Preoperative CT Evaluation of Abdominal Vasculature and the Risk of Surgical Complications in Colorectal Cancer Resection with Anastomosis: A Systematic Review and Meta-Analysis
Abstract
1. Introduction
2. Methods
2.1. Search Strategy
2.2. Study Selection
2.3. Data Extraction
2.4. Bias and Certainty Assessment
2.5. Statistical Analysis
3. Results
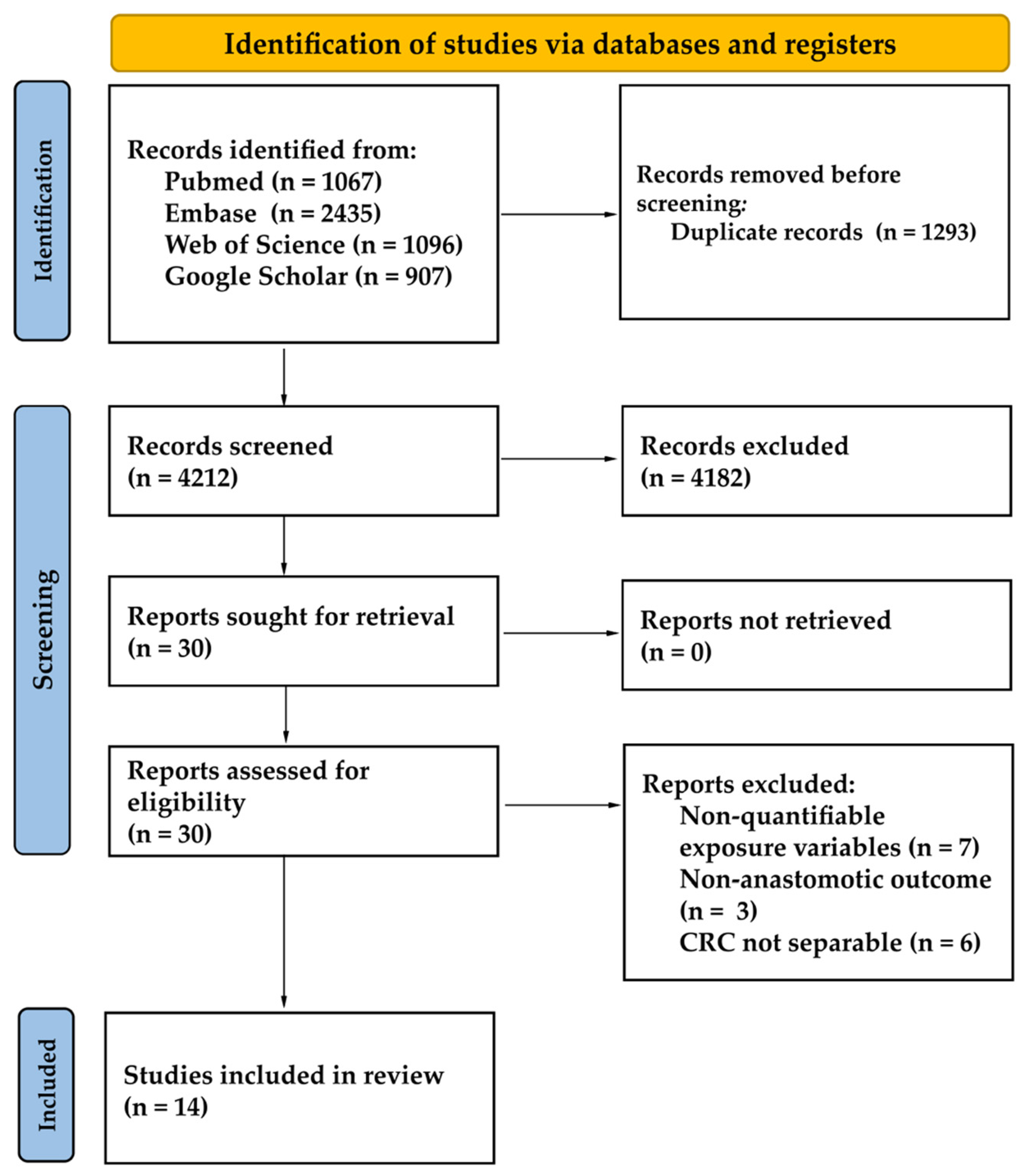
3.1. Study Selection
3.2. Characteristics of the Included Studies
Risk of Bias Assessment
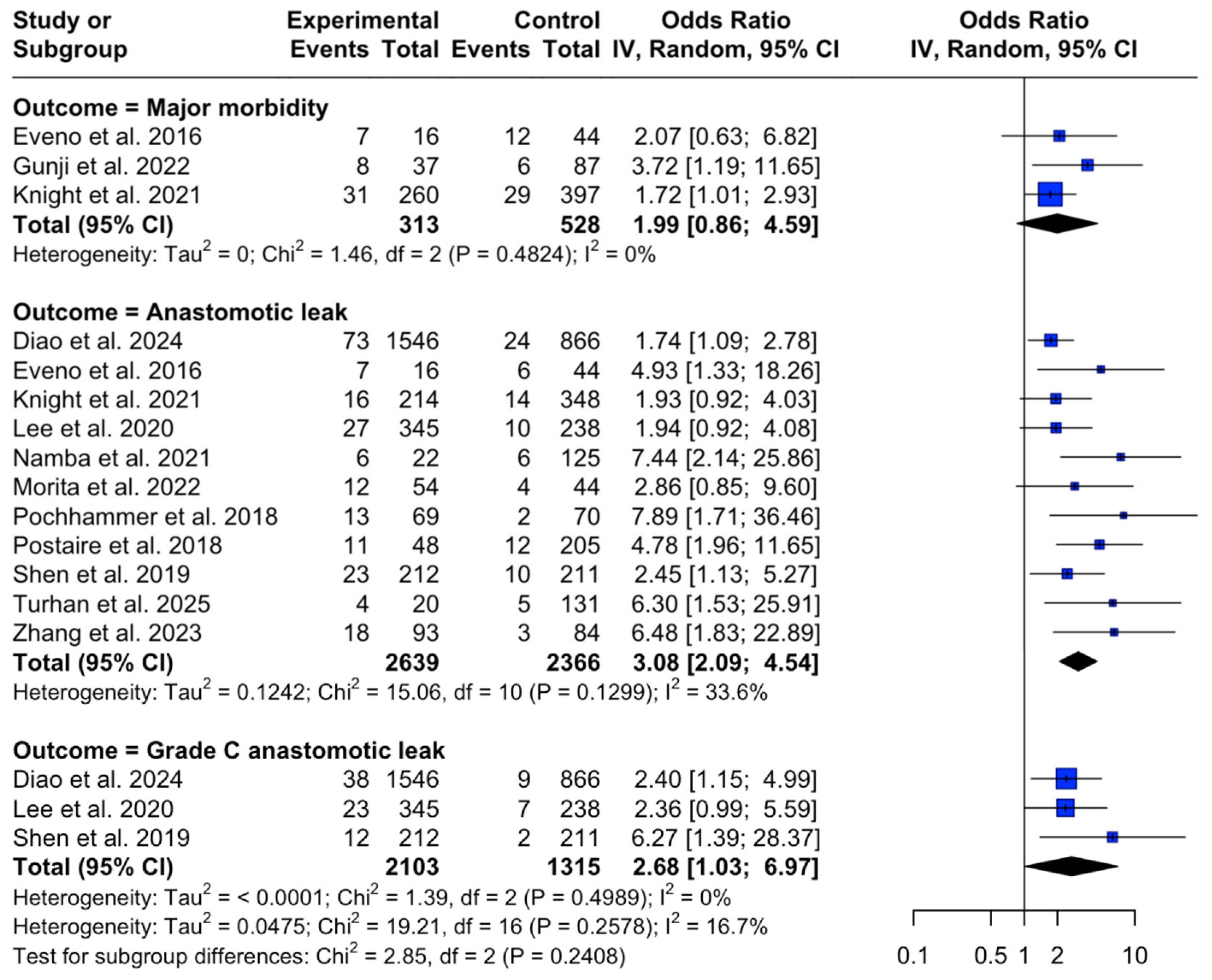
3.3. Summary of Findings
3.4. Relationship Between Calcium Score and Surgical Complications
3.4.1. Sensitivity Analysis
3.4.2. Publication Bias
3.5. Relationship Between Abdominal Vessel Stenosis and Surgical Complications
3.6. Relationship Between Abdominal Vascular Disease and Mortality
4. Discussion
4.1. Strengths and Limitations
4.2. Future Directions
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| AAC | Abdominal aortic calcification |
| ACI | Aorto-iliac calcification index |
| ACS | Abdominal aortic calcification score |
| AL | Anastomotic leakage |
| BMI | Body mass index |
| CA | Celiac artery |
| CC | Case–control |
| CAVI | Cardio-ankle vascular index |
| C-D | Clavien–Dindo (classification) |
| CECT | Contrast-enhanced computed tomography |
| CI | Confidence interval |
| CORREA | ESCP CORREA 2022 audit |
| CRC | Colorectal cancer |
| CT | Computed tomography |
| ESCP | European Society of Coloproctology |
| GLOBOCAN | Global Cancer Observatory database |
| GRADE | Grading of Recommendations Assessment, Development and Evaluation |
| ICG-FA | Indocyanine green fluorescence angiography |
| IMA | Inferior mesenteric artery |
| I2 | I-squared heterogeneity statistic |
| ISREC | International Study Group of Rectal Cancer |
| MEDLINE | Medical Literature Analysis and Retrieval System Online |
| MRI | Magnetic resonance imaging |
| NOS | Newcastle–Ottawa Scale |
| OR | Odds ratio |
| PC | Prospective cohort |
| PCS | Prospective cross-sectional |
| PEO | Patient, exposure, outcome |
| PET | Positron emission tomography |
| PRISMA | Preferred Reporting Items for Systematic Reviews and Meta-Analyses |
| PROSPERO | International Prospective Register of Systematic Reviews |
| RC | Retrospective cohort |
| RCT | Randomized controlled trial |
| ROB | Risk of bias |
| ROC | Receiver operating characteristic |
| SMA | Superior mesenteric artery |
| WoS | Web of Science Core Collection |
| τ2 | Between-study variance (tau-squared) |
Appendix A. Search Strategy
| Block | String | Number of Results |
|---|---|---|
| Cancer—1 | “Colorectal Neoplasms” [mh] OR “Colonic Neoplasms” [mh] OR “Rectal Neoplasms” [mh] OR “Cecal Neoplasms” [mh] OR ((“Intestine, Large” [mh] OR “Cecum” [mh] OR “Colon” [mh] OR “Rectum” [mh] OR colorectal * [tiab] OR colon * [tiab] OR rectal * [tiab] OR rectum [tiab] OR anal [tiab] OR anus [tiab] OR appendi * [tiab] OR cecum * [tiab] OR coecum * [tiab] OR caecum * [tiab] OR cecal * [tiab] OR coecal * [tiab] OR caecal * [tiab] OR sigmoid * [tiab] OR colorectal [tiab] OR colon [tiab] OR colonic [tiab] OR rectal [tiab] OR rectum [tiab] OR “large bowel” [tiab] OR CRC [tiab]) AND (cancer * [tiab] OR carcinoma * [tiab] OR tumor * [tiab] OR tumour * [tiab] OR malignan * [tiab] OR “bowel cancer” [tiab] OR neoplas * [tiab] OR tumour * [tiab] OR cancer * [tiab] OR oncolog * [tiab] OR adenoma * [tiab] OR malignan * [tiab]) | 502,433 results |
| Surgery—2 | “Colorectal Surgery” [mh] OR “colorectal surg *” [tiab] OR “colorectal resect *” [tiab] OR “hemicolectomy” [tiab] OR “colon resect *” [tiab] OR “colorectal anastomos *” [tiab] OR “LAR surgery” [tiab] OR “low anterior resection” [tiab] OR “abdominoperineal resec t*” [tiab] OR “abdominoperineal surg *” [tiab] OR “bowel obstruction surg *” [tiab] OR “proctocolectomy” [tiab] OR “ileocolectomy” [tiab] OR “sigmoidectomy” [tiab] OR “colectomy *” [tiab] OR “proctectomy” [tiab] OR “left colectomy” [tiab] OR “right colectomy” [tiab] OR “laparoscopic colectomy” [tiab] OR “colon surg *” [tiab] OR “rectal surg *” [tiab] OR “proctology *” [tiab] OR “coloproctotom *” [tiab] OR “proctocolonic” [tiab] OR ((“operat *” [tiab] OR “surg *” [tiab] OR “resect *” [tiab]) AND (“Intestine, Large” [mh] OR “Cecum” [mh] OR “Colon” [mh] OR “Rectum” [mh] OR colo * [tiab] OR recta * [tiab] OR rectu * [tiab] OR anal [tiab] OR anus [tiab] OR appendi * [tiab] OR cecum * [tiab] OR coecum * [tiab] OR caecum * [tiab] OR cecal * [tiab] OR coecal * [tiab] OR caecal * [tiab] OR sigmoid * [tiab]) | 276,803 results |
| Imaging—3 | “Diagnostic Imaging” [mh] OR “Tomography, X-Ray Computed” [mh] OR “Computed Tomography Angiography” [mh] OR “CT scan *” [tiab] OR “CAT scan *” [tiab] OR “compute * tomograph*” [tiab] OR “X-ray comput *” [tiab] OR CT [tiab] OR CTA [tiab] OR “CT perfusion” [tiab] OR “perfusion CT” [tiab] OR CTP [tiab] OR “computed tomograph * angiograph*” [tiab] OR “spiral CT” [tiab] OR “helical CT” [tiab] OR “multidetector CT” [tiab] OR “dual-energy CT” [tiab] | 3,460,819 results |
