A Screening Method for Determining Left Ventricular Systolic Function Based on Spectral Analysis of a Single-Channel Electrocardiogram Using Machine Learning Algorithms
Abstract
1. Introduction
2. Materials and Methods
Statistical Analysis
| Train Set | Validation Set | Test Set | |
|---|---|---|---|
| Scikit-learn models and ensemble | 436 (22 targets (5%)) | - | 188 (6 targets (3.2%)) |
| Neural network (PyTorch) | 305 (16 targets (5%)) | 131 (6 targets (4%)) | 188 (6 targets (3.2%)) |
3. Results
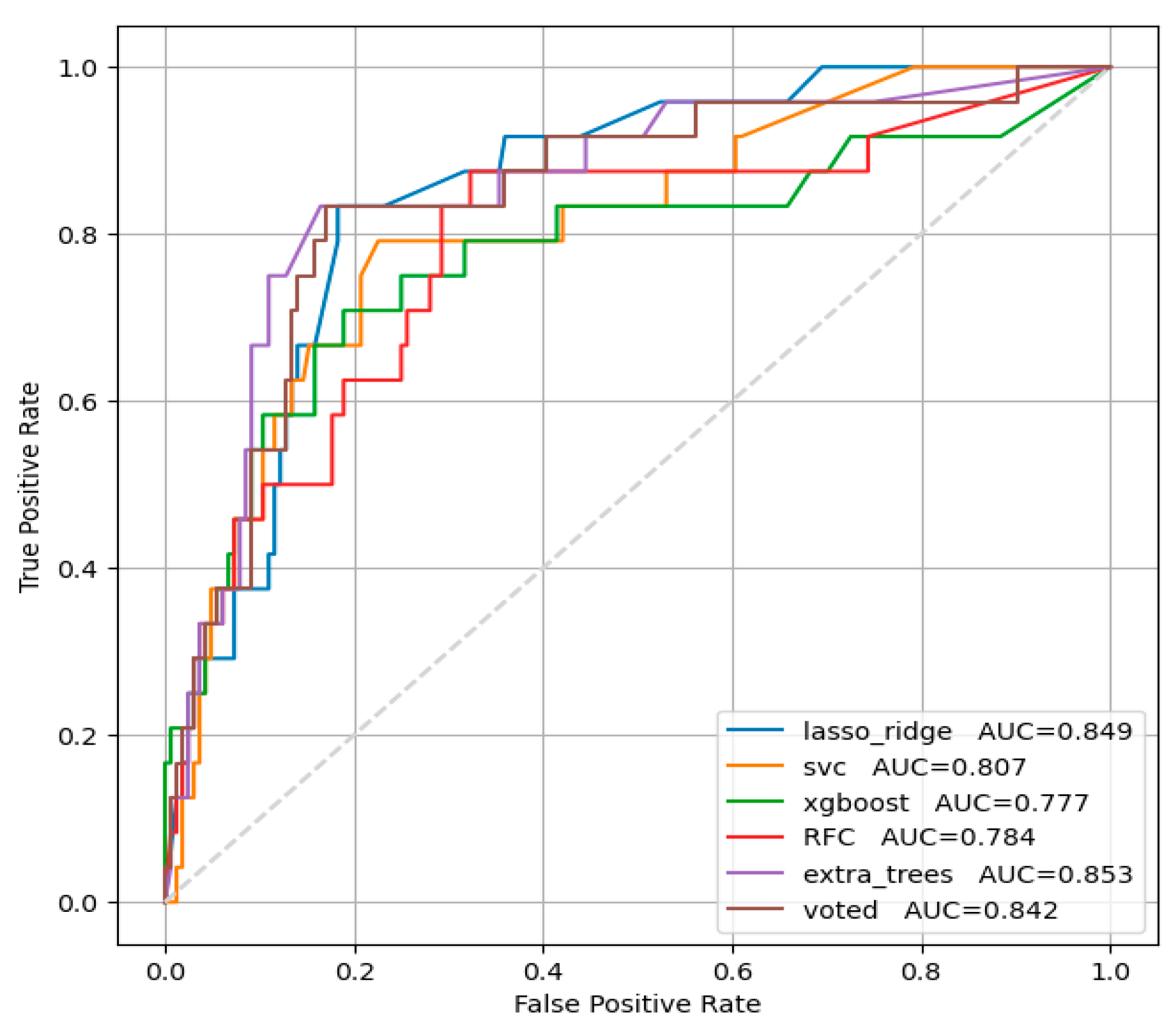
3.1. Left Ventricular Ejection Fraction Below Normal
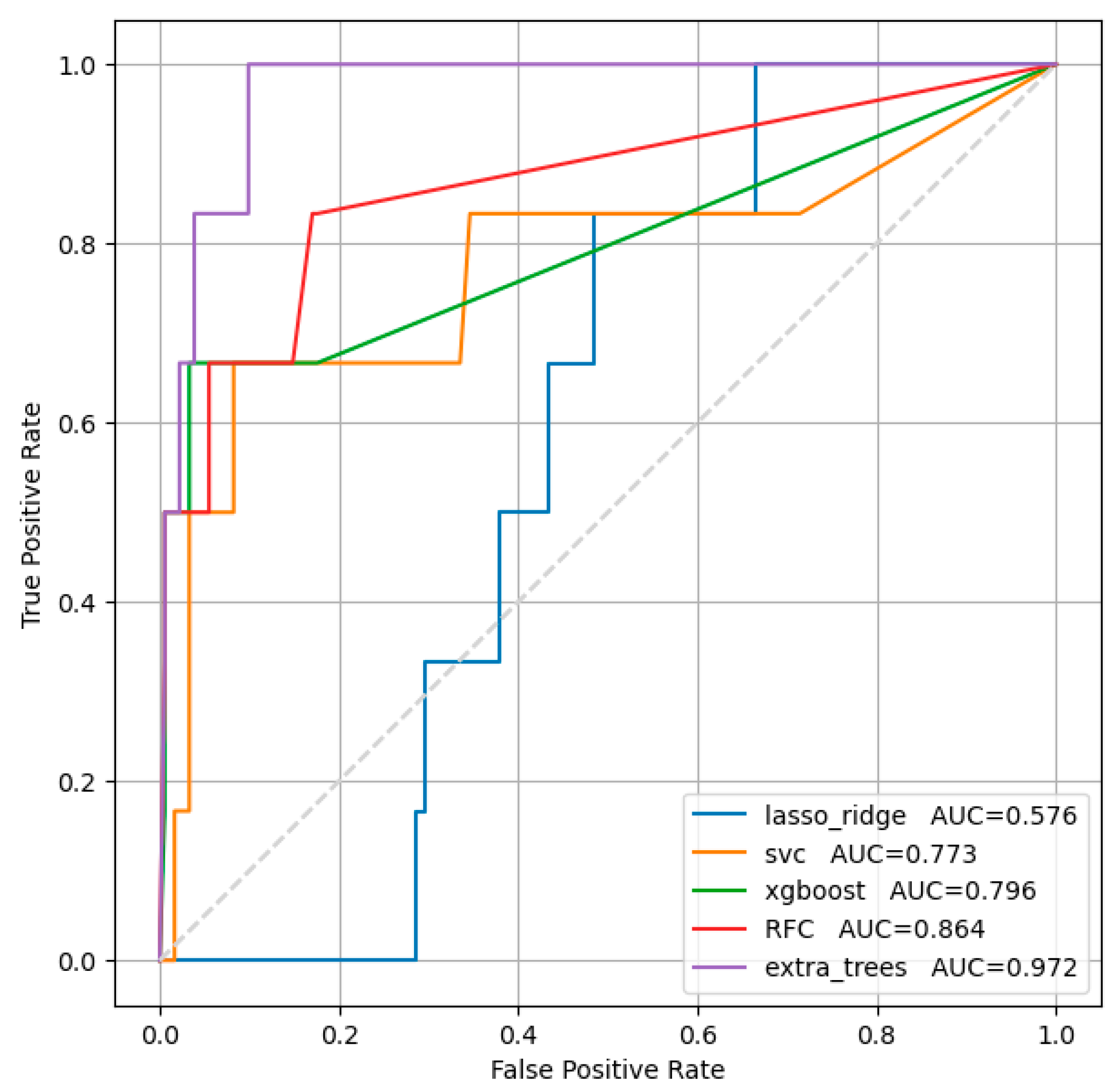
3.2. Left Ventricular Ejection Fraction Below 40%
4. Discussion
The Limits of Our Research
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| AI | Artificial intelligence |
| DD | Diastolic dysfunction |
| ECHO | Echocardiography |
| EF | Ejection fraction |
| ECG | Electrocardiogram |
| HF | Heart failure |
| HFmrEF | Heart failure with mildly reduced ejection fraction |
| HFpEF | Heart failure with preserved ejection fraction |
| HFrEF | Heart failure with reduced ejection fraction |
| LV | Left ventricular |
| LVEF | Left ventricular ejection fraction |
| LVSD | Left ventricular systolic dysfunction |
| LVSF | Left ventricular systolic function |
| NYHA | New York Heart Association |
| SVC | Support vector machines classifier |
| TTE | Transthoracic echocardiography |
| VTI | Velocity time integral |
References
- Lang, R.M.; Badano, L.P.; Mor-Avi, V.; Afilalo, J.; Armstrong, A.; Ernande, L.; Flachskampf, F.A.; Foster, E.; Goldstein, S.A.; Kuznetsova, T.; et al. Recommendations for Cardiac Chamber Quantification by Echocardiography in Adults: An Update from the American Society of Echocardiography and the European Association of Cardiovascular Imaging. J. Am. Soc. Echocardiogr. Uary 2015, 28, 1–39.e14. [Google Scholar] [CrossRef]
- Mosterd, A.; Hoes, A.W. Clinical epidemiology of heart failure. Heart 2007, 93, 1137–1146. [Google Scholar] [CrossRef]
