Ultrasound-Based Techniques for Visualization of Dermal Microvasculature: A Scoping Review
Abstract
1. Introduction
2. Materials and Methods
2.1. Protocol Registration and Search Strategy
2.2. Study Selection and Data Extraction
2.3. Deviations from Protocol
3. Results
3.1. Study Characteristics
3.1.1. Ultrasound Techniques
3.1.2. Clinical and Pathological Focus Areas
3.1.3. Key Findings
3.1.4. Reporting Limitations
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| AP | Angiography Mode (advanced Doppler angiographic mode) |
| AR-PAM | Acoustic-Resolution Photoacoustic Microscopy |
| BCC | Basal Cell Carcinoma |
| B-mode | Brightness Mode ultrasound |
| CFU | Center for Fast Ultrasound Imaging |
| CDI | Color Doppler Imaging |
| CDFI | Color Doppler Flow Imaging |
| Doppler US | Doppler Ultrasound |
| Hb | Deoxygenated Hemoglobin |
| HbO2 | Oxygenated Hemoglobin |
| HbT | Total Hemoglobin |
| HFUS | High-Frequency Ultrasound |
| MeSH | Medical Subject Headings |
| MIP | Maximum Intensity Projection |
| MSOT | Multispectral Optoacoustic Tomography |
| mSO2 | Optoacoustic-derived oxygen saturation |
| MV-Flow | Microvascular Flow imaging (advanced Doppler technique) |
| MicroV | Microvascular Imaging |
| OR-PAM | Optical-Resolution Photoacoustic Microscopy |
| OAM | Optoacoustic Mesoscopy |
| OCT | Optical Coherence Tomography |
| OSF | Open Science Framework |
| PA | Photoacoustic |
| PAI | Photoacoustic Imaging |
| PAM | Photoacoustic Microscopy |
| PAT | Photoacoustic Tomography |
| PAUS | Photoacoustic and Ultrasound imaging |
| PDI | Power Doppler Imaging |
| PRISMA-ScR | Preferred Reporting Items for Systematic Reviews and Meta-Analyses extension for Scoping Reviews |
| ROI | Region of Interest |
| SCC | Squamous Cell Carcinoma |
| SVD | Singular Value Decomposition |
| SMI | Superb Microvascular Imaging |
| SURE | Super-Resolution Ultrasound using Erythrocytes |
| SWE | Shear Wave Elastography |
| US | Ultrasound |
| VHFUS | Very High-Frequency Ultrasound |
| Vs | Vascular density parameter |
Appendix A. Search Strategy
| Set | Search Statement |
| 1. | exp microvessels/ |
| 2. | microcirculation/or microvascular density/ |
| 3. | skin/or dermis/or epidermis/ |
| 4. | (skin* or derm* or cutan* or cutis* or mole or moles or nevi or naevi or nevus or melanoma*).ab,kf,ti. |
| 5. | (“ultrasound localization microscopy” or ULM or “super-resolution ultrasound imaging*” or “SRI” or “SURE” or “super-resolution ultrasound localization*” or “localization-based ultrasound imaging” or “microscale ultrasound imaging” or “ultrasound microvascular imaging*” or “super-resolution vascular ultrasound” or “high-resolution ultrasound microscopy” or “microbubble-enhanced ultrasound imaging*” or “ultrasound molecular imaging*” or “microbubble ultrasound localization microscopy*” or “vascular ultrasound localization imaging” or “super-resolution microvessel imaging” or “contrast-enhanced ultrasound*” or “CEUS” or “super-resolved ultrasound*” or “high-resolution microvascular imaging*” or “advanced microvascular imaging” or “superb microvascular imaging” or “SMI”).ab,kf,ti. |
| 6. | (microvascula* or microvessel*).ab,kf,ti. |
| 7. | (skin* or dermis or cutan* or cutis*).ab,kf,ti. |
| 8. | ultrasonography/or elasticity imaging techniques/or microscopy, acoustic/or ultrasonography, Doppler/ |
| 9. | (“Ultrafast imaging” or ultrasound or “Plane-wave imaging” or “Fast ultrasound imaging”).ab,kf,ti. |
| 10. | 1 or 2 or 6 |
| 11. | 3 or 4 or 7 |
| 12. | 5 or 8 or 9 |
| 13. | 10 and 11 and 12 |
| 1. | (ultrasonography or elasticity imaging or microscopy, acoustic or ultrasonography, doppler).ab,fx,hw,kf,ot,ti. |
| 2. | (ultrasound or “ultrafast imaging” or “plane-wave imaging” or “fast ultrasound imaging”).ab,fx,hw,kf,ot,ti. |
| 3. | (“ultrasound localization microscopy” or ULM or “super-resolution ultrasound imaging*” or SRI or SURE or “localization-based ultrasound imaging” or “microscale ultrasound imaging” or “ultrasound microvascular imaging*” or “super-resolution vascular ultrasound” or “high-resolution ultrasound microscopy” or “microbubble-enhanced ultrasound imaging*” or “ultrasound molecular imaging*” or “microbubble ultrasound localization microscopy*” or “vascular ultrasound localization imaging” or “super-resolution microvessel imaging” or “contrast-enhanced ultrasound*” or CEUS or “super-resolved” or “advanced microvascular imaging” or “superb microvascular imaging” or SMI).ab,fx,hw,kf,ot,ti. |
| 4. | (skin* or derm* or cutan* or cutis* or mole or moles or nevi or naevi or nevus or melanoma*).ab,fx,hw,kf,ot,ti. |
| 5. | (microcirculation or “microvascular density”).ab,fx,hw,kf,ot,ti. |
| 6. | (microvessels or microvascula* or microvessel*).ab,fx,hw,kf,ot,ti. |
| 7. | exp microvascular density/ |
| 8. | skin blood flow/or skin carcinogenesis/or skin carcinoma/or skin blood vessel/or skin capillary/or skin/ |
| 9. | microcirculation/ |
| 10. | 5 or 6 or 7 or 9 |
| 11. | 4 or 8 |
| 12. | 1 or 2 or 3 |
| 13. | 10 and 11 and 12 |
Appendix B
| Study No. | Title | Author | Year of Publication | Country in Which the Study Conducted | Study Design | Population Description | Sample Size | Method of Intervention | Health Focus (e.g., Melanoma) | Conclusions |
