Integrating Clinical Factors and Parity-Specific Models with Molecular Biomarkers to Better Predict the Risk of Preterm Birth in Asymptomatic Women
Abstract
1. Introduction
2. Materials and Methods
2.1. Cohorts and Study Design
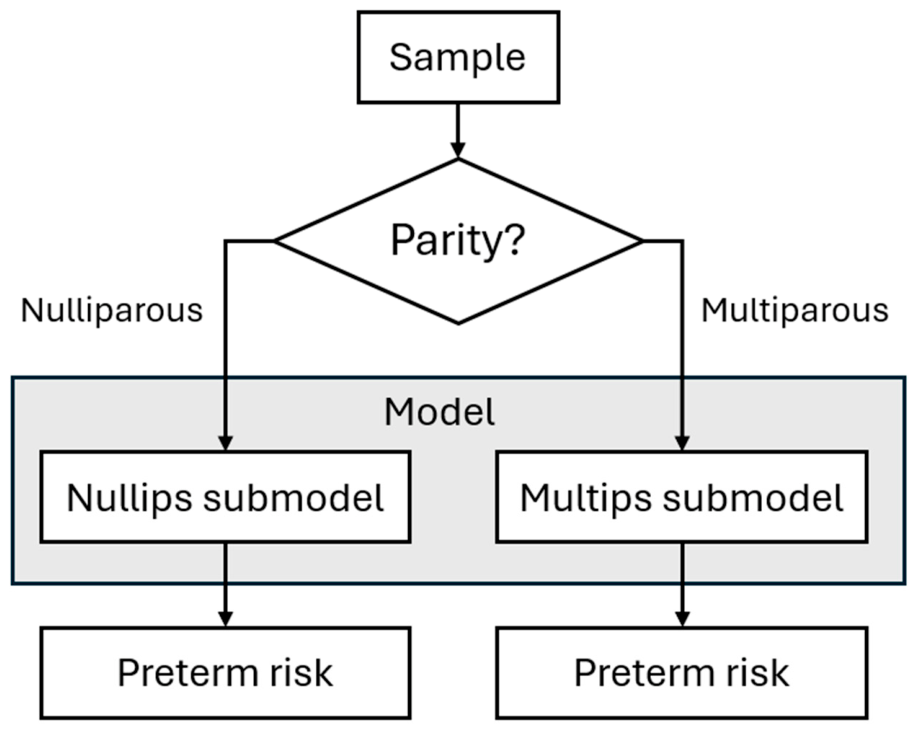
2.2. Model Development
2.3. Model Validation
2.4. Clinical Utility
2.5. Statistical Analyses
3. Results
3.1. Study Participant Demographics
3.2. Performance of Parity-Specific Submodels
3.3. Performance of the Model
3.4. Assessment of Clinical Utility
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| AUC | Area Under the Receiver Operating Characteristic Curve |
| BMI | Body Mass Index |
| CI | Confidence Interval |
| ECI | Estimated Calibration Index |
| GABD | Gestational Age at Blood Draw |
| IGFBP4 | Insulin-like Growth Factor Binding Protein 4 |
| LOS | Length of (Hospital) Stay |
| LR | Likelihood Ratio |
| NICU | Neonatal Intensive Care Unit |
| NNLOS | Neonatal Length of (Hospital) Stay |
| NNS | Number Needed to Screen |
| NPV | Negative Predictive Value |
| PAPR | Proteomic Assessment of Preterm Birth |
| PE | Preeclampsia |
| PPV | Positive Predictive Value |
| PTB | Preterm Birth |
| SD | Standard Deviation |
| SHBG | Sex Hormone Binding Globulin |
| sPTB | Spontaneous Preterm Birth |
References
- Vogel, J.P.; Chawanpaiboon, S.; Moller, A.B.; Watananirun, K.; Bonet, M.; Lumbiganon, P. The global epidemiology of preterm birth. Best Pract. Res. Clin. Obstet. Gynaecol. 2018, 52, 3–12. [Google Scholar] [CrossRef] [PubMed]
- American College of Obstetricians and Gynecologists. Practice Bulletin No. 171: Management of Preterm Labor. Obstet. Gynecol. 2016, 128, e155–e164. [Google Scholar] [CrossRef]
- Allin, M.; Rooney, M.; Griffiths, T.; Cuddy, M.; Wyatt, J.; Rifkin, L.; Murray, R. Neurological abnormalities in young adults born preterm. J. Neurol. Neurosurg. Psychiatry 2006, 77, 495–499. [Google Scholar] [CrossRef]
- Cao, G.; Liu, J.; Liu, M. Global, Regional, and National Incidence and Mortality of Neonatal Preterm Birth, 1990–2019. JAMA Pediatr. 2022, 176, 787–796. [Google Scholar] [CrossRef]
- Fernandez de Gamarra-Oca, L.; Ojeda, N.; Ontanon, J.M.; Loureiro-Gonzalez, B.; Gomez-Gastiasoro, A.; Pena, J.; Ibarretxe-Bilbao, N.; Garcia-Guerrero, M.A.; Zubiaurre-Elorza, L. Long-term brain structural and cognitive outcomes in a low-risk preterm-born sample. Sci. Rep. 2024, 14, 21110. [Google Scholar] [CrossRef]
- Hassan, S.S.; Romero, R.; Vidyadhari, D.; Fusey, S.; Baxter, J.K.; Khandelwal, M.; Vijayaraghavan, J.; Trivedi, Y.; Soma-Pillay, P.; Sambarey, P.; et al. Vaginal progesterone reduces the rate of preterm birth in women with a sonographic short cervix: A multicenter, randomized, double-blind, placebo-controlled trial. Ultrasound Obstet. Gynecol. 2011, 38, 18–31. [Google Scholar] [CrossRef]
