Pediatric Cutaneous Anaplastic Lymphoma Kinase-Positive Histiocytosis with DCTN1::ALK Fusion: A Case Report and Literature Search
Abstract
1. Introduction
2. Materials and Methods
3. Case Report
3.1. Clinical Presentation, Management, and Follow-Up
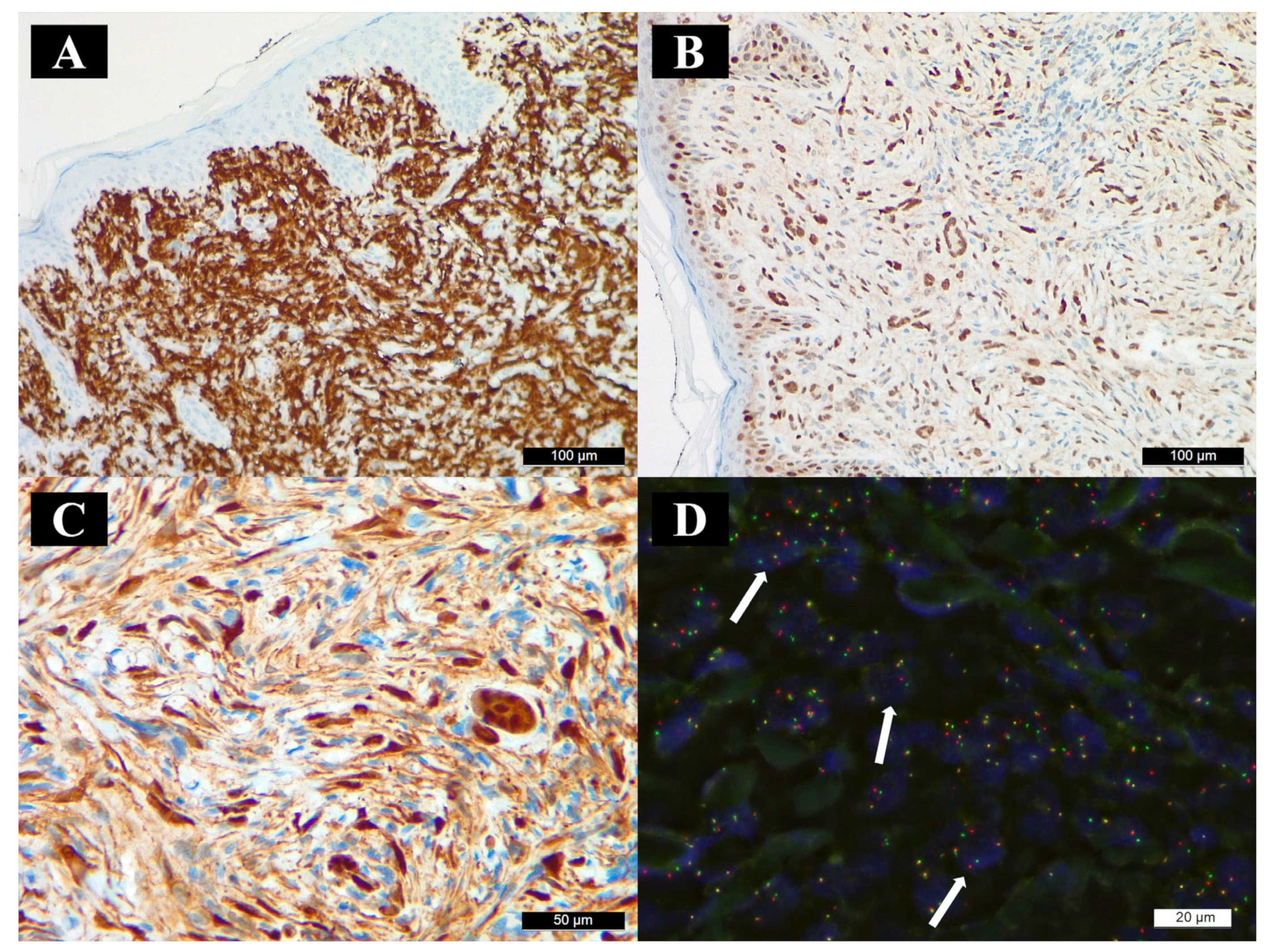
3.2. Histopathology and Immunohistochemistry
3.3. Molecular Findings
3.4. Imaging and Laboratory Findings
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| ALK | Anaplastic lymphoma kinase |
| CSF1R | Colony-stimulating factor 1 receptor |
| CT | Computed tomography |
| ERK | Extracellular signal-regulated kinase |
| FISH | Fluorescence in situ hybridization |
| HMB-45 | Human melanoma black |
| JAK | Janus kinase |
| LCH | Langerhans cell histiocytosis |
| MAPK | Mitogen-activated protein kinase |
| MBC | Molecular barcode |
| MRI | Magnetic resonance imaging |
| MTOR | Mammalian target of rapamycin |
| NGS | Next-generation sequencing |
| NPM | Nucleophosmin |
| NTRK1 | Neurotrophic receptor tyrosine kinase 1 |
| PI3K | Phosphatidylinositol 3-kinase |
| STAT | Signal transducer and activator of transcription |
| VAF | Variant allele frequency |
References
- Durham, B.H.; Lopez Rodrigo, E.; Picarsic, J.; Abramson, D.; Rotemberg, V.; De Munck, S.; Pannecoucke, E.; Lu, S.X.; Pastore, A.; Yoshimi, A.; et al. Activating mutations in CSF1R and additional receptor tyrosine kinases in histiocytic neoplasms. Nat. Med. 2019, 25, 1839–1842. [Google Scholar] [CrossRef] [PubMed]
- Pulford, K.; Lamant, L.; Morris, S.W.; Butler, L.H.; Wood, K.M.; Stroud, D.; Delsol, G.; Mason, D.Y. Detection of anaplastic lymphoma kinase (ALK) and nucleolar protein nucleophosmin (NPM)-ALK proteins in normal and neoplastic cells with the monoclonal antibody ALK1. Blood 1997, 89, 1394–1404. [Google Scholar] [CrossRef] [PubMed]
- Della Corte, C.M.; Viscardi, G.; Di Liello, R.; Fasano, M.; Martinelli, E.; Troiani, T.; Ciardiello, F.; Morgillo, F. Role and targeting of anaplastic lymphoma kinase in cancer. Mol. Cancer 2018, 17, 30. [Google Scholar] [CrossRef] [PubMed]
- Du, X.; Shao, Y.; Qin, H.F.; Tai, Y.H.; Gao, H.J. ALK-rearrangement in non-small-cell lung cancer (NSCLC). Thorac. Cancer 2018, 9, 423–430. [Google Scholar] [CrossRef]
