The Role of Neutrophil Extracellular Traps in the Outcome of Malignant Epitheliomas: Significance of CA215 Involvement
Abstract
1. Introduction
2. Tumor Microenvironment
3. NETs as Part of the TME
4. The Roles of NETs in Cancers
4.1. NETs Facilitate the Endothelial-to-Mesenchymal Transition (EndMT)
4.2. NETs Are Shown to Have a Positive Effect on Tumor Progression, Invasion, and Growth
4.3. NETs Have a Positive Angiogenic Effect
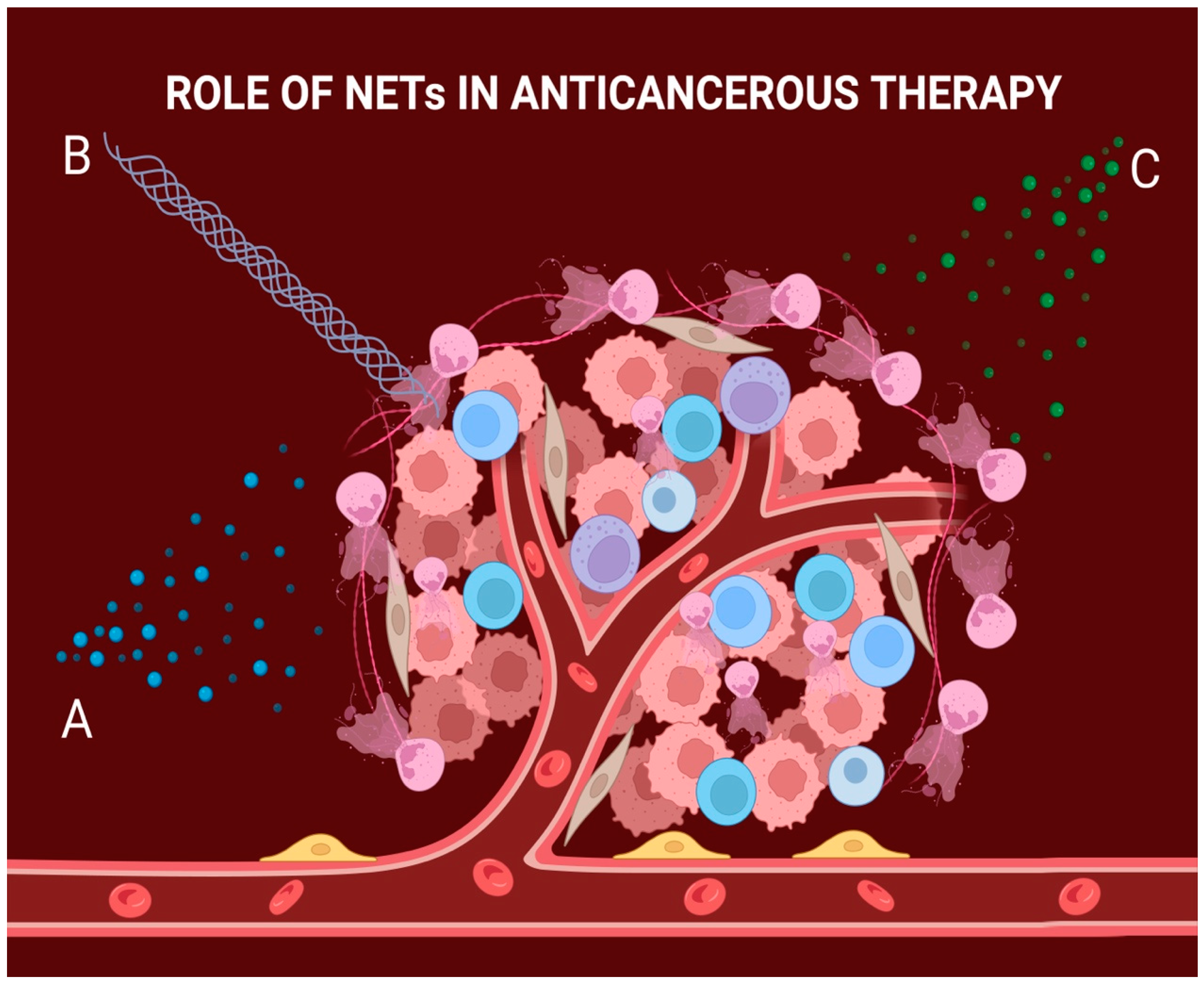
5. NETs Can Severely Affect the Outcome of Cancer Therapy
5.1. NETs Can Provide Resistance to Chemotherapy
5.2. NETs Can Provide Resistance to Immunotherapy
5.3. NETs Can Provide Resistance to Radiotherapy
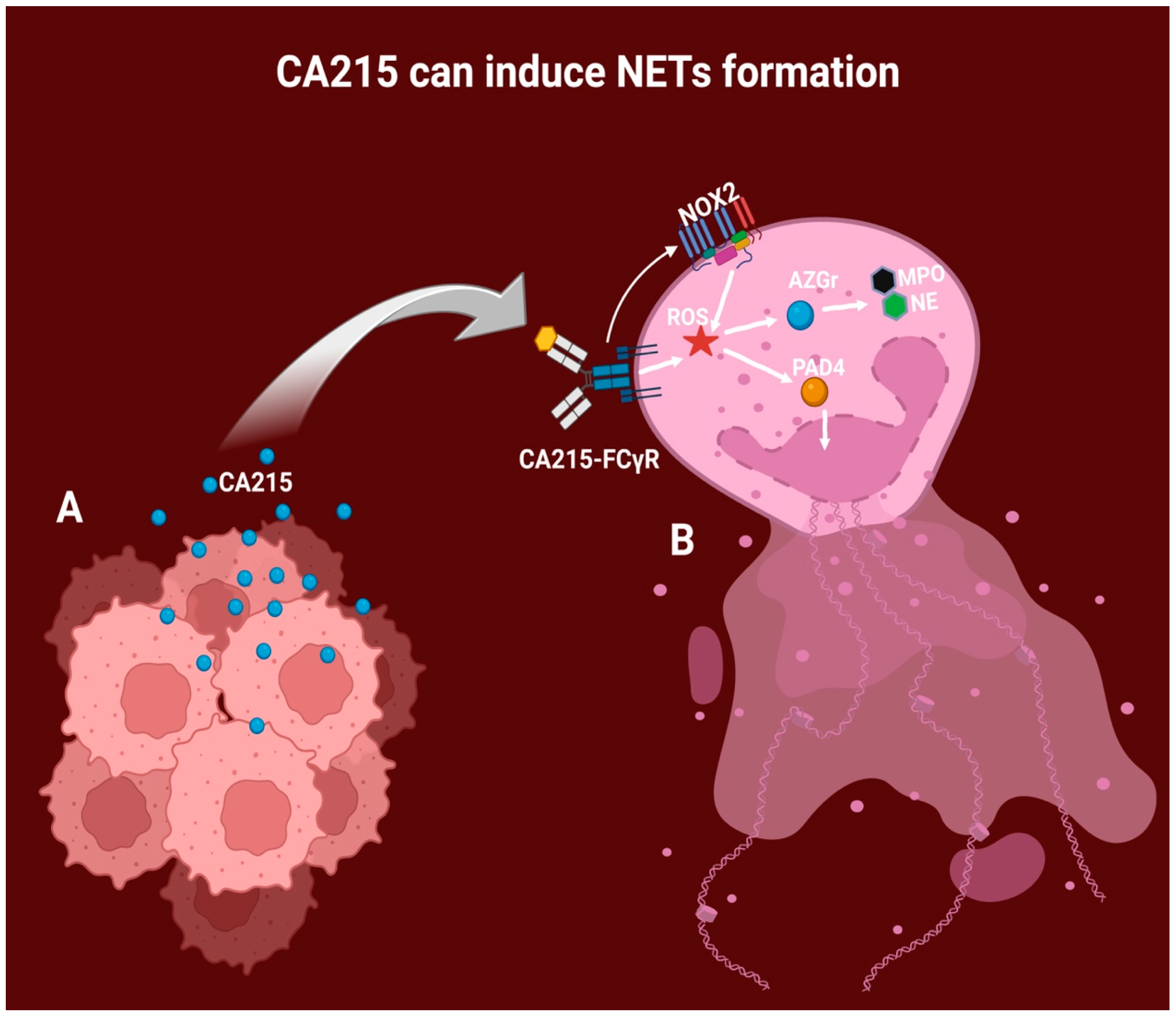
6. CA215 as an Immunoglobulin G Variant
7. Cancer Cells and CA215 Production
8. NETs Assessment
8.1. Quantitative Assays
8.2. Qualitative Assays
8.3. Conclusions and Future Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviation
References
- Brinkmann, V.; Reichard, U.; Goosmann, C.; Fauler, B.; Uhlemann, Y.; Weiss, D.S.; Weinrauch, Y.; Zychlinsky, A. Neutrophil Extracellular Traps Kill Bacteria. Science 2004, 303, 1532–1535. [Google Scholar] [CrossRef] [PubMed]
- Bedard, K.; Krause, K.H. The NOX Family of ROS-Generating NADPH Oxidases: Physiology and Pathophysiology. Physiol. Rev. 2007, 87, 245–313. [Google Scholar] [CrossRef] [PubMed]
- Lambeth, J.D. NOX Enzymes and the Biology of Reactive Oxygen. Nat. Rev. Immunol. 2004, 4, 181–189. [Google Scholar] [CrossRef] [PubMed]
- Rosales, C. Neutrophils at the Crossroads of Innate and Adaptive Immunity. J. Leukoc. Biol. 2020, 108, 377–396. [Google Scholar] [CrossRef] [PubMed]
- Vorobjeva, N.V.; Chernyak, B.V. NETosis: Molecular Mechanisms, Role in Physiology and Pathology. Biochemistry 2020, 85, 1178–1190. [Google Scholar] [CrossRef] [PubMed]
- Mbeunkui, F.; Johann, D.J. Cancer and the Tumor Microenvironment: A Review of an Essential Relationship. Cancer Chemother. Pharmacol. 2009, 63, 571–582. [Google Scholar] [CrossRef] [PubMed]
- Bissell, M.J.; Radisky, D. Putting Tumours in Context. Nat. Rev. Cancer 2001, 1, 46–54. [Google Scholar] [CrossRef]
- Rayes, R.F.; Mouhanna, J.G.; Nicolau, I.; Bourdeau, F.; Giannias, B.; Rousseau, S.; Quail, D.; Walsh, L.; Sangwan, V.; Bertos, N.; et al. Primary Tumors Induce Neutrophil Extracellular Traps with Targetable Metastasis-Promoting Effects. JCI Insight 2019, 4, e128008. [Google Scholar] [CrossRef]
- Quail, D.F.; Joyce, J.A. Microenvironmental Regulation of Tumor Progression and Metastasis. Nat. Med. 2013, 19, 1423–1437. [Google Scholar] [CrossRef]
- Hanahan, D.; Coussens, L.M. Accessories to the Crime: Functions of Cells Recruited to the Tumor Microenvironment. Cancer Cell 2012, 21, 309–322. [Google Scholar] [CrossRef]
- Szczerba, B.M.; Castro-Giner, F.; Vetter, M.; Krol, I.; Gkountela, S.; Landin, J.; Scheidmann, M.C.; Donato, C.; Scherrer, R.; Singer, J.; et al. Neutrophils Escort Circulating Tumour Cells to Enable Cell Cycle Progression. Nature 2019, 566, 553–557. [Google Scholar] [CrossRef] [PubMed]
- Jaillon, S.; Ponzetta, A.; Di Mitri, D.; Santoni, A.; Bonecchi, R.; Mantovani, A. Neutrophil Diversity and Plasticity in Tumour Progression and Therapy. Nat. Rev. Cancer 2020, 20, 485–503. [Google Scholar] [CrossRef] [PubMed]