- Fonseca, E.B.; Celik, E.; Parra, M.; Singh, M.; Nicolaides, K.H. Fetal Medicine Foundation Second Trimester Screening Group. Progesterone and the risk of preterm birth among women with a short cervix. N. Engl. J. Med. 2007, 357, 462–469. [Google Scholar] [CrossRef] [PubMed]
- Hoffman, M.K.; Goudar, S.S.; Kodkany, B.S.; Metgud, M.; Somannavar, M.; Okitawutshu, J.; Lokangaka, A.; Tshefu, A.; Bose, C.L.; Mwapule, A.; et al. Low-dose aspirin for the prevention of preterm delivery in nulliparous women with a singleton pregnancy (ASPIRIN): A randomised, double-blind, placebo-controlled trial. Lancet 2020, 395, 285–293. [Google Scholar] [CrossRef] [PubMed]
- Garite, T.J.; Manuck, T.A. Should case management be considered a component of obstetrical interventions for pregnancies at risk of preterm birth? Am. J. Obstet. Gynecol. 2023, 228, 430–437. [Google Scholar] [CrossRef]
- Romero, R.; Conde-Agudelo, A.; Da Fonseca, E.; O’Brien, J.M.; Cetingoz, E.; Creasy, G.W.; Hassan, S.S.; Nicolaides, K.H. Vaginal progesterone for preventing preterm birth and adverse perinatal outcomes in singleton gestations with a short cervix: A meta-analysis of individual patient data. Am. J. Obstet. Gynecol. 2018, 218, 161–180. [Google Scholar] [CrossRef]
- Cobo, T.; Kacerovsky, M.; Jacobsson, B. Risk factors for spontaneous preterm delivery. Int. J. Gynaecol. Obstet. 2020, 150, 17–23. [Google Scholar] [CrossRef]
- Gulersen, M.; Divon, M.Y.; Krantz, D.; Chervenak, F.A.; Bornstein, E. The risk of spontaneous preterm birth in asymptomatic women with a short cervix (≤25 mm) at 23–28 weeks’ gestation. Am. J. Obstet. Gynecol. MFM 2020, 2, 100059. [Google Scholar] [CrossRef]
- Petrini, J.R.; Callaghan, W.M.; Klebanoff, M.; Green, N.S.; Lackritz, E.M.; Howse, J.L.; Schwarz, R.H.; Damus, K. Estimated effect of 17 alpha-hydroxyprogesterone caproate on preterm birth in the United States. Obstet. Gynecol. 2005, 105, 267–272. [Google Scholar] [CrossRef] [PubMed]
- Esplin, M.S.; Elovitz, M.A.; Iams, J.D.; Parker, C.B.; Wapner, R.J.; Grobman, W.A.; Simhan, H.N.; Wing, D.A.; Haas, D.M.; Silver, R.M.; et al. Predictive Accuracy of Serial Transvaginal Cervical Lengths and Quantitative Vaginal Fetal Fibronectin Levels for Spontaneous Preterm Birth Among Nulliparous Women. JAMA 2017, 317, 1047–1056. [Google Scholar] [CrossRef] [PubMed]
- Berghella, V.; Saccone, G. Cervical assessment by ultrasound for preventing preterm delivery. Cochrane Database Syst. Rev. 2019, 9, CD007235. [Google Scholar] [CrossRef]
- Esplin, M.S.; O’Brien, E.; Fraser, A.; Kerber, R.A.; Clark, E.; Simonsen, S.E.; Holmgren, C.; Mineau, G.P.; Varner, M.W. Estimating recurrence of spontaneous preterm delivery. Obstet. Gynecol. 2008, 112, 516–523. [Google Scholar] [CrossRef]
- Biggio, J.; Society for Maternal-Fetal Medicine (SMFM); SMFM Publications Committee. SMFM Consult Series #70: Management of short cervix in individuals without a history of spontaneous preterm birth. Am. J. Obstet. Gynecol. 2024, 231, B2–B13. [Google Scholar] [CrossRef] [PubMed]
