Optical Coherence Tomography-Guided vs. Angiography-Guided Percutaneous Coronary Intervention for Complex Coronary Lesions: A Systematic Review and Meta-Analysis
Abstract
1. Introduction
2. Materials and Methods
2.1. Data Sources and Search Strategy
2.2. Study Selection
2.3. Data Extraction
2.4. Outcomes of Interest
2.5. Assessment of Study Quality
2.6. Statistical Analysis
| Trial Name | Country | Definition of MACE | Definition of Complex Lesion | Primary Outcome/Major Outcomes |
|---|---|---|---|---|
| ILLUMEN IV [12] | Europe, North America, Asia, Oceania | The Composite of Cardiac Death, Target-Vessel MI, or Definite/Probable Stent Thrombosis | Long or Multiple Lesions with intended Total Stent Length more than or equal to 28 mm, Bifurcation Lesion with Stenting Intended in both the Main and Side Branches more than or equal to 2.5 mm in Diameter, Severe Target Lesion Calcification, defined as Angiographically Visible Calcification on both sides of the Vessel Wall in the absence of Cardiac Motion, Chronic Total Occlusion (CTO) or Diffuse or Multifocal In-Stent Restenosis. | Final Post-PCI Minimal Stent Area (MSA) assessed by OCT in both groups, The 2-Year Outcome of MACE, The 2-Year Effectiveness Outcome of Target-Vessel Failure (TVF). |
| OCTOBER [13] | Europe | Composite of Death from a Cardiac Cause, Target Lesion Myocardial Infarction, or ischemia-Driven Target Lesion Revascularization at a median follow-up of 2 years. | Complex Coronary Artery Bifurcation Lesion. | MACE |
| OCCUPI [14] | South Korea | Composite of Cardiac Death, Myocardial Infarction, Stent Thrombosis, or Ischaemia-Driven Target-Vessel Revascularization 1 Year after PCI. | Acute Myocardial Infarction, Chronic Total Occlusion, Long Lesion [Expected Stent Length ≥28 mm based on Angiography], Calcified Lesion, Bifurcation Lesion, Unprotected Left Main Disease, Small Vessel Diseases [Vessel Diameter <2.5 mm], Intracoronary Thrombus visible on Angiography, Stent Thrombosis, In-Stent Restenosis or Bypass Graft Lesion. | MACE |
| CALIPSO [15] | France | Composite of Cardiovascular death, any Myocardial Infarction or need for Clinically Driven Reintervention on the Target Lesion. | Stable Moderate-to-Severe Calcified Coronary Lesions on Coronary Angiography Scheduled for PCI. | Minimal Stent Area (MSA) on the Qualifying OCT run. |
| RENOVATE- COMPLEX- PCI [16,17] | South Korea | - | Complex Coronary Artery Lesions were defined as true Bifurcation Lesions according to the Medina classification system12 with a Side-Branch Diameter of at least 2.5 mm; a Chronic Total Occlusion; Unprotected Left Main Coronary Artery Disease; Long Coronary Artery Lesions that would involve an expected Stent Length of at least 38 mm; Multi Vessel PCI involving at least two Major Epicardial Coronary Arteries being treated at the same time; a lesion that would necessitate the use of multiple stents (at least three planned stents); a lesion involving In-Stent Restenosis; a Severely Calcified Lesion; or Ostial Lesions of a Major Epicardial Coronary Artery. | The primary end point was Target-Vessel Failure, which was defined as a composite of death from Cardiac Causes, Target-Vessel–Related Myocardial Infarction, or Clinically Driven Target-Vessel Revascularization |
3. Results
3.1. Study Characteristics and Baseline Demographics
3.2. Quality Assessment
3.3. Primary Endpoints
3.4. Secondary Endpoints
4. Discussion
Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| ACS | Acute coronary syndrome |
| BMI | Body mass index |
| CTO | Chronic total occlusion |
| DM | Diabetes mellitus |
| HTN | Hypertension |
| LVEF | Left ventricular ejection fraction. |
| MACEs | Major adverse cardiovascular events |
| MI | Myocardial infarction |
| OCT | Optical coherence tomography |
| PCI | Percutaneous coronary intervention |
| RCTs | Randomized controlled trials |
| RR | Risk ratio |
| STEMI | ST-elevation myocardial infarction |
| TLR | Target lesion revascularization |
| CKD | Chronic kidney disease |
| CA-AKI | Contrast-associated acute kidney injury |
References
- Risk Factors for Coronary Artery Disease—PubMed. Available online: https://pubmed.ncbi.nlm.nih.gov/32119297/ (accessed on 13 October 2024).