| Intervention—I | “Vascular Calcification” [mh] OR “Atherosclerosis/diagnostic imaging” [mh] OR “Mesenteric Arteries/diagnostic imaging” [mh] OR “Aorta, Abdominal/diagnostic imaging” [mh] OR “Iliac Artery/diagnostic imaging” [mh] OR radiomic * [tiab] OR “radiomic signature *” [tiab] OR “texture analysis” [tiab] OR “imaging biomarker *” [tiab] OR “imaging feature *” [tiab] OR calcif * [tiab] OR calcific * [tiab] OR “calcification” [tiab] OR “calcifications” [tiab] OR “calcified plaque” [tiab] OR “calcium score” [tiab] OR “calcium scoring” [tiab] OR “Agatston score” [tiab] OR Agatston [tiab] OR “aortic calcification” [tiab] OR “abdominal aortic calcification” [tiab] OR “iliac calcification” [tiab] OR “vascular calcification” [tiab] OR stenos * [tiab] OR stenosis [tiab] OR stenoses [tiab] OR occlus * [tiab] OR “occlusion” [tiab] OR “occlusive disease” [tiab] OR “arterial stenosis” [tiab] OR “arterial occlusion” [tiab] OR “arterial obstruction” [tiab] OR “mesenteric stenosis” [tiab] OR “mesenteric occlusion” [tiab] OR “mesenteric ischemia” [tiab] OR SMA [tiab] OR IMA [tiab] OR perfus * [tiab] OR “blood flow” [tiab] OR “tissue perfusion” [tiab] OR microperfusion [tiab] OR “CT perfusion” [tiab] OR “perfusion CT” [tiab] OR CTP [tiab] | 1,018,018 results |
| Outcome—O | post-operat * [tiab] OR postoperat * [tiab] OR surger * [tiab] OR surgical * [tiab] OR operat * [tiab]) AND (complication * [tiab]) OR “Postoperative Complications” [mh] OR “Anastomotic Leak” [mh] OR “Anastomosis, Surgical/adverse effects” [mh] OR “Surgical Wound Infection” [mh] OR “Surgical Wound Dehiscence” [mh] OR “Ileus” [mh] OR “Mortality” [mh] OR “Reoperation” [mh] OR “Patient readmission” [mh] OR infect * [tiab] OR outcome * [tiab] OR leak * [tiab] OR hernia * [tiab] OR complicat * [tiab] OR readmi * [tiab] OR prognos * [tiab] OR predict * [tiab] OR abcess [tiab] OR abscess [tiab] OR impact [tiab] OR morbidity [tiab] OR mortality [tiab] OR adverse [tiab] OR recur * [tiab] OR recover * [tiab] OR emergen * [tiab] OR urgen * [tiab] OR death [tiab] OR survival [tiab] OR “acute kidney injury” [tiab] OR AKI [tiab] OR risk [tiab] OR peritonit * [tiab] OR adhesion [tiab] OR ischemia * [tiab] OR perforat * [tiab] OR repair [tiab] OR anastomo * [tiab] OR bleeding [tiab] OR stricture * [tiab] OR stenos * [tiab] OR “postoperative outcome *” [tiab] OR “surgical outcome *” [tiab] OR “treatment outcome *” [tiab] OR “postoperative mortality” [tiab] OR “anastomotic leak” [tiab] OR “anastomotic leakage” [tiab] OR “anastomotic failure” [tiab] OR “anastomotic dehiscence” [tiab] OR “anastomotic insufficiency” [tiab] OR “anastomotic fistula” [tiab] OR “surgical site infection” [tiab] OR SSI [tiab] OR “wound infection” [tiab] OR “intra-abdominal infection” [tiab] OR abscess* [tiab] OR ileus [tiab] OR “postoperative ileus” [tiab] OR “paralytic ileus” [tiab] OR “organ dysfunction” [tiab] OR “organ failure” [tiab] OR “multiple organ dysfunction” [tiab] OR sepsis [tiab] OR reoperate * [tiab] OR “re-operation” [tiab] OR readmiss * [tiab] OR “hospital readmission” [tiab] OR “Clavien-Dindo” [tiab] OR Clavien [tiab] OR “complication grade” [tiab] | 14,872,887 results |
| Population—P | 1 AND 2 AND 3 | 126,080 results |
| Total | P AND I AND O | 1717 results |
| Total—study exclusion | Total NOT (Review [Publication Type] OR “Case reports” [Publication Type]) NOT (animals [Mesh] NOT humans [Mesh]) | 1067 results |
| String | Results Screened |
|---|---|
| “abdominal aortic calcification” CT colorectal anastomotic leak | 72 |
| “aortic calcification index” CT rectal anastomotic leak | 20 |
| “aortoiliac calcification” CT rectal surgery anastomotic leak | 29 |
| “iliac artery calcification” CT rectal resection anastomotic leak | 9 |
| “calcium score” aortic CT colorectal anastomotic leak | 93 |
| Agatston CT rectal cancer surgery anastomotic leak | 71 |
| “vascular calcification” CT colorectal surgery | 113 |
| “inferior mesenteric artery” stenosis CT rectal anastomotic leak | 200 |
| “inferior mesenteric artery” occlusion CT rectal anastomotic leak | 200 |
| “superior mesenteric artery” stenosis CT right colectomy anastomotic leak | 200 |
| “celiac trunk” stenosis CT right colectomy anastomotic leak | 200 |
| “mesenteric artery stenosis” CT colorectal surgery complication | 149 |
| “mesenteric occlusive disease” CT colorectal anastomotic leak | 28 |
Appendix B. Newcastle–Ottawa Scores
| Study | Selection | Comparability | Outcome/Exposure | Total 0–9 | Study Quality | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| S1 | S2 | S3 | S4 | C1 | C2 | O1/E1 | O2/E2 | O3/E3 | |||
| Arron et al. 2022 [19] | ☆ | ☆ | ☆ | ☆ | ☆ | ☆ | ☆ | 7 | Good | ||
| Diao et al. 2024 [20] | ☆ | ☆ | ☆ | ☆ | ☆ | ☆ | ☆ | ☆ | 8 | Good | |
| Eveno et al. 2016 [21] | ☆ | ☆ | ☆ | ☆ | ☆ | ☆ | ☆ | 7 | Good | ||
| Gunji et al. 2022 [27] | ☆ | ☆ | ☆ | ☆ | ☆ | ☆ | 6 | Fair | |||
| Knight et al. 2021 [28] | ☆ | ☆ | ☆ | ☆ | ☆ | ☆ | ☆ | ☆ | 8 | Good | |
| Lee et al. 2020 [29] | ☆ | ☆ | ☆ | ☆ | ☆ | ☆ | 6 | Fair | |||
| Morita et al. 2022 [30] | ☆ | ☆ | ☆ | ☆ | ☆ | ☆ | 6 | Fair | |||
| Namba et al. 2021 [31] | ☆ | ☆ | ☆ | ☆ | ☆ | ☆ | ☆ | 7 | Good | ||
| Norooz et al. 2016 [32] | ☆ | ☆ | ☆ | ☆ | ☆ | 5 | Fair | ||||
| Pochhammer et al. 2018 [33] | ☆ | ☆ | ☆ | ☆ | ☆ | ☆ | ☆ | 7 | Good | ||
| Postaire et al. 2018 [34] | ☆ | ☆ | ☆ | ☆ | ☆ | ☆ | 6 | Fair | |||
| Shen et al. 2019 [35] | ☆ | ☆ | ☆ | ☆ | ☆ | ☆ | 6 | Fair | |||
| Turhan et al. 2025 [36] | ☆ | ☆ | ☆ | ☆ | ☆ | ☆ | ☆ | 7 | Good | ||
| Zhang et al. 2023 [37] | ☆ | ☆ | ☆ | ☆ | ☆ | ☆ | 6 | Fair | |||
