Elevated Serum Chromogranin-A and Characteristic Duodenal Enteroendocrine Cell Distribution in Pancreatic Fibrosis and Chronic Pancreatitis Compared with Other Dyspeptic Disorders: A Case Series Study
Abstract
1. Introduction
2. Methods
3. Results
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Ford, A.C.; Marwaha, A.; Sood, R.; Moayyedi, P. Global prevalence of, and risk factors for, uninvestigated dyspepsia: A meta-analysis. Gut 2015, 64, 1049–1057. [Google Scholar] [CrossRef] [PubMed]
- Wechsler, E.V.; Ahuja, N.K.; Brenner, D.; Chan, W.; Chang, L.; Chey, W.D.; Lembo, A.J.; Moshiree, B.; Nee, J.; Shah, S.C.; et al. Up-Front Endoscopy Maximizes Cost-Effectiveness and Cost-Satisfaction in Uninvestigated Dyspepsia. Clin. Gastroenterol. Hepatol. 2023, 21, 2378–2388.e28. [Google Scholar] [PubMed]
- Ito, K.; Takasaki, Y.; Fujisawa, T.; Ishii, S.; Tomishima, K.; Takahashi, S.; Ikoma, I.; Jimbo, Y.; Ota, H.; Kabemura, D.; et al. Current Situation and Problems in Diagnosis of Early Chronic Pancreatitis. Pancreas 2023, 52, e275–e281. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.-T.; Lee, T.-H.; Lin, C.-K.; Chen, C.-Y.; Yang, Y.-F.; Liang, Y.-J. Pancreatic Fibrosis (Early Chronic Pancreatitis) as Emerging Diagnosis in Structural Causes of Dyspepsia: Evidence from Endoscopic Ultrasonography and Shear Wave Elastography. Diagnostics 2021, 11, 1252. [Google Scholar] [CrossRef]
- Yamawaki, H.; Futagami, S.; Kaneko, K.; Agawa, S.; Higuchi, K.; Murakami, M.; Wakabayashi, M.; Sakasegawa, N.; Kodaka, Y.; Ueki, N.; et al. Camostat Mesilate, Pancrelipase, and Rabeprazole Combination Therapy Improves Epigastric Pain in Early Chronic Pancreatitis and Functional Dyspepsia with Pancreatic Enzyme Abnormalities. Digestion 2019, 99, 283–292. [Google Scholar] [CrossRef]
- Huang, C.T.; Lin, C.K.; Lee, T.H.; Liang, Y.J. Pancreatic Fibrosis and Chronic Pancreatitis: Mini-Review of Non-Histologic Diagnosis for Clinical Applications. Diagnostics 2020, 10, 87. [Google Scholar] [CrossRef]
- Higuchi, K.; Futagami, S.; Yamawaki, H.; Murakami, M.; Kirita, K.; Agawa, S.; Ikeda, G.; Noda, H.; Kodaka, Y.; Ueki, N.; et al. Endosonographic features in patients with non-alcoholic early chronic pancreatitis improved with treatment at one year follow up. J. Clin. Biochem. Nutr. 2021, 68, 86–94. [Google Scholar] [CrossRef]
- Hristov, B.; Andonov, V.; Doykov, D.; Tsvetkova, S.; Doykova, K.; Doykov, M. Evaluation of Ultrasound-Based Point Shear Wave Elastography for Differential Diagnosis of Pancreatic Diseases. Diagnostics 2022, 12, 841. [Google Scholar] [CrossRef]
- Wekerle, M.; Murillo, K.; vonBoscamp, M.; Hauber, V.; Ebert, M.P.; Antoni, C.; Hirth, M. Point-shear wave elastography generated by acoustic radiation force impulse in chronic pancreatitis. United Eur. Gastroenterol. J. 2024, 12, 667–677. [Google Scholar]
- Saint-Denis, E.; Frintu, B.; Goldsmith, M.; Ramos, G.P.; Zeve, D. Enteroendocrine cell differentiation: Implications for human disease. Mol. Cell. Endocrinol. 2025, 607, 112607. [Google Scholar] [CrossRef]
- Massironi, S.; Zilli, A.; Cavalcoli, F.; Conte, D.; Peracchi, M. Chromogranin A and other enteroendocrine markers in inflammatory bowel disease. Neuropeptides 2016, 58, 127–134. [Google Scholar] [CrossRef]
- Huang, C.T.; Liang, Y.J. Comparison of Duodenal Mucosal Chromogranin-A Expression in Non-Alcoholic Fatty Pancreas Dyspeptic Patients with and without Endosonography-Diagnosed Early Chronic Pancreatitis: A Case Series Study. Case Rep. Gastroenterol. 2019, 13, 102–112. [Google Scholar] [CrossRef] [PubMed]