- Iftikhar, S.F.; Bishop, M.A.; Hu, P. Complex Coronary Artery Lesions. StatPearls. Available online: https://www.ncbi.nlm.nih.gov/books/NBK539899/ (accessed on 13 October 2024).
- Abubakar, M.; Javed, I.; Rasool, H.F.; Raza, S.; Basavaraju, D.; Abdullah, R.M.; Ahmed, F.; Salim, S.S.; Faraz, M.A.; Hassan, K.M.; et al. Advancements in Percutaneous Coronary Intervention Techniques: A Comprehensive Literature Review of Mixed Studies and Practice Guidelines. Cureus 2023, 15, e41311. Available online: https://www.cureus.com/articles/167429-advancements-in-percutaneous-coronary-intervention-techniques-a-comprehensive-literature-review-of-mixed-studies-and-practice-guidelines (accessed on 13 October 2024). [CrossRef] [PubMed]
- Kubo, T.; Shinke, T.; Okamura, T.; Hibi, K.; Nakazawa, G.; Morino, Y.; Shite, J.; Ino, Y.; Kitabata, H.; Shimokawa, T.; et al. Comparison between Optical Coherence tomography guidance and Angiography guidance in percutaneous coronary intervention (COCOA): Study protocol for a randomized controlled trial. J. Cardiol. 2018, 72, 170–175. [Google Scholar] [CrossRef]
- Ghafari, C.; Carlier, S. Stent visualization methods to guide percutaneous coronary interventions and assess long-term patency. World J. Cardiol. 2021, 13, 416. Available online: https://pmc.ncbi.nlm.nih.gov/articles/PMC8462039/ (accessed on 13 October 2024). [CrossRef] [PubMed]
- Tu, S.; Jing, J.; Holm, N.R.; Onsea, K.; Zhang, T.; Adriaenssens, T.; Dubois, C.; Desmet, W.; Thuesen, L.; Chen, Y.; et al. In vivo assessment of bifurcation optimal viewing angles and bifurcation angles by three-dimensional (3D) quantitative coronary angiography. Int. J. Cardiovasc. Imaging 2012, 28, 1617–1625. Available online: https://pubmed.ncbi.nlm.nih.gov/22169957/ (accessed on 13 October 2024). [CrossRef] [PubMed]
- Baumgart, D.; Haude, M.; Von Birgelen, C.; Ge, J.; Gorge, G.; Erbel, R. Assessment of ambiguous coronary lesions by intravascular ultrasound. Int. J. Cardiovasc. Intervent. 1999, 2, 3–12. Available online: https://pubmed.ncbi.nlm.nih.gov/12623381/ (accessed on 13 October 2024). [CrossRef] [PubMed]
- Ali, Z.A.; Landmesser, U.; Maehara, A.; Matsumura, M.; Shlofmitz, R.A.; Guagliumi, G.; Price, M.J.; Hill, J.M.; Akasaka, T.; Prati, F.; et al. Optical Coherence Tomography–Guided versus Angiography-Guided PCI. N. Engl. J. Med. 2023, 389, 1466–1476. [Google Scholar] [CrossRef]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. BMJ 2021, 372. Available online: https://www.bmj.com/content/372/bmj.n71 (accessed on 28 July 2024).
