Irisin as an Associative Marker of Metabolic Dysregulation in Obesity: Comparative Profiling of IL-6, IL-15, IL-1β, and CCL2
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Population
2.2. Quantitative Measurement of IL-6, IL-15, IL-1β, CCL2 and Irisin Concentrations
2.3. Statistical Analysis
3. Results
4. Discussion
5. Limitations
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- World Health Organization. Obesity and Overweight. Available online: https://www.who.int/news-room/fact-sheets/detail/obesity-and-overweight (accessed on 24 June 2025).
- National Academies of Sciences, Engineering, and Medicine; Health and Medicine Division; Food and Nutrition Board; Roundtable on Obesity Solutions; Callahan, E.A. The Science, Strengths, and Limitations of Body Mass Index. In Translating Knowledge of Foundational Drivers of Obesity into Practice; The National Academies Press: Washington, DC, USA, 2023. [Google Scholar] [CrossRef]
- Edwards, B.A.; Powell, J.R.; McGaffey, A.; Wislo, V.M.P.; Boron, E.; D’AMico, F.J.; Hogan, L.; Hughes, K.; Jewell, I.K.; Abatemarco, D.J. Fitwits TM Leads to Improved Parental Recognition of Childhood Obesity and Plans to Encourage Change. J. Am. Board Fam. Med. 2017, 30, 178–188. [Google Scholar] [CrossRef]
- Mayoral, L.P.-C.; Andrade, G.M.; Mayoral, E.P.-C.; Huerta, T.H.; Canseco, S.P.; Canales, F.J.R.; Cabrera-Fuentes, H.A.; Cruz, M.M.; Santiago, A.D.P.; Alpuche, J.J.; et al. Obesity subtypes, related biomarkers & heterogeneity. Indian J. Med. Res. 2020, 151, 11–21. [Google Scholar] [CrossRef]
- Al-Ibraheem, A.M.T.; Hameed, A.A.Z.; Marsool, M.D.M.; Jain, H.; Prajjwal, P.; Khazmi, I.; Nazzal, R.S.; Al-Najati, H.M.H.; Al-Zuhairi, B.H.Y.K.; Razzaq, M.; et al. Exercise-Induced cytokines, diet, and inflammation and their role in adipose tissue metabolism. Health Sci. Rep. 2024, 7, e70034. [Google Scholar] [CrossRef] [PubMed]
- Zatterale, F.; Longo, M.; Naderi, J.; Raciti, G.A.; Desiderio, A.; Miele, C.; Beguinot, F. Chronic Adipose Tissue Inflammation Linking Obesity to Insulin Resistance and Type 2 Diabetes. Front. Physiol. 2020, 10, 1607. [Google Scholar] [CrossRef] [PubMed]
- Wu, H.; Ballantyne, C.M. Metabolic Inflammation and Insulin Resistance in Obesity. Circ. Res. 2020, 126, 1549–1564. [Google Scholar] [CrossRef]