- Zeng, Y.; Feldman, A.L. Genetics of anaplastic large cell lymphoma. Leuk. Lymphoma 2016, 57, 21–27. [Google Scholar] [CrossRef]
- Ou, S.I.; Zhu, V.W.; Nagasaka, M. Catalog of 5′ Fusion Partners in ALK-positive NSCLC Circa 2020. JTO Clin. Res. Rep. 2020, 1, 100015. [Google Scholar] [CrossRef]
- Morris, S.W.; Kirstein, M.N.; Valentine, M.B.; Dittmer, K.; Shapiro, D.N.; Look, A.T.; Saltman, D.L. Fusion of a kinase gene, ALK, to a nucleolar protein gene, NPM, in non-Hodgkin’s lymphoma. Science 1995, 267, 316–317. [Google Scholar] [CrossRef]
- Pulford, K.; Lamant, L.; Espinos, E.; Jiang, Q.; Xue, L.; Turturro, F.; Delsol, G.; Morris, S.W. The emerging normal and disease-related roles of anaplastic lymphoma kinase. Cell. Mol. Life Sci. 2004, 61, 2939–2953. [Google Scholar] [CrossRef]
- Chan, J.K.; Lamant, L.; Algar, E.; Delsol, G.; Tsang, W.Y.; Lee, K.C.; Tiedemann, K.; Chow, C.W. ALK+ histiocytosis: A novel type of systemic histiocytic proliferative disorder of early infancy. Blood 2008, 112, 2965–2968. [Google Scholar] [CrossRef]
- Phillips, G.S.; Knapp, M.; Olsen, K.C.; Martin, W.; Hayes-Lattin, B.; Chung, J. Multisystem ALK-Positive Histiocytosis With DCTN1::ALK Fusion in an Adult, Responsive to Alectinib: Case Report and Literature Review. J. Cutan. Pathol. 2024, 52, 63–71. [Google Scholar] [CrossRef]
- Kemps, P.G.; Picarsic, J.; Durham, B.H.; Helias-Rodzewicz, Z.; Hiemcke-Jiwa, L.; van den Bos, C.; van de Wetering, M.D.; van Noesel, C.J.M.; van Laar, J.A.M.; Verdijk, R.M.; et al. ALK-positive histiocytosis: A new clinicopathologic spectrum highlighting neurologic involvement and responses to ALK inhibition. Blood 2022, 139, 256–280. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Liu, H.J.; Wang, W.Y.; Tang, Y.; Zhao, S.; Zhang, W.Y.; Yan, J.Q.; Liu, W.P. Multisystem ALK-positive histiocytosis: A multi-case study and literature review. Orphanet J. Rare Dis. 2023, 18, 53. [Google Scholar] [CrossRef] [PubMed]
- Emile, J.F.; Abla, O.; Fraitag, S.; Horne, A.; Haroche, J.; Donadieu, J.; Requena-Caballero, L.; Jordan, M.B.; Abdel-Wahab, O.; Allen, C.E.; et al. Revised classification of histiocytoses and neoplasms of the macrophage-dendritic cell lineages. Blood 2016, 127, 2672–2681. [Google Scholar] [CrossRef] [PubMed]