- RoB 2: A Revised Cochrane Risk-of-Bias Tool for Randomized Trials|Cochrane Bias [Internet]. Available online: https://methods.cochrane.org/bias/resources/rob-2-revised-cochrane-risk-bias-tool-randomized-trials (accessed on 27 September 2023).
- Higgins, J.P.T.; Thompson, S.G.; Deeks, J.J.; Altman, D.G. Measuring inconsistency in meta-analyses. BMJ 2003, 327, 557–560. Available online: https://www.bmj.com/content/327/7414/557 (accessed on 28 July 2024). [CrossRef] [PubMed]
- Ali, Z.A.; Landmesser, U.; Maehara, A.; Shin, D.; Sakai, K.; Matsumura, M.; Shlofmitz, R.A.; Leistner, D.; Canova, P.; Alfonso, F.; et al. OCT-Guided vs Angiography-Guided Coronary Stent Implantation in Complex Lesions: An ILUMIEN IV Substudy. J. Am. Coll. Cardiol. 2024, 84, 368–378. Available online: https://pubmed.ncbi.nlm.nih.gov/38759907/ (accessed on 13 October 2024). [CrossRef]
- Holm, N.R.; Andreasen, L.N.; Neghabat, O.; Laanmets, P.; Kumsars, I.; Bennett, J.; Olsen, N.T.; Odenstedt, J.; Hoffmann, P.; Dens, J.; et al. OCT or Angiography Guidance for PCI in Complex Bifurcation Lesions. N. Engl. J. Med. 2023, 389, 1477–1487. Available online: https://www.nejm.org/doi/full/10.1056/NEJMoa2307770 (accessed on 13 October 2024). [CrossRef]
- Hong, S.J.; Lee, S.J.; Lee, S.H.; Lee, J.Y.; Cho, D.K.; Kim, J.W.; Kim, S.M.; Hur, S.H.; Heo, J.H.; Jang, J.Y.; et al. Optical coherence tomography-guided versus angiography-guided percutaneous coronary intervention for patients with complex lesions (OCCUPI): An investigator-initiated, multicentre, randomised, open-label, superiority trial in South Korea. Lancet 2024, 404, 1029–1039. Available online: http://www.thelancet.com/article/S0140673624014545/fulltext (accessed on 13 October 2024). [CrossRef]
- Amabile, N.; Rangé, G.; Landolff, Q.; Bressollette, E.; Meneveau, N.; Lattuca, B.; Levesque, S.; Boueri, Z.; Adjedj, J.; Casassus, F.; et al. OCT vs Angiography for Guidance of Percutaneous Coronary Intervention of Calcified Lesions: The CALIPSO Randomized Clinical Trial. JAMA Cardiol. 2025, 10, 666–675. Available online: https://jamanetwork.com/journals/jamacardiology/fullarticle/2832995 (accessed on 22 June 2025). [CrossRef]