| 1 | Combined ultrasound and photoacoustic system for real-time high-contrast imaging using a linear array transducer | Jaeger et al. [8] | 2017 | Switzerland | Technology development and experimental proof-of-concept | Healthy volunteer | 1 | Photoacoustic-based | Normal skin | A combined US/PA linear-array system visualized microvascular structures in real time with high contrast. Demonstrates technical feasibility; clinical applications were proposed but not evaluated. |
| 2 | Photoacoustic and ultrasound [PAUS) dermoscope with high sensitivity and penetration depth by using a bimorph transducer | Wang et al. [15] | 2020 | China | Non-randomized experimental study | Healthy volunteer | 1 | Photoacoustic-based | General dermal imaging | A PA/US dermoscope prototype improved sensitivity and penetration for vascular imaging in skin. Microvascular visualization is technically promising, but clinical performance was not evaluated. |
| 3 | Handheld optical-resolution photoacoustic microscopy | Lin et al. [16] | 2017 | United States | Non-randomized experimental study | Healthy volunteer | 1 | Photoacoustic-based | General dermal microvasculature. Mole, cuticle, and normal skin | A handheld OR-PAM system visualized superficial microvasculature with high resolution in a human case. Feasibility is strong, but clinical usefulness and diagnostic performance remain untested. |
| 4 | In vivo photoacoustic microscopy of human cutaneous microvasculature and a nevus | Favazza et al. [17] | 2011 | United States | Non-randomized experimental study | Healthy volunteers | 3 | Photoacoustic-based | Pigmented lesion (nevus) and melanoma | Photoacoustic microscopy visualized detailed dermal microvasculature in normal skin and a nevus. Strong microvascular depiction, but diagnostic performance for lesions (including melanoma) was not assessed. Histopathologic confirmation. |
| 5 | Full-view in vivo skin and blood vessels profile segmentation in photoacoustic imaging based on deep learning | Ly et al. [18] | 2022 | Republic of Korea | Non-randomized experimental study | Healthy volunteer | 1 | Photoacoustic-based | Normal skin and subcutaneous vascular structures | Photoacoustic imaging visualized skin/subcutaneous vasculature and deep-learning improved vessel profile extraction. Useful for analysis automation, but clinical benefit and generalizability are not yet demonstrated. |
| 6 | Linear-array-based photoacoustic imaging of human microcirculation with a range of high frequency transducer probes | Zafar et al. [19] | 2015 | Ireland | Non-randomized experimental study | Healthy volunteer | Not clearly reported | Photoacoustic-based | Normal skin | Linear-array photoacoustic imaging produced 3D microcirculation images in human skin, with performance dependent on probe choice. Feasibility is shown, but clinical utility and reproducibility were not tested. |
| 7 | Photoacoustic imaging of the human forearm using 40 MHz linear-array transducer | Zafar et al. [20] | 2014 | Ireland | Case report | Healthy volunteer | 1 | Photoacoustic-based | Normal skin | A 40 MHz linear-array photoacoustic setup visualized superficial skin microvasculature in a healthy volunteer. Feasibility only; no clinical validation or comparative performance assessment. |
| 8 | Fast raster-scan optoacoustic mesoscopy enables assessment of human melanoma microvasculature in vivo | He et al. [21] | 2022 | Germany | Case control study | Healthy volunteers and patients | 22 | Photoacoustic-based | Normal skin, psoriasis, melanomas, and benign nevi | Optoacoustic mesoscopy visualized and quantified microvascular differences between melanoma and benign pigmented lesions. Potentially useful as an adjunct for differentiation, but requires validation in larger, clinically integrated studies. |
| 9 | Implications of ultrasound frequency in optoacoustic mesoscopy of the skin | Schwarz et al. [22] | 2015 | Germany | Non-randomized experimental study | Healthy volunteer | 1 | Photoacoustic-based | Normal skin (glabrous and hairy skin) and benign nevus | Frequency selection strongly affected depiction of dermal microvasculature and superficial layers in optoacoustic mesoscopy. Useful as methodological guidance for optimizing microvascular visibility, not as clinical evidence. |
| 10 | Design of a high-frequency array based photoacoustic microscopy system for micro-vascular imaging | Bitton et al. [23] | 2007 | United States | Non-randomized experimental study | Healthy volunteer | 1 | Photoacoustic-based | Normal skin | An early high-frequency array PA system imaged microvessels in human skin (reported <100 µm). Demonstrates feasibility only and has minimal clinical applicability without further validation. |
| 11 | A fast all-optical 3D photoacoustic scanner for clinical vascular imaging | Huynh et al. [24] | 2024 | UK | Non-randomized experimental study | Healthy volunteers and patients | Not clearly reported | Photoacoustic-based | Skin inflammation, diabetes, rheumatoid arthritis | The system enabled rapid 3D visualization of vessels (including deeper structures) with reduced motion artifacts. Microvasculature is clearly visible and clinically promising, but current evidence remains exploratory with limited validation. |