Appendix C. Adjusted Analyses
| Study | Outcome | CT Vascular Marker (Cut Point) | Adjustment/Matching (Covariates) | Adjusted/Matched Estimate |
|---|---|---|---|---|
| Arron et al. 2022 [19] | Any AL | IMA stenosis ≥ 70% or occlusion | Matched CC (1:1) on age, BMI, cardiovascular comorbidity | Clinically significant IMA stenosis: 21.2% (AL cases) vs. 1.9% (controls); p = 0.01 |
| Diao et al. 2024 [20] | Any AL | AAC (present vs. absent) | Multivariable logistic regression; sex, HTN, T2DM, tumor location, tumor stage, smoking, drinking | OR 1.443 (95% CI 0.871–2.358); p = 0.157 |
| Gunji et al. 2022 [27] | Major complications (C-D ≥ III) | AAC score (continuous variable) | Multivariable logistic regression; covariates (p < 0.25 in univariate): sex, ASA ≥ 3, hyperlipidemia, chronic renal failure, stage, CAVI, laparoscopic surgery, anterior resection, operative time, AAC score | OR 1.083 (95% CI 1.009–1.163); p = 0.026 |
| Knight et al. 2021 [28] | AL Major complications (C-D III–V) | Distal aortic calcification (None/Minor/Major) | Multivariable logistic regression; covariates: significant univariate predictors (sex, tumor site, surgical approach, smoking) | Any complications: OR 1.05 (95% CI 0.83–1.33); p = 0.70. Major complications: OR 1.32 (95% CI 0.90–1.94); p = 0.15 |
| Lee et al. 2020 [29] | Grade C AL | Aortoiliac calcification score ≥ 3 vs. <3 | Backward stepwise logistic regression; sex, ASA score, operative time ≥ 180 min, AICS ≥ 3 | OR 2.677 (95% CI 1.040–6.890); p = 0.041 |
| Namba et al. 2021 [31] | AL | Agatston score (high vs. low) | Multivariable logistic regression; male sex, lower rectal tumor, preop WBC, Agatston score | OR 6.09 (95% CI 1.48–25.0); p = 0.012 |
| Pochhammer et al. 2018 [33] | AL | Internal iliac artery calcification (volume score ≥ 30 vs. <30; ROC-derived) | Logistic regression; multivariable model retained internal iliac volume score + renal disease | Multivariable: calcification not independent (p = 0.08); renal disease p = 0.001 a. |
| Shen et al. 2019 [35] | AL | Aortic Calcification Index (ACI) > 4.8% | Multivariable logistic regression (after LASSO selection); CAD, TG > 1.7, glucose > 6.1, UICC stage III-IV, op time > 240 min, diverting ileostomy, ACI | OR 2.391 (95% CI 1.040–5.494); p = 0.040 |
| Turhan et al. 2025 [36] | AL | Aortic calcification >50% | Multivariable logistic regression; TNM stage, HTN, cardiovascular disease, neoadjuvant therapy | OR 10.38 (95% CI 1.243–92.118); p = 0.032 |
| Zhang et al. 2023 [37] | AL | SMA calcium volume score (continuous variable) | Multivariable logistic regression; tumor location, albumin, lymphocyte count, NLR, SMA calcium volume score | OR 6.810 (95% CI 1.870–24.801); p = 0.004 |
Appendix D. Sensitivity Analysis
| Moderator (Comparison) | k (Ref/Comp) | Test of Moderator | p-Value | Exp(β) (Ratio of ORs) (95% CI for OR Exp(β)) | Heterogeneity (Residual τ2; R2) |
|---|---|---|---|---|---|
| Cut point type (ROC-derived vs. non-ROC-derived) a | 7/4 | 5.42 | 0.044 | 2.32 (1.02–5.24) | 0.0217/82.5% |
| Exposure measurement (visual vs. quantitative) b | 5/6 | 2.07 | 0.184 | 0.61 (0.28–1.33) | 0.82/34% |
| Vascular territory (non-Aorta-only vs. Aorta-only) c | 6/5 | 0.026 | 0.875 | 1.06 (0.46–2.45) | 0.16/0% |
| NOS score (good vs. fair) | 6/5 | 0.0055 | 0.942 | 0.97 (0.42–2.25) | 0.16/0% |
| Outcome/ Exposure | Studies/ Participants | Primary Model: REML + Hartung–Knapp OR (95% CI) | Primary Model: DerSimonian–Laird OR (95% CI) | Effect of Estimator Choice |
|---|---|---|---|---|
| Major morbidity/AAC | k = 3/n = 841 | 1.99 (0.86–4.59) | 1.99 (1.27–3.11) | DerSimonian–Laird produced a narrower CI and changed nominal statistical significance |
| Any anastomotic leak/AAC | k = 11/n = 5005 | 3.08 (2.09–4.54) | 3.04 (2.15–4.31) | DerSimonian–Laird produced a narrower CI without changing statistical significance |
| Grade C anastomotic leak/AAC | k = 3/n = 3418 | 2.68 (1.03–6.97) | 2.68 (1.59–4.52) | DerSimonian–Laird produced a narrower CI without changing statistical significance |
| Moderator | Studies Included | Coding | Ratio of ORs | 95% CI | p-Value | Residual τ2 Residual I2 R2 | Interpretation |
|---|---|---|---|---|---|---|---|
| Sample size | 11 | Per 100-participant increase | 0.963 | 0.932–0.994 | 0.024 | 0.0187 6.65% 84.9% | Larger studies reported smaller effect estimates consistent with small study effects |
| Publication year | 11 | Per additional calendar year within the observed range | 0.94 | 0.801–1.103 | 0.402 | 0.1009 30.07% 18.7% | Publication year did not clearly explain heterogeneity |
| Geographic region | 11 | Asia vs. Europe | 1.488 | 0.668–3.310 | 0.290 | 0.1085 32.81% 12.7% | No clear regional effect modification was observed |
Appendix E. Exploratory Analysis of Mixed-Indication Abdominal Arterial Stenosis (CA, SMA and IMA) and AL

Appendix F. Domain-Level GRADE Evidence Decision Table
| Starting Certainty | Risk of Bias | Inconsistency | Indirectness | Imprecision | Publication Bias | Upgrade Factors | Final Certainty |
|---|---|---|---|---|---|---|---|
| Major morbidity/abdominal aortic calcification score k = 3/n = 841 OR 1.99, 95% CI 0.86–4.59 | |||||||
| Low | Serious; downgraded one level | Not serious | Not serious | Serious; downgraded one level | Not assessable | None | ⊕◯◯◯ Very low |