- Lee, J.M.; Kim, H.; Lee, J.Y.; Choi, K.H.; Song, Y.B.; Lee, S.J.; Lee, S.Y.; Kim, S.M.; Yun, K.H.; Cho, J.Y.; et al. Optical Coherence Tomography Compared With Intravascular Ultrasound and Angiography in Complex Coronary Artery Lesions. JACC Cardiovasc. Imaging 2024, 17, 336–338. Available online: https://pubmed.ncbi.nlm.nih.gov/37943231/ (accessed on 22 June 2025). [CrossRef]
- Lee, J.M.; Choi, K.H.; Bin Song, Y.; Lee, J.-Y.; Lee, S.-J.; Lee, S.Y.; Kim, S.M.; Yun, K.H.; Cho, J.Y.; Kim, C.J.; et al. Intravascular Imaging–Guided or Angiography-Guided Complex PCI. N. Engl. J. Med. 2023, 388, 1668–1679. Available online: https://www.nejm.org/doi/pdf/10.1056/NEJMoa2216607 (accessed on 22 June 2025). [CrossRef]
- Hong, D.; Kim, S.M.; Lee, S.Y.; Choi, K.H.; Bin Song, Y.; Lee, J.-Y.; Lee, S.-J.; Yun, K.H.; Cho, J.Y.; Kim, C.J.; et al. Prognostic Impact of Intravascular Imaging-Guided Percutaneous Coronary Intervention in Chronic Total Occlusion. Circulation 2023, 148, 903–905. Available online: https://www.ahajournals.org/doi/10.1161/CIRCULATIONAHA.123.065876 (accessed on 13 October 2024). [CrossRef] [PubMed]
- Tian, N.-L.; Gami, S.-K.; Ye, F.; Zhang, J.-J.; Liu, Z.-Z.; Lin, S.; Ge, Z.; Shan, S.-J.; You, W.; Chen, L.; et al. Angiographic and clinical comparisons of intravascular ultrasound- versus angiography-guided drug-eluting stent implantation for patients with chronic total occlusion lesions: Two-year results from a randomised AIR-CTO study. EuroIntervention 2015, 10, 1409–1417. Available online: https://pubmed.ncbi.nlm.nih.gov/25912391/ (accessed on 13 October 2024). [CrossRef]
- Kim, B.-K.; Shin, D.-H.; Hong, M.-K.; Park, H.S.; Rha, S.-W.; Mintz, G.S.; Kim, J.-S.; Kim, J.S.; Lee, S.-J.; Kim, H.-Y.; et al. Clinical Impact of Intravascular Ultrasound-Guided Chronic Total Occlusion Intervention With Zotarolimus-Eluting Versus Biolimus-Eluting Stent Implantation: Randomized Study. Circ. Cardiovasc. Interv. 2015, 8, e002592. Available online: https://pubmed.ncbi.nlm.nih.gov/26156151 (accessed on 13 October 2024). [CrossRef]
- Gao, X.-F.; Ge, Z.; Kong, X.-Q.; Kan, J.; Han, L.; Lu, S.; Tian, N.-L.; Lin, S.; Lu, Q.-H.; Wang, X.-Y.; et al. 3-Year Outcomes of the ULTIMATE Trial Comparing Intravascular Ultrasound Versus Angiography-Guided Drug-Eluting Stent Implantation. JACC Cardiovasc. Interv. 2021, 14, 247–257. Available online: https://pubmed.ncbi.nlm.nih.gov/33541535/ (accessed on 13 October 2024). [CrossRef]
