Dual-Energy and Photon-Counting Computed Tomography in Vascular Applications—Technical Background and Post-Processing Techniques
Abstract
1. Introduction
2. Techniques of DECT
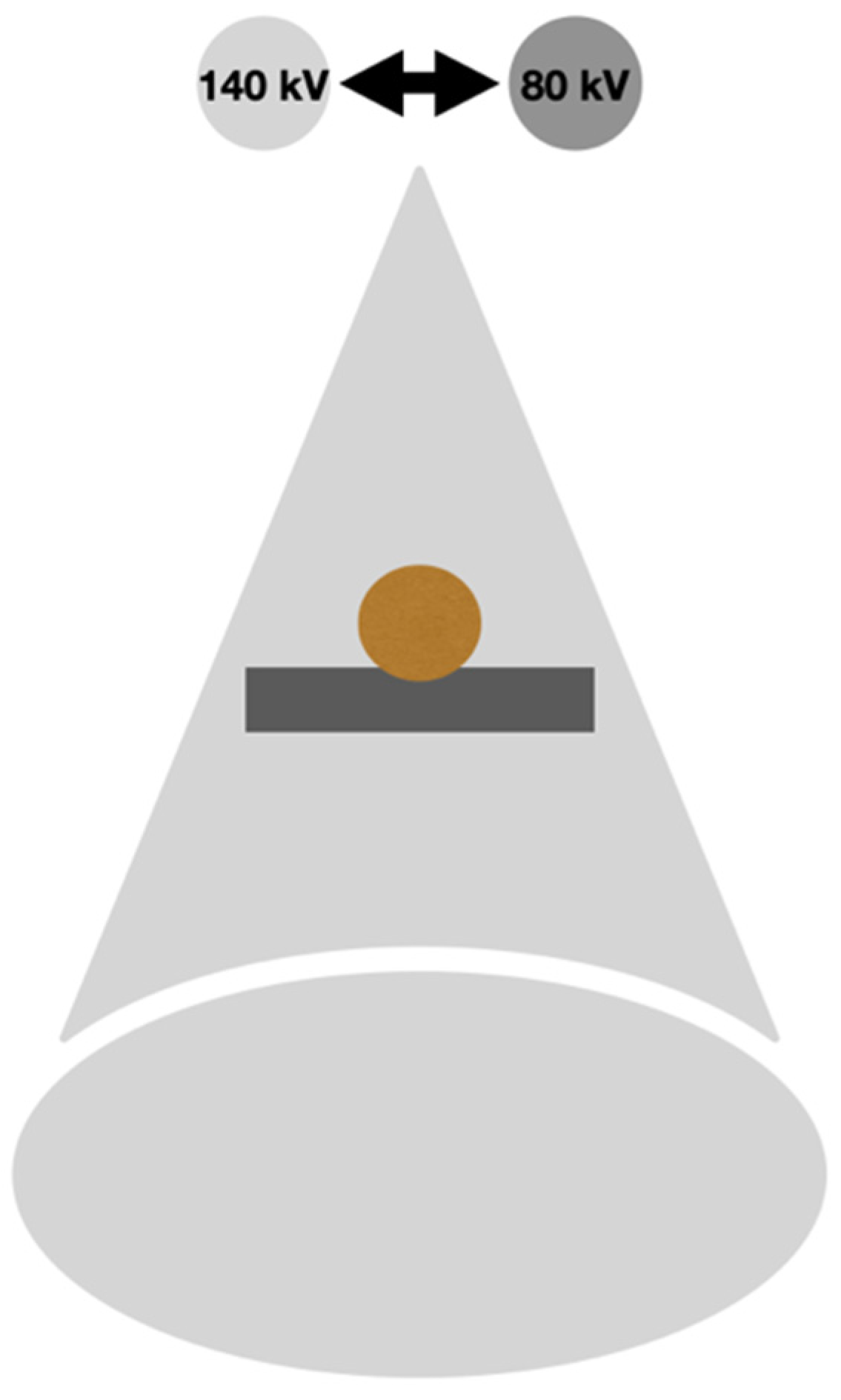
2.1. Rapid-kVp Switching DECT
2.2. Dual-Source DECT
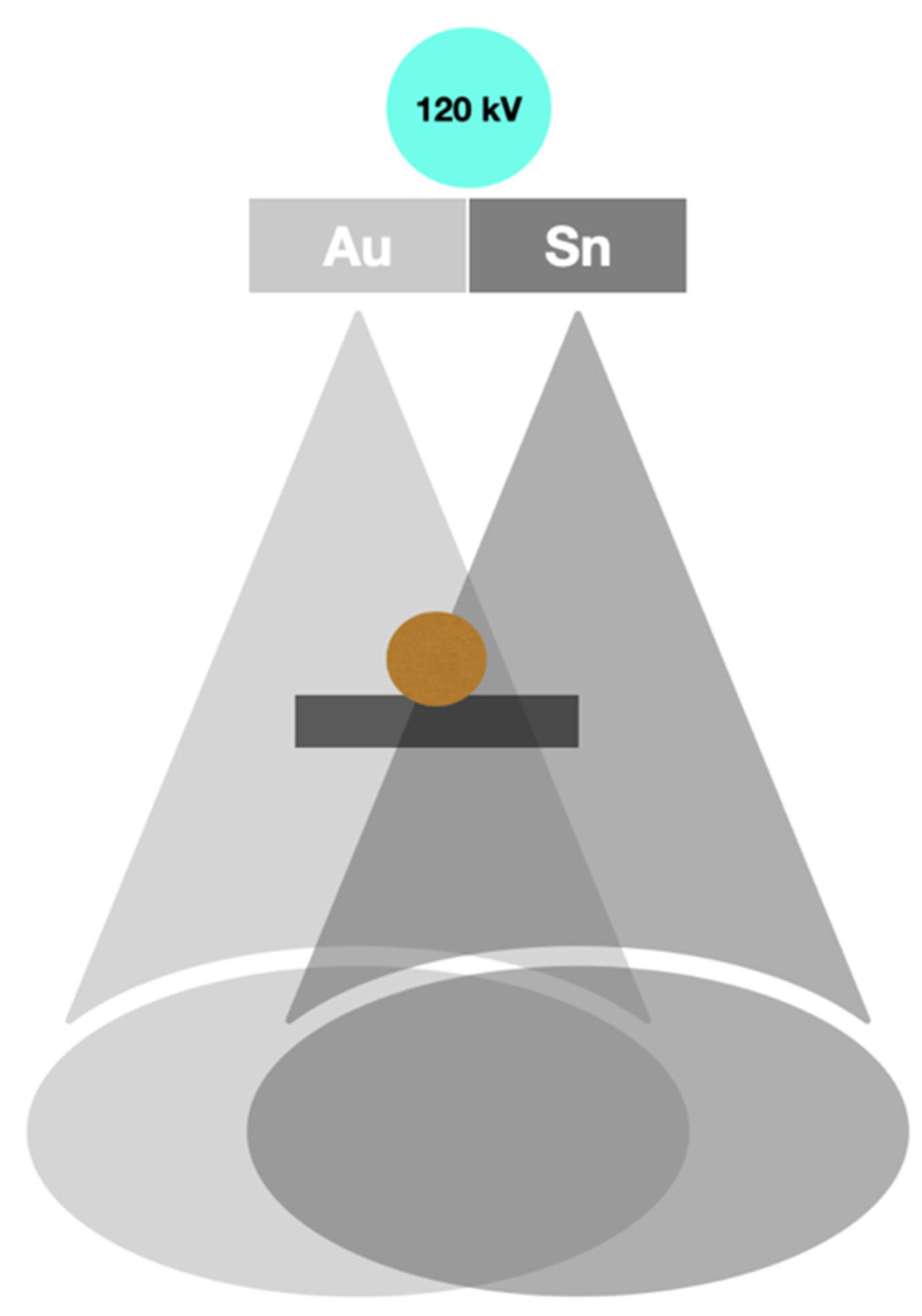
2.3. Split-Filter DECT
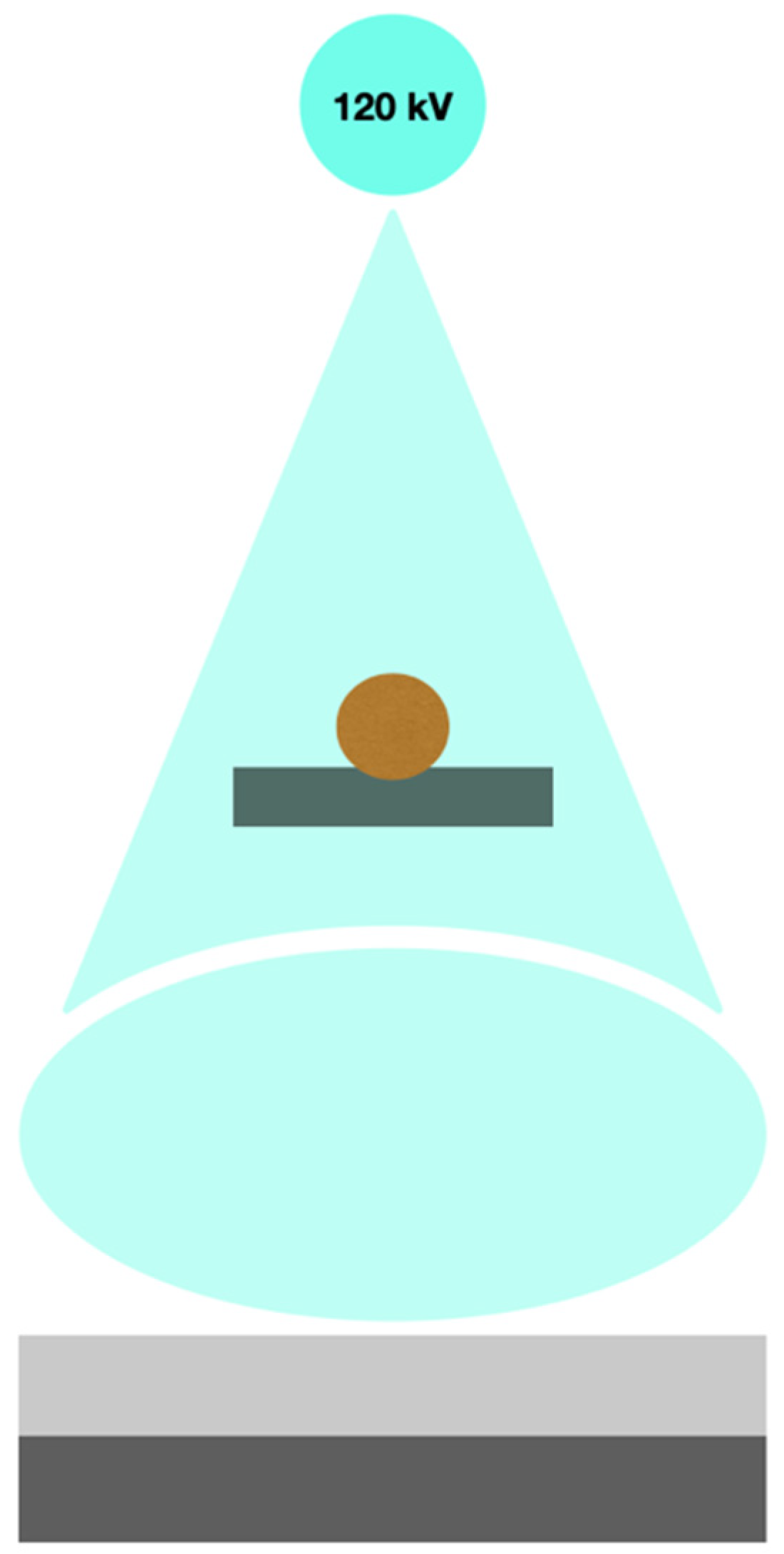
2.4. Multilayer Detector CT
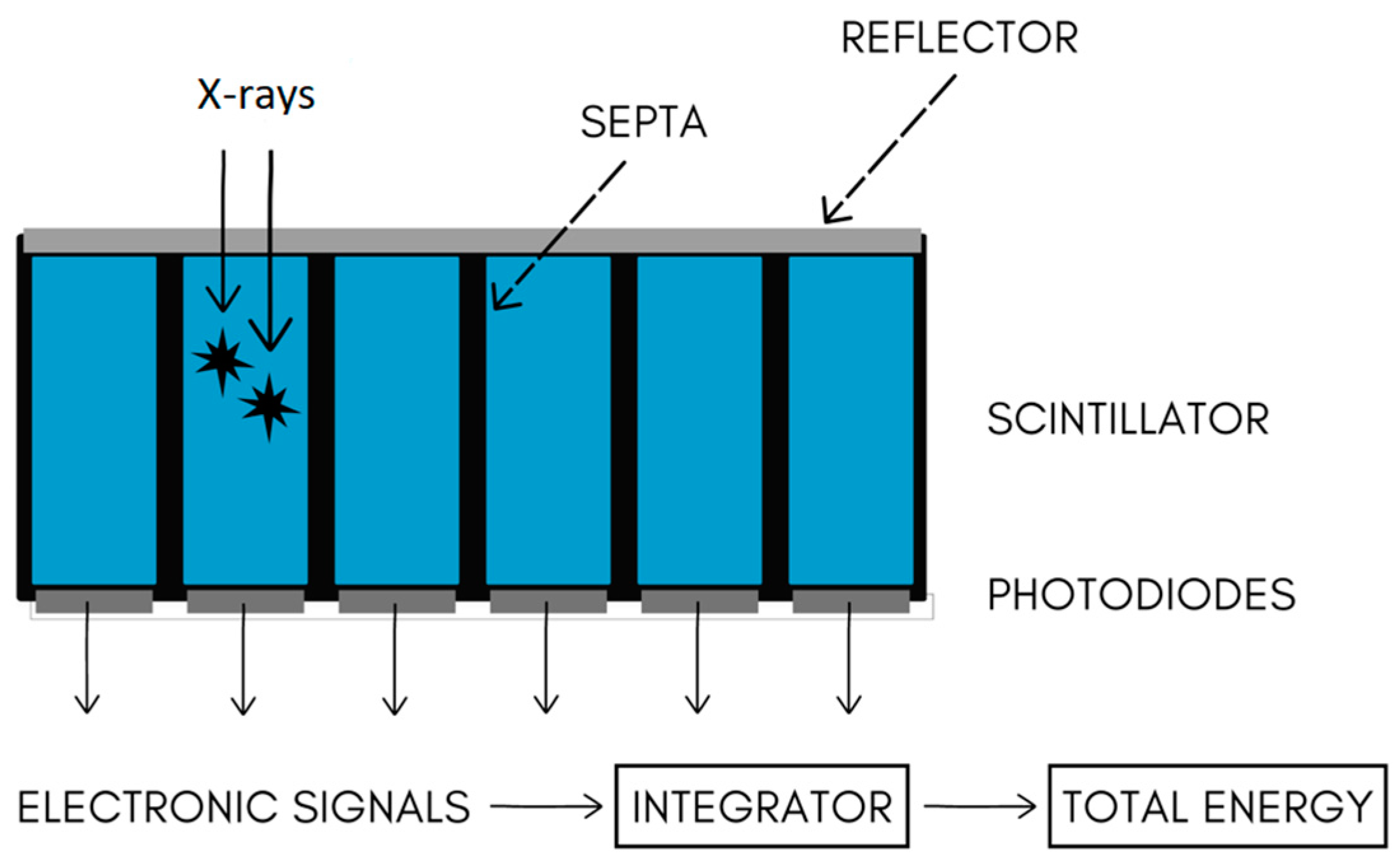
3. Photon-Counting CT
Technical Background
4. Postprocessing Techniques
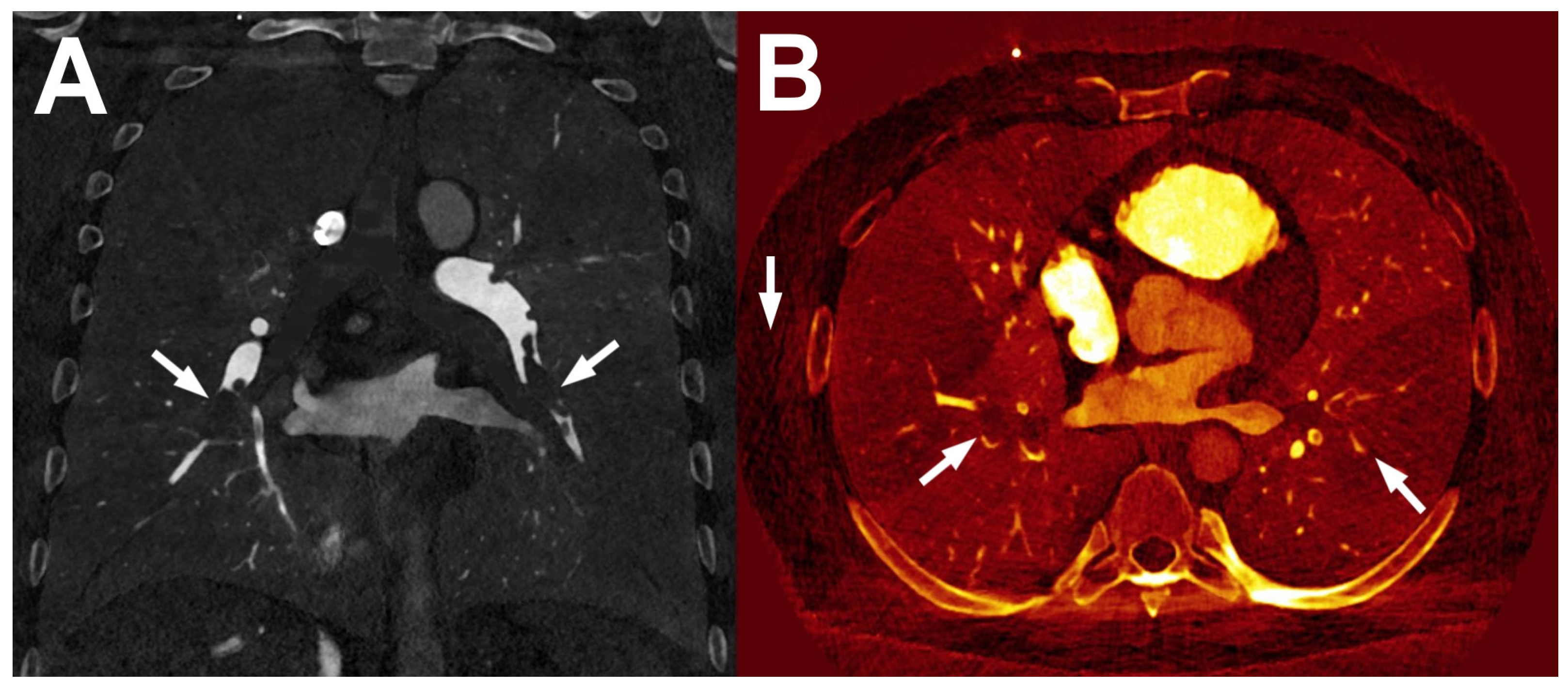
4.1. Material Decomposition
4.1.1. DECT
4.1.2. PCCT
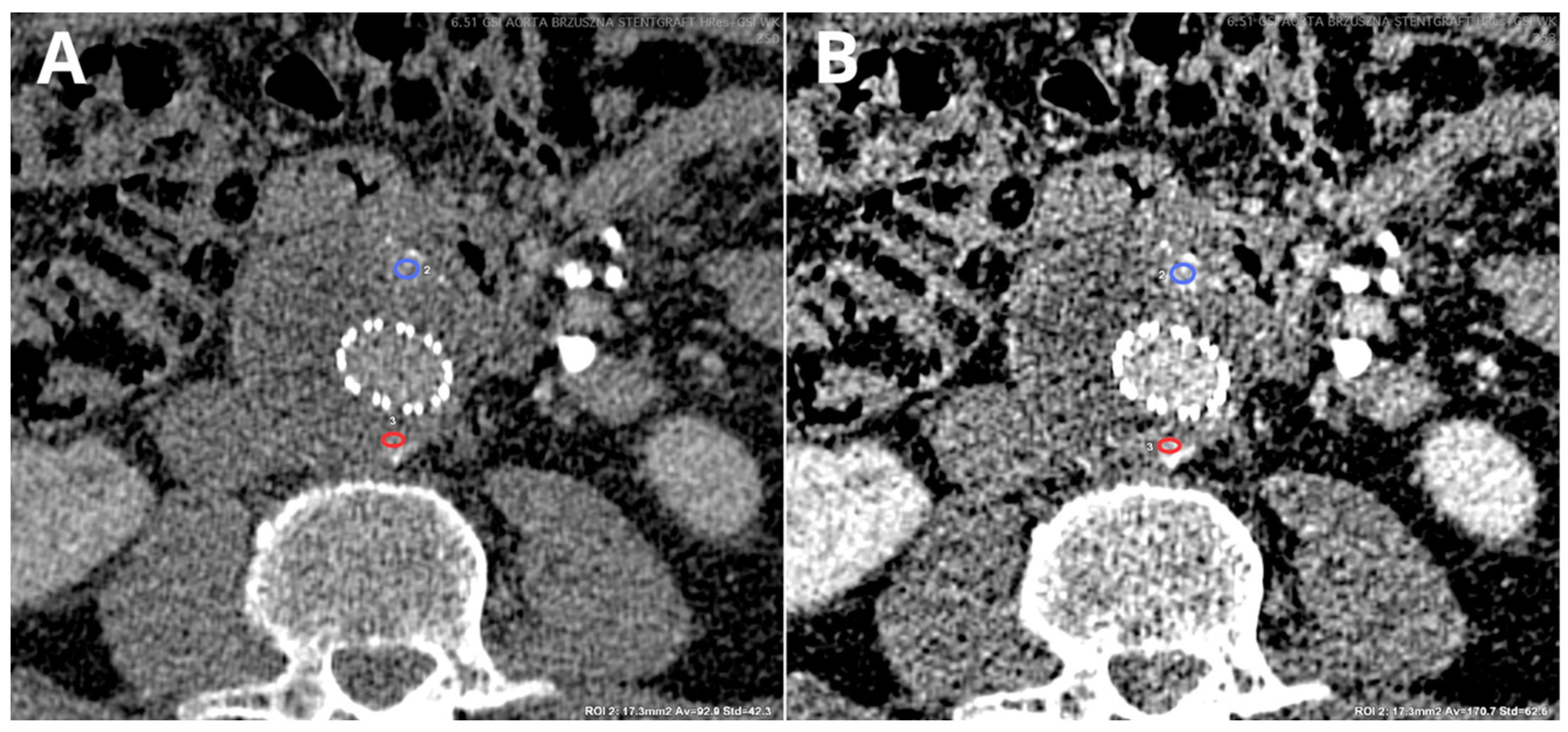
4.2. Virtual Non-Contrast
4.2.1. DECT
4.2.2. PCCT
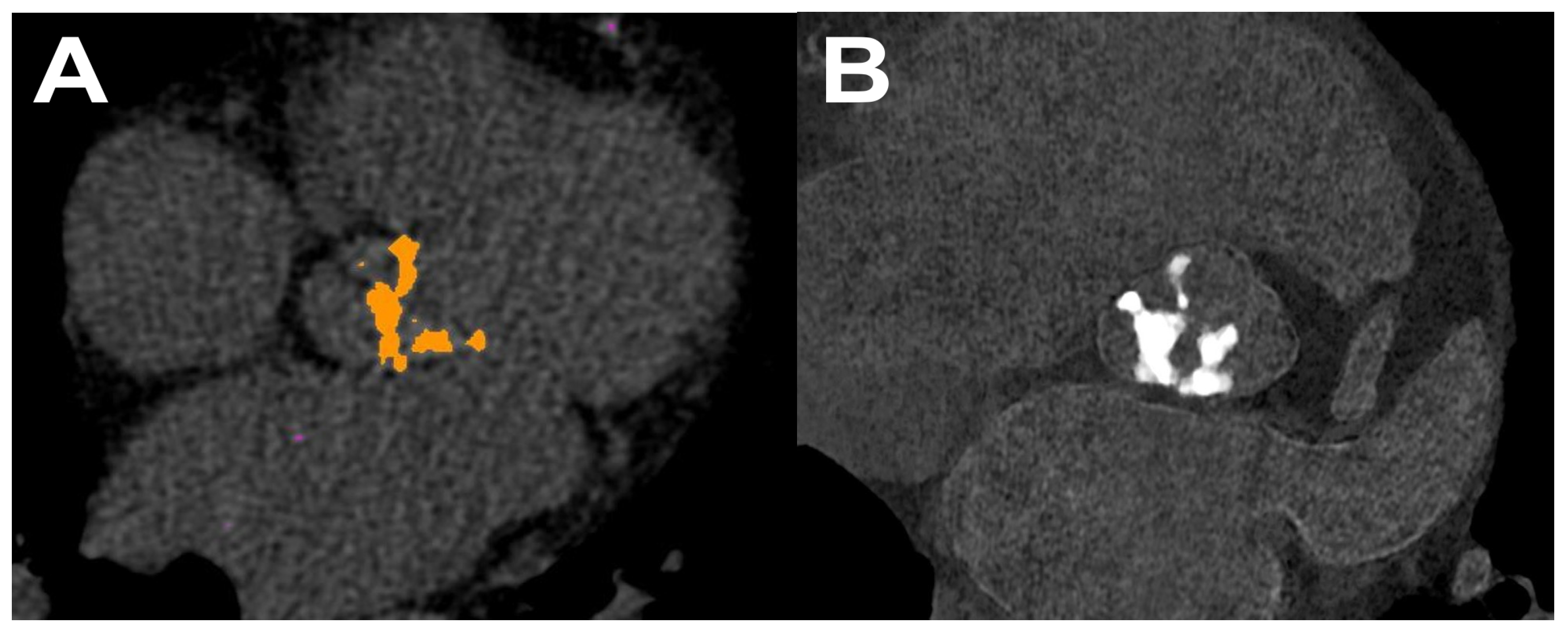
4.3. Virtual Non Calcium (VNCa)
4.3.1. DECT
4.3.2. PCCT
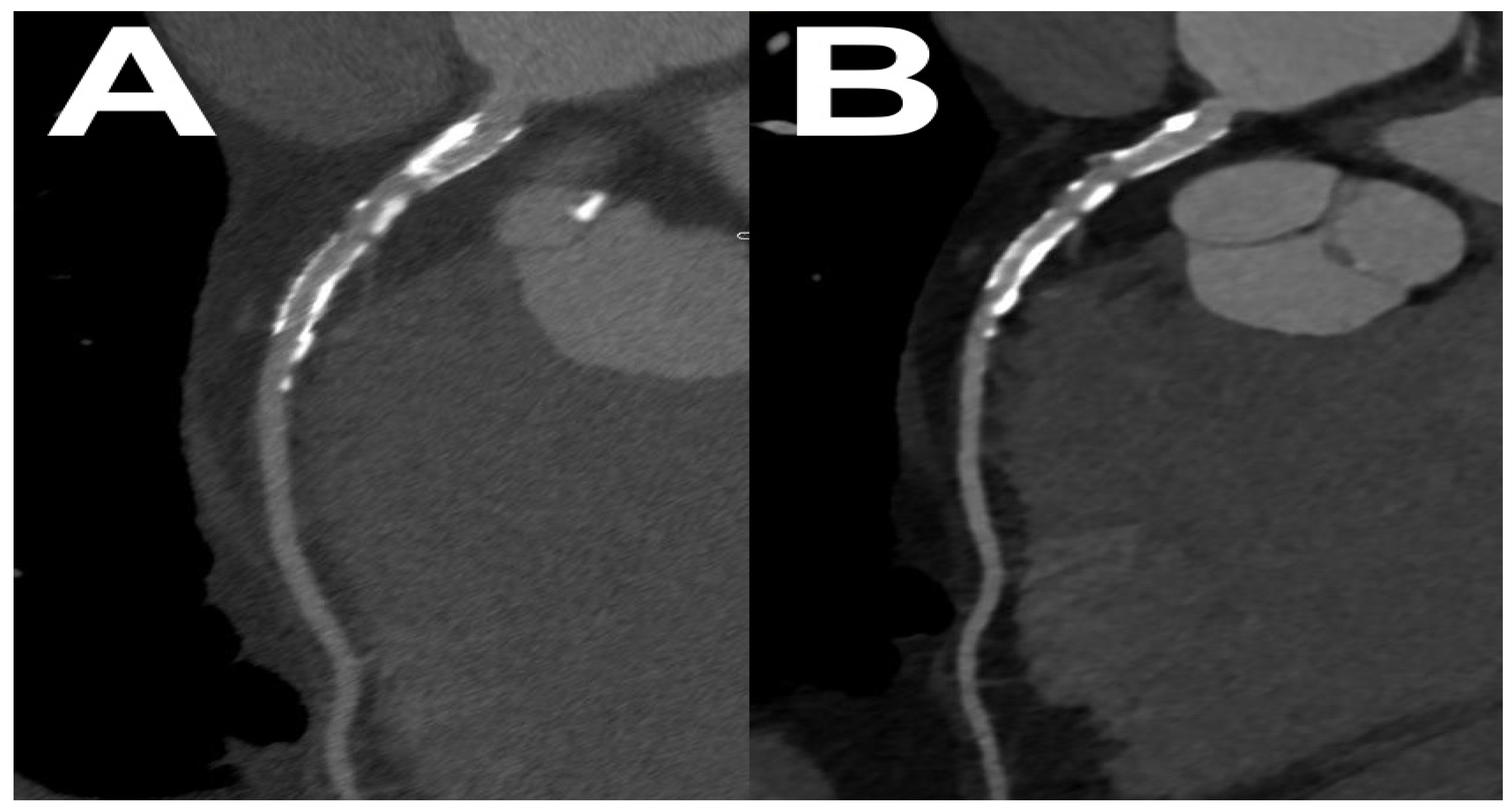
4.4. Virtual Monoenergetic Images
4.4.1. DECT
4.4.2. PCCT
5. Limitations
6. Conclusions
Funding
Conflicts of Interest
Abbreviations
References
- Hounsfield, G.N. Computerized Transverse Axial Scanning (Tomography): Part 1. Description of System. Br. J. Radiol. 1973, 46, 1016–1022. [Google Scholar] [CrossRef] [PubMed]
- McCollough, C.H.; Leng, S.; Yu, L.; Fletcher, J.G. Dual- and Multi-Energy CT: Principles, Technical Approaches, and Clinical Applications. Radiology 2015, 276, 637–653. [Google Scholar] [CrossRef] [PubMed]
- Kazimierczak, W.; Kazimierczak, N.; Serafin, Z. Review of Clinical Applications of Dual-Energy CT in Patients after Endovascular Aortic Repair. J. Clin. Med. 2023, 12, 7766. [Google Scholar] [CrossRef] [PubMed]
- Parakh, A.; Lennartz, S.; An, C.; Rajiah, P.; Yeh, B.M.; Simeone, F.J.; Sahani, D.V.; Kambadakone, A.R. Dual-Energy CT Images: Pearls and Pitfalls. Radiographics 2021, 41, 98–119. [Google Scholar] [CrossRef]
- Willemink, M.J.; Persson, M.; Pourmorteza, A.; Pelc, N.J.; Fleischmann, D. Photon-Counting CT: Technical Principles and Clinical Prospects. Radiology 2018, 289, 293–312. [Google Scholar] [CrossRef] [PubMed]
- Albrecht, M.H.; Vogl, T.J.; Martin, S.S.; Nance, J.W.; Duguay, T.M.; Wichmann, J.L.; Cecco, C.N.D.; Varga-Szemes, A.; van Assen, M.; Tesche, C.; et al. Review of Clinical Applications for Virtual Monoenergetic Dual-Energy CT. Radiology 2019, 293, 260–271. [Google Scholar] [CrossRef] [PubMed]
- Siegel, M.J.; Ramirez-Giraldo, J.C. Dual-Energy CT in Children: Imaging Algorithms and Clinical Applications. Radiology 2019, 291, 182289. [Google Scholar] [CrossRef] [PubMed]