- Chang, K.T.E.; Tay, A.Z.E.; Kuick, C.H.; Chen, H.; Algar, E.; Taubenheim, N.; Campbell, J.; Mechinaud, F.; Campbell, M.; Super, L.; et al. ALK-positive histiocytosis: An expanded clinicopathologic spectrum and frequent presence of KIF5B-ALK fusion. Mod. Pathol. 2019, 32, 598–608. [Google Scholar] [CrossRef]
- Keeney, M.G.; Flotte, T.J.; Macon, W.R. Cutaneous ALK-positive histiocytosis. J. Hematop. 2021, 14, 89–91. [Google Scholar] [CrossRef]
- Wang, H.; Wang, Y.; Wang, R.; Li, X. Case Report: Two Infant Cases of Langerhans Cell Histiocytosis Involving the Digestive Tract. Front. Pediatr. 2021, 9, 545771. [Google Scholar] [CrossRef]
- Schroer, T.A. Dynactin. Annu. Rev. Cell Dev. Biol. 2004, 20, 759–779. [Google Scholar] [CrossRef]
- Konno, T.; Ross, O.A.; Teive, H.A.G.; Slawek, J.; Dickson, D.W.; Wszolek, Z.K. DCTN1-related neurodegeneration: Perry syndrome and beyond. Park. Relat. Disord. 2017, 41, 14–24. [Google Scholar] [CrossRef]
- Vidrine, D.W.; Berry, J.F.; Garbuzov, A.; Falcon, C.; Tubbs, R.S.; Bui, C.J. DCTN1-ALK gene fusion in inflammatory myofibroblastic tumor (IMT) of the CNS. Childs Nerv. Syst. 2021, 37, 2147–2151. [Google Scholar] [CrossRef]
- Vendrell, J.A.; Taviaux, S.; Beganton, B.; Godreuil, S.; Audran, P.; Grand, D.; Clermont, E.; Serre, I.; Szablewski, V.; Coopman, P.; et al. Detection of known and novel ALK fusion transcripts in lung cancer patients using next-generation sequencing approaches. Sci. Rep. 2017, 7, 12510. [Google Scholar] [CrossRef]
- Fung, C.K.; Chow, C.; Chan, W.K.; Choi, E.W.K.; To, K.F.; Chan, J.K.C.; Cheuk, W. Spindle cell/sclerosing rhabdomyosarcoma with DCTN1::ALK fusion: Broadening the molecular spectrum with potential therapeutic implications. Virchows Arch. 2022, 480, 927–932. [Google Scholar] [CrossRef] [PubMed]
- Murakami, N.; Okuno, Y.; Yoshida, K.; Shiraishi, Y.; Nagae, G.; Suzuki, K.; Narita, A.; Sakaguchi, H.; Kawashima, N.; Wang, X.; et al. Integrated molecular profiling of juvenile myelomonocytic leukemia. Blood 2018, 131, 1576–1586. [Google Scholar] [CrossRef]
- Shimada, Y.; Kohno, T.; Ueno, H.; Ino, Y.; Hayashi, H.; Nakaoku, T.; Sakamoto, Y.; Kondo, S.; Morizane, C.; Shimada, K.; et al. An Oncogenic ALK Fusion and an RRAS Mutation in KRAS Mutation-Negative Pancreatic Ductal Adenocarcinoma. Oncologist 2017, 22, 158–164. [Google Scholar] [CrossRef] [PubMed]
