, A.; Geusau, A.; Haumer, M.; Weber, M. Prevalence of nephrogenic systemic fibrosis following intravenous gadolinium contrast media administration in dialysis patients with end-stage renal disease. Eur. J. Radiol. 2010, 76, 129–134. [Google Scholar] [CrossRef]
- Woolen, S.A.; Shankar, P.R.; Gagnier, J.J.; MacEachern, M.P.; Singer, L.; Davenport, M.S. Risk of Nephrogenic Systemic Fibrosis in Patients with Stage 4 or 5 Chronic Kidney Disease Receiving a Group II Gadolinium-Based Contrast Agent: A Systematic Review and Meta-analysis. JAMA Intern. Med. 2020, 180, 223–230. [Google Scholar] [CrossRef]
- Kanda, T.; Ishii, K.; Kawaguchi, H.; Kitajima, K.; Takenaka, D. High signal intensity in the dentate nucleus and globus pallidus on unenhanced T1-weighted MR images: Relationship with increasing cumulative dose of a gadolinium-based contrast material. Radiology 2014, 270, 834–841. [Google Scholar] [CrossRef] [PubMed]
- de Campos, R.O.; Heredia, V.; Ramalho, M.; De Toni, M.S.; Lugo-Somolinos, A.; Fuller, E.R.; Semelka, R.C. Quarter-dose (0.025 mmol/kg) gadobenate dimeglumine for abdominal MRI in patients at risk for nephrogenic systemic fibrosis: Preliminary observations. AJR Am. J. Roentgenol. 2011, 196, 545–552. [Google Scholar] [CrossRef] [PubMed]
- Mathur, M.; Jones, J.R.; Weinreb, J.C. Gadolinium deposition and nephrogenic systemic fibrosis: A radiologist’s primer. Radiographics 2020, 40, 153–162. [Google Scholar] [CrossRef] [PubMed]
- Larson, K.N.; Gagnon, A.L.; Darling, M.D.; Patterson, J.W.; Cropley, T.G. Nephrogenic systemic fibrosis manifesting a decade after exposure to gadolinium. JAMA Dermatol. 2015, 151, 1117–1120. [Google Scholar] [CrossRef]


| Variables | CKD 3 (n = 95) | CKD 4 (n = 62) | CKD 5 (n = 31) | Hemodialysis (n = 100) | Overall (n = 288) |
|---|---|---|---|---|---|
| Male, n (%) | 73 (76.8) | 41 (66.1) | 15 (48.4) | 58 (58.0) | 187 (64.9) |
| Age (years) | 66.7 ± 11.4 | 68.0 ± 11.0 | 62.0 ± 12.1 | 63.2 ± 12.0 | 64.6 ± 11.7 |
| Follow-up period (months) | 26.2 ± 30.8 (range 1–120) | 34.7 ± 31.3 (range 1–120) | 27.8 ± 37.4 (range 1–120) | 24.1 ± 28.8 (range 1–120) | 27.5 ± 31.0 (range 1–120) |
| No. of GBCA-Enhanced MRI Examination | CKD 3 (n = 95) | CKD 4 (n = 62) | CKD 5 (n = 31) | Hemodialysis (n = 100) |
|---|---|---|---|---|
| 1 exam | 30 | 32 | 19 | 55 |
| 2 exams | 22 | 11 | 6 | 25 |
| 3 exams | 10 | 9 | 2 | 10 |
| ≥4 exams | 33 | 10 | 4 | 10 |
| Renal Function | CKD 4 | CKD 5 or Hemodialysis | ||
|---|---|---|---|---|
| Serum Bilirubin Levels | Normal † | Elevated ‡ | Normal § | Elevated ∥ |
| Abdomen | 88 | 2 | 67 | 5 |
| Neuro (Brain/Spine) | 46 | 1 | 109 | 4 |
| MSK, Others (Chest, Breast) | 18 | 2 | 66 | 1 |
| Total | 152 | 5 | 242 | 10 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Kim, J.W.; Lee, C.H.; Ko, G.-J.; Suh, S.-I. Safety of a Tailored Gadolinium-Based Contrast Agent Protocol Considering Excretion Pathways in Patients with Renal Impairment. Diagnostics 2026, 16, 451. https://doi.org/10.3390/diagnostics16030451
Kim JW, Lee CH, Ko G-J, Suh S-I. Safety of a Tailored Gadolinium-Based Contrast Agent Protocol Considering Excretion Pathways in Patients with Renal Impairment. Diagnostics. 2026; 16(3):451. https://doi.org/10.3390/diagnostics16030451
Chicago/Turabian StyleKim, Jeong Woo, Chang Hee Lee, Gang-Jee Ko, and Sang-Il Suh. 2026. "Safety of a Tailored Gadolinium-Based Contrast Agent Protocol Considering Excretion Pathways in Patients with Renal Impairment" Diagnostics 16, no. 3: 451. https://doi.org/10.3390/diagnostics16030451
APA StyleKim, J. W., Lee, C. H., Ko, G.-J., & Suh, S.-I. (2026). Safety of a Tailored Gadolinium-Based Contrast Agent Protocol Considering Excretion Pathways in Patients with Renal Impairment. Diagnostics, 16(3), 451. https://doi.org/10.3390/diagnostics16030451