- Dommel, S.; Blüher, M. Does C-C Motif Chemokine Ligand 2 (CCL2) Link Obesity to a Pro-Inflammatory State? Int. J. Mol. Sci. 2021, 22, 1500. [Google Scholar] [CrossRef]
- Kanda, H.; Tateya, S.; Tamori, Y.; Kotani, K.; Hiasa, K.-I.; Kitazawa, R.; Kitazawa, S.; Miyachi, H.; Maeda, S.; Egashira, K.; et al. MCP-1 contributes to macrophage infiltration into adipose tissue, insulin resistance, and hepatic steatosis in obesity. J. Clin. Investig. 2006, 116, 1494–1505. [Google Scholar] [CrossRef]
- Wang, Y.-T.; Zheng, S.-Y.; Jiang, S.-D.; Luo, Y.; Wu, Y.-X.; Naranmandakh, S.; Li, Y.-S.; Liu, S.-G.; Xiao, W.-F. Irisin in degenerative musculoskeletal diseases: Functions in system and potential in therapy. Pharmacol. Res. 2024, 210, 107480. [Google Scholar] [CrossRef]
- Iglesias, P. Muscle in Endocrinology: From Skeletal Muscle Hormone Regulation to Myokine Secretion and Its Implications in Endocrine–Metabolic Diseases. J. Clin. Med. 2025, 14, 4490. [Google Scholar] [CrossRef] [PubMed]
- Li, G.; Zhang, L.; Wang, D.; Aiqudsy, L.; Jiang, J.X.; Xu, H.; Shang, P. Muscle-bone crosstalk and potential therapies for sarco-osteoporosis. J. Cell. Biochem. 2019, 120, 14262–14273. [Google Scholar] [CrossRef]
- Graf, C.; Ferrari, N. Metabolic Health—The Role of Adipo-Myokines. Int. J. Mol. Sci. 2019, 20, 6159. [Google Scholar] [CrossRef] [PubMed]
- Street, M.E.; Moghetti, P.; Chiarelli, F.; Balakrishnan, R.; Thurmond, D.C. Mechanisms by Which Skeletal Muscle Myokines Ameliorate Insulin Resistance. Int. J. Mol. Sci. 2022, 23, 4636. [Google Scholar] [CrossRef] [PubMed]
- Nadeau, L.; Patten, D.; Caron, A.; Garneau, L.; Pinault-Masson, E.; Foretz, M.; Haddad, P.; Anderson, B.; Quinn, L.; Jardine, K.; et al. IL-15 improves skeletal muscle oxidative metabolism and glucose uptake in association with increased respiratory chain supercomplex formation and AMPK pathway activation. Biochim. Biophys. Acta (BBA) Gen. Subj. 2019, 1863, 395–407. [Google Scholar] [CrossRef]
- Li, Y.; Xu, Z. Association between irisin and metabolic parameters in nondiabetic, nonobese adults: A meta-analysis. Diabetol. Metab. Syndr. 2022, 14, 152. [Google Scholar] [CrossRef]
- Chen, J.-Q.; Huang, Y.-Y.; Gusdon, A.M.; Qu, S. Irisin: A new molecular marker and target in metabolic disorder. Lipids Health Dis. 2015, 14, 2. [Google Scholar] [CrossRef]