- Voisin, M.B.; Nourshargh, S. Neutrophil Trafficking to Lymphoid Tissues: Physiological and Pathological Implications. J. Pathol. 2019, 247, 662–671. [Google Scholar] [CrossRef]
- Demers, M.; Krause, D.S.; Schatzberg, D.; Martinod, K.; Voorhees, J.R.; Fuchs, T.A.; Scadden, D.T.; Wagner, D.D. Cancers Predispose Neutrophils to Release Extracellular DNA Traps That Contribute to Cancer-Associated Thrombosis. Proc. Natl. Acad. Sci. USA 2012, 109, 13076–13081. [Google Scholar] [CrossRef]
- Park, J.; Wysocki, R.W.; Amoozgar, Z.; Maiorino, L.; Fein, M.R.; Jorns, J.; Schott, A.F.; Kinugasa-Katayama, Y.; Lee, Y.; Won, N.H.; et al. Cancer Cells Induce Metastasis-Supporting Neutrophil Extracellular DNA Traps. Sci. Transl. Med. 2016, 8, 361ra138. [Google Scholar] [CrossRef]
- Ronchetti, L.; Boubaker, N.S.; Barba, M.; Vici, P.; Gurtner, A.; Piaggio, G. Neutrophil Extracellular Traps in Cancer: Not Only Catching Microbes. J. Exp. Clin. Cancer Res. 2021, 40, 231. [Google Scholar] [CrossRef]
- Veglia, F.; Tyurin, V.A.; Blasi, M.; De Leo, A.; Kossenkov, A.V.; Donthireddy, L.; To, T.K.J.; Schug, Z.; Basu, S.; Wang, F.; et al. Fatty Acid Transport Protein 2 Reprograms Neutrophils in Cancer. Nature 2019, 569, 73–78. [Google Scholar] [CrossRef] [PubMed]
- Cedervall, J.; Zhang, Y.; Huang, H.; Zhang, L.; Femel, J.; Dimberg, A.; Olsson, A.K. Neutrophil Extracellular Traps Accumulate in Peripheral Blood Vessels and Compromise Organ Function in Tumor-Bearing Animals. Cancer Res. 2015, 75, 2653–2662. [Google Scholar] [CrossRef]
- Hidalgo, A.; Libby, P.; Soehnlein, O.; Aramburu, I.V.; Papayannopoulos, V.; Silvestre-Roig, C. Neutrophil Extracellular Traps: From Physiology to Pathology. Cardiovasc. Res. 2022, 118, 2737–2753. [Google Scholar] [CrossRef]
- Snoderly, H.T.; Boone, B.A.; Bennewitz, M.F. Neutrophil Extracellular Traps in Breast Cancer and beyond: Current Perspectives on NET Stimuli, Thrombosis and Metastasis, and Clinical Utility for Diagnosis and Treatment. Breast Cancer Res. 2019, 21, 145. [Google Scholar] [CrossRef]
- Tohme, S.; Yazdani, H.O.; Al-Khafaji, A.B.; Chidi, A.P.; Loughran, P.; Mowen, K.; Wang, Y.; Simmons, R.L.; Huang, H.; Tsung, A. Neutrophil Extracellular Traps Promote the Development and Progression of Liver Metastases after Surgical Stress. Cancer Res. 2016, 76, 1367–1380. [Google Scholar] [CrossRef] [PubMed]
- Cristinziano, L.; Modestino, L.; Antonelli, A.; Marone, G.; Simon, H.U.; Varricchi, G.; Galdiero, M.R. Neutrophil Extracellular Traps in Cancer. Semin. Cancer Biol. 2022, 79, 91–104. [Google Scholar] [CrossRef] [PubMed]
- Kaltenmeier, C.; Simmons, R.L.; Tohme, S.; Yazdani, H.O. Neutrophil Extracellular Traps (Nets) in Cancer Metastasis. Cancers 2021, 13, 6131. [Google Scholar] [CrossRef] [PubMed]
- Söderberg, D.; Segelmark, M. Neutrophil Extracellular Traps in Vasculitis, Friend or Foe? Curr. Opin. Rheumatol. 2018, 30, 16–23. [Google Scholar] [CrossRef] [PubMed]
- Kaltenmeier, C.; Yazdani, H.O.; Morder, K.; Geller, D.A.; Simmons, R.L.; Tohme, S. Neutrophil Extracellular Traps Promote T Cell Exhaustion in the Tumor Microenvironment. Front. Immunol. 2021, 12, 785222. [Google Scholar] [CrossRef] [PubMed]
- Li, P.; Lu, M.; Shi, J.; Hua, L.; Gong, Z.; Li, Q.; Shultz, L.D.; Ren, G. Dual Roles of Neutrophils in Metastatic Colonization Are Governed by the Host NK Cell Status. Nat. Commun. 2020, 11, 4387. [Google Scholar] [CrossRef] [PubMed]