| 12 | Photoacoustic tomography of fingerprint and underlying vasculature for improved biometric identification | Zheng et al. [26] | 2021 | United States | Non-randomized experimental study | Healthy volunteer | 1 | Photoacoustic-based | Fingerprint | Photoacoustic imaging visualized subsurface vasculature beneath fingerprint patterns to support biometric identification. Vascular structures are visible, but this is not a dermatologic diagnostic application. |
| 13 | Miniaturized photoacoustic probe for in vivo imaging of subcutaneous microvessels within human skin | Zhang et al. [27] | 2019 | China | Non-randomized experimental study | Healthy volunteers | Not reported | Photoacoustic-based | General microvascular visualization in skin | A miniaturized PA probe visualized cutaneous/subcutaneous microvessels with micrometer-scale resolution and millimeter penetration (lateral resolution of ~8.9 µm and imaging depth of ~2.4 mm). Strong technical feasibility for microvessel depiction, but no clinical or pathological validation. |
| 14 | In vivo photoacoustic microscopy of human cuticle microvasculature with single-cell resolution | Hsu et al. [29] | 2016 | United States | Non-randomized experimental study | Healthy volunteers | 9 | Photoacoustic-based | Normal skin (microvasculature) | OR-PAM visualized capillary loops and even individual RBC-related signals, enabling functional hemodynamic readouts. Highly effective for microvascular depiction, but clinical utility in disease remains untested. |
| 15 | Visualization of vasculature using a hand-held photoacoustic probe: phantom and in vivo validation | Heres et al. [30] | 2017 | Netherlands | Non-randomized experimental study | Healthy volunteers | 4 | Photoacoustic-based | General vascular visualization | A handheld photoacoustic probe visualized superficial dermal vasculature in vivo in real time. Demonstrates feasibility for bedside vascular imaging, but clinical applicability and quantitative validation remain limited. |
| 16 | Noninvasive and high-resolving photoacoustic dermoscopy of human skin | Xu et al. [31] | 2016 | China | Non-randomized experimental study | Not reported | Not reported | Photoacoustic-based | Normal skin | Photoacoustic imaging differentiated microvascular patterns between port-wine stain lesions and normal skin (e.g., larger/deeper vessels in lesions). Potentially useful for lesion characterization/monitoring, but not validated against histology. |
| 17 | Combined multi-modal photoacoustic tomography, optical coherence tomography [OCT), and OCT angiography system with an articulated probe for in vivo human skin structure and vasculature imaging | Liu et al. [32] | 2016 | Austria | Non-randomized experimental study | Healthy volunteers | Not reported | Photoacoustic-based | Normal skin, nevus | Multimodal imaging visualized dermal microvasculature and skin structure, showing complementary vascular/morphologic information. Feasibility is demonstrated, but clinical performance and diagnostic value were not evaluated. |
| 18 | Optoacoustic imaging offers new insights into in vivo human skin vascular physiology | Rodrigues et al. [33] | 2022 | Portugal | Non-randomized experimental study | Healthy volunteers | 10 | Photoacoustic-based | Normal skin with reactive hyperemia after inflation of a cuff to occlude the brachial artery | Optoacoustic imaging captured depth-dependent microvascular perfusion dynamics during occlusion and reactive hyperemia. Useful for functional/physiological assessment, but tested only in healthy volunteers and without outcome validation. |
| 19 | Ultrasound array photoacoustic microscopy for dynamic in vivo 3D imaging | Song et al. [34] | 2010 | United States | Non-randomized experimental study | Healthy volunteers | 1 | Photoacoustic-based | Normal skin | Photoacoustic microscopy visualized superficial dermal microvasculature and demonstrated pulsatile signal dynamics. Primarily a feasibility demonstration; diagnostic or clinical usefulness was not established. |
| 20 | High-frequency ultrasound (22MHz) in the evaluation of malignant cutaneous neoplasms | Barcaui et al. [2] | 2014 | Brazil | Text and opinion | BCC, SCC, and melanoma | Not reported | HFUS | Malignant skin neoplasms (BCC, SCC, and melanoma) | HFUS with Doppler is described as providing structural and vascular information that may assist preoperative planning and lesion characterization. Evidence is narrative without quantified microvascular performance or diagnostic accuracy. |
| 21 | Region-based SVD processing of high-frequency ultrafast ultrasound to visualize cutaneous vascular networks | Bhatti et al. [6] | 2023 | Japan | Non-randomized experimental study | Healthy volunteers | Not clearly reported | HFUS | Normal skin | Region-based SVD processing improved visualization of dermal microvessels and subcutaneous vessels compared with standard processing. Promising for enhancing microvascular visibility, but lacks patient data and reference-standard validation. |
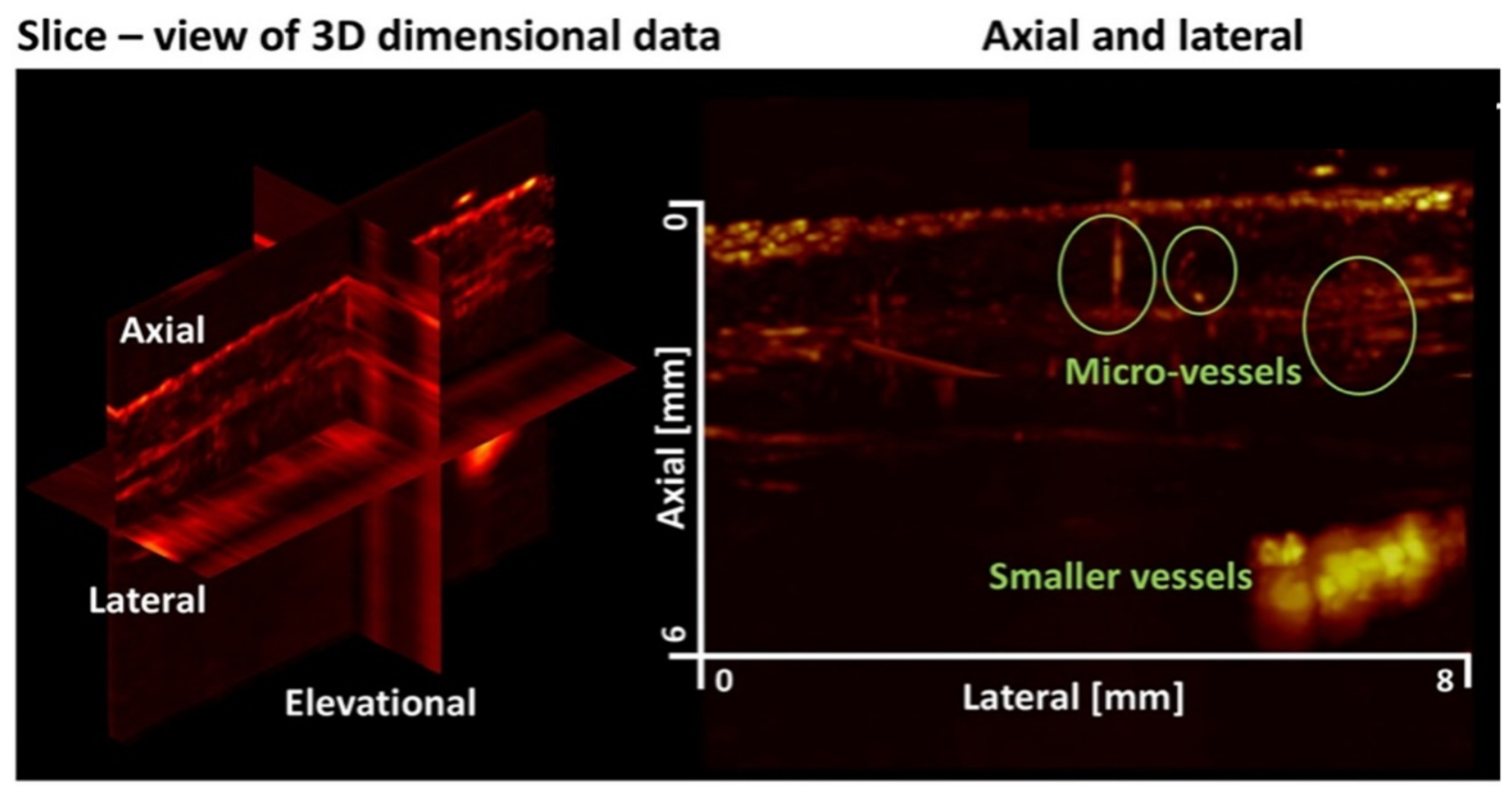
| 22 | Volumetric visualization of the dermal vasculature with signal and image-based feature extraction on a high-frequency ultrafast ultrasound dataset | Bhatti et al. [37] | 2025 | Japan | Non-randomized experimental study | Healthy volunteer | 1 | HFUS | Normal skin | This methodological study used high-frequency ultrafast ultrasound combined with region-based singular value decomposition filtering to visualize dermal and subdermal microvasculature in healthy human skin. The approach enabled 3D visualization of vascular networks and extraction of vessel-related features. |