| Explanation: | Low starting certainty because the evidence was based on observational studies. ROB was judged serious because of retrospective designs and incomplete confounder control. Serious imprecision on account of the CI crossing the null and including both no important effect and clinically important harm. | ||||||
| Any anastomotic leak/abdominal aortic calcification score k = 11/n = 5005 OR 3.08, 95% CI 2.09–4.54 | |||||||
| Low | Serious; downgraded one level | Not serious | Not serious | Not serious | Suspected; concern but no formal downgrade | Large magnitude of effect; upgraded one level | ⊕⊕◯◯ Low |
| Explanation: | Low starting certainty because the evidence was based on observational studies. ROB was judged serious because most studies were retrospective and incompletely adjusted. Moderate heterogeneity but leave-one-out and sensitivity analyses preserved the direction of effect. Publication bias was suspected, but, since trim-and-fill did not eliminate the association, no additional formal downgrade was applied. Upgraded for large magnitude of effect. | ||||||
| Grade C anastomotic leak/abdominal aortic calcification score k = 3/n = 3418 OR 2.68, 95% CI 1.03–6.97 | |||||||
| Low | Serious; downgraded one level | Not serious | Not serious | Not serious | Not assessable | None | ⊕◯◯◯ Very low |
| Explanation: | Low starting certainty because the evidence was based on observational studies. Serious ROB because of retrospective and incompletely adjusted evidence. | ||||||
| Mortality/abdominal aortic calcification score k = 2/n = 184; additional deaths mentioned without exposure-stratified data Not pooled/not estimable | |||||||
| Low | Serious; downgraded one level | Not reliably assessable | Serious; downgraded one level | Serious; downgraded one level | Not assessable | None | ⊕◯◯◯ Very low |
| Explanation: | Effect estimate not pooled; downgraded for serious imprecision because mortality events were sparsely reported and some studies did not stratify mortality by exposure, hindering the extraction of stable mortality estimates. Downgraded for indirectness because post-operative and all-cause mortality were not separated. Only two AAC studies provided mortality information compatible with vascular exposure assessment, while additional studies reported deaths without stratification by calcium score category or without perioperative timing. | ||||||
| Any anastomotic leak/clinically significant IMA stenosis k = 1/n = 104 OR 13.68, 95% CI 1.69–110.4 | |||||||
| Low | Serious; downgraded one level | Not assessable; single-study | Not serious | Very serious; downgraded two levels | Not assessable | None | ⊕◯◯◯ Very low |
| Explanation: | Evidence came from a single CRC-specific matched case–control study. The association was large, but the estimate was very imprecise with a wide CI and few exposed events. Inconsistency and publication bias could not be assessed. No upgrade was applied because the evidence was single-study and the association was imprecise. Mixed-indication stenosis studies were not used to determine this certainty rating. | ||||||
References
- Bray, F.; Laversanne, M.; Sung, H.; Ferlay, J.; Siegel, R.L.; Soerjomataram, I.; Jemal, A. Global Cancer Statistics 2022: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J. Clin. 2024, 74, 229–263. [Google Scholar] [CrossRef]
- Argilés, G.; Tabernero, J.; Labianca, R.; Hochhauser, D.; Salazar, R.; Iveson, T.; Laurent-Puig, P.; Quirke, P.; Yoshino, T.; Taieb, J.; et al. Localised Colon Cancer: ESMO Clinical Practice Guidelines for Diagnosis, Treatment and Follow-Up. Ann. Oncol. 2020, 31, 1291–1305. [Google Scholar] [CrossRef] [PubMed]
- Hofheinz, R.-D.; Fokas, E.; Benhaim, L.; Price, T.J.; Arnold, D.; Beets-Tan, R.; Guren, M.G.; Hospers, G.A.P.; Lonardi, S.; Nagtegaal, I.D.; et al. Localised Rectal Cancer: ESMO Clinical Practice Guideline for Diagnosis, Treatment and Follow-Up. Ann. Oncol. 2025, 36, 1007–1024. [Google Scholar] [CrossRef] [PubMed]
- 2022 European Society of Coloproctology (ESCP) CORREA Collaborating Group. Towards Safer Colorectal Surgery Worldwide: Outcomes and Benchmarks from the ESCP CORREA 2022 Audit. Color. Dis. 2025, 27, e70281. [Google Scholar] [CrossRef] [PubMed]
- Dindo, D.; Demartines, N.; Clavien, P.-A. Classification of Surgical Complications: A New Proposal with Evaluation in a Cohort of 6336 Patients and Results of a Survey. Ann. Surg. 2004, 240, 205. [Google Scholar] [CrossRef]
- Rahbari, N.N.; Weitz, J.; Hohenberger, W.; Heald, R.J.; Moran, B.; Ulrich, A.; Holm, T.; Wong, W.D.; Tiret, E.; Moriya, Y.; et al. Definition and Grading of Anastomotic Leakage Following Anterior Resection of the Rectum: A Proposal by the International Study Group of Rectal Cancer. Surgery 2010, 147, 339–351. [Google Scholar] [CrossRef]
- Kube, R.; Mroczkowski, P.; Granowski, D.; Benedix, F.; Sahm, M.; Schmidt, U.; Gastinger, I.; Lippert, H. Anastomotic Leakage after Colon Cancer Surgery: A Predictor of Significant Morbidity and Hospital Mortality, and Diminished Tumour-Free Survival. Eur. J. Surg. Oncol. 2010, 36, 120–124. [Google Scholar] [CrossRef]
- Mirnezami, A.; Mirnezami, R.; Chandrakumaran, K.; Sasapu, K.; Sagar, P.; Finan, P. Increased Local Recurrence and Reduced Survival from Colorectal Cancer Following Anastomotic Leak: Systematic Review and Meta-Analysis. Ann. Surg. 2011, 253, 890. [Google Scholar] [CrossRef]