- Martin, J.A.; Hamilton, B.E.; Sutton, P.D.; Ventura, S.J.; Menacker, F.; Munson, M.L. Births: Final data for 2002. Natl. Vital. Stat. Rep. 2003, 52, 10. [Google Scholar]
- Saade, G.R.; Boggess, K.A.; Sullivan, S.A.; Markenson, G.R.; Iams, J.D.; Coonrod, D.V.; Pereira, L.M.; Esplin, M.S.; Cousins, L.M.; Lam, G.K.; et al. Development and validation of a spontaneous preterm delivery predictor in asymptomatic women. Am. J. Obstet. Gynecol. 2016, 214, 633.e1–633.e24. [Google Scholar] [CrossRef]
- Markenson, G.R.; Saade, G.R.; Laurent, L.C.; Heyborne, K.D.; Coonrod, D.V.; Schoen, C.N.; Baxter, J.K.; Haas, D.M.; Longo, S.; Grobman, W.A.; et al. Performance of a proteomic preterm delivery predictor in a large independent prospective cohort. Am. J. Obstet. Gynecol. MFM 2020, 2, 100140. [Google Scholar] [CrossRef]
- Forbes, K.; Westwood, M. Maternal growth factor regulation of human placental development and fetal growth. J. Endocrinol. 2010, 207, 1–16. [Google Scholar] [CrossRef]
- Qiu, Q.; Bell, M.; Lu, X.; Yan, X.; Rodger, M.; Walker, M.; Wen, S.W.; Bainbridge, S.; Wang, H.; Gruslin, A. Significance of IGFBP-4 in the development of fetal growth restriction. J. Clin. Endocrinol. Metab. 2012, 97, E1429–E1439. [Google Scholar] [CrossRef]
- Nawathe, A.R.; Christian, M.; Kim, S.H.; Johnson, M.; Savvidou, M.D.; Terzidou, V. Insulin-like growth factor axis in pregnancies affected by fetal growth disorders. Clin. Epigenet. 2016, 8, 11. [Google Scholar] [CrossRef]
- Gladych-Macioszek, A.; Ozegowska, K.; Radzicka-Mularczyk, S.; Sibiak, R.; Desoye, G.; Wender-Ozegowska, E. Insulin-Like Growth Factors, Binding Proteins and Their Role in Pregnancy in Patients With Diabetes. J. Diabetes Res. 2025, 2025, 3330482. [Google Scholar] [CrossRef] [PubMed]
- Preston, M.; Hall, M.; Shennan, A.; Story, L. The role of placental insufficiency in spontaneous preterm birth: A literature review. Eur. J. Obstet. Gynecol. Reprod. Biol. 2024, 295, 136–142. [Google Scholar] [CrossRef]
- Habelrih, T.; Augustin, T.L.; Mauffette-Whyte, F.; Ferri, B.; Sawaya, K.; Cote, F.; Gallant, M.; Olson, D.M.; Chemtob, S. Inflammatory mechanisms of preterm labor and emerging anti-inflammatory interventions. Cytokine Growth Factor Rev. 2024, 78, 50–63. [Google Scholar] [CrossRef] [PubMed]
- Cappelletti, M.; Della Bella, S.; Ferrazzi, E.; Mavilio, D.; Divanovic, S. Inflammation and preterm birth. J. Leukoc. Biol. 2016, 99, 67–78. [Google Scholar] [CrossRef]
- American College of Obstetricians and Gynecologists. ACOG Practice Bulletin No. 234. Obstet. Gynecol. 2021, 138, e65–e90. [Google Scholar] [CrossRef]
- Hoffman, M.K.; Kitto, C.; Zhang, Z.; Shi, J.; Walker, M.G.; Shahbaba, B.; Ruhstaller, K. Neonatal Outcomes after Maternal Biomarker-Guided Preterm Birth Intervention: The AVERT PRETERM Trial. Diagnostics 2024, 14, 1462. [Google Scholar] [CrossRef]