- Hong, S.J.; Kim, B.K.; Shin, D.H.; Nam, C.M.; Kim, J.S.; Ko, Y.G.; Choi, D.; Kang, T.S.; Kang, W.C.; Her, A.Y.; et al. Effect of Intravascular Ultrasound-Guided vs Angiography-Guided Everolimus-Eluting Stent Implantation: The IVUS-XPL Randomized Clinical Trial. JAMA 2015, 314, 2155–2163. Available online: https://pubmed.ncbi.nlm.nih.gov/26556051/ (accessed on 13 October 2024). [CrossRef] [PubMed]
- Hong, S.J.; Mintz, G.S.; Ahn, C.M.; Kim, J.S.; Kim, B.K.; Ko, Y.G.; Kang, T.S.; Kang, W.C.; Kim, Y.H.; Hur, S.H.; et al. Effect of Intravascular Ultrasound-Guided Drug-Eluting Stent Implantation: 5-Year Follow-Up of the IVUS-XPL Randomized Trial. JACC Cardiovasc. Interv. 2020, 13, 62–71. Available online: https://pubmed.ncbi.nlm.nih.gov/31918944/ (accessed on 13 October 2024). [CrossRef] [PubMed]
- Gupta, A.; Shrivastava, A.; Vijayvergiya, R.; Chhikara, S.; Datta, R.; Aziz, A.; Meena, D.S.; Nath, R.K.; Kumar, J.R. Optical Coherence Tomography: An Eye Into the Coronary Artery. Front. Cardiovasc. Med. 2022, 9, 854554. Available online: https://pmc.ncbi.nlm.nih.gov/articles/PMC9130606/ (accessed on 13 October 2024). [CrossRef] [PubMed]
- Tea, I.; Gilani, S.; Kleiman, N.S. When Prevention is Truly Better than Cure: Contrast-Associated Acute Kidney Injury in Percutaneous Coronary Intervention. Methodist DeBakey Cardiovasc. J. 2022, 18, 73–85. [Google Scholar] [CrossRef]
- Kurogi, K.; Ishii, M.; Sakamoto, K.; Kusaka, H.; Yamamoto, N.; Takashio, S.; Arima, Y.; Yamamoto, E.; Kaikita, K.; Tsujita, K. Minimum-Contrast Percutaneous Coronary Intervention Guided by Optical Coherence Tomography Using Low–Molecular Weight Dextran. JACC Cardiovasc. Interv. 2020, 13, 1270–1272. [Google Scholar] [CrossRef]
- Almajid, F.; Kang, D.-Y.; Ahn, J.-M.; Park, S.-J.; Park, D.-W. Optical coherence tomography to guide percutaneous coronary intervention. EuroIntervention 2024, 20, e1202–e1216. [Google Scholar] [CrossRef] [PubMed]
- Hong, S.J.; Zhang, J.J.; Mintz, G.S.; Ahn, C.M.; Kim, J.S.; Kim, B.K.; Ko, Y.G.; Choi, D.; Jang, Y.; Kan, J.; et al. Improved 3-Year Cardiac Survival After IVUS-Guided Long DES Implantation: A Patient-Level Analysis From 2 Randomized Trials. JACC Cardiovasc. Interv. 2022, 15, 208–216. Available online: https://pubmed.ncbi.nlm.nih.gov/35057991/ (accessed on 13 October 2024). [CrossRef]