- Rashid, F.A.; Abbas, H.J.; Naser, N.A.; Ali, H.A. Effect of Long-Term Moderate Physical Exercise on Irisin between Normal Weight and Obese Men. Sci. World J. 2020, 2020, 1897027. [Google Scholar] [CrossRef]
- Hejazi, J.; Ghobadian, B.; Ghasemi, N.; Sadeh, H.; Abedimanesh, N.; Rahimlou, M. Relationship of serum irisin levels, physical activity, and metabolic syndrome biomarkers in obese individuals with low-calorie intake and non-obese individuals with high-calorie intake. J. Health Popul. Nutr. 2025, 44, 2. [Google Scholar] [CrossRef]
- Mai, S.; Grugni, G.; Mele, C.; Vietti, R.; Vigna, L.; Sartorio, A.; Aimaretti, G.; Scacchi, M.; Marzullo, P. Irisin levels in genetic and essential obesity: Clues for a potential dual role. Sci. Rep. 2020, 10, 1020. [Google Scholar] [CrossRef]
- Huerta, A.E.; Prieto-Hontoria, P.L.; Fernández-Galilea, M.; Sáinz, N.; Cuervo, M.; Martínez, J.A.; Moreno-Aliaga, M.J. Circulating irisin and glucose metabolism in overweight/obese women: Effects of α-lipoic acid and eicosapentaenoic acid. J. Physiol. Biochem. 2015, 71, 547–558. [Google Scholar] [CrossRef] [PubMed]
- Qiu, S.; Cai, X.; Yin, H.; Zügel, M.; Sun, Z.; Steinacker, J.M.; Schumann, U. Association between circulating irisin and insulin resistance in non-diabetic adults: A meta-analysis. Metabolism 2016, 65, 825–834. [Google Scholar] [CrossRef]
- Werida, R.H.; El-Gharbawy, N.M.; Mostafa, T.M. Circulating IL-6, clusterin and irisin in obese subjects with different grades of obesity: Association with insulin resistance and sexual dimorphism. Arch. Endocrinol. Metab. 2021, 65, 126–136. [Google Scholar] [CrossRef]
- Elizondo-Montemayor, L.; Gonzalez-Gil, A.M.; Tamez-Rivera, O.; Toledo-Salinas, C.; Peschard-Franco, M.; Rodríguez-Gutiérrez, N.A.; Silva-Platas, C.; Garcia-Rivas, G. Association between Irisin, hs-CRP, and Metabolic Status in Children and Adolescents with Type 2 Diabetes Mellitus. Mediat. Inflamm. 2019, 2019, 6737318. [Google Scholar] [CrossRef]
- Eslampour, E.; Ebrahimzadeh, F.; Abbasnezhad, A.; Khosroshahi, M.Z.; Choghakhori, R.; Asbaghi, O. Association between Circulating Irisin and C-Reactive Protein Levels: A Systematic Review and Meta-Analysis. Endocrinol. Metab. 2019, 34, 140–149. [Google Scholar] [CrossRef]