- Polyakov, D.S.; Fomin, I.V.; Belenkov, Y.N.; Mareev, V.Y.; Ageev, F.T.; Artemjeva, E.G.; Badin, Y.V.; Bakulina, E.V.; Vinogradova, N.G.; Galyavich, A.S.; et al. Chronic heart failure in the Russian Federation: What has changed over 20 years of observation? Results of the EPOCH–CHF study. Cardiology 2021, 61, 4–14. [Google Scholar] [CrossRef]
- McDonagh, T.A.; Metra, M.; Adamo, M.; Gardner, R.S.; Baumbach, A.; Böhm, M.; Burri, H.; Butler, J.; Čelutkienė, J.; Chioncel, O.; et al. 2021 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure: Developed by the Task Force for the diagnosis and treatment of acute and chronic heart failure of the European Society of Cardiology (ESC) With the special contribution of the Heart Failure Association (HFA) of the ESC. Eur. Heart J. 2021, 42, 3599–3726. [Google Scholar] [CrossRef] [PubMed]
- Savarese, G.; Stolfo, D.; Sinagra, G.; Lund, L.H. Heart failure with mid-range or mildly reduced ejection fraction. Nat. Rev. Cardiol. 2022, 19, 100–116. [Google Scholar] [CrossRef] [PubMed]
- Kuznetsova, N.; Sagirova, Z.; Suvorov, A.; Dhif, I.; Gognieva, D.; Afina, B.; Poltavskaya, M.; Sedov, V.; Chomakhidze, P.; Kopylov, P. A screening method for predicting left ventricular dysfunction based on spectral analysis of a single-channel electrocardiogram using machine learning algorithms. Biomed. Signal Process. Control. 2023, 86 Pt B, 105219. [Google Scholar] [CrossRef]
- Kuznetsova, N.; Gubina, A.; Sagirova, Z.; Dhif, I.; Gognieva, D.; Melnichuk, A.; Orlov, O.; Syrkina, E.; Sedov, V.; Chomakhidze, P.; et al. Left Ventricular Diastolic Dysfunction Screening by a Smartphone-Case Based on Single Lead ECG. Clin. Med. Insights Cardiol. 2022, 16, 11795468221120088. [Google Scholar] [CrossRef]
- Van Rossum, G.; Drake, F.L., Jr. Python Reference Manual; Centrum voor Wiskunde en Informatica: Amsterdam, The Netherlands, 1995. [Google Scholar]
- Paszke, A.; Gross, S.; Massa, F.; Lerer, A. PyTorch: An imperative style, high-performance deep learning library. In Proceedings of the 33rd International Conference on Neural Information Processing Systems NIPS’19, Vancouver, BC, Canada, 8–14 December 2019; Curran Associates Inc.: Red Hook, NY, USA, 2019. [Google Scholar] [CrossRef]