- Ribatti, D.; Tamma, R.; Annese, T. Epithelial-Mesenchymal Transition in Cancer: A Historical Overview. Transl. Oncol. 2020, 13, 100773. [Google Scholar] [CrossRef] [PubMed]
- Pieterse, E.; Rother, N.; Garsen, M.; Hofstra, J.M.; Satchell, S.C.; Hoffmann, M.; Loeven, M.A.; Knaapen, H.K.; Van Der Heijden, O.W.H.; Berden, J.H.M.; et al. Neutrophil Extracellular Traps Drive Endothelial-to-Mesenchymal Transition. Arterioscler. Thromb. Vasc. Biol. 2017, 37, 1371–1379. [Google Scholar] [CrossRef]
- van Buul, J.D.; van Alphen, F.P.; Hordijk, P.L. The Presence of Alpha-Catenin in the VE-Cadherin Complex Is Required for Efficient Transendothelial Migration of Leukocytes. Int. J. Biol. Sci. 2009, 5, 695–705. [Google Scholar] [CrossRef]
- Zhu, T.; Zou, X.; Yang, C.; Li, L.; Wang, B.; Li, R.; Li, H.; Xu, Z.; Huang, D.; Wu, Q. Neutrophil Extracellular Traps Promote Gastric Cancer Metastasis by Inducing Epithelial-Mesenchymal Transition. Int. J. Mol. Med. 2021, 48, 127. [Google Scholar] [CrossRef]
- Demers, M.; Wong, S.L.; Martinod, K.; Gallant, M.; Cabral, J.E.; Wang, Y.; Wagner, D.D. Priming of Neutrophils toward NETosis Promotes Tumor Growth. Oncoimmunology 2016, 5, e1134073. [Google Scholar] [CrossRef]
- Miller-Ocuin, J.L.; Liang, X.; Boone, B.A.; Doerfler, W.R.; Singhi, A.D.; Tang, D.; Kang, R.; Lotze, M.T.; Zeh, H.J. DNA Released from Neutrophil Extracellular Traps (NETs) Activates Pancreatic Stellate Cells and Enhances Pancreatic Tumor Growth. Oncoimmunology 2019, 8, e1605822. [Google Scholar] [CrossRef]
- Yang, L.Y.; Luo, Q.; Lu, L.; Zhu, W.W.; Sun, H.T.; Wei, R.; Lin, Z.F.; Wang, X.Y.; Wang, C.Q.; Lu, M.; et al. Increased Neutrophil Extracellular Traps Promote Metastasis Potential of Hepatocellular Carcinoma via Provoking Tumorous Inflammatory Response. J. Hematol. Oncol. 2020, 13, 3. [Google Scholar] [CrossRef]
- Milette, S.; Quail, D.F.; Spicer, J.D. Neutrophil DNA Webs Untangled. Cancer Cell 2020, 38, 164–166. [Google Scholar] [CrossRef]
- Yang, L.; Liu, Q.; Zhang, X.; Liu, X.; Zhou, B.; Chen, J.; Huang, D.; Li, J.; Li, H.; Chen, F.; et al. DNA of Neutrophil Extracellular Traps Promotes Cancer Metastasis via CCDC25. Nature 2020, 583, 133–138. [Google Scholar] [CrossRef]
- Masucci, M.T.; Minopoli, M.; Carriero, M.V. Tumor Associated Neutrophils. Their Role in Tumorigenesis, Metastasis, Prognosis and Therapy. Front. Oncol. 2019, 9, 1146. [Google Scholar] [CrossRef]
- Aldabbous, L.; Abdul-Salam, V.; Mckinnon, T.; Duluc, L.; Pepke-Zaba, J.; Southwood, M.; Ainscough, A.J.; Hadinnapola, C.; Wilkins, M.R.; Toshner, M.; et al. Neutrophil Extracellular Traps Promote Angiogenesis. Arterioscler. Thromb. Vasc. Biol. 2016, 36, 2078–2087. [Google Scholar] [CrossRef]
- Lavoie, S.S.; Dumas, E.; Vulesevic, B.; Neagoe, P.-E.; White, M.; Sirois, M.G. Synthesis of Human Neutrophil Extracellular Traps Contributes to Angiopoietin-Mediated In Vitro Proinflammatory and Proangiogenic Activities. J. Immunol. 2018, 200, 3801–3813. [Google Scholar] [CrossRef] [PubMed]
- Papayannopoulos, V. Neutrophil Extracellular Traps in Immunity and Disease. Nat. Rev. Immunol. 2018, 18, 134–147. [Google Scholar] [CrossRef] [PubMed]
- Webb, N.J.A.; Myers, C.R.; Watson, C.J.; Bottomley, M.J.; Brenchley, P.E.C. Activated Human Neutrophils Express Vascular Endothelial Growth Factor (VEGF). Cytokine 1998, 10, 254–257. [Google Scholar] [CrossRef] [PubMed]