- Khajebishak, Y.; Faghfouri, A.H.; Soleimani, A.; Peyrovi, S.; Payahoo, L. The Potential Relationship Between Serum Irisin Concentration With Inflammatory Cytokines, Oxidative Stress Biomarkers, Glycemic Indices and Lipid Profiles in Obese Patients with Type 2 Diabetes Mellitus: A Pilot Study. J. ASEAN Fed. Endocr. Soc. 2023, 38, 45. [Google Scholar] [CrossRef]
- Ashraf, H.; Laway, B.A.; Afroze, D.; Wani, A.I. Evaluation of Proinflammatory Cytokines in Obese vs Non-obese Patients with Metabolic Syndrome. Indian J. Endocrinol. Metab. 2018, 22, 751–756. [Google Scholar] [CrossRef]
- Su, Z.; Efremov, L.; Mikolajczyk, R. Differences in the levels of inflammatory markers between metabolically healthy obese and other obesity phenotypes in adults: A systematic review and meta-analysis. Nutr. Metab. Cardiovasc. Dis. 2024, 34, 251–269. [Google Scholar] [CrossRef]
- Tarantino, G.; Citro, V.; Balsano, C.; Capone, D. Age and Interleukin-15 Levels Are Independently Associated with Intima-Media Thickness in Obesity-Related NAFLD Patients. Front. Med. 2021, 8, 634962. [Google Scholar] [CrossRef]
- Silva, F.M.; Duarte-Mendes, P.; Teixeira, A.M.; Soares, C.M.; Ferreira, J.P. The effects of combined exercise training on glucose metabolism and inflammatory markers in sedentary adults: A systematic review and meta-analysis. Sci. Rep. 2024, 14, 1936. [Google Scholar] [CrossRef]
- Di Ludovico, A.; La Bella, S.; Ciarelli, F.; Chiarelli, F.; Breda, L.; Mohn, A. Skeletal muscle as a pro- and anti-inflammatory tissue: Insights from children to adults and ultrasound findings. J. Ultrasound 2024, 27, 769–779. [Google Scholar] [CrossRef]
- Neira, G.; Gómez-Ambrosi, J.; Cienfuegos, J.A.; Ramírez, B.; Becerril, S.; Rodríguez, A.; Burrell, M.A.; Baixauli, J.; Mentxaka, A.; Casado, M.; et al. Increased expression of IL-1β in adipose tissue in obesity influences the development of colon cancer by promoting inflammation. J. Physiol. Biochem. 2025, 81, 471–482. [Google Scholar] [CrossRef]
- Yan, J.; Zhang, P.; Liu, X.; Pan, C.; Shi, G.; Ye, P.; Zou, X.; Li, X.; Zheng, X.; Liu, Y.; et al. Obesity modulates hematopoietic stem cell fate decision via IL-1β induced p38/MAPK signaling pathway. Stem Cell Res. Ther. 2024, 15, 336. [Google Scholar] [CrossRef]