- Pedregosa, F.; Varoquaux, G.; Gramfort, A.; Michel, V.; Thirion, B.; Grisel, O.; Blondel, M.; Prettenhofer, P.; Weiss, R.; Dubourg, V.; et al. Scikit-learn: Machine Learning in Python. J. Mach. Learn. Res. 2011, 12, 2825–2830. [Google Scholar]
- Collins, G.; Reitsma, J.; Altman, D.; Moons, K. Transparent Reporting of a multivariable prediction model for Individual Prognosis or Diagnosis (TRIPOD): The TRIPOD Statement. Eur. J. Clin. Investig. 2015, 32, 204–214. [Google Scholar] [CrossRef]
- Bjerkén, L.V.; Rønborg, S.N.; Jensen, M.T.; Ørting, S.N.; Nielsen, O.W. Artificial intelligence enabled ECG screening for left ventricular systolic dysfunction: A systematic review. Heart Fail Rev. 2023, 28, 419–430. [Google Scholar] [CrossRef]
- Choi, J.; Lee, S.; Chang, M.; Lee, Y.; Oh, G.C.; Lee, H.Y. Deep learning of ECG waveforms for diagnosis of heart failure with a reduced left ventricular ejection fraction. Sci. Rep. 2022, 12, 14235, Correction in Sci. Rep. 2022, 12, 17191. https://doi.org/10.1038/s41598-022-22012-7. [Google Scholar] [CrossRef]
- Jentzer, J.C.; Kashou, A.H.; Attia, Z.I.; Lopez-Jimenez, F.; Kapa, S.; Friedman, P.A.; Noseworthy, P.A. Left ventricular systolic dysfunction identification using artificial intelligence-augmented electrocardiogram in cardiac intensive care unit patients. Int. J. Cardiol. 2021, 326, 114–123. [Google Scholar] [CrossRef]
- Kashou, A.H.; Medina-Inojosa, J.R.; Noseworthy, P.A.; Rodeheffer, R.J.; Lopez-Jimenez, F.; Attia, I.Z.; Kapa, S.; Scott, C.G.; Lee, A.T.; Friedman, P.A.; et al. Artificial Intelligence-Augmented Electrocardiogram Detection of Left Ventricular Systolic Dysfunction in the General Population. Mayo. Clin. Proc. 2021, 96, 2576–2586. [Google Scholar] [CrossRef] [PubMed]
- Katsushika, S.; Kodera, S.; Nakamoto, M.; Ninomiya, K.; Inoue, S.; Sawano, S.; Kakuda, N.; Takiguchi, H.; Shinohara, H.; Matsuoka, R.; et al. The Effectiveness of a Deep Learning Model to Detect Left Ventricular Systolic Dysfunction from Electrocardiograms. Int. Heart J. 2021, 62, 1332–1341. [Google Scholar] [CrossRef]