- Iriye, B.K.; O’Brien, J.M.; Ennen, C.S.; Barrilleaux, P.S.; Berkin, J.A.; Palatnik, A.; Son, M.; Gyamfi-Bannerman, C.; McDonnold, M.; Markenson, G.R.; et al. Neonatal impact of maternal biomarker screening for risk of preterm birth with targeted interventions (PRIME): A multicenter, randomized, controlled trial. Pregnancy 2026, 2, e70202. [Google Scholar] [CrossRef]
- Grabner, M.; Burchard, J.; Nguyen, C.; Chung, H.; Gangan, N.; Boniface, J.J.; Zupancic, J.A.F.; Stanek, E. Cost-Effectiveness of a Proteomic Test for Preterm Birth Prediction. Clin. Outcomes Res. 2021, 13, 809–820. [Google Scholar] [CrossRef]
- American College of Obstetricians and Gynecologists. Updated Clinical Guidance for the Use of Progestogen Supplementation for the Prevention of Recurrent Preterm Birth. 2025. Available online: https://www.acog.org/clinical/clinical-guidance/practice-advisory/articles/2023/04/updated-guidance-use-of-progestogen-supplementation-for-prevention-of-recurrent-preterm-birth (accessed on 20 March 2026).
- Society for Maternal-Fetal Medicine (SMFM); SMFM Publications Committee. Society for Maternal-Fetal Medicine Statement: Response to the Food and Drug Administration’s withdrawal of 17-alpha hydroxyprogesterone caproate. Am. J. Obstet. Gynecol. 2023, 229, B2–B6. [Google Scholar] [CrossRef]
- Ballering, G.; Leijnse, J.; Eijkelkamp, N.; Peeters, L.; de Heus, R. First-trimester placental vascular development in multiparous women differs from that in nulliparous women. J. Matern. Fetal Neonatal Med. 2018, 31, 209–215. [Google Scholar] [CrossRef]
- Haizler-Cohen, L.; Wang, G.; Habtewold, T.D.; Wijesiriwardhana, P.; Grantz, K.L.; Tekola-Ayele, F. Placental gene expression signatures based on maternal parity. Placenta 2026, 174, 214–224. [Google Scholar] [CrossRef]
- Ding, W.; Lau, S.L.; Wang, C.C.; Zhang, T.; Getsko, O.; Lee, N.M.W.; Chim, S.S.C.; Wong, C.K.; Leung, T.Y. Dynamic changes in maternal immune biomarkers during labor in nulliparous vs multiparous women. Am. J. Obstet. Gynecol. 2022, 227, 627.e1–627.e23. [Google Scholar] [CrossRef] [PubMed]
- Wolf, M.; Shah, A.; Lam, C.; Martinez, A.; Smirnakis, K.V.; Epstein, F.H.; Taylor, R.N.; Ecker, J.L.; Karumanchi, S.A.; Thadhani, R. Circulating levels of the antiangiogenic marker sFLT-1 are increased in first versus second pregnancies. Am. J. Obstet. Gynecol. 2005, 193, 16–22. [Google Scholar] [CrossRef]
- Quezada, M.S.; Rodriguez-Calvo, J.; Villalain, C.; Gomez-Arriaga, P.I.; Galindo, A.; Herraiz, I. sFlt-1/PlGF ratio and timing of delivery in early-onset fetal growth restriction with antegrade umbilical artery flow. Ultrasound Obstet. Gynecol. 2020, 56, 549–556. [Google Scholar] [CrossRef]