- Blandin, A.F.; Giglio, R.; Graham, M.S.; Garcia, G.; Malinowski, S.; Woods, J.K.; Ramkissoon, S.; Ramkissoon, L.; Dubois, F.; Schoolcraft, K.; et al. ALK Amplification and Rearrangements Are Recurrent Targetable Events in Congenital and Adult Glioblastoma. Clin. Cancer Res. 2023, 29, 2651–2667. [Google Scholar] [CrossRef] [PubMed]
- Wiesner, T.; He, J.; Yelensky, R.; Esteve-Puig, R.; Botton, T.; Yeh, I.; Lipson, D.; Otto, G.; Brennan, K.; Murali, R.; et al. Kinase fusions are frequent in Spitz tumours and spitzoid melanomas. Nat. Commun. 2014, 5, 3116. [Google Scholar] [CrossRef]
- DeSimone, M.S.; Odintsov, I.; Tsai, H.K.; Dickson, B.C.; Alomari, A.K.; Hornick, J.L.; Fletcher, C.D.M.; Papke, D.J., Jr. Epithelioid Fibrous Histiocytoma Is on a Continuum With Superficial ALK-rearranged Myxoid Spindle Cell Neoplasm: A Clinicopathologic Series of 35 Cases Including Alternate RET and NTRK3 Fusions. Am. J. Surg. Pathol. 2024. [Google Scholar] [CrossRef]
- Dermarkarian, C.R.; Patel, K.R.; Fuller, M.Y.; Bernini, J.C.; Chevez-Barrios, P.; Allen, R.C. Inflammatory Myofibroblastic Tumor of the Orbit in an 8-Month Old. Ophthalmic Plast. Reconstr. Surg. 2020, 36, e65–e68. [Google Scholar] [CrossRef]
- Wang, B.; Chen, R.; Wang, C.; Guo, J.; Yuan, M.; Chen, H.; Xia, X.; Zhong, D. Identification of novel ALK fusions using DNA/RNA sequencing in immunohistochemistry/RT-PCR discordant NSCLC patients. Hum. Pathol. 2021, 114, 90–98. [Google Scholar] [CrossRef]
- Wang, X.; Krishnan, C.; Nguyen, E.P.; Meyer, K.J.; Oliveira, J.L.; Yang, P.; Yi, E.S.; Erickson-Johnson, M.R.; Yaszemski, M.J.; Maran, A.; et al. Fusion of dynactin 1 to anaplastic lymphoma kinase in inflammatory myofibroblastic tumor. Hum. Pathol. 2012, 43, 2047–2052. [Google Scholar] [CrossRef]
- Wang, Z.; Han, Y.; Tao, H.; Xu, M.; Liu, Z.; Zhu, J.; Li, W.; Ma, J.; Liu, Z.; Wang, W.; et al. Molecular characterization of genomic breakpoints of ALK rearrangements in non-small cell lung cancer. Mol. Oncol. 2023, 17, 765–778. [Google Scholar] [CrossRef]

| ANTIBODY | ANTIGEN RETRIEVAL | IHC STAINERS | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Name | Vendor | Host | Clone | Dil. | Incubation Time/Temp | Method | Time/Temp | Visualization System | Platform |