- Sreenivasan, J.; Reddy, R.K.; Jamil, Y.; Malik, A.; Chamie, D.; Howard, J.P.; Nanna, M.G.; Mintz, G.S.; Maehara, A.; Ali, Z.A.; et al. Intravascular Imaging-Guided Versus Angiography-Guided Percutaneous Coronary Intervention: A Systematic Review and Meta-Analysis of Randomized Trials. J. Am. Heart Assoc. 2024, 13, e031111. Available online: https://pubmed.ncbi.nlm.nih.gov/38214263/ (accessed on 13 October 2024). [CrossRef] [PubMed]
- Khan, S.U.; Agarwal, S.; Arshad, H.B.; Akbar, U.A.; Mamas, M.A.; Arora, S.; Baber, U.; Goel, S.S.; Kleiman, N.S.; Shah, A.R. Intravascular imaging guided versus coronary angiography guided percutaneous coronary intervention: Systematic review and meta-analysis. BMJ 2023, 383, e077848. Available online: https://www.bmj.com/content/383/bmj-2023-077848 (accessed on 13 October 2024). [CrossRef] [PubMed]
- Attar, A.; Hosseinpour, A.; Azami, P.; Kohansal, E.; Javaheri, R. Clinical outcomes of optical coherence tomography versus conventional angiography guided percutaneous coronary intervention: A meta-analysis. Curr. Probl. Cardiol. 2024, 49, 102224. Available online: https://pubmed.ncbi.nlm.nih.gov/38040219/ (accessed on 13 October 2024). [CrossRef]
- Ahmed, M.; Javaid, H.; Maniya, M.T.; Shafiq, A.; Shahbaz, H.; Singh, P.; Jain, H.; Basit, J.; Hamza, M.; Nashwan, A.J.; et al. Optical coherence tomography-guided versus angiography-guided percutaneous coronary intervention: A meta-analysis of randomized controlled trials. IJC Heart Vasc. 2024, 52, 101405. Available online: http://creativecommons.org/licenses/by/4.0/ (accessed on 13 October 2024). [CrossRef]
- Ishihara, T.; Okada, K.; Kida, H.; Tsujimura, T.; Iida, O.; Okuno, S.; Hata, Y.; Toyoshima, T.; Higashino, N.; Kikuchi, A.; et al. Long-Term Outcomes and Clinical Predictors of Mortality Following Occurrence of Stent Thrombosis. J. Am. Heart Assoc. Cardiovasc. Cerebrovasc. Dis. 2022, 11, 23276. Available online: https://pubmed.ncbi.nlm.nih.gov/35377181/ (accessed on 13 October 2024). [CrossRef]
- Bae, Y.; Kang, S.-J.; Kim, G.; Lee, J.-G.; Min, H.-S.; Cho, H.; Kang, D.-Y.; Lee, P.H.; Ahn, J.-M.; Park, D.-W.; et al. Prediction of coronary thin-cap fibroatheroma by intravascular ultrasound-based machine learning. Atherosclerosis 2019, 288, 168–174. Available online: https://pubmed.ncbi.nlm.nih.gov/31130215/ (accessed on 9 December 2024). [CrossRef]