- Atanga, R.; Singh, V.; In, J.G. Intestinal Enteroendocrine Cells: Present and Future Druggable Targets. Int. J. Mol. Sci. 2023, 24, 8836. [Google Scholar] [CrossRef] [PubMed]
- Shen, R.; Huang, Y.; Kong, D.; Ma, W.; Liu, J.; Zhang, H.; Cheng, S.; Feng, L. Spatial distribution pattern of immune cells is associated with patient prognosis in colorectal cancer. J. Transl. Med. 2024, 22, 606. [Google Scholar] [CrossRef] [PubMed]
- Parra, E.R.; Zhang, J.; Jiang, M.; Tamegnon, A.; Pandurengan, R.K.; Behrens, C.; Solis, L.; Haymaker, C.; Heymach, J.V.; Moran, C.; et al. Immune cellular patterns of distribution affect outcomes of patients with non-small cell lung cancer. Nat. Commun. 2023, 14, 2364. [Google Scholar] [CrossRef]
- Marín-Serrano, E.; Barbado Cano, A.; Fernández Martos, R.; Abadía Barno, M.; Olveira Martín, A.; Martín Arranz, M.D. Protocol for acquisition of images and measurement of transabdominal ultrasound pancreatic two-dimensional shear wave elastography (2D-SWE). Gastroenterol. Hepatol. 2024, 47, 502198. [Google Scholar] [CrossRef]
- Shintani, S.; Inatomi, O.; Okamoto, T.; Hiroe, K.; Eguchi, T.; Tomozawa, Y.; Inoue, A.; Kimura, H.; Nishida, A.; Tsuji, Y.; et al. Diagnostic accuracy of endoscopic ultrasonographic shear wave elastography for assessing early chronic pancreatitis using the Japanese diagnostic criteria 2019. DEN Open 2025, 5, e387. [Google Scholar] [CrossRef]
- Gut, P.; Czarnywojtek, A.; Fischbach, J.; Bączyk, M.; Ziemnicka, K.; Wrotkowska, E.; Gryczyńska, M.; Ruchała, M. Chromogranin A—Unspecific neuroendocrine marker. Clinical utility and potential diagnostic pitfalls. Arch. Med. Sci. 2016, 12, 1–9. [Google Scholar] [CrossRef]
- Romano, E.; Rinzivillo, M.; Lamberti, G.; Marasco, M.; Esposito, G.; Campana, D.; Panzuto, F. Rethinking chromogranin A: Unveiling gastrointestinal factors beyond neuroendocrine neoplasms-a narrative review. Transl. Gastroenterol. Hepatol. 2025, 10, 57. [Google Scholar] [CrossRef]
- Fu, Y.; Qu, H.; Qu, D.; Zhao, M. Trajectory Inference with Cell-Cell Interactions (TICCI): Intercellular communication improves the accuracy of trajectory inference methods. Bioinformatics 2025, 41, btaf027. [Google Scholar] [CrossRef]
- Adriaenssens, A.E.; Reimann, F.; Gribble, F.M. Distribution and Stimulus Secretion Coupling of Enteroendocrine Cells along the Intestinal Tract. Compr. Physiol. 2018, 8, 1603–1638. [Google Scholar] [CrossRef] [PubMed]
- Monachese, M.; Lee, P.J.; Harris, K.; Jang, S.; Bhatt, A.; Chahal, P.; Lopez, R.; Stevens, T. EUS and secretin endoscopic pancreatic function test predict evolution to overt structural changes of chronic pancreatitis in patients with nondiagnostic baseline imaging. Endosc. Ultrasound 2021, 10, 116–123. [Google Scholar] [PubMed]
- Hashimoto, S.; Futagami, S.; Yamawaki, H.; Kaneko, K.; Kodaka, Y.; Wakabayashi, M.; Sakasegawa, N.; Agawa, S.; Higuchi, K.; Akimoto, T.; et al. Epigastric pain syndrome accompanying pancreatic enzyme abnormalities was overlapped with early chronic pancreatitis using endosonography. J. Clin. Biochem. Nutr. 2017, 61, 140–145. [Google Scholar] [CrossRef] [PubMed]
- Huyghe, P.; Ceulemans, M.; Keita, Å.V.; Söderholm, J.; Depoortere, I.; Tack, J.; Wauters, L.; Vanuytsel, T. The Duodenal Microenvironment in Functional Dyspepsia. J. Neurogastroenterol. Motil. 2025, 31, 186–198. [Google Scholar] [CrossRef]