- Meloni, A.; Frijia, F.; Panetta, D.; Degiorgi, G.; Gori, C.D.; Maffei, E.; Clemente, A.; Positano, V.; Cademartiri, F. Photon-Counting Computed Tomography (PCCT): Technical Background and Cardio-Vascular Applications. Diagnostics 2023, 13, 645. [Google Scholar] [CrossRef] [PubMed]
- Flohr, T.; Petersilka, M.; Henning, A.; Ulzheimer, S.; Ferda, J.; Schmidt, B. Photon-Counting CT Review. Phys. Med. 2020, 79, 126–136. [Google Scholar] [CrossRef]
- Decker, J.A.; Bette, S.; Scheurig-Muenkler, C.; Jehs, B.; Risch, F.; Woźnicki, P.; Braun, F.M.; Haerting, M.; Wollny, C.; Kroencke, T.J.; et al. Virtual Non-Contrast Reconstructions of Photon-Counting Detector CT Angiography Datasets as Substitutes for True Non-Contrast Acquisitions in Patients after EVAR—Performance of a Novel Calcium-Preserving Reconstruction Algorithm. Diagnostics 2022, 12, 558. [Google Scholar] [CrossRef]
- Alvarez, R.E.; Macovski, A. Energy-Selective Reconstructions in X-Ray Computerised Tomography. Phys. Med. Biol. 1976, 21, 733–744. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, Y.; Higaki, T.; Kondo, S.; Kawashita, I.; Takahashi, I.; Awai, K. An Introduction to Photon-Counting Detector CT (PCD CT) for Radiologists. Jpn. J. Radiol. 2023, 41, 266–282. [Google Scholar] [CrossRef] [PubMed]
- Sartoretti, T.; Wildberger, J.E.; Flohr, T.; Alkadhi, H. Photon-Counting Detector CT: Early Clinical Experience Review. Br. J. Radiol. 2023, 96, 20220544. [Google Scholar] [CrossRef] [PubMed]
- Kazimierczak, W.; Kazimierczak, N.; Serafin, Z. Quality of Virtual-Non-Contrast Phases Derived from Arterial and Delayed Phases of Fast-KVp Switching Dual-Energy CT in Patients after Endovascular Aortic Repair. Int. J. Cardiovasc. Imaging 2023, 39, 1805–1813. [Google Scholar] [CrossRef] [PubMed]
- Connolly, M.J.; McInnes, M.D.F.; El-Khodary, M.; McGrath, T.A.; Schieda, N. Diagnostic Accuracy of Virtual Non-Contrast Enhanced Dual-Energy CT for Diagnosis of Adrenal Adenoma: A Systematic Review and Meta-Analysis. Eur. Radiol. 2017, 27, 4324–4335. [Google Scholar] [CrossRef] [PubMed]
- Lehti, L.; Söderberg, M.; Höglund, P.; Nyman, U.; Gottsäter, A.; Wassélius, J. Reliability of Virtual Non-Contrast Computed Tomography Angiography: Comparing It with the Real Deal. Acta Radiol. Open 2018, 7, 2058460118790115. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, N.; Hartman, R.P.; Vrtiska, T.J.; Kawashima, A.; Primak, A.N.; Dzyubak, O.P.; Mandrekar, J.N.; Fletcher, J.G.; McCollough, C.H. Dual-Energy CT Iodine-Subtraction Virtual Unenhanced Technique to Detect Urinary Stones in an Iodine-Filled Collecting System: A Phantom Study. Am. J. Roentgenol. 2008, 190, 1169–1173. [Google Scholar] [CrossRef] [PubMed]