| 23 | High-frequency power Doppler ultrasonography in predicting burn depth: a preliminary case report | Saijo et al. [38] | 2024 | Japan | Case report | Patients | 2 | HFUS | Deep fire burns and burn depth assessment | Presence/absence of dermal perfusion on high-frequency power Doppler aligned with tissue viability and burn depth. Potentially useful as an adjunct bedside tool, but evidence is limited to small case data without reference validation. |
| 24 | In vivo microcirculation mapping of human skin keloid by 40-MHz ultrafast ultrasound imaging | Chen et al. [39] | 2017 | Taiwan | Non-randomized experimental study | Volunteer with a keloid | 1 | HFUS | Keloid (benign dermal fibrosis and microvascular architecture) | Ultrafast Doppler with SVD filtering detected microvascular flow within a keloid scar, demonstrating feasibility of allowing microcirculation mapping. Clinical usefulness is exploratory and needs reproducibility and reference-standard validation. |
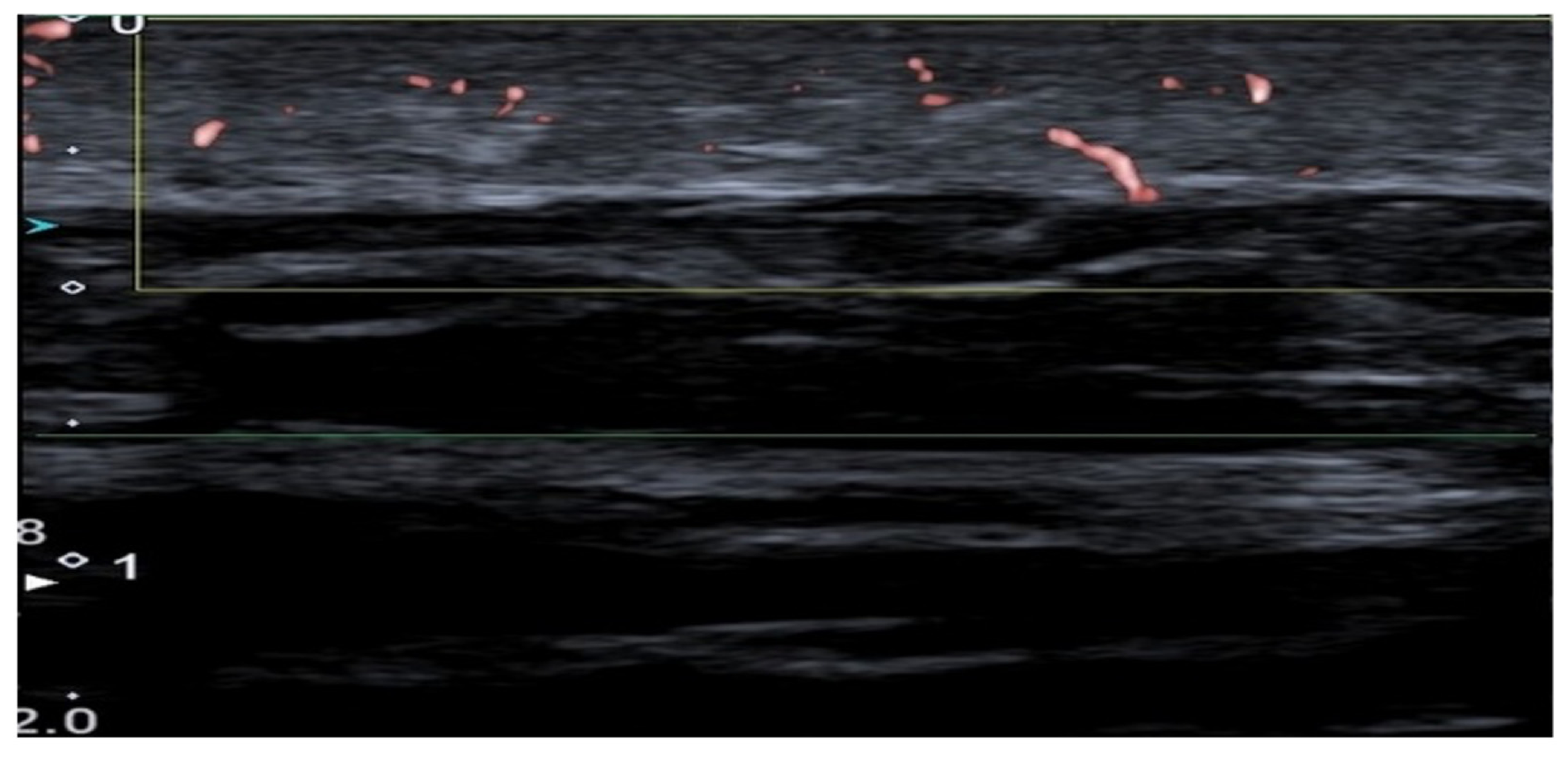
| 25 | Seeing the unseen with superb microvascular imaging: Ultrasound depiction of normal dermis vessels | Corvino et al. [10] | 2022 | Italy | Non-randomized experimental study | Healthy volunteers | 30 | SMI | Normal skin | SMI detected low-flow dermal vessels in all assessed body areas and yielded higher vascular index scores than power Doppler. Useful for demonstrating normal dermal microvasculature and establishing reference vascularity, but evaluated only in healthy subjects. |
| 26 | Assessing scleroderma patterns with superb microvascular imaging: is it possible? New prospects for ultrasound | Jasionyte et al. [35] | 2023 | Lithuania | Case series | Healthy volunteer and patients | 4 | SMI | Systemic sclerosis (SSc) and Raynaud’s phenomenon | SMI visualized altered nailfold microvascular patterns and avascular areas consistent with systemic sclerosis. Potentially useful as a supplementary real-time tool, but evidence is limited to a very small case series. |
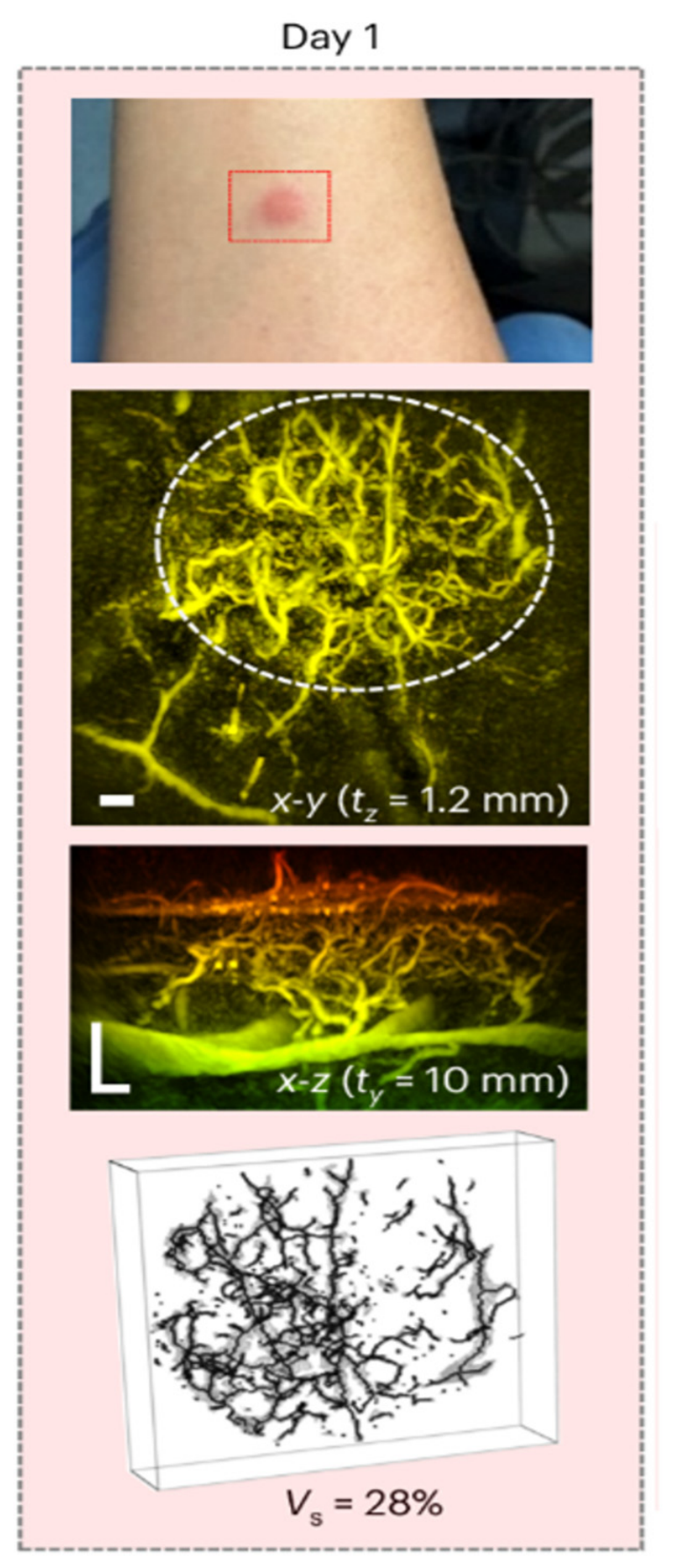
| 27 | Human mole contrast-free microvascular imaging using erythrocytes | Salari et al. [12] | 2025 | Denmark | Case report | Healthy volunteer | 1 | Super-resolution ultrasound imaging using erythrocytes (SURE) | Moles | They successfully visualized the microvasculature in a human mole and estimated the velocity map and velocity profile of vessels as small as 65 µm in diameter, with flow velocities as low as 3.2 mm/s. |
| 28 | Dermatology ultrasound imaging technique, tips and tricks, high-resolution anatomy | Catalano et al. [4] | 2020 | Italy and Chile | Systematic review | Not reported (review) | Not reported | HFUS, VHFUS, CDI, elastography | The skin in general | Educational review illustrating dermatologic ultrasound techniques, including examples of microvascular imaging (e.g., SMI). Useful for implementation and interpretation guidance, but does not provide new microvascular outcome evidence. |
| 29 | Use of new microcirculation software allows the demonstration of dermis vascularization | Catalano et al. [10] | 2023 | Italy | Non-randomized experimental study | Healthy volunteers | 50 | SMI and MV-Flow (HFUS, CDI, PDI, and advanced Doppler techniques) | Normal skin (dermal microvascularization) | Advanced Doppler (SMI/MV-Flow) visualized dermal vessels more consistently than power Doppler, which often showed no flow. Clinically useful for improving low-flow dermal microvascular detection, though site- and artifact-sensitive. |
| 30 | Shear wave elastography and microvascular ultrasound in response evaluation to calcipotriol + betamethasone foam in plaque psoriasis | Guazzaroni et al. [11] | 2021 | Italy | Cohort study | Patients | 26 | MicroV, SWE | Mild to moderate plaque psoriasis, PASI ≥ 4 | Microvascular ultrasound showed reduced vascularity in responding plaques and may detect early treatment response alongside SWE. Potentially useful for non-invasive monitoring, but conclusions are limited by small samples and minimal imaging follow-up data. |
| 31 | Three-dimensional multistructural quantitative photoacoustic and US imaging of human feet in vivo | Choi et al. [25] | 2022 | South Korea | Non-randomized experimental study | Healthy volunteers | 6 | Photoacoustic imaging, US, 3D image reconstruction | Normal skin | 3D PA/US provided microvascular images and quantitative parameters (e.g., vessel density/depth) in the foot, including responses to venous occlusion. Useful for peripheral microcirculation assessment, but tested only in healthy subjects without gold-standard comparison. |
| 32 | Visualization of skin morphology and microcirculation with high-frequency ultrasound and dual-wavelength photoacoustic microscope | Saijo et al. [28] | 2019 | Japan | Non-randomized experimental study | Healthy volunteers | 40 | HFUS and photoacoustic imaging | Normal skin (skin aging and dermal microstructure) | The integrated HFUS/PA system visualized skin structure and microcirculation in 3D and supported functional assessment (e.g., oxygenation-related imaging). Promising for microvascular assessment, but clinical validation metrics are missing. |