- Wilimitis, D.; Walsh, C.G. Practical Considerations and Applied Examples of Cross-Validation for Model Development and Evaluation in Health Care: Tutorial. JMIR AI 2023, 2, e49023. [Google Scholar] [CrossRef] [PubMed]
- Steyerberg, E.W. Clinical Prediction Models: A Practical Approach to Development, Validation, and Updating; Springer: New York, NY, USA, 2009. [Google Scholar]
- Hastie, T.; Tibshirani, R.; Friedman, J. The Elembents of Statistical Learning; Springer: New York, NY, USA, 2009. [Google Scholar]
- Obuchowski, N.A.; McClish, D.K. Sample size determination for diagnostic accuracy studies involving binormal ROC curve indices. Stat. Med. 1997, 16, 1529–1542. [Google Scholar] [CrossRef]
- Van Calster, B.; Nieboer, D.; Vergouwe, Y.; De Cock, B.; Pencina, M.J.; Steyerberg, E.W. A calibration hierarchy for risk models was defined: From utopia to empirical data. J. Clin. Epidemiol. 2016, 74, 167–176. [Google Scholar] [CrossRef]
- R Foundation for Statistical Computing. R: A Language and Environment for Statistical Computing. 2021. Available online: https://www.R-project.org/ (accessed on 3 February 2026).
- Burchard, J.; Polpitiya, A.D.; Fox, A.C.; Randolph, T.L.; Fleischer, T.C.; Dufford, M.T.; Garite, T.J.; Critchfield, G.C.; Boniface, J.J.; Saade, G.R.; et al. Clinical Validation of a Proteomic Biomarker Threshold for Increased Risk of Spontaneous Preterm Birth and Associated Clinical Outcomes: A Replication Study. J. Clin. Med. 2021, 10, 5088. [Google Scholar] [CrossRef] [PubMed]
- ACOG Practice Bulletin. Clinical management guidelines for obstetrician-gynecologists. No. 82 June 2007. Management of herpes in pregnancy. Obstet. Gynecol. 2007, 109, 1489–1498. [Google Scholar] [CrossRef]
- Mercer, B.M.; Goldenberg, R.L.; Das, A.; Moawad, A.H.; Iams, J.D.; Meis, P.J.; Copper, R.L.; Johnson, F.; Thom, E.; McNellis, D.; et al. The preterm prediction study: A clinical risk assessment system. Am. J. Obstet. Gynecol. 1996, 174, 1885–1893; discussion 1893–1895. [Google Scholar] [CrossRef]


| Chronic Diabetes | BMI ≥ 30 | Maternal Age ≥ 35 Years | Prior sPTB | Prior PE | Chronic Hypertension | |
|---|---|---|---|---|---|---|
| Nulliparous | ✓ | |||||
| Multiparous | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ |
| Clinical Variable | Nulliparous | Multiparous | Combined |
|---|---|---|---|
| NTotal | 356 | 620 | 976 |
| N(%) sPTB < 37 Outcome | 27 (7.6%) | 57 (9.2%) | 84 (8.6%) |
| N(%) PTB < 37 Outcome | 49 (13.8%) | 93 (15%) | 142 (14.5%) |
| N(%) sPTB < 37 & NNLOS ≥ 5 days | 13 (3.7%) | 23 (3.7%) | 36 (3.7%) |
| N(%) PTB < 37 & NNLOS ≥ 5 days | 29 (8.1%) | 36 (5.8%) | 65 (6.7%) |
| N(%) Chronic Diabetes | 24 (6.7%) | 31 (5.0%) | 55 (5.6%) |
| N(%) Chronic Hypertension | 17 (4.8%) | 44 (7.1%) | 61 (6.3%) |
| N(%) Prior PE | N/A | 60 (9.7%) | 60 (6.1%) |
| N(%) Prior PTB | N/A | 165 (26.6%) | 165 (16.9%) |
| NNLOS (mean, median, SD, min, max) | 4.07, 3, 6.99, 0, 99 | 4.06, 3, 7.93, 0, 88 | 4.06, 3, 7.6, 0, 99 |