- Koedam, T.W.A.; Bootsma, B.T.; Deijen, C.L.; van de Brug, T.; Kazemier, G.; Cuesta, M.A.; Fürst, A.; Lacy, A.M.; Haglind, E.; Tuynman, J.B.; et al. Oncological Outcomes After Anastomotic Leakage After Surgery for Colon or Rectal Cancer: Increased Risk of Local Recurrence. Ann. Surg. 2022, 275, e420. [Google Scholar] [CrossRef]
- Nijssen, D.J.; Wienholts, K.; Postma, M.J.; Tuynman, J.; Bemelman, W.A.; Laméris, W.; Hompes, R. The Economic Impact of Anastomotic Leakage after Colorectal Surgery: A Systematic Review. Tech. Coloproctol. 2024, 28, 55. [Google Scholar] [CrossRef]
- Caruso, D.; Polici, M.; Bellini, D.; Laghi, A. ESR Essentials: Imaging in Colorectal Cancer-Practice Recommendations by ESGAR. Eur. Radiol. 2024, 34, 5903–5910. [Google Scholar] [CrossRef]
- Expert Panel on Gastrointestinal Imaging; Fowler, K.J.; Kaur, H.; Cash, B.D.; Feig, B.W.; Gage, K.L.; Garcia, E.M.; Hara, A.K.; Herman, J.M.; Kim, D.H.; et al. ACR Appropriateness Criteria® Pretreatment Staging of Colorectal Cancer. J. Am. Coll. Radiol. 2017, 14, S234–S244. [Google Scholar] [CrossRef] [PubMed]
- Staal, F.C.R.; van der Reijd, D.J.; Taghavi, M.; Lambregts, D.M.J.; Beets-Tan, R.G.H.; Maas, M. Radiomics for the Prediction of Treatment Outcome and Survival in Patients with Colorectal Cancer: A Systematic Review. Clin. Colorectal Cancer 2021, 20, 52–71. [Google Scholar] [CrossRef]
- Bates, D.; Pickhardt, P. CT-Derived Body Composition Assessment as a Prognostic Tool in Oncologic Patients: From Opportunistic Research to Artificial Intelligence–Based Clinical Implementation. Am. J. Roentgenol. 2022, 219, 671–680. [Google Scholar] [CrossRef]
- van Helsdingen, C.P.M.; van Wijlick, J.G.A.; de Vries, R.; Bouvy, N.D.; Leeflang, M.M.G.; Hemke, R.; Derikx, J.P.M. Association of Computed Tomography-Derived Body Composition and Complications after Colorectal Cancer Surgery: A Systematic Review and Meta-Analysis. J. Cachexia Sarcopenia Muscle 2024, 15, 2234–2269. [Google Scholar] [CrossRef] [PubMed]
- Imaoka, Y.; Ohira, M.; Akabane, M.; Sasaki, K.; Ohdan, H. Abdominal Aortic Calcification among Gastroenterological and Transplant Surgery. Ann. Gastroenterol. Surg. 2024, 8, 987–998. [Google Scholar] [CrossRef] [PubMed]
- Harmankaya, D.; Egberts, K.J.V.; Metz, F.M.; Vaassen, H.G.M.; Petersman, S.S.E.; Brusse-Keizer, M.; le Haen, P.; Bruno, M.J.; Geelkerken, B.H.; Noord, D. van Mesenteric Artery Stenosis Is a Risk Factor for Anastomotic Leakage in Colorectal Surgery. Eur. J. Vasc. Endovasc. Surg. 2025, 69, 628–637. [Google Scholar] [CrossRef]
- Kornmann, V.N.N.; van Werkum, M.H.; Bollen, T.L.; van Ramshorst, B.; Boerma, D. Compromised Visceral Circulation Does Not Affect the Outcome of Colorectal Surgery. Surg. Today 2014, 44, 1220–1226. [Google Scholar] [CrossRef]
- Arron, M.N.N.; Broek, R.P.G.T.; Adriaansens, C.M.E.M.; Bluiminck, S.; van Wely, B.J.; Ferenschild, F.T.J.; Smits, H.F.M.; van Goor, H.; de Wilt, J.H.W.; van Petersen, A.S. Mesenteric Occlusive Disease of the Inferior Mesenteric Artery Is Associated with Anastomotic Leak after Left-Sided Colon and Rectal Cancer Surgery: A Retrospective Cohort Study. Int. J. Colorectal Dis. 2022, 37, 631–638. [Google Scholar] [CrossRef]
- Diao, Y.-H.; Chen, J.; Liu, Y.; Peng, D.; Yang, D. Does Aortic Calcification Really Affect Anastomotic Leakage after Rectal Cancer Surgery? Medicine 2024, 103, e38860. [Google Scholar] [CrossRef]
- Eveno, C.; Latrasse, V.; Gayat, É.; Lo Dico, R.; Dohan, A.; Pocard, M. Colorectal Anastomotic Leakage Can Be Predicted by Abdominal Aortic Calcification on Preoperative CT Scans: A Pilot Study. J. Visc. Surg. 2016, 153, 253–257. [Google Scholar] [CrossRef] [PubMed]
- Roviello, F.; Andreucci, E.; Carbone, L.; Calomino, N.; Piccioni, S.; Bobbio, L.; Piagnerelli, R.; Fontani, A.; Marrelli, D. Preoperative Injection of Indocyanine Green Fluorescence at the Anorectal Junction Safely Identifies the Inferior Mesenteric Artery in a Prospective Case-Series Analysis of Colorectal Cancer Patients. Gastrointest. Disord. 2025, 7, 76. [Google Scholar] [CrossRef]
- Page, M.J.; Moher, D.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. PRISMA 2020 Explanation and Elaboration: Updated Guidance and Exemplars for Reporting Systematic Reviews. BMJ 2021, 372, n160. [Google Scholar] [CrossRef] [PubMed]
- Clark, J.M.; Sanders, S.; Carter, M.; Honeyman, D.; Cleo, G.; Auld, Y.; Booth, D.; Condron, P.; Dalais, C.; Bateup, S.; et al. Improving the Translation of Search Strategies Using the Polyglot Search Translator: A Randomized Controlled Trial. J. Med. Libr. Assoc. 2020, 108, 195–207. [Google Scholar] [CrossRef]
- Schünemann, H.J.; Mustafa, R.A.; Brozek, J.; Santesso, N.; Bossuyt, P.M.; Steingart, K.R.; Leeflang, M.; Lange, S.; Trenti, T.; Langendam, M.; et al. GRADE Guidelines: 22. The GRADE Approach for Tests and Strategies-from Test Accuracy to Patient-Important Outcomes and Recommendations. J. Clin. Epidemiol. 2019, 111, 69–82. [Google Scholar] [CrossRef]
- Chapter 10: Analysing Data and Undertaking Meta-Analyses|Cochrane. Available online: https://www.cochrane.org/authors/handbooks-and-manuals/handbook/current/chapter-10 (accessed on 19 April 2026).