| Study ID, Year | Clinical Trial Identifier | Number of Patients | Mean Age; Mean ± SD or Median (IQR) | Female Sex, n (%) | BMI; Mean ± SD or Median (IQR) | DM; n (%) | HTN; n (%) | Smoking; n (%) | LVEF; Mean (SD) or Median (IQR) | Follow-Up (Months) | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| OCT | Angiography | OCT | Angiography | OCT | Angiography | OCT | Angiography | OCT | Angiography | OCT | Angiography | OCT | Angiography | OCT | Angiography | |||
| ILUMIEN IV [12] | NCT03507777 | 992 | 981 | 65.6 ± 10.5 | 65.7 ± 10.4 | 200 (20.2) | 231 (23.5) | - | - | 360 (36.3) | 342 (34.9) | 707 (71.3) | 734 (74.8) | 198 (20) | 192 (19.6) | 55.1 (8.5) | 55 (8.7) | 2 years |
| OCTOBER [13] | NCT03171311 | 600 | 601 | 66.4 ± 10.5 | 66.2 ± 9.9 | 127 (21.2) | 126 (21.0) | 28.0 ± 4.6 | 28.2 ± 4.9 | 103 (17.2) | 97 (16.1) | 422 (70.3) | 448 (74.5) | 77 (12.8) | 85 (14.1) | 59.5 (50–60) | 58.0 (50–60) | 2 years |
| OCCUPI [14] | NCT03625908 | 803 | 801 | 64 (57−70) | 64 (58−70) | 157 (20%) | 157 (20%) | 24.8 (23.0−26.6) | 24.6 (22.9−26.6) | 261 (33%) | 262 (33%) | 466 (58%) | 451 (56%) | 149 (19%) | 158 (20%) | 59·5% (8·8) | 59·7% (10·1) | 1 year |
| Calipso Trial [15] | NCT05301218 | 65 | 69 | 72.0 (65.0−76.5) | 74 (68.5−79.0) | 13 (20) | 12 (17) | 26.8 (24.6–28.9) | 26.0 (24.0–29.1) | 24 (37) | 27 (39) | 46 (71) | 43 (62) | 8 (12) | 10 (15) | 60 (50–65) | 60 (50–64) | 1 month and 12 months |
| Renovate Complex PCI [16,17] * | NCT03381872 | 278 | 547 | 65.3 | 66 | 20.4 | 21.2 | - | - | 36.1 | 40.8 | 62.5 | 59 | 19.4 | 17.4 | 58.4 | 59.3 | 2.1 years |
| Outcome | Studies | Effect Estimate (95% CI) | p Value | Figure | Interpretation |
|---|---|---|---|---|---|
| Major Adverse Cardiovascular Events (MACEs) | 5 | RR = 0.71 (0.59–0.84) | 0.0001 | Figure 3a | OCT-guided PCI reduced MACE risk by 29% |
| Cardiac Mortality | 4 | RR = 0.43 (0.24–0.76) | 0.003 | Figure 3b | OCT-guided PCI lowered cardiac mortality by 57% |
| Target Lesion Revascularization (TLR) | 3 | RR = 0.53 (0.33–0.84) | 0.007 | Figure 3c | OCT-guided PCI decreased TLR by 47% |
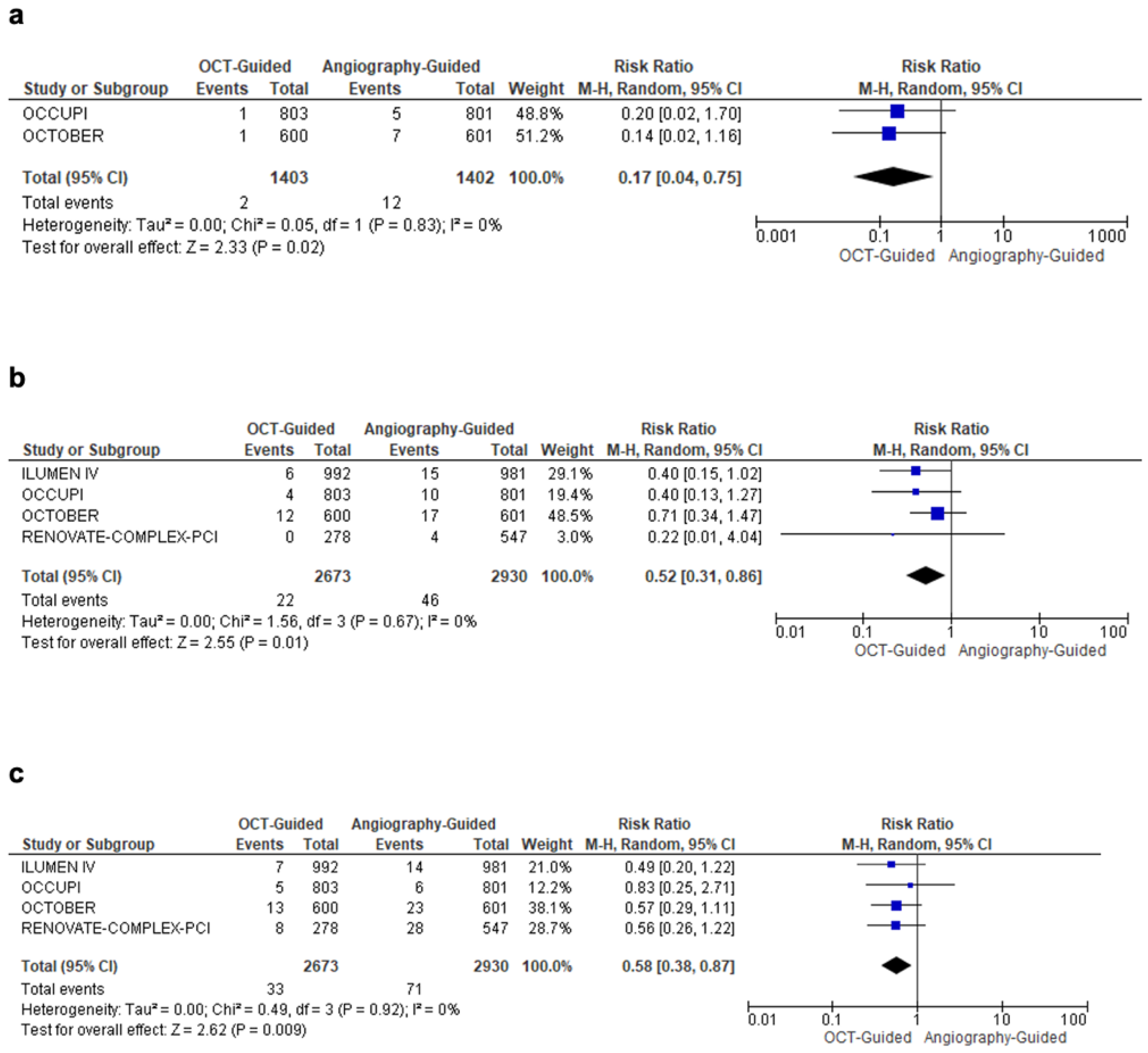
| Stroke | 2 | RR = 0.17 (0.04–0.75) | 0.02 | Figure 4a | OCT-guided PCI reduced stroke risk by 83% |
| Stent Thrombosis | 4 | RR = 0.52 (0.31–0.86) | 0.01 | Figure 4b | OCT-guided PCI lowered stent thrombosis by 48% |