| N(%) NNLOS ≥ 5 days | 53 (14.9%) | 72 (11.6%) | 125 (12.8%) |
| Maternal Age (mean, median, SD, min, max) | 25.29, 24, 5.81, 18, 46 | 28.98, 28, 5.59, 18, 44 | 27.64, 27, 5.94, 18, 46 |
| N(%) Maternal Age ≥ 35 | 31 (8.7%) | 122 (19.7%) | 153 (15.7%) |
| BMI (mean, median, SD, min, max) | 27.45, 25.8, 7.23, 15.2, 58.1 | 29.26, 28.3, 7.59, 15.8, 75.6 | 28.6, 27.3, 7.51, 15.2, 75.6 |
| N(%) BMI ≥ 30 | 108 (30.3%) | 248 (40.0%) | 356 (36.5%) |
| N(%) BMI ≥ 21 | 301 (84.6%) | 554 (89.4%) | 855 (87.6%) |
| N(%) White | 257 (72.2%) | 450 (72.6%) | 707 (72.4%) |
| N(%) Black | 68 (19.1%) | 101 (16.3%) | 169 (17.3%) |
| N(%) Asian | 6 (1.7%) | 7 (1.1%) | 13 (1.3%) |
| N(%) Hispanic | 98 (27.5%) | 257 (41.5%) | 355 (36.4%) |
| N(%) Other Race | 25 (7.0%) | 62 (10.0%) | 87 (8.9%) |
| Model | Training AUC (95% CI) | Mean AUC of Cross Validation Measurements | Wilcoxon p-Value 1 | ECI | R2 |
|---|---|---|---|---|---|
| Nulliparous submodel | 0.74 (0.62–0.86) | 0.73 | <0.001 | 0.12 | 0.99 |
| Multiparous submodel | 0.78 (0.71–0.84) | 0.74 | <0.001 | 0.07 | 0.99 |
| Model | AUC | AUC 95% CI | Wilcoxon p-Value 1 | ECI | R2 |
|---|---|---|---|---|---|
| Nulliparous submodel | 0.71 | 0.60–0.82 | <0.001 | 0.11 | 0.88 |
| Multiparous submodel | 0.72 | 0.63–0.80 | <0.001 | 0.09 | 0.72 |
| Model (Combined Parity) | 0.70 | 0.64–0.76 | <0.001 | 0.10 | 0.85 |
| Subgroup | Outcome | AUC | AUC 95% CI | Wilcoxon p-Value 1 |
|---|---|---|---|---|
| Full GABD range, all BMIs | sPTB | 0.71 | 0.65–0.77 | <0.001 |
| PTB | 0.73 | 0.69–0.78 | <0.001 | |
| Restricted GABD range and BMI | sPTB | 0.77 | 0.70–0.85 | <0.001 |
| PTB | 0.79 | 0.72–0.85 | <0.001 |
| Subgroup | Outcome | Fisher’s p-Value 1 | Sensitivity (95% CI) | Specificity (95% CI) | PPV (95% CI) | NPV (95% CI) | LR+ (95% CI) | LR− (95% CI) | NNS (95% CI) |
|---|---|---|---|---|---|---|---|---|---|
| Full GABD range, all BMIs | sPTB | <0.001 | 61.9% (50.6–72.1) | 76.7% (73.7–79.4) | 20.0% (16.9–23.5) | 95.5% (94.2–96.6) | 2.65 (2.16–3.26) | 0.5 (0.38–0.65) | 19 (16–23) |
| PTB | <0.001 | 63.4% (54.8–71.2) | 76.7% (73.7–79.5) | 31.7% (28.0–35.6) | 92.5% (90.8–93.9) | 2.72 (2.29–3.25) | 0.48 (0.38–0.59) | 11 (10–13) | |
| Restricted GABD range and BMI | sPTB | <0.001 | 77.1% (59.4–89.0) | 74.4% (69.7–78.6) | 21.4% (17.6–25.9) | 97.3% (95.1–98.5) | 3.02 (2.35–3.86) | 0.31 (0.17–0.57) | 16 (14–20) |
| PTB | <0.001 | 76.8% (63.3–86.6) | 74.6% (69.7–78.9) | 31.6% (26.9–36.7) | 95.5% (92.9–97.1) | 3.02 (2.41–3.79) | 0.31 (0.19–0.5) | 10 (9–12) |
| sPTB Predictor | Sensitivity | PPV | NNS | LR+ | LR− |
|---|---|---|---|---|---|
| Model | 77.1% | 21.4% | 16 | 3.0 | 0.3 |
| PreTRM Test | 75.0% [19] | 14.6% [19] | 16 | 2.9 [28] | 0.3 [28] |
| Cervical Length Screening | 8% [6,17,28] | 16.2% [6,17,28] | 151 | 3.7 [14] | 0.9 [14] |
| Prior sPTB | 11% [13,17,18] | 22.5% [13,17,18] | 109 | 3.4 [13] * | 0.9 [13] * |