- Gao, H.; Lan, X.; Li, S.; Xue, Y. Relationships of MMP-9, E-Cadherin, and VEGF Expression with Clinicopathological Features and Response to Chemosensitivity in Gastric Cancer. Tumor Biol. 2017, 39, 1010428317698368. [Google Scholar] [CrossRef]
- Scapini, P.; Calzetti, F.; Cassatella, M.A. On the Detection of Neutrophil-Derived Vascular Endothelial Growth Factor (VEGF). J. Immunol. Methods 1999, 232, 121–129. [Google Scholar] [CrossRef]
- Zhang, Y.; Hu, Y.; Ma, C.; Sun, H.; Wei, X.; Li, M.; Wei, W.; Zhang, F.; Yang, F.; Wang, H.; et al. Diagnostic, Therapeutic Predictive, and Prognostic Value of Neutrophil Extracellular Traps in Patients With Gastric Adenocarcinoma. Front. Oncol. 2020, 10, 1036. [Google Scholar] [CrossRef]
- Joshi, B.H.; Shahzad, M.H.; Feng, L.; Su, X.; Brassard, A.; Dhoparee-Doomah, I.; Ferri, L.E.; Spicer, J.D.; Cools-Lartigue, J.J. Neutrophil Extracellular Traps in Cancer Therapy Resistance. Cancers 2022, 14, 1359. [Google Scholar] [CrossRef]
- Jaboury, S.; Wang, K.; O’Sullivan, K.M.; Ooi, J.D.; Ho, G.Y. NETosis as an Oncologic Therapeutic Target: A Mini Review. Front. Immunol. 2023, 14, 1170603. [Google Scholar] [CrossRef]
- Ramachandran, I.R.; Condamine, T.; Lin, C.; Herlihy, S.E.; Garfall, A.; Vogl, D.T.; Gabrilovich, D.I.; Nefedova, Y. Bone Marrow PMN-MDSCs and Neutrophils Are Functionally Similar in Protection of Multiple Myeloma from Chemotherapy. Cancer Lett. 2016, 371, 117–124. [Google Scholar] [CrossRef] [PubMed]
- Lin, C.; Herlihy, S.E.; Li, M.; Deng, H.; Bernabei, L.; Gabrilovich, D.I.; Vogl, D.T.; Nefedova, Y. NETs Promote Tumor Resistance to Anthracyclines. Cancer Res. 2019, 79, 2103. [Google Scholar] [CrossRef]
- Heinhuis, K.M.; Ros, W.; Kok, M.; Steeghs, N.; Beijnen, J.H.; Schellens, J.H.M. Enhancing Anti-Tumor Response by Combining Immune Checkpoint Inhibitors with Chemotherapy in Solid Tumors. Ann. Oncol. 2019, 30, 219–235. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Liu, G.; Sun, M.; Lu, X. Targeting and Exploitation of Tumor-Associated Neutrophils to Enhance Immunotherapy and Drug Delivery for Cancer Treatment. Cancer Biol. Med. 2020, 17, 32–43. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Chandra, V.; Sanchez, E.R.; Dutta, P.; Quesada, P.R.; Rakoski, A.; Zoltan, M.; Arora, N.; Baydogan, S.; Horne, W.; et al. Interleukin-17-Induced Neutrophil Extracellular Traps Mediate Resistance to Checkpoint Blockade in Pancreatic Cancer. J. Exp. Med. 2020, 217, e20190354. [Google Scholar] [CrossRef]
- Teijeira, Á.; Garasa, S.; Gato, M.; Alfaro, C.; Migueliz, I.; Cirella, A.; de Andrea, C.; Ochoa, M.C.; Otano, I.; Etxeberria, I.; et al. CXCR1 and CXCR2 Chemokine Receptor Agonists Produced by Tumors Induce Neutrophil Extracellular Traps That Interfere with Immune Cytotoxicity. Immunity 2020, 52, 856–871. [Google Scholar] [CrossRef] [PubMed]
- Moding, E.J.; Kastan, M.B.; Kirsch, D.G. Strategies for Optimizing the Response of Cancer and Normal Tissues to Radiation. Nat. Rev. Drug Discov. 2013, 12, 526–542. [Google Scholar] [CrossRef] [PubMed]
- Wisdom, A.J.; Hong, C.S.; Lin, A.J.; Xiang, Y.; Cooper, D.E.; Zhang, J.; Xu, E.S.; Kuo, H.C.; Mowery, Y.M.; Carpenter, D.J.; et al. Neutrophils Promote Tumor Resistance to Radiation Therapy. Proc. Natl. Acad. Sci. USA 2019, 116, 18584–18589. [Google Scholar] [CrossRef] [PubMed]