- Futagami, S.; Yamawaki, H.; Agawa, S.; Ikeda, G.; Noda, H.; Kirita, K.; Higuchi, K.; Gudis, K.; Murakami, M.; Kodaka, Y.; et al. Comparison of Functional Dyspepsia and Early Chronic Pancreatitis. J. Nippon. Med. Sch. 2020, 87, 2–6. [Google Scholar]
- Futagami, S.; Wakabayashi, M. Pancreatic Dysfunction and Duodenal Inflammatory Responses Coordinate with Refractory Epigastric Pain Including Functional Dyspepsia: A Narrative Review. J. Nippon. Med. Sch. 2022, 89, 255–262. [Google Scholar]
- Moin, A.S.M.; Cory, M.; Choi, J.; Ong, A.; Dhawan, S.; Dry, S.M.; Butler, P.C.; Rizza, R.A.; Butler, A.E. Increased Chromogranin A-Positive Hormone-Negative Cells in Chronic Pancreatitis. J. Clin. Endocrinol. Metab. 2018, 103, 2126–2135. [Google Scholar] [CrossRef]
- Ferraro, S.; Borille, S.; Panteghini, M. Reference intervals for the Kryptor second-generation chromogranin A assay. Clin. Chem. Lab. Med. 2016, 54, e335–e337. [Google Scholar] [CrossRef]




| CgA < 50 (ng/mL) | CgA ≥ 50 (ng/mL) | p Value | |
|---|---|---|---|
| Serum CgA range | 29~35 | 60~132 | <0.01 |
| Total no. of cases | N = 4 | N = 11 | # |
| Age (years) | 37~55 | 52~68 | <0.01 |
| Gender (male/female) | 2/2 | 7/4 | # |
| Functional dyspepsia | 1 | 5 | 0.46 |
| Duodenal ulcers | 2 | 0 | 0.05 |
| Gallstone | 2 | 2 | 0.27 |
| Pancreatic fibrosis | 0 | 3 | 0.36 |
| Chronic pancreatitis | 0 | 1 | 0.73 |
| Diabetes mellitus | 0 | 2 | # |
| H. pylori infection (pathology) | 2 | 1 | # |
| Renal insufficiency | 0 | 0 | # |
| Atrophic gastritis and intestinal metaplasia (pathology) | 1 | 4 | # |
| Proton pump inhibitor use | 1 | 1 | # |
| Classification | p Value | |
|---|---|---|
| CgA < 50 ng/mL group (biopsy specimen n = 12) | CgA > 50 ng/mL group (biopsy specimen n = 33) | |
| Discrete, fewer 83.3% (10/12) | Centralized, clustered 84.8% (28/33) | <0.01 |
| Consistency of the same individual | Consistency of the same individual | |
| 3 of 3 specimens: 2 cases | 3 of 3 specimens: 6 cases | |
| 2 of 3 specimens: 2 cases | 2 of 3 specimens: 5 cases | |
| CgA comparison between specific diseases | ||
| Duodenal ulcer (n = 2) | Pancreatic fibrosis, and chronic pancreatitis (n = 4) | |
| 29, 35 | 62, 78, 119, 121 | <0.01 |
EEC distribution pattern | EEC distribution pattern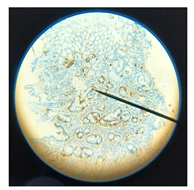 | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Huang, C.-T.; Liang, Y.-J. Elevated Serum Chromogranin-A and Characteristic Duodenal Enteroendocrine Cell Distribution in Pancreatic Fibrosis and Chronic Pancreatitis Compared with Other Dyspeptic Disorders: A Case Series Study. Diagnostics 2025, 15, 2428. https://doi.org/10.3390/diagnostics15192428
Huang C-T, Liang Y-J. Elevated Serum Chromogranin-A and Characteristic Duodenal Enteroendocrine Cell Distribution in Pancreatic Fibrosis and Chronic Pancreatitis Compared with Other Dyspeptic Disorders: A Case Series Study. Diagnostics. 2025; 15(19):2428. https://doi.org/10.3390/diagnostics15192428
Chicago/Turabian StyleHuang, Chung-Tsui, and Yao-Jen Liang. 2025. "Elevated Serum Chromogranin-A and Characteristic Duodenal Enteroendocrine Cell Distribution in Pancreatic Fibrosis and Chronic Pancreatitis Compared with Other Dyspeptic Disorders: A Case Series Study" Diagnostics 15, no. 19: 2428. https://doi.org/10.3390/diagnostics15192428
APA StyleHuang, C.-T., & Liang, Y.-J. (2025). Elevated Serum Chromogranin-A and Characteristic Duodenal Enteroendocrine Cell Distribution in Pancreatic Fibrosis and Chronic Pancreatitis Compared with Other Dyspeptic Disorders: A Case Series Study. Diagnostics, 15(19), 2428. https://doi.org/10.3390/diagnostics15192428