- Phan, C.M.; Yoo, A.J.; Hirsch, J.A.; Nogueira, R.G.; Gupta, R. Differentiation of Hemorrhage from Iodinated Contrast in Different Intracranial Compartments Using Dual-Energy Head CT. Am. J. Neuroradiol. 2012, 33, 1088–1094. [Google Scholar] [CrossRef]
- Yamada, Y.; Jinzaki, M.; Okamura, T.; Yamada, M.; Tanami, Y.; Abe, T.; Kuribayashi, S. Feasibility of Coronary Artery Calcium Scoring on Virtual Unenhanced Images Derived from Single-Source Fast KVp-Switching Dual-Energy Coronary CT Angiography. J. Cardiovasc. Comput. Tomogr. 2014, 8, 391–400. [Google Scholar] [CrossRef]
- Ascenti, G.; Sofia, C.; Mazziotti, S.; Silipigni, S.; D’Angelo, T.; Pergolizzi, S.; Scribano, E. Dual-Energy CT with Iodine Quantification in Distinguishing between Bland and Neoplastic Portal Vein Thrombosis in Patients with Hepatocellular Carcinoma. Clin. Radiol. 2016, 71, 938.e1–938.e9. [Google Scholar] [CrossRef]
- Chau, M. Dual-Energy Computed Tomography Can Replace the Standard Triphasic Protocol in Follow-Up Imaging of Endovascular Aneurysm Repair: A Review. Cardiovasc. Imaging Asia 2019, 3, 68. [Google Scholar] [CrossRef]
- Lehti, L.; Söderberg, M.; Höglund, P.; Wassélius, J. Comparing Arterial- and Venous-Phase Acquisition for Optimization of Virtual Noncontrast Images From Dual-Energy Computed Tomography Angiography. J. Comput. Assist. Tomogr. 2019, 43, 770–774. [Google Scholar] [CrossRef] [PubMed]
- Heye, T.; Nelson, R.C.; Ho, L.M.; Marin, D.; Boll, D.T. Dual-Energy CT Applications in the Abdomen. AJR Am. J. Roentgenol. 2012, 199, S64–S70. [Google Scholar] [CrossRef] [PubMed]
- Virarkar, M.K.; Vulasala, S.S.R.; Gupta, A.V.; Gopireddy, D.; Kumar, S.; Hernandez, M.; Lall, C.; Bhosale, P. Virtual Non-Contrast Imaging in The Abdomen and The Pelvis: An Overview. Semin. Ultrasound CT MRI 2022, 43, 293–310. [Google Scholar] [CrossRef] [PubMed]
- Ascenti, G.; Mazziotti, S.; Lamberto, S.; Bottari, A.; Caloggero, S.; Racchiusa, S.; Mileto, A.; Scribano, E. Dual-Energy CT for Detection of Endoleaks After Endovascular Abdominal Aneurysm Repair: Usefulness of Colored Iodine Overlay. Am. J. Roentgenol. 2011, 196, 1408–1414. [Google Scholar] [CrossRef] [PubMed]