- Gunji, T.; Tomita, K.; Koganezawa, I.; Nakagawa, M.; Yokozuka, K.; Ochiai, S.; Kobayashi, T.; Sano, T.; Tabuchi, S.; Chiba, N.; et al. Impact of Atherosclerosis on the Postoperative Complications of Colorectal Surgery in Older Patients with Colorectal Cancer. BMC Gastroenterol. 2022, 22, 519. [Google Scholar] [CrossRef]
- Knight, K.A.; Fei, C.H.; Boland, K.F.; Dolan, D.R.; Golder, A.M.; McMillan, D.C.; Horgan, P.G.; Black, D.H.; Park, J.H.; Roxburgh, C.S.D. Aortic Calcification Is Associated with Non-Infective Rather than Infective Postoperative Complications Following Colorectal Cancer Resection: An Observational Cohort Study. Eur. Radiol. 2021, 31, 4319–4329. [Google Scholar] [CrossRef]
- Lee, S.Y.; Yeom, S.-S.; Kim, C.H.; Kim, Y.J.; Kim, H.R. A New Aortoiliac Calcification Scoring System to Predict Grade C Anastomotic Leak Following Rectal Cancer Surgery. Tech. Coloproctol. 2020, 24, 843–849. [Google Scholar] [CrossRef]
- Morita, S.; Tsuruta, M.; Okabayashi, K.; Shigeta, K.; Seishima, R.; Monno, M.; Itano, O.; Kitagawa, Y. Evaluation of Abdominal Aortic Calcification by Plain CT Predicts Anastomotic Leakage in Laparoscopic Surgery for Colorectal Cancer. Jpn. J. Clin. Oncol. 2022, 52, 122–127. [Google Scholar] [CrossRef]
- Namba, Y.; Mukai, S.; Saito, Y.; Moriuchi, T.; Bekki, T.; Okimoto, S.; Oishi, K.; Fujisaki, S.; Takahashi, M.; Fukuda, T.; et al. Risk Factors for Anastomotic Leakage after Colorectal Surgery with Double-Staple Technique Anastomosis: Impact of the Agatston Score. J. Anus Rectum Colon 2021, 5, 181–187. [Google Scholar] [CrossRef]
- Norooz, M.T.; Moradi, H.; Safdarian, M.; Jahangiri, F.; Amoli, H.A. Does Calcium Score in Great Pelvic Vessels Predict Colorectal Anastomotic Leakage? A Prospective Study of One Hundred Anastomoses. Acta Gastro-Enterol. Belg. 2016, 79, 415–420. [Google Scholar]
- Pochhammer, J.; Tröster, F.; Blumenstock, G.; Closset, J.; Lang, S.; Weller, M.-P.; Schäffer, M. Calcification of the Iliac Arteries: A Marker for Leakage Risk in Rectal Anastomosis—A Blinded Clinical Trial. Int. J. Colorectal Dis. 2018, 33, 163–170. [Google Scholar] [CrossRef]
- Postaire, B.; Abet, E.; Montigny, P.; Vent, P.A. Does the Degree of Calcification of the Celiac Trunk and Superior Mesenteric Artery on Preoperative Computerized Tomography Predict the Risk of Anastomotic Leak after Right Colectomy? A Single Center Retrospective Study. J. Visc. Surg. 2019, 156, 191–195. [Google Scholar] [CrossRef]
- Shen, Z.; An, Y.; Shi, Y.; Yin, M.; Xie, Q.; Gao, Z.; Jiang, K.; Wang, S.; Ye, Y. The Aortic Calcification Index Is a Risk Factor Associated with Anastomotic Leakage after Anterior Resection of Rectal Cancer. Colorectal Dis. 2019, 21, 1397–1404. [Google Scholar] [CrossRef]
- Turhan, V.B.; Karacif, O.; Tutan, M.B.; Kartal, B.; Şahin, F.; Kendirci, M.; Alkurt, E.G. The Impact of Aortic Calcification on Surgical Outcomes in Colorectal Cancer Patients: A Retrospective Analysis Focused on Anastomotic Leakage. Medicina 2025, 61, 606. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Sun, W.; Wang, J.; Deng, Y.; Yan, Y.; Li, D.; Fu, W. A Nomogram to Predict the Risk of Colorectal Anastomotic Leakage Combining Inflammatory-Nutritional and Abdominal Aorta Calcium Index. Front. Surg. 2023, 9, 1008448. [Google Scholar] [CrossRef] [PubMed]
- Nepal, P.; Mori, S.; Kita, Y.; Tanabe, K.; Baba, K.; Sasaki, K.; Kurahara, H.; Arigami, T.; Ohtsuka, T. Anatomical Study of the Inferior Mesenteric Vein Using Three-Dimensional Computed Tomography Angiography in Laparoscopy-Assisted Surgery for Left-Sided Colorectal Cancer. Surg. Today 2021, 51, 1665–1670. [Google Scholar] [CrossRef]
- Nozawa, H.; Okamoto, K.; Kawai, K.; Sasaki, K.; Emoto, S.; Murono, K.; Sonoda, H.; Ishihara, S. Anatomical Features of Inferior Mesenteric and Left Colic Arteries and Surgery in Colorectal Cancer Patients with Persistent Descending Mesocolon. ANZ J. Surg. 2022, 92, 1760–1765. [Google Scholar] [CrossRef]
- Suh, J.W.; Park, J.; Lee, J.; Yang, I.J.; Ahn, H.-M.; Oh, H.-K.; Kim, D.-W.; Kang, S.-B. Clinical Impact of Inferior Mesenteric Vein Preservation during Left Hemicolectomy with Low Ligation of the Inferior Mesenteric Artery for Distal Transverse and Descending Colon Cancers: A Comparative Study Based on Computed Tomography. Front. Oncol. 2022, 12, 986516. [Google Scholar] [CrossRef]
- Wang, Y. Imaging diagnosis and imaging risk factors of anastomotic leakage after rectal cancer surgery. Chin. J. Gastrointest. Surg. 2018, 21, 404–408. [Google Scholar]
- Yotsov, T.; Karamanliev, M.; Maslyankov, S.; Iliev, S.; Ramadanov, N.; Dimitrov, D. Mesenteric Vascular Evaluation with Pre-Operative Multidetector Computed Tomographic Angiography and Intraoperative Indocyanine Green Angiography to Reduce Anastomotic Leaks after Minimally Invasive Surgery for Colorectal Cancer. JSLS 2022, 26, e2022.00022. [Google Scholar] [CrossRef]
- Kearns, E.C.; Moynihan, A.; Khan, M.F.; Lawler, L.; Cahill, R.A. Comparison and Impact of Preoperative 3D Virtual Vascular Modelling with Intraoperative Indocyanine Green Perfusion Angiography for Personalized Proximal Colon Cancer Surgery. Eur. J. Surg. Oncol. 2025, 51, 109581. [Google Scholar] [CrossRef] [PubMed]
- Huang, J.; Zhou, J.; Wan, Y.; Lin, Y.; Deng, Y.; Zhou, Z.; Qiu, J.; Wang, J.; Huang, M. Influences of inferior mesenteric artery types and Riolan artery arcade absence on the incidence of anastomotic leakage after laparoscopic resection of rectal cancer. Chin. J. Gastrointest. Surg. 2016, 19, 1113–1118. [Google Scholar]
- Kono, Y.; Yamamoto, M.; Yasui, C.; Ishiguro, R.; Yagyu, T.; Kihara, K.; Matsunaga, T.; Takano, S.; Tokuyasu, N.; Sakamoto, T.; et al. Preoperative High Agatston Score in Aorta Leads to Postoperative High-Output Ileostomy. J. Surg. Res. 2025, 314, 139–145. [Google Scholar] [CrossRef]
- Yu, A.; Li, Y.; Zhang, H.; Hu, G.; Zhao, Y.; Guo, J.; Wei, M.; Yu, W.; Yan, Z. Development and Validation of a Preoperative Nomogram for Predicting the Surgical Difficulty of Laparoscopic Colectomy for Right Colon Cancer: A Retrospective Analysis. Int. J. Surg. 2023, 109, 870–878. [Google Scholar] [CrossRef] [PubMed]
- Son, G.M.; Kim, T.U.; Park, B.-S.; Jung, H.J.; Lee, S.S.; Yoon, J.-U.; Lee, J.W. Colonic Hypoperfusion Following Ligation of the Inferior Mesenteric Artery in Rectosigmoid Colon Cancer Patients. Ann. Surg. Treat. Res. 2019, 97, 74–82. [Google Scholar] [CrossRef]