- Shaul, M.E.; Fridlender, Z.G. Tumour-Associated Neutrophils in Patients with Cancer. Nat. Rev. Clin. Oncol. 2019, 16, 601–620. [Google Scholar] [CrossRef] [PubMed]
- Shinde-Jadhav, S.; Mansure, J.J.; Rayes, R.F.; Marcq, G.; Ayoub, M.; Skowronski, R.; Kool, R.; Bourdeau, F.; Brimo, F.; Spicer, J.; et al. Role of Neutrophil Extracellular Traps in Radiation Resistance of Invasive Bladder Cancer. Nat. Commun. 2021, 12, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Tadie, J.M.; Bae, H.B.; Jiang, S.; Park, D.W.; Bell, C.P.; Yang, H.; Pittet, J.F.; Tracey, K.; Thannickal, V.J.; Abraham, E.; et al. HMGB1 Promotes Neutrophil Extracellular Trap Formation through Interactions with Toll-like Receptor 4. Am. J. Physiol. Lung Cell. Mol. Physiol. 2013, 304, 8–12. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.W.; Lee, H.; Lee, H.K.; Kim, I.D.; Lee, J.K. Neutrophil Extracellular Trap Induced by HMGB1 Exacerbates Damages in the Ischemic Brain. Acta Neuropathol. Commun. 2019, 7, 94. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Gu, J. Immunoglobulin G Expression in Carcinomas and Cancer Cell Lines. FASEB J. 2007, 21, 2931–2938. [Google Scholar] [CrossRef]
- Qiu, X.; Zhu, X.; Zhang, L.; Mao, Y.; Zhang, J.; Hao, P.; Li, G.; Lv, P.; Li, Z.; Sun, X.; et al. Human Epithelial Cancers Secrete Immunoglobulin G with Unidentified Specificity to Promote Growth and Survival of Tumor Cells. Cancer Res. 2003, 63, 6488–6495. [Google Scholar]
- Lee, G.; Azadi, P. Peptide Mapping and Glycoanalysis of Cancer Cell-Expressed Glycoproteins CA215 Recognized by RP215 Monoclonal Antibody. J. Carbohydr. Chem. 2012, 31, 10–30. [Google Scholar] [CrossRef]
- Granger, V.; Peyneau, M.; Chollet-Martin, S.; de Chaisemartin, L. Neutrophil Extracellular Traps in Autoimmunity and Allergy: Immune Complexes at Work. Front. Immunol. 2019, 10, 2824. [Google Scholar] [CrossRef]
- Lee, G.; Laflamme, E.; Chien, C.H.; Hong, H.T. Molecular Identity of a Pan Cancer Marker, CA215. Cancer Biol. Ther. 2008, 7, 2007–2014. [Google Scholar] [CrossRef]
- Thiel, A.; Schott, M. Immunoglobulin G Expression and Its Colocalization with Complement Proteins in Papillary Thyroid Cancer. Yearb. Endocrinol. 2012, 2012, 158–160. [Google Scholar] [CrossRef]
- Liu, Y.; Chen, Z.; Niu, N.; Chang, Q.; Deng, R.; Korteweg, C.; Gu, J. IgG Gene Expression and Its Possible Significance in Prostate Cancers. Prostate 2012, 72, 690–701. [Google Scholar] [CrossRef] [PubMed]
- Wan, X.; Lei, Y.; Li, Z.; Wang, J.; Chen, Z.; McNutt, M.; Lin, D.; Zhao, C.; Jiang, C.; Li, J.; et al. Pancreatic Expression of Immunoglobulin G in Human Pancreatic Cancer and Associated Diabetes. Pancreas 2015, 44, 1304–1313. [Google Scholar] [CrossRef] [PubMed]
- Ma, C.; Wang, Y.; Zhang, G.; Chen, Z.; Qiu, Y.; Li, J.; Luo, J.; Huang, B.; Jiang, C.; Huang, G.; et al. Immunoglobulin G Expression and Its Potential Role in Primary and Metastatic Breast Cancers. Curr. Mol. Med. 2013, 13, 429–437. [Google Scholar] [CrossRef] [PubMed]
- Martins-Cardoso, K.; Almeida, V.H.; Bagri, K.M.; Rossi, M.I.D.; Mermelstein, C.S.; König, S.; Monteiro, R.Q. Neutrophil Extracellular Traps (Nets) Promote pro-Metastatic Phenotype in Human Breast Cancer Cells through Epithelial–Mesenchymal Transition. Cancers 2020, 12, 1542. [Google Scholar] [CrossRef] [PubMed]