- Sartoretti, T.; Mergen, V.; Higashigaito, K.; Eberhard, M.; Alkadhi, H.; Euler, A. Virtual Noncontrast Imaging of the Liver Using Photon-Counting Detector Computed Tomography. Investig. Radiol. 2022, 57, 488–493. [Google Scholar] [CrossRef] [PubMed]
- Niehoff, J.H.; Woeltjen, M.M.; Saeed, S.; Michael, A.E.; Boriesosdick, J.; Borggrefe, J.; Kroeger, J.R. Assessment of Hepatic Steatosis Based on Virtual Non-Contrast Computed Tomography: Initial Experiences with a Photon Counting Scanner Approved for Clinical Use. Eur. J. Radiol. 2022, 149, 110185. [Google Scholar] [CrossRef] [PubMed]
- Niehoff, J.H.; Woeltjen, M.M.; Laukamp, K.R.; Borggrefe, J.; Kroeger, J.R. Virtual Non-Contrast versus True Non-Contrast Computed Tomography: Initial Experiences with a Photon Counting Scanner Approved for Clinical Use. Diagnostics 2021, 11, 2377. [Google Scholar] [CrossRef]
- Krauss; Grant, B.; Allmendinger, K. Modified Dual Energy-Based Three Material Decomposition for Calcium Plaque Removal without Compromising Iodine Contrast. In Proceedings of the Radiology Society of Nothern America Annual Meeting, Chicago, IL, USA, 27 November–2 December 2016. [Google Scholar]
- Mannil, M.; Ramachandran, J.; de Martini, I.V.; Wegener, S.; Schmidt, B.; Flohr, T.; Krauss, B.; Valavanis, A.; Alkadhi, H.; Winklhofer, S. Modified Dual-Energy Algorithm for Calcified Plaque Removal. Investig. Radiol. 2017, 52, 680–685. [Google Scholar] [CrossRef]
- Hachamovitch, R.; Nutter, B.; Hlatky, M.A.; Shaw, L.J.; Ridner, M.L.; Dorbala, S.; Beanlands, R.S.B.; Chow, B.J.W.; Branscomb, E.; Chareonthaitawee, P.; et al. Patient Management After Noninvasive Cardiac Imaging Results From SPARC (Study of Myocardial Perfusion and Coronary Anatomy Imaging Roles in Coronary Artery Disease). J. Am. Coll. Cardiol. 2012, 59, 462–474. [Google Scholar] [CrossRef]
- Silvennoinen, H.M.; Ikonen, S.; Soinne, L.; Railo, M.; Valanne, L. CT Angiographic Analysis of Carotid Artery Stenosis: Comparison of Manual Assessment, Semiautomatic Vessel Analysis, and Digital Subtraction Angiography. Am. J. Neuroradiol. 2007, 28, 97–103. [Google Scholar] [PubMed]
- Wildberger, J.E.; Alkadhi, H. New Horizons in Vascular Imaging With Photon-Counting Detector CT. Investig. Radiol. 2023, 58, 499–504. [Google Scholar] [CrossRef] [PubMed]
- Mergen, V.; Rusek, S.; Civaia, F.; Rossi, P.; Rajagopal, R.; Bättig, E.; Manka, R.; Candreva, A.; Eberhard, M.; Alkadhi, H. Virtual Calcium Removal in Calcified Coronary Arteries with Photon-Counting Detector CT—First in-Vivo Experience. Front. Cardiovasc. Med. 2024, 11, 1367463. [Google Scholar] [CrossRef] [PubMed]