| ALK (D5F300A0045) XP® | Cell Signaling Technology Europe, B.V., Leiden, The Netherlands | rabbit | D5F3 | 1/200 | 16′/36 °C | CC1 solution, pH 8.5 | 48′, 100 °C | OptiView DAB Detection, 6396500001 | VENTANA BenchMark ULTRA |
| Phospho-p44/42 MAPK (Erk1/2) (Thr202/Tyr204) | D13.14.4E | 32′/36 °C | 36′, 100 °C | Ultraview Universal DAB Detection Kit, 5269806001 | |||||
| Cyclin D1 | Roche (Hungary) Ltd., Budapest, Hungary | SP4-R | RTU | 48′, 100 °C | OptiView DAB Detection 6396500001 | ||||
| CD163 | Sigma-Aldrich Chemie GmbH, Taufkirchen, Germany | mouse | MRQ-26 | 1/200 | 36′, 100 °C | ||||
| CD34 | Leica Biosystems Nussloch GmbH, Nussloch, Germany | QBEnd/10 | 1/2000 | Ultraview Universal DAB Detection Kit, 5269806001 | |||||
| S-100 | polyclonal | 1/200 | 32′/RT | 20′, 100 °C | |||||
| CD10 | 56C6 | 1/100 | 30′/RT | HIER BERS2, pH 9 | Bond Polymer Refine Detection | Leica-Bond Max | |||
| Ki-67 | Dako, an Agilent Technologies Company, Glostrup, Denmark | MIB1 | 1/200 | ||||||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Korpás, K.L.; Mokánszki, A.; Beke, L.; Méhes, G.; Chang Chien, Y.-C. Pediatric Cutaneous Anaplastic Lymphoma Kinase-Positive Histiocytosis with DCTN1::ALK Fusion: A Case Report and Literature Search. Diagnostics 2025, 15, 1057. https://doi.org/10.3390/diagnostics15091057
Korpás KL, Mokánszki A, Beke L, Méhes G, Chang Chien Y-C. Pediatric Cutaneous Anaplastic Lymphoma Kinase-Positive Histiocytosis with DCTN1::ALK Fusion: A Case Report and Literature Search. Diagnostics. 2025; 15(9):1057. https://doi.org/10.3390/diagnostics15091057
Chicago/Turabian StyleKorpás, Kristóf Levente, Attila Mokánszki, Lívia Beke, Gábor Méhes, and Yi-Che Chang Chien. 2025. "Pediatric Cutaneous Anaplastic Lymphoma Kinase-Positive Histiocytosis with DCTN1::ALK Fusion: A Case Report and Literature Search" Diagnostics 15, no. 9: 1057. https://doi.org/10.3390/diagnostics15091057
APA StyleKorpás, K. L., Mokánszki, A., Beke, L., Méhes, G., & Chang Chien, Y.-C. (2025). Pediatric Cutaneous Anaplastic Lymphoma Kinase-Positive Histiocytosis with DCTN1::ALK Fusion: A Case Report and Literature Search. Diagnostics, 15(9), 1057. https://doi.org/10.3390/diagnostics15091057