- Ghanbari, M.; Momen Maragheh, S.; Aghazadeh, A.; Mehrjuyan, S.R.; Hussen, B.M.; Abdoli Shadbad, M.; Dastmalchi, N.; Safaralizadeh, R. Interleukin-1 in obesity-related low-grade inflammation: From molecular mechanisms to therapeutic strategies. Int. Immunopharmacol. 2021, 96, 107765. [Google Scholar] [CrossRef]
- Belizário, J.E.; Fontes-Oliveira, C.C.; Borges, J.P.; Kashiabara, J.A.; Vannier, E. Skeletal muscle wasting and renewal: A pivotal role of myokine IL-6. SpringerPlus 2016, 5, 619. [Google Scholar] [CrossRef] [PubMed]
- Mir, M.M.; Alfaifi, J.; Sohail, S.K.; Rizvi, S.F.; Akhtar, T.; Alghamdi, M.A.A.; Mir, R.; Wani, J.I.; Sabah, Z.U.; Alhumaydhi, F.A.; et al. Role of Pro-Inflammatory Chemokines CCL-1, 2, 4, and 5 in the Etiopathogenesis of Type 2 Diabetes Mellitus in Subjects from the Asir Region of Saudi Arabia: Correlation with Different Degrees of Obesity. J. Pers. Med. 2024, 14, 743. [Google Scholar] [CrossRef]
- Pan, X.; Kaminga, A.C.; Wen, S.W.; Liu, A. Chemokines in Prediabetes and Type 2 Diabetes: A Meta-Analysis. Front. Immunol. 2021, 12, 622438. [Google Scholar] [CrossRef] [PubMed]
- Alexaki, V.I. Adipose tissue-derived mediators of systemic inflammation and metabolic control. Curr. Opin. Endocr. Metab. Res. 2024, 37, 100560. [Google Scholar] [CrossRef]
- Singhai, M.; Faizy, A.F.; Goyal, R.; Siddiqui, S.S. Evaluation of TNF-α and IL-6 Levels in Obese and Non-obese Diabetics: Pre- and Postinsulin Effects. N. Am. J. Med. Sci. 2012, 4, 180. [Google Scholar] [CrossRef] [PubMed]
- Reddy, V.K.K.; Shiddapur, G.; Jagdale, N.; Kondapalli, M.P.; Adapa, S. Investigating Interleukin-6 Levels in Type 2 Diabetes Mellitus Patients With and Without Diabetic Nephropathy. Cureus 2024, 16, e67014. [Google Scholar] [CrossRef] [PubMed]
- Fang, P.; She, Y.; Yu, M.; Min, W.; Shang, W.; Zhang, Z. Adipose–Muscle crosstalk in age-related metabolic disorders: The emerging roles of adipo-myokines. Ageing Res. Rev. 2023, 84, 101829. [Google Scholar] [CrossRef]
- Zhang, Y.; He, X.; Wang, K.; Xue, Y.; Hu, S.; Jin, Y.; Zhu, G.; Shi, Q.; Rui, Y. Irisin alleviates obesity-induced bone loss by inhibiting interleukin 6 expression via TLR4/MyD88/NF-κB axis in adipocytes. J. Adv. Res. 2025, 69, 343–359. [Google Scholar] [CrossRef] [PubMed]
- Mu, W.-J.; Zhu, J.-Y.; Chen, M.; Guo, L. Exercise-Mediated Browning of White Adipose Tissue: Its Significance, Mechanism and Effectiveness. Int. J. Mol. Sci. 2021, 22, 11512. [Google Scholar] [CrossRef] [PubMed]