- Deguelte, S.; Besson, R.; Job, L.; Hoeffel, C.; Jolly, D.; Kianmanesh, R. Assessing Abdominal Aortic Calcifications before Performing Colocolic or Colorectal Anastomoses: A Case–Control Study. J. Res. Med. Sci. 2021, 26, 110. [Google Scholar] [CrossRef]
- Komen, N.; Klitsie, P.; Dijk, J.W.; Slieker, J.; Hermans, J.; Havenga, K.; Oudkerk, M.; Weyler, J.; Kleinrensink, G.-J.; Lange, J.F. Calcium Score: A New Risk Factor for Colorectal Anastomotic Leakage. Am. J. Surg. 2011, 201, 759–765. [Google Scholar] [CrossRef]
- Boersema, G.S.A.; Vakalopoulos, K.A.; Kock, M.C.J.M.; Van Ooijen, P.M.A.; Havenga, K.; Kleinrensink, G.J.; Jeekel, J.; Lange, J.F. Is Aortoiliac Calcification Linked to Colorectal Anastomotic Leakage? A Case-Control Study. Int. J. Surg. 2016, 25, 123–127. [Google Scholar] [CrossRef]
- Krzeszowiak, J.; Wróbel, S.; Del Carmen Yika, A.; Rudnicki, W.; Kenig, J. Evaluation of Individual and Composite CT-Based Markers in Frailty Diagnostics in Older Patients with Cancer Undergoing Major Abdominal Surgery. BMC Geriatr. 2025, 26, 44. [Google Scholar] [CrossRef] [PubMed]
- Chan, D.K.H.; Lee, S.K.F.; Ang, J.J. Indocyanine Green Fluorescence Angiography Decreases the Risk of Colorectal Anastomotic Leakage: Systematic Review and Meta-Analysis. Surgery 2020, 168, 1128–1137. [Google Scholar] [CrossRef]
- Watanabe, J.; Takemasa, I.; Kotake, M.; Noura, S.; Kimura, K.; Suwa, H.; Tei, M.; Takano, Y.; Munakata, K.; Matoba, S.; et al. Blood Perfusion Assessment by Indocyanine Green Fluorescence Imaging for Minimally Invasive Rectal Cancer Surgery (EssentiAL Trial): A Randomized Clinical Trial. Ann. Surg. 2023, 278, e688–e694. [Google Scholar] [CrossRef]
- Faber, R.A.; Meijer, R.P.J.; Droogh, D.H.M.; Jongbloed, J.J.; Bijlstra, O.D.; Boersma, F.; Braak, J.P.B.M.; Meershoek-Klein Kranenbarg, E.; Putter, H.; Holman, F.A.; et al. Indocyanine Green Near-Infrared Fluorescence Bowel Perfusion Assessment to Prevent Anastomotic Leakage in Minimally Invasive Colorectal Surgery (AVOID): A Multicentre, Randomised, Controlled, Phase 3 Trial. Lancet Gastroenterol. Hepatol. 2024, 9, 924–934. [Google Scholar] [CrossRef]
- Jafari, M.D.; Lee, K.H.; Halabi, W.J.; Mills, S.D.; Carmichael, J.C.; Stamos, M.J.; Pigazzi, A. The Use of Indocyanine Green Fluorescence to Assess Anastomotic Perfusion during Robotic Assisted Laparoscopic Rectal Surgery. Surg. Endosc. Interv. Tech. 2013, 27, 3003–3008. [Google Scholar] [CrossRef]
- Rinne, J.K.A.; Huhta, H.; Pinta, T.; Turunen, A.; Mattila, A.; Tahkola, K.; Helminen, O.; Ohtonen, P.; Rautio, T.; Kössi, J. Indocyanine Green Fluorescence Imaging in Prevention of Colorectal Anastomotic Leakage: A Randomized Clinical Trial. JAMA Surg. 2025, 160, 486–493. [Google Scholar] [CrossRef] [PubMed]
- Imaoka, K.; Shimomura, M.; Okuda, H.; Yano, T.; Akabane, S.; Ohira, M.; Imaoka, Y.; Mochizuki, T.; Hattori, M.; Ohdan, H. Abdominal Aortic Calcification as a Predictor of Incomplete Adjuvant Chemotherapy in Stage III Colorectal Cancer: A Retrospective Cohort Study. Cureus 2024, 16, e71288. [Google Scholar] [CrossRef] [PubMed]
- Tao, W.; Cheng, Y.-X.; Zou, Y.-Y.; Peng, D.; Zhang, W. Aorta Calcification Increases the Risk of Anastomotic Leakage After Gastrectomy in Gastric Cancer Patients. Cancer Manag. Res. 2021, 13, 3857–3865. [Google Scholar] [CrossRef] [PubMed]
- Kakizawa, N.; Noda, H.; Watanabe, F.; Ichida, K.; Suzuki, K.; Rikiyama, T. A High Abdominal Aortic Calcification Score on CT Is a Risk Factor for Postoperative Pancreatic Fistula in Elderly Patients Undergoing Pancreaticoduodenectomy. World J. Surg. 2018, 42, 1129–1137. [Google Scholar] [CrossRef]
- Ito, T.; Kuriyama, N.; Kaluba, B.; Teraoka, S.; Komatsubara, H.; Sakamoto, T.; Noguchi, D.; Hayasaki, A.; Fujii, T.; Iizawa, Y.; et al. Impact of Aortic Calcification at the Origin of Celiac Artery on Post-Operative Outcomes of Major Hepatectomy: A Significant Risk Factor for Posthepatectomy Liver Failure. Langenbecks Arch. Surg. 2025, 410, 129. [Google Scholar] [CrossRef]
- Hoek, V.T.; Edomskis, P.P.; Menon, A.G.; Kleinrensink, G.-J.; Lagarde, S.M.; Lange, J.F.; Wijnhoven, B.P.L. Arterial Calcification Is a Risk Factor for Anastomotic Leakage after Esophagectomy: A Systematic Review and Meta-Analysis. Eur. J. Surg. Oncol. 2020, 46, 1975–1988. [Google Scholar] [CrossRef]
- Aboumsallem, J.P.; Moslehi, J.; de Boer, R.A. Reverse Cardio-Oncology: Cancer Development in Patients with Cardiovascular Disease. J. Am. Heart Assoc. 2020, 9, e013754. [Google Scholar] [CrossRef]
- Bell, C.F.; Lei, X.; Haas, A.; Baylis, R.A.; Gao, H.; Luo, L.; Giordano, S.H.; Wehner, M.R.; Nead, K.T.; Leeper, N.J. Risk of Cancer After Diagnosis of Cardiovascular Disease. Cardio Oncol. 2023, 5, 431–440. [Google Scholar] [CrossRef]
- Paolisso, P.; Foà, A.; Magnani, I.; Bergamaschi, L.; Graziosi, M.; Angeli, F.; Chiti, C.; Fabrizio, M.; Rinaldi, A.; Stefanizzi, A.; et al. Development and Validation of a Diagnostic Echocardiographic Mass Score in the Approach to Cardiac Masses. Cardiovasc. Imaging 2022, 15, 2010–2012. [Google Scholar] [CrossRef]
- Lyon, A.R.; López-Fernández, T.; Couch, L.S.; Asteggiano, R.; Aznar, M.C.; Bergler-Klein, J.; Boriani, G.; Cardinale, D.; Cordoba, R.; Cosyns, B.; et al. 2022 ESC Guidelines on Cardio-Oncology Developed in Collaboration with the European Hematology Association (EHA), the European Society for Therapeutic Radiology and Oncology (ESTRO) and the International Cardio-Oncology Society (IC-OS): Developed by the Task Force on Cardio-Oncology of the European Society of Cardiology (ESC). Eur. Heart J. 2022, 43, 4229–4361. [Google Scholar] [CrossRef]
- Knight, K.A.; Horgan, P.G.; McMillan, D.C.; Roxburgh, C.S.D.; Park, J.H. The Relationship between Aortic Calcification and Anastomotic Leak Following Gastrointestinal Resection: A Systematic Review. Int. J. Surg. 2020, 73, 42–49. [Google Scholar] [CrossRef] [PubMed]
- Tong, L.; Xie, D.; Song, X.; Wu, X.; Wen, S.; Liu, A. Is Abdominal Vascular Calcification Score Valuable in Predicting the Occurrence of Colorectal Anastomotic Leakage? A Meta-Analysis. Int. J. Colorectal Dis. 2020, 35, 641–653. [Google Scholar] [CrossRef] [PubMed]
- Hoek, V.T.; Edomskis, P.P.; Menon, A.G.; Dwarkasing, R.S.; Kleinrensink, G.-J.; Jeekel, J.; Lange, J.F. Calcification of the Aorta-Iliac Trajectory as a Risk Factor for Anastomotic Leakage in Colorectal Surgery: Individual Patient Data Meta-Analysis and Systematic Review. Surg. Technol. Int. 2021, 39, 155–165. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.-R.; Liu, F.; Zhang, W.; Peng, D. The Aortic Calcification Is a Risk Factor for Colorectal Anastomotic Leakage. Updat. Surg. 2023, 75, 1857–1865. [Google Scholar] [CrossRef]