| 33 | Multimodal ultrasound assessment for monitoring keloid severity and treatment response | Zhou et al. [36] | 2025 | China | Cohort study | Patients | 31 | HFUS, SWE, AP, CDFI, PDI | Keloid scars | Significant differences among mild, moderate, and severe keloid groups in thickness, stiffness (SWE parameters), and blood flow (AP mode) (p < 0.001). AP imaging was more sensitive than CDFI and PDI in detecting microvascular changes and differentiating keloid severity levels. |
| 34 | Morphological aspects of basal cell carcinoma vascularization | Bungărdean et al. [7] | 2023 | Romania | Review, without systematic methodology | Not reported | 3 | HFUS and CDI | BCC | Describes typical BCC vascular patterns (often avascular center with peripheral vascularity) and suggests that Doppler may aid characterization. Provides supportive context, but no primary data or validated microvascular outcomes. |
| 35 | Advanced multimodal ultrasound for pre-operative assessment of skin tumours: a case series | Kho et al. [40] | 2024 | Singapore | Case series | Patients | 11 | B-mode, CDI, SMI, strain elastography, SWE | BCC, SCC | Multimodal ultrasound, especially SMI, revealed tumor-associated microvascular networks and supported preoperative planning. Microvascular depiction appears useful as an adjunct, but evidence is limited to a small observational series. |
| 36 | Microvenous reflux in the skin of limbs with superficial venous incompetence | Govind et al. [41] | 2018 | New Zealand | Case control study | Patients and healthy volunteers | Not clearly reported | SMI and CDI | Chronic venous disease | SMI detected more low-flow microvenous reflux than conventional color Doppler. Improves sensitivity for superficial microvascular abnormalities and may aid earlier assessment, but evidence is limited by sample size and missing variability data. |
Appendix C
| Study No | Author and Year of Publication | Modality | System/Platform | Probe/Transducer | Frequency/Software Mode |
|---|---|---|---|---|---|
| 1 | Barcaui et al. 2014 [2] | HFUS | NR | Linear probe | 22 MHz |
| 2 | Catalano 2020 [4] | HFUS, VHFUS, CDI, Elastography | NR | NR | Review; no single study device |
| 3 | Bhatti 2023 [6] | HFUS/ultrafast US | Vantage 256 | L38–22v CMUT | 31.5 MHz Center frequency |
| 4 | Bungărdean 2023 [7] | Review | NR | NR | NR |
| 5 | Jaeger 2017 [8] | Combined US + PA | Sigma 5000 IMAGIC | 128-element array transducer | Center frequency 7.5 MHz; 40 MHz frequency |
| 6 | Catalano 2023 [9] | SMI, MV-Flow | Aplio i800 (Canon) and RS85 Prestige (Samsung Medison) | 22-MHz linear probe | NR |
| 7 | Corvino 2022 [10] | SMI | Aplio i800 (Canon) | 22-MHz linear probe | NR |
| 8 | Guazzaroni 2021 [11] | MicroV + SWE | Esaote MyLab 9 (Genova, Italy) | High-frequency linear probe 4–15 MHz | NR |
| 9 | Salari 2025 [12] | Super Resolution Using the Erythrocytes | GE Logiq E9 Verasonics Vantage 256TM | 10 MHz GE L8–18iD | 8Mhz; Matlab 2021B |
| 10 | Wang 2020 [15] | PAUS | Custom PAUS dermoscope | Bimorph transducer Custom | ~33–35 MHz center frequencies |
| 11 | Lin 2017 [16] | OR-PAM | Custom OR-PAM | 50-Mhz V214-BB-RM (Olympus NDT) | 2Hz |
| 12 | Favazza 2011 [17] | PAM | NR | V214-BB-RM (Panametrics) | 50 MHz Center frequency |
| 13 | Ly 2022 [18] | PAM | Dual-fast scanning PAM system (Ohlabs) | Olympus flat transducer | Center Frequency 50 MHz; 200MHz Sampling rate |
| 14 | Zafar 2015 [19] | PAI + US | Vevo LAZR (Fujifilm VisualSonics) | 256-element linear-array probes | Center Frequencies 15, 21, 40 MHz |
| 15 | Zafar 2014 [20] | PAI + US | Vevo 2100 LAZR PAT (VisualSonics) | Linear-array transducer | 40 MHz |
| 16 | He 2022 [21] | FRSOM/RSOM | Fast RSOM (FRSOM) custom platform | Custom transducer | Reconstructed bands 10–40 MHz and 40–120 MHz; |
| 17 | Schwarz 2015 [22] | OAM/RSOM | NR | Ultra-wideband spherical | <25Mhz and between 25 MHz–63.5 MHz and >63.5 MHz |
| 18 | Bitton 2007 [23] | Array-based PAM | Custom PAM system | 30 MHz linear array (custom) | 30 MHz with multichannel receiver system |
| 19 | Huynh 2024 [24] | PAT | Custom build PAT scanner (University College London) | Fabry–Perot polymer film ultrasound sensor | 0.05–35 MHz |
| 20 | Choi 2022 [25] | 3D PA/US | E-CUBE 12R (Alpinion medical systems) + Phocus Mobile (Opotek) | 128-element L3–12 linear array (Alpinion) | 3D foot scanner; 3–12 MHz |
| 21 | Zheng 2021 [26] | PAT | Verasonics 128-channel DAQ | L22–8 linear transducer array (Verasonics) | 15.6 MHz center frequency |
| 22 | Zhang 2019 [27] | PAI | Custom miniaturized PA probe | Planar ultrasound transducer | 15 MHz center frequency |
| 23 | Saijo 2019 [28] | HFUS + PAM | Custom HFUS + dual-wavelength PAM | Concave Ultrasound transducer | 50 MHz center frequency |
| 24 | Hsu 2016 [29] | OR-PAM | Custom OR-PAM system | V214, Olympus NDT | 50 MHz |
| 25 | Heres 2017 [30] | PA imaging + comparator US/PDUS | FULLPHASE prototypecontrolled by MyLab One | SL3323 linear array | 7.5 MHz |
| 26 | Xu 2016 [31] | PA dermoscopy | Custom PA dermoscope | Integrated custom PA probe | 18 MHz Center Frequency |
| 27 | Liu 2016 [32] | PAT + OCT + OCTA | Combined multimodal PAT/OCT/OCTA system | Fabry–Perot polymer film ultrasound sensor | NR |
| 28 | Rodrigues 2022 [33] | MSOT/optoacoustic imaging | MSOT optoacoustic system, iThera Medical GmbH, Munich, Germany | MSOT measurement probe/3D cup probe | NR |