| Model | PreTRM Test | DeLong p-Value 2 | McNemar p-Value 3 | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Metric | AUC (95% CI) | Wilcoxon p-Value 1 | Sensitivity (95% CI) | Specificity (95% CI) | AUC (95% CI) | Wilcoxon p-Value 1 | Sensitivity (95% CI) | Specificity (95% CI) | ||
| LOS ≥ 5 + sPTB | 0.74 (0.66–0.82) | <0.001 | 69.4% (51.7–83.1) | 75.0% (72.1–77.7) | 0.61 (0.50–0.72) | 0.054 | 55.6 (38.3–71.7) | 67.9% (64.8–70.8) | 0.044 | 0.263 |
| LOS ≥ 5 + PTB | 0.76 (0.71–0.82) | <0.001 | 72.3% (59.6–82.3) | 74.0% (71.0–76.8) | 0.62 (0.54–0.70) | 0.002 | 52.3 (39.6–64.7) | 68.4% (65.2–71.4) | <0.001 | 0.009 |
| Study Scenario | Progressive Grade | PreTRM Evidence |
|---|---|---|
| Retrospective case–control study (PAPR) [19] | Grade C (consensus or very limited/poor quality data) | Development of PreTRM Internal, retrospective validation of PreTRM |
| Prospective blinded validation in an independent cohort (TREETOP) [45] | Grade B (consistent evidence but still limited) | External, prospective validation of PreTRM |
| Randomized clinical trial of test-guided management (PRIME) [30] | Grade A (strong recommendation based on consistent RCT and meta-analysis evidence) | PreTRM guides treatment that reduces sPTB |
| Performance Enhancement (the Model presented in the current study) | Grade A (meets performance requirements in ACOG practice Bulletin 234) | Enhanced PPV and outcome prediction |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Polpitiya, A.; Cox, C.; Butler, H.; Badsha, M.B.; Sommerville, L.J.; Boniface, J.J.; Saade, G.; Kearney, P. Integrating Clinical Factors and Parity-Specific Models with Molecular Biomarkers to Better Predict the Risk of Preterm Birth in Asymptomatic Women. Diagnostics 2026, 16, 1487. https://doi.org/10.3390/diagnostics16101487
Polpitiya A, Cox C, Butler H, Badsha MB, Sommerville LJ, Boniface JJ, Saade G, Kearney P. Integrating Clinical Factors and Parity-Specific Models with Molecular Biomarkers to Better Predict the Risk of Preterm Birth in Asymptomatic Women. Diagnostics. 2026; 16(10):1487. https://doi.org/10.3390/diagnostics16101487
Chicago/Turabian StylePolpitiya, Ashoka, Charles Cox, Heather Butler, Md. Bahadur Badsha, Laura J. Sommerville, J. Jay Boniface, George Saade, and Paul Kearney. 2026. "Integrating Clinical Factors and Parity-Specific Models with Molecular Biomarkers to Better Predict the Risk of Preterm Birth in Asymptomatic Women" Diagnostics 16, no. 10: 1487. https://doi.org/10.3390/diagnostics16101487
APA StylePolpitiya, A., Cox, C., Butler, H., Badsha, M. B., Sommerville, L. J., Boniface, J. J., Saade, G., & Kearney, P. (2026). Integrating Clinical Factors and Parity-Specific Models with Molecular Biomarkers to Better Predict the Risk of Preterm Birth in Asymptomatic Women. Diagnostics, 16(10), 1487. https://doi.org/10.3390/diagnostics16101487