- Allmendinger, T.; Nowak, T.; Flohr, T.; Klotz, E.; Hagenauer, J.; Alkadhi, H.; Schmidt, B. Photon-Counting Detector CT-Based Vascular Calcium Removal Algorithm. Investig. Radiol. 2022, 57, 399–405. [Google Scholar] [CrossRef] [PubMed]
- Huda, W.; Scalzetti, E.M.; Levin, G. Technique Factors and Image Quality as Functions of Patient Weight at Abdominal CT1. Radiology 2000, 217, 430–435. [Google Scholar] [CrossRef]
- Hu, D.; Yu, T.; Duan, X.; Peng, Y.; Zhai, R. Determination of the Optimal Energy Level in Spectral CT Imaging for Displaying Abdominal Vessels in Pediatric Patients. Eur. J. Radiol. 2014, 83, 589–594. [Google Scholar] [CrossRef] [PubMed]
- Siegel, M.J.; Kaza, R.K.; Bolus, D.N.; Boll, D.T.; Rofsky, N.M.; Cecco, C.N.D.; Foley, W.D.; Morgan, D.E.; Schoepf, U.J.; Sahani, D.V.; et al. White Paper of the Society of Computed Body Tomography and Magnetic Resonance on Dual-Energy CT, Part 1. J. Comput. Assist. Tomogr. 2016, 40, 841–845. [Google Scholar] [CrossRef]
- Kazimierczak, W.; Nowak, E.; Kazimierczak, N.; Jankowski, T.; Jankowska, A.; Serafin, Z. The Value of Metal Artifact Reduction and Iterative Algorithms in Dual Energy CT Angiography in Patients after Complex Endovascular Aortic Aneurysm Repair. Heliyon 2023, 9, e20700. [Google Scholar] [CrossRef]
- Cecco, C.N.D.; Caruso, D.; Schoepf, U.J.; Santis, D.D.; Muscogiuri, G.; Albrecht, M.H.; Meinel, F.G.; Wichmann, J.L.; Burchett, P.F.; Varga-Szemes, A.; et al. A Noise-Optimized Virtual Monoenergetic Reconstruction Algorithm Improves the Diagnostic Accuracy of Late Hepatic Arterial Phase Dual-Energy CT for the Detection of Hypervascular Liver Lesions. Eur. Radiol. 2018, 28, 3393–3404. [Google Scholar] [CrossRef]
- Zhang, X.; Zhang, G.; Xu, L.; Bai, X.; Lu, X.; Yu, S.; Sun, H.; Jin, Z. Utilisation of Virtual Non-Contrast Images and Virtual Mono-Energetic Images Acquired from Dual-Layer Spectral CT for Renal Cell Carcinoma: Image Quality and Radiation Dose. Insights Imaging 2022, 13, 12. [Google Scholar] [CrossRef]
- Kazimierczak, W.; Kazimierczak, N.; Lemanowicz, A.; Nowak, E.; Migdalski, A.; Jawien, A.; Jankowski, T.; Serafin, Z. Improved Detection of Endoleaks in Virtual Monoenergetic Images in Dual-Energy CT Angiography Following EVAR. Acad. Radiol. 2023, 30, 2813–2824. [Google Scholar] [CrossRef] [PubMed]