| 29 | Song 2010 [34] | Ultrasound-array photoacoustic microscopy | Custom-built UA-PAM | High Frequency 30 MHz Linear array | 30 MHz |
| 30 | Jasionyte 2023 [35] | SMI | Canon TUS-AI800, Canon Medical Systems | 24 MHz linear transducer | 19 MHz |
| 31 | Zhou 2025 [36] | CDFI, PDI, AP, SWE | Aixplorer (SuperSonic Imaging) | Linear probe | 4–15 MHz |
| 32 | Bhatti 2025 [37] | Volumetric HFUS/ultrafast workflow | Vantage 256 High Frequency Configuration, Verasonics | L38–22v, KOLO Medical | 30 MHz Center frequency |
| 33 | Saijo 2024 [38] | HF Power Doppler | LOGIQ e ultrasound device, GE Healthcare | L10–22-RS | 22MHz |
| 34 | Chen 2017 [39] | Ultrafast HFUS | Vantage 256 (Verasonics) | MS550D array transducer (Fujifilm VisualSonics) | 40 MHz |
| 35 | Kho 2024 [40] | B-mode, CDI, SMI, spectral Doppler, SE, SWE | Canon i800 (Canon Medical) | Linear-matrix transducer | 24 and 18 MHz |
| 36 | Govind 2018 [41] | SMI + color Doppler | Toshiba Aplio 500 | Linear array PLB-1005BT transducer | 18 MHz |
References
- Siegel, R.L.; Miller, K.D.; Jemal, A. Cancer statistics, 2020. CA Cancer J. Clin. 2020, 70, 7–30. [Google Scholar] [CrossRef]
- Barcaui, E.O.; Carvalho, A.C.P.; Piñeiro-Maceira, J.; Valiante, P.M.; Barcaui, C.B. Highfrequency ultrasound (22MHz) in the evaluation of malignant cutaneous neoplasms. Surg. Cosmet. Dermatol. 2014, 6, 105–110. [Google Scholar]
- Reginelli, A.; Belfiore, M.P.; Russo, A.; Turriziani, F.; Moscarella, E.; Troiani, T.; Brancaccio, G.; Ronchi, A.; Giunta, E.F.; Sica, A.; et al. A Preliminary Study for Quantitative Assessment with HFUS (High-Frequency Ultrasound) of Nodular Skin Melanoma Breslow Thickness in Adults Before Surgery: Interdisciplinary Team Experience. Curr. Radiopharm. 2020, 13, 48–55. [Google Scholar] [CrossRef]
- Catalano, O.; Wortsman, X. Dermatology Ultrasound. Imaging Technique, Tips and Tricks, High-Resolution Anatomy. Ultrasound Q. 2020, 36, 321–327. [Google Scholar] [CrossRef]
- Sellyn, G.E.; Lopez, A.A.; Ghosh, S.; Topf, M.C.; Chen, H.; Tkaczyk, E.; Powers, J.G. High-frequency ultrasound accuracy in preoperative cutaneous melanoma assessment: A meta-analysis. J. Eur. Acad. Dermatol. Venereol. 2025, 39, 86–96. [Google Scholar] [CrossRef]
- Bhatti, A.; Ishii, T.; Kanno, N.; Ikeda, H.; Funamoto, K.; Saijo, Y. Region-based SVD processing of high-frequency ultrafast ultrasound to visualize cutaneous vascular networks. Ultrasonics 2023, 129, 106907. [Google Scholar] [CrossRef] [PubMed]
- Bungărdean, R.-M.; Stoia, M.-A.; Pop, B.; Crişan, M. Morphological aspects of basal cell carcinoma vascularization. Rom. J. Morphol. Embryol. 2023, 64, 15–23. [Google Scholar] [CrossRef]
- Jaeger, M.; Frenz, M. Combined ultrasound and photoacoustic system for real-time high-contrast imaging using a linear array transducer. In Photoacoustic Imaging and Spectroscopy; CRC Press: Boca Raton, FL, USA, 2017; pp. 289–298. [Google Scholar] [CrossRef]
- Catalano, O.; Corvino, A.; Basile, L.; Catalano, F.; Varelli, C. Use of new microcirculation software allows the demonstration of dermis vascularization. J. Ultrasound 2023, 26, 169–174. [Google Scholar] [CrossRef]
- Corvino, A.; Varelli, C.; Cocco, G.; Corvino, F.; Catalano, O. Seeing the unseen with superb microvascular imaging: Ultrasound depiction of normal dermis vessels. J. Clin. Ultrasound 2022, 50, 121–127. [Google Scholar] [PubMed]
- Guazzaroni, M.; Ferrari, D.; Lamacchia, F.; Marisi, V.; Tatulli, D.; Marsico, S.; Orlacchio, A.; Floris, R.; Bianchi, L.; Dattola, A. Shear wave elastography and microvascular ultrasound in response evaluation to calcipotriol+betamethasone foam in plaque psoriasis. Postgrad. Med. J. 2021, 97, 16–22. [Google Scholar] [CrossRef] [PubMed]
- Salari, A.; Baarts, R.; Naji, M.A.; Tomov, B.G.; Nielsen, M.B.; Jensen, J.A. Human Mole Contrast-Free Microvascular Imaging using Erythrocytes. In Proceedings of the 2025 IEEE International Ultrasonics Symposium (IUS); IEEE: Utrecht, The Netherlands, 2025; pp. 1–4. [Google Scholar] [CrossRef]
- von Knorring, T.; Blanche, P.; Holstad Pedersen, H.; Rosenkrantz Hölmich, L.; Løth Mårtensson, N.; Reichl, C.; Karmisholt, K.; Mogensen, M. Diagnostic accuracy of expeditious bedside evaluation of cutaneous malignant melanoma using photoacoustic imaging. J. Eur. Acad. Dermatol. Venereol. 2025, 39, e23–e25. [Google Scholar]
- Tricco, A.C.; Lillie, E.; Zarin, W.; O’Brien, K.K.; Colquhoun, H.; Levac, D.; Moher, D.; Peters, M.D.J.; Horsley, T.; Weeks, L.; et al. PRISMA Extension for Scoping Reviews (PRISMAScR): Checklist and Explanation. Ann. Intern. Med. 2018, 169, 467–473. [Google Scholar] [CrossRef]
- Wang, Z.; Yang, F.; Ma, H.; Cheng, Z.; Yang, S. Photoacoustic and ultrasound (PAUS) dermoscope with high sensitivity and penetration depth by using a bimorph transducer. J. Biophotonics 2020, 13, e202000145. [Google Scholar] [CrossRef]
- Lin, L.; Zhang, P.; Xu, S.; Shi, J.; Li, L.; Yao, J.; Wang, L.; Zou, J.; Wang, L.V. Handheld optical-resolution photoacoustic microscopy. J. Biomed. Opt. 2017, 22, 41002. [Google Scholar] [CrossRef] [PubMed]
- Favazza, C.P.; Jassim, O.; Cornelius, L.A.; Wang, L.V. In vivo photoacoustic microscopy of human cutaneous microvasculature and a nevus. J. Biomed. Opt. 2011, 16, 016015. [Google Scholar] [CrossRef]