- Pinho, S.S.; Reis, C.A. Glycosylation in Cancer: Mechanisms and Clinical Implications. Nat. Rev. Cancer 2015, 15, 540–555. [Google Scholar] [CrossRef]
- Xue, J.; Zhu, L.P.; Wei, Q. IgG-Fc N-Glycosylation at Asn297 and IgA O-Glycosylation in the Hinge Region in Health and Disease. Glycoconj. J. 2013, 30, 735–745. [Google Scholar] [CrossRef] [PubMed]
- Tang, J.; Zhang, J.; Liu, Y.; Liao, Q.; Huang, J.; Geng, Z.; Xu, W.; Sheng, Z.; Lee, G.; Zhang, Y.; et al. Lung Squamous Cell Carcinoma Cells Express Non-Canonically Glycosylated IgG That Activates Integrin-FAK Signaling. Cancer Lett. 2018, 430, 148–159. [Google Scholar] [CrossRef]
- Kdimati, S.; Mullins, C.S.; Linnebacher, M. Cancer-Cell-Derived Igg and Its Potential Role in Tumor Development. Int. J. Mol. Sci. 2021, 22, 11597. [Google Scholar] [CrossRef]
- Jiang, C.; Huang, T.; Wang, Y.; Huang, G.; Wan, X.; Gu, J. Immunoglobulin G Expression in Lung Cancer and Its Effects on Metastasis. PLoS ONE 2014, 9, e97359. [Google Scholar] [CrossRef] [PubMed]
- Lee, G. Cancerous Immunoglobulins and CA215: Implications in Cancer Immunology. Am. J. Immunol. 2012, 8, 101–116. [Google Scholar] [CrossRef]
- Chen, Q.; Wang, J.; Zhang, Q.; Zhang, J.; Lou, Y.; Yang, J.; Chen, Y.; Wei, T.; Zhang, J.; Fu, Q.; et al. Tumour Cell-Derived Debris and IgG Synergistically Promote Metastasis of Pancreatic Cancer by Inducing Inflammation via Tumour-Associated Macrophages. Br. J. Cancer 2019, 121, 786–795. [Google Scholar] [CrossRef]
- Kimoto, Y. Expression of Heavy-Chain Constant Region of Immunoglobulin and T-Cell Receptor Gene Transcripts in Human Non-Hematopoietic Tumor Cell Lines. Genes Chromosom. Cancer 1998, 22, 83–86. [Google Scholar] [CrossRef]
- Pan, B.; Zheng, S.; Liu, C.; Xu, Y. Suppression of IGHG1 Gene Expression by SiRNA Leads to Growth Inhibition and Apoptosis Induction in Human Prostate Cancer Cell. Mol. Biol. Rep. 2013, 40, 27–33. [Google Scholar] [CrossRef]
- Cui, M.; Huang, J.; Zhang, S.; Liu, Q.; Liao, Q.; Qiu, X. Immunoglobulin Expression in Cancer Cells and Its Critical Roles in Tumorigenesis. Front. Immunol. 2021, 12, 613530. [Google Scholar] [CrossRef]
- Chen, T.; Li, Y.; Sun, R.; Hu, H.; Liu, Y.; Herrmann, M.; Zhao, Y.; Muñoz, L.E. Receptor-Mediated NETosis on Neutrophils. Front. Immunol. 2021, 12, 775267. [Google Scholar] [CrossRef] [PubMed]
- de Buhr, N.; von Köckritz-Blickwede, M. Detection, Visualization, and Quantification of Neutrophil Extracellular Traps (NETs) and NET Markers. Methods Mol. Biol. 2020, 2087, 425–442. [Google Scholar] [CrossRef]
- Zhang, S.; Jia, X.; Zhang, Q.; Zhang, L.; Yang, J.; Hu, C.; Shi, J.; Jiang, X.; Lu, J.; Shen, H. Neutrophil Extracellular Traps Activate Lung Fibroblast to Induce Polymyositis-Related Interstitial Lung Diseases via TLR9-MiR-7-Smad2 Pathway. J. Cell. Mol. Med. 2020, 24, 1658–1669. [Google Scholar] [CrossRef]
- Kolman, J.P.; Pagerols Raluy, L.; Müller, I.; Nikolaev, V.O.; Trochimiuk, M.; Appl, B.; Wadehn, H.; Dücker, C.M.; Stoll, F.D.; Boettcher, M.; et al. NET Release of Long-Term Surviving Neutrophils. Front. Immunol. 2022, 13, 815412. [Google Scholar] [CrossRef]
- Li, Q.; Chen, W.; Li, Q.; Mao, J.; Chen, X. A Novel Neutrophil Extracellular Trap Signature to Predict Prognosis and Immunotherapy Response in Head and Neck Squamous Cell Carcinoma. Front. Immunol. 2022, 13, 1019967. [Google Scholar] [CrossRef]