- Khan, S.M.; Wells, C.; Christou, N.; Tan, C.Y.; Mathur, P.; El-Hussuna, A. Preoperative Assessment of Blood Supply and Its Role in Predicting Anastomotic Leak. Surgery 2023, 174, 46–51. [Google Scholar] [CrossRef]
- Berger, J.S.; Lyu, C.; Iturrate, E.; Westerhoff, M.; Gyftopoulos, S.; Dane, B.; Zhong, J.; Recht, M.; Bredella, M.A. Opportunistic Assessment of Abdominal Aortic Calcification Using Artificial Intelligence (AI) Predicts Coronary Artery Disease and Cardiovascular Events. Am. Heart J. 2025, 288, 122–130. [Google Scholar] [CrossRef]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. BMJ 2021, 372, n71. [Google Scholar] [CrossRef]




| Study | Design and Setting | Population | Tumor Location | Primary Outcome - Incidence | Secondary Outcomes a | Risk of Bias (Newcastle–Ottawa Score) |
|---|---|---|---|---|---|---|
| Arron et al. 2022 [19] | RC and nested CC Multi-center Netherlands 2009–2018 | n = 1273 Mean age = 67 Gender not reported | Left colon Rectum | Any AL—6% of subjects (52 cases vs. 52 controls) | - | 7/9 |
| Diao et al. 2024 [20] | RC Single-center China 2011–2020 | n = 2412 Mean age = 62 62.4% M | Colon Rectum | Any AL—4% of subjects | Grade C AL | 8/9 |
| Eveno et al. 2016 [21] | RC Single-center France 2007–2010 | n = 60 Mean age = 70.5 50% M/F | Left colon Rectum | Any AL—21.6% of subjects | Major complications (C-D grade ≥III) Mortality | 7/9 |
| Gunji et al. 2022 [27] | PC Single-center Japan 2017–2020 | n = 124 Mean age = 75.3 61.3% M | Colon Rectum | Major complications (C-D grade ≥ III)—11.3% of subjects | Major complications (C-D grade ≥III) Mortality | 6/9 |
| Knight et al. 2021 [28] | RC Single-center United Kingdom 2008–2016 | n = 657 Mean age = 68 56% M | Colon Rectum | Infective (28%) vs. non-infective (20%) complications | All complications Major complications (C-D grade ≥III) Any AL (5.3%) | 8/9 |
| Lee et al. 2020 [29] | RC Single-center South Korea 2013–2015 | n = 583 Mean age = 66 68.8% M | Rectum | Grade C AL—5.1% of subjects | Any AL (6.3%) | 6/9 |
| Morita et al. 2022 [30] | RC Single-center Japan 2014–2016 | n = 98 Mean age = 69 56% M | Colon Rectum | Any AL—16.3% of subjects | - | 6/9 |
| Namba et al. 2021 [31] | RC Single-center Japan 2015–2020 | n = 147 Mean age = 73.4 55.5% M | Colon Rectum | Any AL—8.16% of subjects | - | 7/9 |
| Norooz et al. 2016 [32] | PC Single-center Iran 2012–2014 | n = 100 Mean age = 63.7 55% M | Colon Rectum | Any AL—20% of subjects | - | 5/9 |
| Pochhammer et al. 2018 [33] | PC Single-center Germany 2014–2016 | n = 139 Mean age = 73 52.5% M | Colon Rectum | Any AL—11% of subjects | - | 7/9 |
| Postaire et al. 2018 [34] | RC Single-center France 2011–2016 | n = 253 Mean age = 72 Gender not reported | Right colon | Any AL—9.1% of subjects | - | 6/9 |
| Shen et al. 2019 [35] | RC Single-center China 2009–2017 | n = 423 Mean age = 64 62.4% M | Rectum | Any AL—7.8% of subjects | Grade C AL | 6/9 |
| Turhan et al. 2025 [36] | RC Single-center Turkey 2020–2023 | n = 151 Mean age = 65.4 61.6% M | Colon Rectum | Any AL—5.96% of subjects | - | 7/9 |
| Zhang et al. 2023 [37] | RC Single-center China 2018–2021 | n = 292 Mean age = 67 57.7% M | Colon Rectum | AL—12.67% of subjects | - | 6/9 |
| Outcome (Exposure) | Assumed Risk Without Substantial Vascular Disease | Assumed Risk with Substantial Vascular Disease Present | Relative Effect (OR, 95% CI) | Participants (Studies) | Certainty (GRADE) | Reasoning |
|---|---|---|---|---|---|---|
| Major morbidity (AAC score) | ~89 per 1000 | 177 per 1000 (95% CI 76.54–408.51) | 1.99 (95% CI 0.86–4.59) | 841 (k = 3) | ⊕◯◯◯Very low | Downgraded for ROB and imprecision a. |
| Any anastomotic leak (AAC score) | ~40 per 1000 | 123 per 1000 (95% CI 83.6–181.6) | 3.08 (95% CI 2.09–4.54) | 5005 (k = 11) | ⊕⊕◯◯Low | Downgraded for ROB a. Publication bias was suspected and retained as a residual concern b. Sensitivity analyses informed the decision not to downgrade further for inconsistency or imprecision. Upgraded for large magnitude of effect c. |
| Grade C anastomotic leak (AAC score) | ~14 per 1000 | 37 per 1000 (95% CI 14.42–97.58) | 2.68 (95% CI 1.03–6.97) | 3418 (k = 3) | ⊕◯◯◯Very low | Downgraded for ROB a. |
| Mortality (AAC score) d | Not estimable | Not estimable | Not pooled | Vascular exposure mortality data available from two AAC studies, n = 184 | ⊕◯◯◯Very low | Downgraded for ROB, indirectness from inconsistent reporting and very serious imprecision. Deaths were rare and mortality not consistently stratified by CT vascular exposure. |
| Any anastomotic leak (clinically significant IMA stenosis) e | Not estimable f | Not estimable | 13.68 (95% CI 1.69–110.4) | 104 (k = 1) | ⊕◯◯◯Very low | Evidence came from one CRC-specific case–control analysis. Downgraded for ROB and very serious imprecision. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Nicoară, M.-I.; Socaciu, M.A.; Ursu, D.; Ciocan, A.; Al Hajjar, N. Preoperative CT Evaluation of Abdominal Vasculature and the Risk of Surgical Complications in Colorectal Cancer Resection with Anastomosis: A Systematic Review and Meta-Analysis. Diagnostics 2026, 16, 1449. https://doi.org/10.3390/diagnostics16101449
Nicoară M-I, Socaciu MA, Ursu D, Ciocan A, Al Hajjar N. Preoperative CT Evaluation of Abdominal Vasculature and the Risk of Surgical Complications in Colorectal Cancer Resection with Anastomosis: A Systematic Review and Meta-Analysis. Diagnostics. 2026; 16(10):1449. https://doi.org/10.3390/diagnostics16101449
Chicago/Turabian StyleNicoară, Mihnea-Ionuț, Mihai Adrian Socaciu, Diana Ursu, Andra Ciocan, and Nadim Al Hajjar. 2026. "Preoperative CT Evaluation of Abdominal Vasculature and the Risk of Surgical Complications in Colorectal Cancer Resection with Anastomosis: A Systematic Review and Meta-Analysis" Diagnostics 16, no. 10: 1449. https://doi.org/10.3390/diagnostics16101449
APA StyleNicoară, M.-I., Socaciu, M. A., Ursu, D., Ciocan, A., & Al Hajjar, N. (2026). Preoperative CT Evaluation of Abdominal Vasculature and the Risk of Surgical Complications in Colorectal Cancer Resection with Anastomosis: A Systematic Review and Meta-Analysis. Diagnostics, 16(10), 1449. https://doi.org/10.3390/diagnostics16101449