| All-Cause Mortality | 4 | RR = 0.58 (0.38–0.87) | 0.009 | Figure 4c | OCT-guided PCI reduced all-cause mortality by 42% |
| Procedural Duration | 4 | MD = 16.14 min (6.67–25.61) | 0.0008 | Figure S1 | OCT-guided PCI prolonged procedure time by ~16 min |
| Ischemia-Driven TVR | 4 | RR = 0.67 (0.44–1.03) | 0.07 | Figure S2 | No significant reduction (trend toward benefit) |
| Minimal Stent Area (MSA) | 2 | MD = 0.93 (–0.37–2.23) | 0.16 | Figure S3 | No significant improvement in MSA |
| Myocardial Infarction (MI) | 4 | RR = 0.77 (0.57–1.03) | 0.07 | Figure S4 | No significant reduction in MI |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shuja, M.H.; Ahmed, M.; Hannat, R.; Khurram, L.; Hasnain Sheikh, H.A.; Shuja, S.H.; Raja, A.; Ahmed, J.; Soni, K.; Wani, S.A.; et al. Optical Coherence Tomography-Guided vs. Angiography-Guided Percutaneous Coronary Intervention for Complex Coronary Lesions: A Systematic Review and Meta-Analysis. Diagnostics 2025, 15, 1907. https://doi.org/10.3390/diagnostics15151907
Shuja MH, Ahmed M, Hannat R, Khurram L, Hasnain Sheikh HA, Shuja SH, Raja A, Ahmed J, Soni K, Wani SA, et al. Optical Coherence Tomography-Guided vs. Angiography-Guided Percutaneous Coronary Intervention for Complex Coronary Lesions: A Systematic Review and Meta-Analysis. Diagnostics. 2025; 15(15):1907. https://doi.org/10.3390/diagnostics15151907
Chicago/Turabian StyleShuja, Muhammad Hamza, Muhammad Ahmed, Ramish Hannat, Laiba Khurram, Hamza Ali Hasnain Sheikh, Syed Hasan Shuja, Adarsh Raja, Jawad Ahmed, Kriti Soni, Shariq Ahmad Wani, and et al. 2025. "Optical Coherence Tomography-Guided vs. Angiography-Guided Percutaneous Coronary Intervention for Complex Coronary Lesions: A Systematic Review and Meta-Analysis" Diagnostics 15, no. 15: 1907. https://doi.org/10.3390/diagnostics15151907
APA StyleShuja, M. H., Ahmed, M., Hannat, R., Khurram, L., Hasnain Sheikh, H. A., Shuja, S. H., Raja, A., Ahmed, J., Soni, K., Wani, S. A., Goyal, A., Pushparaji, B., Hasan, A., Ahmed, R., & Jain, H. (2025). Optical Coherence Tomography-Guided vs. Angiography-Guided Percutaneous Coronary Intervention for Complex Coronary Lesions: A Systematic Review and Meta-Analysis. Diagnostics, 15(15), 1907. https://doi.org/10.3390/diagnostics15151907