- Severinsen, M.C.K.; Pedersen, B.K. Muscle–Organ Crosstalk: The Emerging Roles of Myokines. Endocr. Rev. 2020, 41, 594–609. [Google Scholar] [CrossRef] [PubMed]
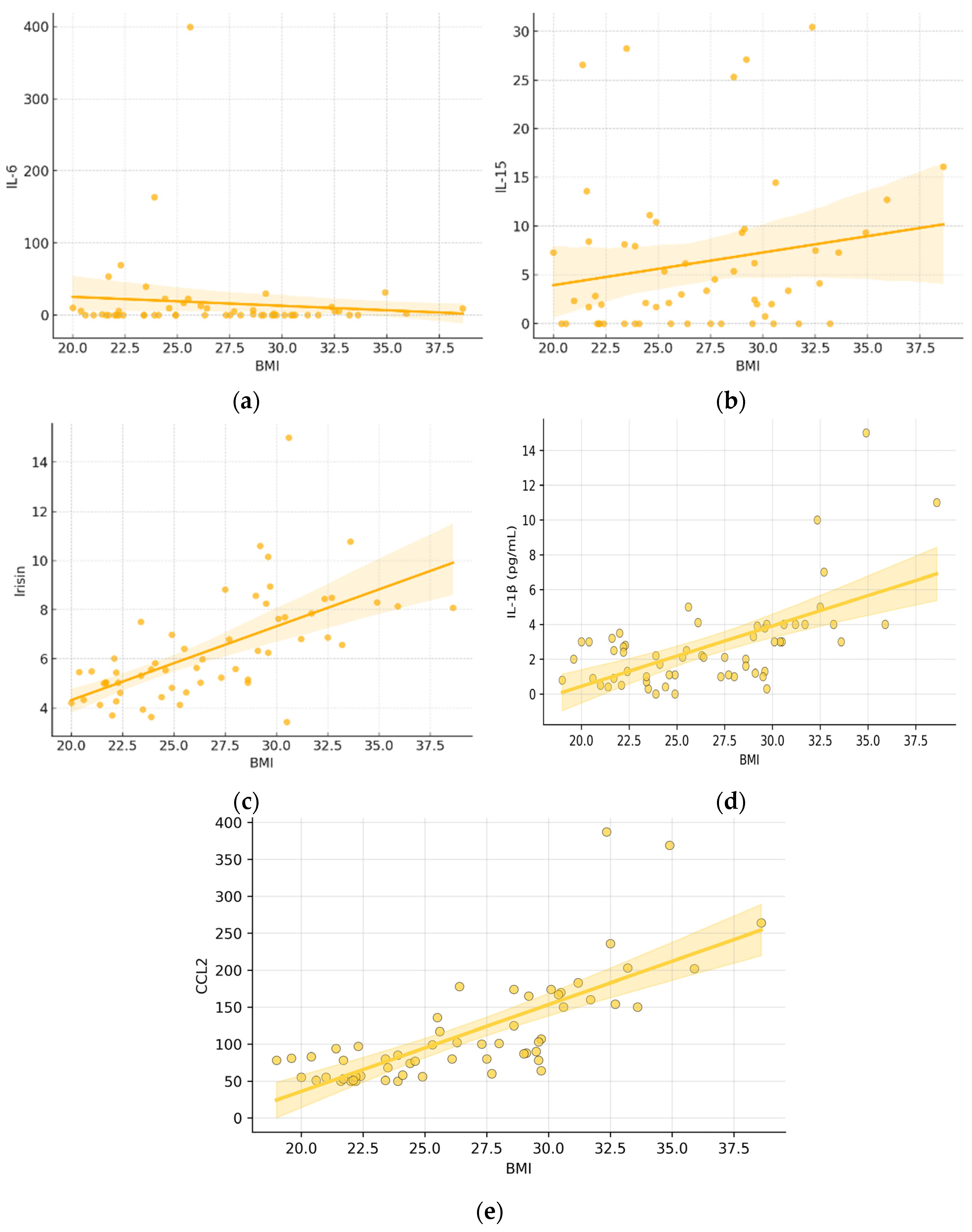
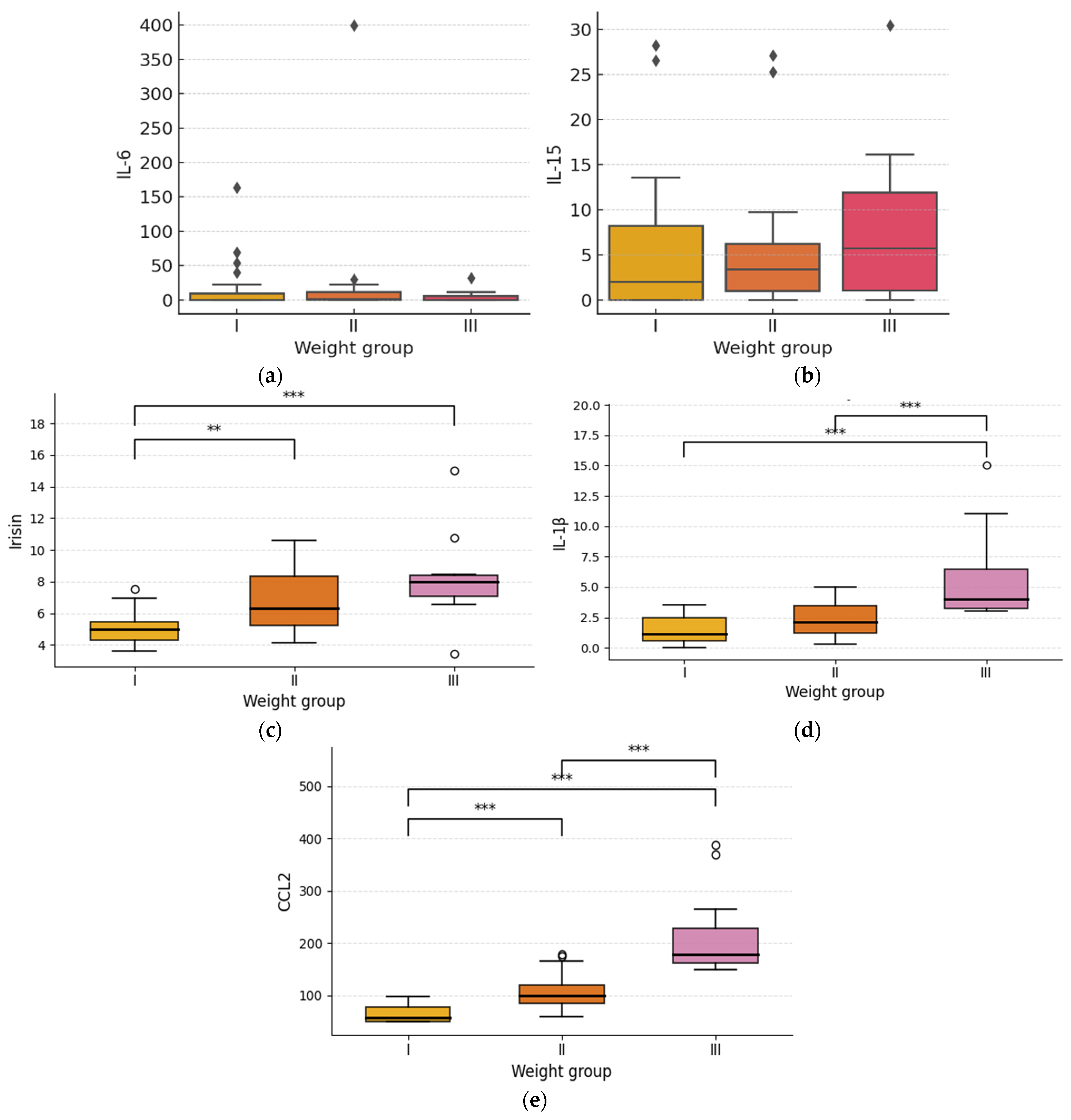


| Parameter | Group I (Normal Weight, n = 20) | Group II (Overweight, n = 20) | Group III (Obese, n = 11) |
|---|---|---|---|
| BMI (kg/m2) | 22 (21–23) | 28 (27–29) | 34 (33–35) |
| WHR | 0.80 (0.76–0.84) | 1.00 (0.97–1.03) | 1.20 (1.15–1.25) |
| Systolic BP (mmHg) | 119 (116–122) | 121 (117–125) | 124 (121–127) |
| Diastolic BP (mmHg) | 78 (74–82) | 80 (77–83) | 83 (80–86) |
| Visceral fat (level) | 4 (3–5) | 8 (6–10) | 12 (9–15) |
| HOMA-IR | 2.0 (1.3–2.7) | 3.1 (1.8–4.4) | 3.5 (2.2–4.8) |
| Total cholesterol (mg/dL) | 193 (170–216) | 253 (248–258) | 204 (199–209) |
| LDL (mg/dL) | 135 (131–139) | 120 (92–148) | 124 (111–137) |
| HDL (mg/dL) | 67 (64–70) | 58 (47–69) | 57 (47–67) |
| Triglycerides (mg/dL) | 51 (50–52) | 158 (136–180) | 114 (66–162) |
| Insulin (µIU/mL) | 8.3 (3.0–13.6) | 12.0 (4.6–19.4) | 11.0 (8.4–13.6) |
| C-peptide (ng/mL) | 1.9 (0.7–3.1) | 3.0 (1.3–4.7) | 2.3 (1.8–2.8) |
| CRP (mg/L) | 1.0 (0.9–1.1) | 5.2 (0–10.9) | 2.5 (0.5–4.5) |
| Fasting glucose (mg/dL) | 97 (95–99) | 91 (89–93) | 99 (84–114) |
| 1 IL-6 | 2 IL-15 | Irisin | IL-1β | CCL2 | |
|---|---|---|---|---|---|
| 3 BMI | 0.0963 | 0.1918 | 0.5442 * | 0.6121 * | 0.7812 * |
| 4 WHR | 0.0423 | 0.1790 | 0.4197 | 0.5525 * | 0.7304 * |
| Visceral tissue | 0.1043 | 0.2040 | 0.4624 | 0.4810 | 0.6145 * |
| Systolic blood pressure | 0.0593 | 0.3847 | 0.2350 | 0.1547 | 0.0875 |
| Diastolic blood pressure | 0.2140 | 0.1754 | 0.2366 | 0.0932 | 0.1455 |
| Glucose | 0.1111 | 0.0611 | 0.2041 | 0.1456 | 0.3054 |
| Insulin | 0.0871 | 0.1842 | 0.7704 * | 0.3202 | 0.37 |
| C-peptide | 0.0878 | 0.1050 | 0.7115 * | 0.3332 | 0.36 |
| 5 HOMA-IR | 0.0732 | 0.1802 | 0.6332 * | 0.3648 | 0.4 |
| Cholesterol | 0.0238 | 0.2482 | 0.3093 | 0.0452 | 0.2457 |
| 6 HDL | 0.0573 | 0.0423 | 0.2286 | 0.0056 | 0.0454 |
| 7 LDL | 0.0136 | 0.2725 | 0.1392 | 0.0458 | 0.1236 |
| 8 TG | 0.0735 | 0.2305 | 0.2050 | 0.2365 | 0.0548 |
| 9 CRP | 0.0743 | 0.0749 | 0.2112 | 0.4235 | 0.41 |
| Biomarker | Test Results | Interpretation |
|---|---|---|
| IL-6 | H-statistic = 0.47 | No significant difference in IL-6 levels across weight groups. |