- Kazimierczak, W.; Kazimierczak, N.; Wilamowska, J.; Wojtowicz, O.; Nowak, E.; Serafin, Z. Enhanced Visualization in Endoleak Detection through Iterative and AI-Noise Optimized Spectral Reconstructions. Sci. Rep. 2024, 14, 3845. [Google Scholar] [CrossRef] [PubMed]
- Michalska, M.; Kazimierczak, W.; Leszczyński, W.; Nadolska, K.; Bryl, Ł. Contemporary Follow-up Imaging after Endovascular Repair of Lower Extremity Atherosclerotic Lesions. Pol. J. Radiol. 2018, 83, e634–e642. [Google Scholar] [CrossRef] [PubMed]
- Ko, S.M.; Choi, J.W.; Song, M.G.; Shin, J.K.; Chee, H.K.; Chung, H.W.; Kim, D.H. Myocardial Perfusion Imaging Using Adenosine-Induced Stress Dual-Energy Computed Tomography of the Heart: Comparison with Cardiac Magnetic Resonance Imaging and Conventional Coronary Angiography. Eur. Radiol. 2011, 21, 26–35. [Google Scholar] [CrossRef] [PubMed]
- Wichmann, J.L.; Bauer, R.W.; Doss, M.; Stock, W.; Lehnert, T.; Bodelle, B.; Frellesen, C.; Vogl, T.J.; Kerl, J.M. Diagnostic Accuracy of Late Iodine–Enhancement Dual-Energy Computed Tomography for the Detection of Chronic Myocardial Infarction Compared With Late Gadolinium–Enhancement 3-T Magnetic Resonance Imaging. Investig. Radiol. 2013, 48, 851–856. [Google Scholar] [CrossRef] [PubMed]
- Albrecht, M.H.; Cecco, C.N.D.; Nance, J.W.; Varga-Szemes, A.; Santis, D.D.; Eid, M.; Tesche, C.; Apfaltrer, G.; von Knebel Doeberitz, P.L.; Jacobs, B.; et al. Cardiac Dual-Energy CT Applications and Clinical Impact. Curr. Radiol. Rep. 2017, 5, 42. [Google Scholar] [CrossRef]
- Mangold, S.; Cannaó, P.M.; Schoepf, U.J.; Wichmann, J.L.; Canstein, C.; Fuller, S.R.; Muscogiuri, G.; Varga-Szemes, A.; Nikolaou, K.; Cecco, C.N.D. Impact of an Advanced Image-Based Monoenergetic Reconstruction Algorithm on Coronary Stent Visualization Using Third Generation Dual-Source Dual-Energy CT: A Phantom Study. Eur. Radiol. 2016, 26, 1871–1878. [Google Scholar] [CrossRef] [PubMed]
- Stehli, J.; Clerc, O.F.; Fuchs, T.A.; Possner, M.; Gräni, C.; Benz, D.C.; Buechel, R.R.; Kaufmann, P.A. Impact of Monochromatic Coronary Computed Tomography Angiography from Single-Source Dual-Energy CT on Coronary Stenosis Quantification. J. Cardiovasc. Comput. Tomogr. 2016, 10, 135–140. [Google Scholar] [CrossRef] [PubMed]
- Winklhofer, S.; Hinzpeter, R.; Stocker, D.; Baltsavias, G.; Michels, L.; Burkhardt, J.-K.; Regli, L.; Valavanis, A.; Alkadhi, H. Combining Monoenergetic Extrapolations from Dual-Energy CT with Iterative Reconstructions: Reduction of Coil and Clip Artifacts from Intracranial Aneurysm Therapy. Neuroradiology 2018, 60, 281–291. [Google Scholar] [CrossRef]