- Sangha, V.; Nargesi, A.A.; Dhingra, L.S.; Khunte, A.; Mortazavi, B.J.; Ribeiro, A.H.; Banina, E.; Adeola, O.; Garg, N.; Brandt, C.A.; et al. Detection of Left Ventricular Systolic Dysfunction from Electrocardiographic Images. Circulation 2023, 148, 765–777. [Google Scholar] [CrossRef]
- Vaid, A.; Johnson, K.W.; Badgeley, M.A.; Somani, S.S.; Bicak, M.; Landi, I.; Russak, A.; Zhao, S.; Levin, M.A.; Freeman, R.S.; et al. Using Deep-Learning Algorithms to Simultaneously Identify Right and Left Ventricular Dysfunction from the Electrocardiogram. JACC Cardiovasc. Imaging. 2022, 15, 395–410. [Google Scholar] [CrossRef] [PubMed]
- Adedinsewo, D.; Carter, R.E.; Attia, Z.; Johnson, P.; Kashou, A.H.; Dugan, J.L.; Albus, M.; Sheele, J.M.; Bellolio, F.; Friedman, P.A.; et al. Artificial Intelligence-Enabled ECG Algorithm to Identify Patients with Left Ventricular Systolic Dysfunction Presenting to the Emergency Department with Dyspnea. Circ. Arrhythm. Electrophysiol. 2020, 13, e008437. [Google Scholar] [CrossRef] [PubMed]
- Anjewierden, S.; O’Sullivan, D.; Mangold, K.E.; Greason, G.; Attia, I.Z.; Lopez-Jimenez, F.; Friedman, P.A.; Asirvatham, S.J.; Anderson, J.; Eidem, B.W.; et al. Detection of Right and Left Ventricular Dysfunction in Pediatric Patients Using Artificial Intelligence-Enabled ECGs. J. Am. Heart Assoc. 2024, 13, e035201. [Google Scholar] [CrossRef]
- Attia, Z.I.; Kapa, S.; Yao, X.; Lopez-Jimenez, F.; Mohan, T.L.; Pellikka, P.A.; Carter, R.E.; Shah, N.D.; Friedman, P.A.; Noseworthy, P.A. Prospective validation of a deep learning electrocardiogram algorithm for the detection of left ventricular systolic dysfunction. J. Cardiovasc. Electrophysiol. 2019, 30, 668–674. [Google Scholar] [CrossRef]
- Attia, Z.I.; Tseng, S.A.; Benavente, D.E.; Medina-Inojosa, J.R.; Clark, T.G.; Malyutina, S.; Kapa, S.; Schirmer, H.; Kudryavtsev, A.V.; Noseworthy, P.A.; et al. External validation of a deep learning electrocardiogram algorithm to detect ventricular dysfunction. Int. J. Cardiol. 2021, 329, 130–135. [Google Scholar] [CrossRef]
- Chiou, Y.A.; Syu, J.Y.; Wu, S.Y.; Lin, L.Y.; Yi, L.T.; Lin, T.T.; Lin, S.-F. Electrocardiogram lead selection for intelligent screening of patients with systolic heart failure. Sci. Rep. 2021, 11, 1948. [Google Scholar] [CrossRef] [PubMed]