- Ly, C.D.; Nguyen, V.T.; Vo, T.H.; Mondal, S.; Park, S.; Choi, J.; Vu, T.T.H.; Kim, C.-S.; Oh, J. Full-view in vivo skin and blood vessels profile segmentation in photoacoustic imaging based on deep learning. Photoacoustics 2022, 25, 100310. [Google Scholar] [CrossRef] [PubMed]
- Zafar, H.; Breathnach, A.; Subhash, H.M.; Leahy, M.J. Linear-array-based photoacoustic imaging of human microcirculation with a range of high frequency transducer probes. J. Biomed. Opt. 2015, 20, 051021. [Google Scholar] [CrossRef]
- Zafar, H.; Breathnach, A.; Subhash, H.M.; Leahy, M.J. Photoacoustic imaging of the human forearm using 40 MHz linear-array transducer. In Proceedings of the Dynamics and Fluctuations in Biomedical Photonics XI; SPIE: San Francisco, CA, USA, 2014; Volume 8942. [Google Scholar]
- He, H.; Schönmann, C.; Schwarz, M.; Hindelang, B.; Berezhnoi, A.; Steimle-Grauer, S.A.; Darsow, U.; Aguirre, J.; Ntziachristos, V. Fast raster-scan optoacoustic mesoscopy enables assessment of human melanoma microvasculature in vivo. Nat. Commun. 2022, 13, 2803. [Google Scholar] [CrossRef]
- Schwarz, M.; Omar, M.; Buehler, A.; Aguirre, J.; Ntziachristos, V. Implications of ultrasound frequency in optoacoustic mesoscopy of the skin. IEEE Trans. Med. Imaging 2015, 34, 672–677. [Google Scholar] [PubMed]
- Bitton, R.; Zemp, R.; Yen, J.; Wang, L.H.; Shung, K.k. Design of a high frequency array based photoacoustic microscopy system for micro-vascular imaging. Annu. Int. Conf. IEEE Eng. Med. Biol. Soc. 2007, 2007, 2175–2178. [Google Scholar]
- Huynh, N.T.; Zhang, E.; Francies, O.; Kuklis, F.; Allen, T.; Zhu, J.; Abeyakoon, O.; Lucka, F.; Betcke, M.; Jaros, J.; et al. A fast all-optical 3D photoacoustic scanner for clinical vascular imaging. Nat. Biomed. Eng. 2024, 9, 638–655. [Google Scholar] [CrossRef]
- Choi, W.; Park, E.Y.; Jeon, S.; Yang, Y.; Park, B.; Ahn, J.; Cho, S.; Lee, C.; Seo, D.K.; Cho, J.H.; et al. Three-dimensional Multistructural Quantitative Photoacoustic and US Imaging of Human Feet in Vivo. Radiology 2022, 303, 467–473. [Google Scholar] [CrossRef] [PubMed]
- Zheng, W.; Lee, D.; Xia, J. Photoacoustic tomography of fingerprint and underlying vasculature for improved biometric identification. Sci. Rep. 2021, 11, 17536. [Google Scholar] [CrossRef]
- Zhang, W.; Ma, H.; Cheng, Z.; Wang, Z.; Zhang, L.; Yang, S. Miniaturized photoacoustic probe for in vivo imaging of subcutaneous microvessels within human skin. Quant. Imaging Med. Surg. 2019, 9, 807–814. [Google Scholar] [CrossRef]
- Saijo, Y.; Ida, T.; Iwazaki, H.; Shintate, R.; Sato, K.; Hiratsuka, T.; Yoshizawa, S.; Umemura, S.-I.; Miyajima, J.; Tang, H. Visualization of skin morphology and microcirculation with high frequency ultrasound and dual-wavelength photoacoustic microscope. In Proceedings of the Photons Plus Ultrasound: Imaging and Sensing 2019; SPIE: San Francisco, CA, USA, 2019; Volume 10878. [Google Scholar]
- Hsu, H.; Wang, L.; Wang, L. In vivo photoacoustic microscopy of human cuticle microvasculature with single-cell resolution. J. Biomed. Opt. 2016, 21, 56004. [Google Scholar] [CrossRef]
- Heres, H.; Arabul, M.; Rutten, M.; Van de Vosse, F.; Lopata, R. Visualization of vasculature using a hand-held photoacoustic probe: Phantom and in vivo validation. J. Biomed. Opt. 2017, 22, 41013. [Google Scholar] [CrossRef] [PubMed]
- Xu, D.; Yang, S.; Wang, Y.; Gu, Y.; Xing, D. Noninvasive and high-resolving photoacoustic dermoscopy of human skin. Biomed. Opt. Express 2016, 7, 2095–2102. [Google Scholar] [CrossRef]
- Liu, M.; Chen, Z.; Zabihian, B.; Sinz, C.; Zhang, E.; Beard, P.C.; Ginner, L.; Hoover, E.; Minneman, M.P.; Leitgeb, R.A.; et al. Combined multi-modal photoacoustic tomography, optical coherence tomography (OCT) and OCT angiography system with an articulated probe for in vivo human skin structure and vasculature imaging. Biomed. Opt. Express 2016, 7, 3390. [Google Scholar] [CrossRef]
- Rodrigues, L.; Granja, T.; de Andrade, S. Optoacoustic Imaging Offers New Insights into In Vivo Human Skin Vascular Physiology. Life 2022, 12, 1628. [Google Scholar] [CrossRef]
- Song, L.; Maslov, K.; Shung, K.; Wang, L. Ultrasound Array Photoacoustic Microscopy for Dynamic In Vivo 3-D Imaging. In Proceedings of the Photons Plus Ultrasound: Imaging and Sensing 2010; SPIE: San Francisco, CA, USA, 2010; Volume 7564. [Google Scholar]
- Jasionyte, G.; Seskute, G.; Rugiene, R.; Butrimiene, I. Assessing scleroderma patterns with superb microvascular imaging: Is it possible? New prospects for ultrasound. Clin. Rheumatol. 2023, 42, 301–306. [Google Scholar] [CrossRef] [PubMed]
- Zhou, L.; Zhou, Q.; Zheng, C.; Wang, Z.; Rao, M. Multimodal ultrasound assessment for monitoring keloid severity and treatment response. Sci. Rep. 2025, 15, 8568. [Google Scholar] [CrossRef] [PubMed]
- Bhatti, A.; Ishii, T.; Lafond, M.; Saijo, Y. Volumetric Visualization of the Dermal Vasculature with Signal and Image-based Feature Extraction on a High-frequency Ultrafast Ultrasound Dataset. Ultrasound Med. Biol. 2025, 51, 1514–1522. [Google Scholar] [CrossRef]
- Saijo, Y.; Akaishi, S.; Kuwahara, H. High-frequency Power Doppler Ultrasonography in Predicting Burn Depth: A Preliminary Case Report. Plast. Reconstr. Surg.-Glob. Open 2024, 12, e5949. [Google Scholar] [CrossRef]