- Vrbaski, S.; Bache, S.; Rajagopal, J.; Samei, E. Quantitative Performance of Photon-counting CT at Low Dose: Virtual Monochromatic Imaging and Iodine Quantification. Med. Phys. 2023, 50, 5421–5433. [Google Scholar] [CrossRef]
- Decker, J.A.; Bette, S.; Lubina, N.; Rippel, K.; Braun, F.; Risch, F.; Woźnicki, P.; Wollny, C.; Scheurig-Muenkler, C.; Kroencke, T.J.; et al. Low-Dose CT of the Abdomen: Initial Experience on a Novel Photon-Counting Detector CT and Comparison with Energy-Integrating Detector CT. Eur. J. Radiol. 2022, 148, 110181. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.P.; Shapira, N.; Chen, A.A.; Shinohara, R.T.; Sahbaee, P.; Schnall, M.; Litt, H.I.; Noël, P.B. First-Generation Clinical Dual-Source Photon-Counting CT: Ultra-Low-Dose Quantitative Spectral Imaging. Eur. Radiol. 2022, 32, 8579–8587. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.P.; Shapira, N.; Sahbaee, P.; Gang, G.J.; Knollman, F.D.; Chen, M.Y.; Litt, H.I.; Noël, P.B. Consistency of Spectral Results in Cardiac Dual-Source Photon-Counting CT. Sci. Rep. 2023, 13, 14895. [Google Scholar] [CrossRef] [PubMed]
- Booij, R.; van der Werf, N.R.; Dijkshoorn, M.L.; van der Lugt, A.; van Straten, M. Assessment of Iodine Contrast-To-Noise Ratio in Virtual Monoenergetic Images Reconstructed from Dual-Source Energy-Integrating CT and Photon-Counting CT Data. Diagnostics 2022, 12, 1467. [Google Scholar] [CrossRef] [PubMed]
- Sartoretti, T.; Mergen, V.; Jungblut, L.; Alkadhi, H.; Euler, A. Liver Iodine Quantification With Photon-Counting Detector CT: Accuracy in an Abdominal Phantom and Feasibility in Patients. Acad. Radiol. 2023, 30, 461–469. [Google Scholar] [CrossRef]
- Baig, M.; Gilbert, G.; Almansoor, Z.; Agrawal, S.; Junejo, S.; Raja, Y. Is Ct Coronary Angiography (ctca) a New Gold Standard for Diagnosis of Coronary Artery Disease?—Comparison of Ctca and Invasive Coronary Angiography (ica). J. Cardiovasc. Comput. Tomogr. 2023, 17, S11. [Google Scholar] [CrossRef]



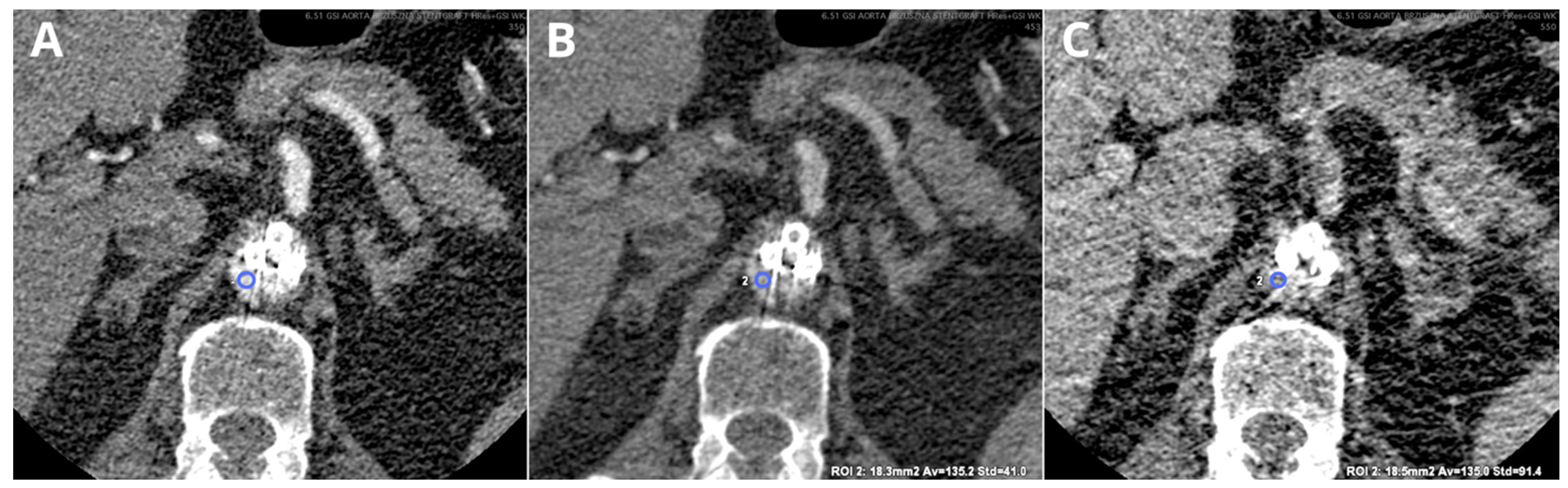
| Siemens Healthineers | GE Healthcare | Philips Healthcare | |
|---|---|---|---|
| Technique | |||
| DECT system | Dual source, split filter | Rapid kVp switching | Dual layer |
| Material decomposition images—iodine | Liver non-calcium, lung perfusion, brain hemorrhage, heart, pulmonary blood volume | Iodine (water), iodine (calcium), iodine (hydroxyapatite), iodine (fat) | Iodine no water, iodine density |