| Characteristics | Results, n (%) |
|---|---|
| Age | 58.0 ± 16.0 |
| Men | 261 (41.8%) |
| Smoking for ≥5 years prior to inclusion | 104 (16.7%) |
| Diabetes mellitus | 82 (13.1%) |
| Hypertension | 371 (59.5%) |
| Stage 1 | 64 (10.3%) |
| Stage 2 | 167 (26.8%) |
| Stage 3 | 140 (22.4%) |
| Ischemic heart disease | 151 (24.2%) |
| Atrial fibrillation at the time of ECG registration | 32 (5.1%) |
| Heart Failure (all classes) | 143 (22.9%) |
| NYHA functional class I | 7 (1.1%) |
| NYHA functional class II | 98 (15.7%) |
| NYHA functional class III | 36 (5.8%) |
| NYHA functional class IV | 2 (0.3%) |
| Aortic insufficiency | |
| Grade 1 | 116 (18.6%) |
| Grade 2 | 13 (2.1%) |
| Aortic stenosis | |
| Grade 1 | 13 (2.1%) |
| Grade 2 | 6 (1.0%) |
| Grade 3 | 2 (0.3%) |
| Mitral insufficiency | |
| Grade 1 | 321 (51.4%) |
| Grade 2 | 45 (7.2%) |
| Grade 3 | 7 (1.1%) |
| Mitral stenosis | |
| Grade 1 | 8 (1.3%) |
| Grade 2 | 1 (0.2%) |
| Left ventricular end diastolic volume | 94.6 ± 37.9 |
| LV ejection fraction <52% for men and LV ejection fraction <54% for women | 126 (20.2%) |
| LV ejection fraction <40% | 68 (10.1%) |
| VTI in LV output tract | 19.5 ± 4.7 |
| LV DD 1–3 grade | 215 (34.5%) |
| LV DD 2–3 grade | 72 (11.5%) |
| Parameters | Description | Results in the General Group |
|---|---|---|
| SBeta | ratio of the maximum modulus of the derivative value at the leading front of the T-wave to the maximum modulus of the value at the trailing front of the T-wave | 0.9 ± 0.6 |
| QRSfi | marker of the end of the QRS complex | 422.1 ± 135.7 |
| Tfi | T-wave end marker | 72.0 ± 241.0 |
| Tpeak | T-wave peak position | 394.7 ± 117.7 |
| Tons | point of maximum slope of the T-wave fore front | 347.6 ± 114.6 |
| Toffs | point of maximum slope of the T-wave back front | 435.6 ± 123.0 |
| ECG Parameter | AUC | Sensitivity | Specificity |
|---|---|---|---|
| Left ventricle ejection fraction < 52 for female and < 54% for male | |||
| TA | 0.822 | 80% | 69% |
| J80A | 0.713 | 77% | 66% |
| RonsF | 0.743 | 81% | 77% |