- Sano, M.; Maejima, Y.; Nakagama, S.; Shiheido-Watanabe, Y.; Tamura, N.; Hirao, K.; Isobe, M.; Sasano, T. Neutrophil Extracellular Traps-Mediated Beclin-1 Suppression Aggravates Atherosclerosis by Inhibiting Macrophage Autophagy. Front. Cell Dev. Biol. 2022, 10, 876147. [Google Scholar] [CrossRef]
- Kong, X.; Zhang, Y.; Xiang, L.; You, Y.; Duan, Y.; Zhao, Y.; Li, S.; Wu, R.; Zhang, J.; Zhou, L.; et al. Fusobacterium Nucleatum-Triggered Neutrophil Extracellular Traps Facilitate Colorectal Carcinoma Progression. J. Exp. Clin. Cancer Res. 2023, 42, 236. [Google Scholar] [CrossRef]
- Jin, L.; Zhang, Z.; Pan, P.; Zhao, Y.; Zhou, M.; Liu, L.; Zhai, Y.; Wang, H.; Xu, L.; Mei, D.; et al. Low-Dose Ethanol Consumption Inhibits Neutrophil Extracellular Traps Formation to Alleviate Rheumatoid Arthritis. Commun. Biol. 2023, 6, 1088. [Google Scholar] [CrossRef] [PubMed]
- Landoni, V.I.; Pittaluga, J.R.; Carestia, A.; Castillo, L.A.; Nebel, M.d.C.; Martire-Greco, D.; Birnberg-Weiss, F.; Schattner, M.; Schierloh, P.; Fernández, G.C. Neutrophil Extracellular Traps Induced by Shiga Toxin and Lipopolysaccharide-Treated Platelets Exacerbate Endothelial Cell Damage. Front. Cell. Infect. Microbiol. 2022, 12, 897019. [Google Scholar] [CrossRef]
- Zhan, X.; Wu, R.; Kong, X.H.; You, Y.; He, K.; Sun, X.Y.; Huang, Y.; Chen, W.X.; Duan, L. Elevated Neutrophil Extracellular Traps by HBV-Mediated S100A9-TLR4/RAGE-ROS Cascade Facilitate the Growth and Metastasis of Hepatocellular Carcinoma. Cancer Commun. 2023, 43, 225–245. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.; Wang, S.S.; Chen, L.; Wang, Z.; Chen, J.; Ni, Q.; Guo, X.; Zhang, L.; Xue, G. Neutrophil Extracellular Traps Delay Diabetic Wound Healing by Inducing Endothelial-to-Mesenchymal Transition via the Hippo Pathway. Int. J. Biol. Sci. 2023, 19, 347–361. [Google Scholar] [CrossRef]
- Saithong, S.; Worasilchai, N.; Saisorn, W.; Udompornpitak, K.; Bhunyakarnjanarat, T.; Chindamporn, A.; Tovichayathamrong, P.; Torvorapanit, P.; Chiewchengchol, D.; Chancharoenthana, W.; et al. Neutrophil Extracellular Traps in Severe SARS-CoV-2 Infection: A Possible Impact of LPS and (1→3)-β-D-Glucan in Blood from Gut Translocation. Cells 2022, 11, 1103. [Google Scholar] [CrossRef] [PubMed]
- Mi, L.; Min, X.; Shi, M.; Liu, L.; Zhang, Y.; Zhu, Y.; Li, P.; Chai, Y.; Chen, F.; Deng, Q.; et al. Neutrophil Extracellular Traps Aggravate Neuronal Endoplasmic Reticulum Stress and Apoptosis via TLR9 after Traumatic Brain Injury. Cell Death Dis. 2023, 14, 374. [Google Scholar] [CrossRef] [PubMed]
- Chu, Z.; Huang, Q.; Ma, K.; Liu, X.; Zhang, W.; Cui, S.; Wei, Q.; Gao, H.; Hu, W.; Wang, Z.; et al. Novel Neutrophil Extracellular Trap-Related Mechanisms in Diabetic Wounds Inspire a Promising Treatment Strategy with Hypoxia-Challenged Small Extracellular Vesicles. Bioact. Mater. 2023, 27, 257–270. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Zhang, X.; Zhang, C.; Xu, W.; Li, W.; Feng, Z.; Zhang, X.; Zhao, K. Edaravone Dexborneol Downregulates Neutrophil Extracellular Trap Expression and Ameliorates Blood-Brain Barrier Permeability in Acute Ischemic Stroke. Mediat. Inflamm. 2022, 2022, 3855698. [Google Scholar] [CrossRef] [PubMed]
- Okamoto, M.; Mizuno, R.; Kawada, K.; Itatani, Y.; Kiyasu, Y.; Hanada, K.; Hirata, W.; Nishikawa, Y.; Masui, H.; Sugimoto, N.; et al. Neutrophil Extracellular Traps Promote Metastases of Colorectal Cancers through Activation of ERK Signaling by Releasing Neutrophil Elastase. Int. J. Mol. Sci. 2023, 24, 1118. [Google Scholar] [CrossRef] [PubMed]