- Chen, P.; Hsueh, Y.; Huang, C. In vivo microcirculation mapping of human skin keloid by 40-MHz ultrafast ultrasound imaging. In Proceedings of the 2017 IEEE International Ultrasonics Symposium (IUS); IEEE: New York, NY, USA, 2017. [Google Scholar]
- Kho, Y.Y.; Ooi, C.C.; Too, C.W.; Ma, V.C.; Abu Bakar, R.; Lim, C.Y.; Tang, P.Y.; Oh, C.C. Advanced multimodal ultrasound for pre-operative assessment of skin tumours: A case series. Ultrasound 2024, 33, 130–138. [Google Scholar] [CrossRef] [PubMed]
- Govind, D.; Thomas, K.N.; Hill, B.G.; van Rij, A.M. Microvenous Reflux in the Skin of Limbs with Superficial Venous Incompetence. Ultrasound Med. Biol. 2018, 44, 756–761. [Google Scholar] [CrossRef]
- Ward, W.H.; Farma, J.M.; Goel, N.; Yu, J.Q.; Farma, J.M. Clinical Presentation and Staging of Melanoma. In Cutaneous Melanoma: Etiology and Therapy; Ward, W.H., Farma, J.M., Eds.; Codon Publications: Brisbane, Australia, 2017; pp. 79–89. [Google Scholar] [CrossRef]
- Naji, M.A.; Panduro, N.S.; Majd, S.M.M.T.; Hansen, L.N.; Taghavi, I.; McDermott, A.; Gundlach, C.; Tomov, B.G.; Nielsen, M.B.; Sørensen, C.M.; et al. Human lymph node microvascular imaging using a fast contrast-free super-resolution ultrasound technique. Sci. Rep. 2025, 15, 23061. [Google Scholar]
- Tew, G.A.; Klonizakis, M.; Crank, H.; Briers, J.D.; Hodges, G.J. Comparison of laser speckle contrast imaging with laser Doppler for assessing microvascular function. Microvasc. Res. 2011, 82, 326–332. [Google Scholar] [CrossRef]
- Briers, D.; Duncan, D.D.; Hirst, E.; Kirkpatrick, S.J.; Larsson, M.; Steenbergen, W.; Stromberg, T.; Thompson, O.B. Laser speckle contrast imaging: Theoretical and practical limitations. J. Biomed. Opt. 2013, 18, 066018. [Google Scholar] [CrossRef] [PubMed]



| Modality | Number of Studies |
|---|---|
| Photoacoustic-based (PAI/PAM/PAT/OAM) | 21 |
| High-frequency ultrasound (HFUS/VHFUS) | 10 |
| Conventional Doppler (CDFI/PDI) | 7 |
| Advanced Doppler (SMI/MicroV/MV-Flow) | 7 |
| Multimodal combinations (≥2 modalities) | 9 |
| Super-Resolution Ultrasound using tracking of the erythrocytes | 1 |
| Clinical and pathological focus areas | |
| Healthy volunteers/normal skin | 19 |
| Inflammatory/vascular disease | 5 |
| Benign fibrotic lesions (keloid) | 2 |
| Benign pigmented lesions (nevi/moles) | 5 |
| Malignant skin tumours | 4 |
| Other pathology | 1 |
| Reported variabilities | |
| Median sample size (participants) | 3 |
| Studies < 20 participants | 21 |
| Studies missing numeric sample size | 9 |
| Studies reporting age | 13 |
| Studies reporting sex | 16 |
| Studies reporting both inclusion and exclusion criteria | 7 |
| Studies informing about histopathology | 1 |
| Modality | Studies (n) | Main Strengths | Main Limitations | Clinical Status |
|---|---|---|---|---|
| Photoacoustic-based imaging | 21 | High microvascular detail; functional imaging possible | Mostly experimental; limited standardization; depth–resolution trade-off; affected by pigmentation/skin type | Mainly experimental |
| High-frequency ultrasound (HFUS/VHFUS) | 10 | Widely available; useful structural imaging | Limited capillary-level detail without added processing | Clinically available, but limited for fine microvascular imaging |
| Conventional Doppler (CDFI/PDI) | 7 | Flow-related information; widely available | Low sensitivity to slow dermal flow; limited fine-vessel depiction | Clinically established, but limited for dermal microvasculature |
| Advanced Doppler (SMI/MicroV/MV-Flow/AP) | 7 | Better sensitivity to low-flow dermal vessels; relatively accessible clinically | Small heterogeneous evidence base; device-dependent; lower detail than highest-resolution techniques | Promising clinical adjunct |
| Super-resolution ultrasound using erythrocytes (SURE) | 1 | Very high spatial detail; contrast-free microvascular imaging | Only one included dermal study; no basis yet for clinical readiness | Very early-stage |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Baarts, R.; Henriksen, A.C.; Panduro, N.S.; Bengtsson, E.K.E.; Salari, A.; Clausen, C.; Hölmich, L.R.; Lönn, L.; Sørensen, C.M.; Jensen, J.A.; et al. Ultrasound-Based Techniques for Visualization of Dermal Microvasculature: A Scoping Review. Diagnostics 2026, 16, 1435. https://doi.org/10.3390/diagnostics16101435
Baarts R, Henriksen AC, Panduro NS, Bengtsson EKE, Salari A, Clausen C, Hölmich LR, Lönn L, Sørensen CM, Jensen JA, et al. Ultrasound-Based Techniques for Visualization of Dermal Microvasculature: A Scoping Review. Diagnostics. 2026; 16(10):1435. https://doi.org/10.3390/diagnostics16101435
Chicago/Turabian StyleBaarts, Rikke, Alexander Cuculiza Henriksen, Nathalie Sarup Panduro, Emma Kanchana Ertner Bengtsson, Ali Salari, Caroline Clausen, Lisbet Rosenkrantz Hölmich, Lars Lönn, Charlotte Mehlin Sørensen, Jørgen Arendt Jensen, and et al. 2026. "Ultrasound-Based Techniques for Visualization of Dermal Microvasculature: A Scoping Review" Diagnostics 16, no. 10: 1435. https://doi.org/10.3390/diagnostics16101435
APA StyleBaarts, R., Henriksen, A. C., Panduro, N. S., Bengtsson, E. K. E., Salari, A., Clausen, C., Hölmich, L. R., Lönn, L., Sørensen, C. M., Jensen, J. A., & Nielsen, M. B. (2026). Ultrasound-Based Techniques for Visualization of Dermal Microvasculature: A Scoping Review. Diagnostics, 16(10), 1435. https://doi.org/10.3390/diagnostics16101435