| RoffsF | 0.729 | 79% | 78% |
| Left ventricle ejection fraction < 41% | |||
| TA | 0.915 | 85% | 83% |
| J80A | 0.717 | 76% | 73% |
| RonsF | 0.844 | 82% | 82% |
| RoffsF | 0.825 | 81% | 79% |
| Train Set | Validation Set | Test Set | |
|---|---|---|---|
| Scikit-learn models and ensemble | 436 (56 targets (12.8%)) | - | 188 (24 targets (12.8%)) |
| Neural network (PyTorch) | 305 (41 targets (13%)) | 131 (15 targets (11%)) | 188 (24 targets (12.8%)) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Kuznetsova, N.; Suvorov, A.; Gognieva, D.; Fashafsha, Z.; Podgalo, D.; Mesitskaya, D.; Shchekochikhin, D.; Sedov, V.; Chomakhidze, P.; Kopylov, P. A Screening Method for Determining Left Ventricular Systolic Function Based on Spectral Analysis of a Single-Channel Electrocardiogram Using Machine Learning Algorithms. Diagnostics 2026, 16, 262. https://doi.org/10.3390/diagnostics16020262
Kuznetsova N, Suvorov A, Gognieva D, Fashafsha Z, Podgalo D, Mesitskaya D, Shchekochikhin D, Sedov V, Chomakhidze P, Kopylov P. A Screening Method for Determining Left Ventricular Systolic Function Based on Spectral Analysis of a Single-Channel Electrocardiogram Using Machine Learning Algorithms. Diagnostics. 2026; 16(2):262. https://doi.org/10.3390/diagnostics16020262
Chicago/Turabian StyleKuznetsova, Natalia, Aleksandr Suvorov, Daria Gognieva, Zaki Fashafsha, Dmitrii Podgalo, Dinara Mesitskaya, Dmitry Shchekochikhin, Vsevolod Sedov, Petr Chomakhidze, and Philippe Kopylov. 2026. "A Screening Method for Determining Left Ventricular Systolic Function Based on Spectral Analysis of a Single-Channel Electrocardiogram Using Machine Learning Algorithms" Diagnostics 16, no. 2: 262. https://doi.org/10.3390/diagnostics16020262
APA StyleKuznetsova, N., Suvorov, A., Gognieva, D., Fashafsha, Z., Podgalo, D., Mesitskaya, D., Shchekochikhin, D., Sedov, V., Chomakhidze, P., & Kopylov, P. (2026). A Screening Method for Determining Left Ventricular Systolic Function Based on Spectral Analysis of a Single-Channel Electrocardiogram Using Machine Learning Algorithms. Diagnostics, 16(2), 262. https://doi.org/10.3390/diagnostics16020262