- Jiang, M.; Wu, W.; Xia, Y.; Wang, X.; Liang, J. Platelet-Derived Extracellular Vesicles Promote Endothelial Dysfunction in Sepsis by Enhancing Neutrophil Extracellular Traps. BMC Immunol. 2023, 24, 22. [Google Scholar] [CrossRef]
- Zhou, Y.; Tao, W.; Shen, F.; Du, W.; Xu, Z.; Liu, Z. The Emerging Role of Neutrophil Extracellular Traps in Arterial, Venous and Cancer-Associated Thrombosis. Front. Cardiovasc. Med. 2021, 8, 786387. [Google Scholar] [CrossRef]
- Buckland, R.L.; Wilson, A.S.; Brüstle, A. Quantification of Neutrophil Extracellular Traps Isolated From Mouse Tissues. Curr. Protoc. Mouse Biol. 2020, 10, e78. [Google Scholar] [CrossRef]
- Zhang, L.; Gao, H.; Yang, L.; Liu, T.; Zhang, Q.; Xun, J.; Li, C.; Cui, L.; Wang, X. FTY720 Induces Neutrophil Extracellular Traps via a NADPH Oxidase-Independent Pathway. Arch. Biochem. Biophys. 2021, 711, 109015. [Google Scholar] [CrossRef]
- Zou, Y.; Chen, X.; He, B.; Xiao, J.; Yu, Q.; Xie, B.; Yang, S.; Dai, L.; Dai, Z.; Chen, Q. Neutrophil Extracellular Traps Induced by Cigarette Smoke Contribute to Airway Inflammation in Mice. Exp. Cell Res. 2020, 389, 111888. [Google Scholar] [CrossRef] [PubMed]
- Takesue, S.; Ohuchida, K.; Shinkawa, T.; Otsubo, Y.; Matsumoto, S.; Sagara, A.; Yonenaga, A.; Ando, Y.; Kibe, S.; Nakayama, H.; et al. Neutrophil Extracellular Traps Promote Liver Micrometastasis in Pancreatic Ductal Adenocarcinoma via the Activation of Cancer-associated Fibroblasts. Int. J. Oncol. 2020, 56, 596–605. [Google Scholar] [CrossRef] [PubMed]

| Role of NETs in Cancer | References |
|---|---|
| Regulate EndMT | [15,16,17] |
| Positive effect on tumor progression, invasion, and growth | [18,19,20,21] |
| Positive effect on angiogenesis | [22,23,24,25] |
| Provide resistance to chemotherapy | [26,27,28] |
| Provide resistance to immunotherapy | [31,32] |
| Provide resistance to radiotherapy | [33,34,35,40,42] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Himcinschi, M.E.; Uscatescu, V.; Gherghe, G.; Stoian, I.; Vlad, A.; Popa, D.C.; Coriu, D.; Anghel, A. The Role of Neutrophil Extracellular Traps in the Outcome of Malignant Epitheliomas: Significance of CA215 Involvement. Diagnostics 2024, 14, 328. https://doi.org/10.3390/diagnostics14030328
Himcinschi ME, Uscatescu V, Gherghe G, Stoian I, Vlad A, Popa DC, Coriu D, Anghel A. The Role of Neutrophil Extracellular Traps in the Outcome of Malignant Epitheliomas: Significance of CA215 Involvement. Diagnostics. 2024; 14(3):328. https://doi.org/10.3390/diagnostics14030328
Chicago/Turabian StyleHimcinschi, Mihai Emanuel, Valentina Uscatescu, Georgiana Gherghe, Irina Stoian, Adelina Vlad, Delia Codruța Popa, Daniel Coriu, and Andrei Anghel. 2024. "The Role of Neutrophil Extracellular Traps in the Outcome of Malignant Epitheliomas: Significance of CA215 Involvement" Diagnostics 14, no. 3: 328. https://doi.org/10.3390/diagnostics14030328
APA StyleHimcinschi, M. E., Uscatescu, V., Gherghe, G., Stoian, I., Vlad, A., Popa, D. C., Coriu, D., & Anghel, A. (2024). The Role of Neutrophil Extracellular Traps in the Outcome of Malignant Epitheliomas: Significance of CA215 Involvement. Diagnostics, 14(3), 328. https://doi.org/10.3390/diagnostics14030328