| Material decomposition images—calcium | Brain hemorrhage, kidney stones | Calcium (water), calcium (iodine), calcium (uric acid), | Uric acid, uric acid removed |
| Material decomposition—urate | Kidney stones, gout | uric acid (calcium), uric acid (hydroxyapatite) | Uric acid, uric acid removed |
| Virtual non-contrast images | Liver virtual non-calcium, virtual unenhanced | Water (iodine), virtual unenhanced | Virtual unenhanced, iodine removed |
| Virtual non-calcium image | Bone removal, hard plaque, brain hemorrhage, blood marrow | Iodine (calcium) | Calcium suppressed |
| Virtual kilo-electronovolt images | Virtual monoergetic images | Virtual monochromatic images | Monoenergetic Images |
| Reconstruction Technique | Advantages | Applications |
| Material decomposition | Decompose materials into their elemental components |
|
| Virtual non-contrast | Non-contrast scan can be omitted |
|
| Virtual non-calcium | Robust calcium subtraction method |
|
| Virtual monoenergetic images | Mimic the attenuation values of an image obtained with single energy |
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Stański, M.; Michałowska, I.; Lemanowicz, A.; Karmelita-Katulska, K.; Ratajczak, P.; Sławińska, A.; Serafin, Z. Dual-Energy and Photon-Counting Computed Tomography in Vascular Applications—Technical Background and Post-Processing Techniques. Diagnostics 2024, 14, 1223. https://doi.org/10.3390/diagnostics14121223
Stański M, Michałowska I, Lemanowicz A, Karmelita-Katulska K, Ratajczak P, Sławińska A, Serafin Z. Dual-Energy and Photon-Counting Computed Tomography in Vascular Applications—Technical Background and Post-Processing Techniques. Diagnostics. 2024; 14(12):1223. https://doi.org/10.3390/diagnostics14121223
Chicago/Turabian StyleStański, Marcin, Ilona Michałowska, Adam Lemanowicz, Katarzyna Karmelita-Katulska, Przemysław Ratajczak, Agata Sławińska, and Zbigniew Serafin. 2024. "Dual-Energy and Photon-Counting Computed Tomography in Vascular Applications—Technical Background and Post-Processing Techniques" Diagnostics 14, no. 12: 1223. https://doi.org/10.3390/diagnostics14121223
APA StyleStański, M., Michałowska, I., Lemanowicz, A., Karmelita-Katulska, K., Ratajczak, P., Sławińska, A., & Serafin, Z. (2024). Dual-Energy and Photon-Counting Computed Tomography in Vascular Applications—Technical Background and Post-Processing Techniques. Diagnostics, 14(12), 1223. https://doi.org/10.3390/diagnostics14121223