| p-value = 0.79 | ||
| IL-15 | H-statistic = 1.34 | No significant difference in IL-15 levels across weight groups. |
| p-value = 0.51 | ||
| Irisin | H-statistic = 21.14 | Highly significant difference in Irisin levels between weight groups. |
| p-value ≈ 2.6 × 10−5 | ||
| IL-1β | H-statistic = 26.26 | Highly significant difference in IL-1β levels between weight groups. |
| p-value ≈ 2.0 × 10−4 | ||
| CCL2 | H-statistic = 42.06 | Highly significant difference in CCL2 levels between weight groups. |
| p-value ≈ 7.3 × 10−6 |
| Outcome | Predictor | β | 95% CI | p-Value | R2 | Adj. R2 |
|---|---|---|---|---|---|---|
| HOMA-IR | Irisin | 0.89 | 0.62 to 1.16 | <0.001 | 0.71 | 0.68 |
| IL-6 | 0.10 | −0.02 to 0.22 | 0.100 | |||
| IL-15 | 0.04 | −0.09 to 0.17 | 0.524 | |||
| IL-1β | −0.07 | −0.32 to 0.18 | 0.586 | |||
| CCL2 | −0.04 | −0.40 to 0.33 | 0.838 | |||
| BMI | Irisin | 0.38 | 0.12 to 0.64 | 0.004 | 0.72 | 0.69 |
| IL-6 | −0.03 | −0.33 to 0.26 | 0.819 | |||
| IL-15 | −0.11 | −0.32 to 0.10 | 0.315 | |||
| IL-1β | −0.07 | −0.44 to 0.31 | 0.723 | |||
| CCL2 | 0.70 | 0.33 to 1.07 | <0.001 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Narloch, W.; Jaskulak, M.; Antoniak-Pietrynczak, K.; Zorena, K. Irisin as an Associative Marker of Metabolic Dysregulation in Obesity: Comparative Profiling of IL-6, IL-15, IL-1β, and CCL2. Diagnostics 2026, 16, 1459. https://doi.org/10.3390/diagnostics16101459
Narloch W, Jaskulak M, Antoniak-Pietrynczak K, Zorena K. Irisin as an Associative Marker of Metabolic Dysregulation in Obesity: Comparative Profiling of IL-6, IL-15, IL-1β, and CCL2. Diagnostics. 2026; 16(10):1459. https://doi.org/10.3390/diagnostics16101459
Chicago/Turabian StyleNarloch, Wiktoria, Marta Jaskulak, Klaudia Antoniak-Pietrynczak, and Katarzyna Zorena. 2026. "Irisin as an Associative Marker of Metabolic Dysregulation in Obesity: Comparative Profiling of IL-6, IL-15, IL-1β, and CCL2" Diagnostics 16, no. 10: 1459. https://doi.org/10.3390/diagnostics16101459
APA StyleNarloch, W., Jaskulak, M., Antoniak-Pietrynczak, K., & Zorena, K. (2026). Irisin as an Associative Marker of Metabolic Dysregulation in Obesity: Comparative Profiling of IL-6, IL-15, IL-1β, and CCL2. Diagnostics, 16(10), 1459. https://doi.org/10.3390/diagnostics16101459








