Role of Cardiac Biomarkers in Non-Small Cell Lung Cancer Patients
Abstract
1. Introduction
2. Materials and Methods
2.1. Patient Population
- -
- The inclusion criteria were as follows:
- -
- basal pre-treatment CT imaging of the thorax;
- -
- cardiac evaluation, including ultrasonography with the calculation of ejection fraction (EF);
- -
- complete anamnesis, including co-pathologies that could interfere with cardiac functionality;
- -
- pharmacological therapy.
2.2. Ethics Approval
2.3. Computed Tomography Imaging
2.4. CAC Score Calculation
2.5. Clinical Variables
2.6. Endpoints and Statistical Analysis
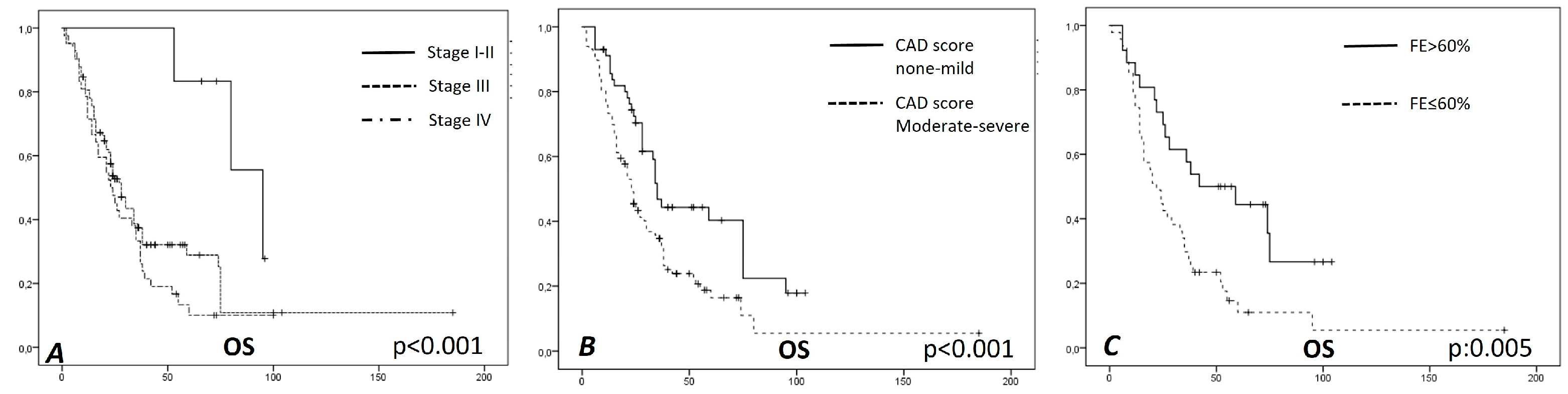
3. Results
Correlation with Outcomes
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Siegel, R.L.; Miller, K.D.; Jemal, A. Cancer Statistics, 2017. CA A Cancer J. Clin. 2017, 67, 7–30. [Google Scholar] [CrossRef] [PubMed]
- Tini, P.; Nardone, V.; Pastina, P.; Pirtoli, L.; Correale, P.; Giordano, A. The effects of radiotherapy on the survival of patients with unresectable non-small cell lung cancer. Expert Rev. Anticancer. Ther. 2018, 18, 593–602. [Google Scholar] [CrossRef] [PubMed]
- Pastina, P.; Nardone, V.; Botta, C.; Croci, S.; Tini, P.; Battaglia, G.; Ricci, V.; Cusi, M.G.; Gandolfo, C.; Misso, G.; et al. Radiotherapy prolongs the survival of advanced non-small-cell lung cancer patients undergone to an immune-modulating treatment with dose-fractioned cisplatin and metronomic etoposide and bevacizumab (mPEBev). Oncotarget 2017, 8, 75904–75913. [Google Scholar] [CrossRef] [PubMed]
- Pastina, P.; Nardone, V.; Croci, S.; Battaglia, G.; Vanni, F.; Bellan, C.; Barbarino, M.; Ricci, V.; Costantini, S.; Capone, F.; et al. Anti-cancer activity of dose-fractioned mPE +/- bevacizumab regimen is paralleled by immune-modulation in advanced squamous NSLC patients. J. Thorac. Dis. 2017, 9, 3123–3131. [Google Scholar] [CrossRef]
- Reginelli, A.; Capasso, R.; Petrillo, M.; Rossi, C.; Faella, P.; Grassi, R.; Belfiore, M.P.; Rossi, G.; Muto, M.; Muto, P.; et al. Looking for Lepidic Component inside Invasive Adenocarcinomas Appearing as CT Solid Solitary Pulmonary Nodules (SPNs): CT Morpho-Densitometric Features and 18-FDG PET Findings. Biomed. Res. Int. 2019, 2019, 7683648. [Google Scholar] [CrossRef]
- Singh, N.; Temin, S.; Baker, S., Jr.; Blanchard, E.; Brahmer, J.R.; Celano, P.; Duma, N.; Ellis, P.M.; Elkins, I.B.; Haddad, R.Y.; et al. Therapy for Stage IV Non-Small-Cell Lung Cancer Without Driver Alterations: ASCO Living Guideline. J. Clin. Oncol. Off. J. Am. Soc. Clin. Oncol. 2022, 40, 3323–3343. [Google Scholar] [CrossRef]
- Singh, N.; Temin, S.; Baker, S., Jr.; Blanchard, E.; Brahmer, J.R.; Celano, P.; Duma, N.; Ellis, P.M.; Elkins, I.B.; Haddad, R.Y.; et al. Therapy for Stage IV Non-Small-Cell Lung Cancer With Driver Alterations: ASCO Living Guideline. J. Clin. Oncol. Off. J. Am. Soc. Clin. Oncol. 2022, 40, 3310–3322. [Google Scholar] [CrossRef]
- Nardone, V.; Giannicola, R.; Bianco, G.; Giannarelli, D.; Tini, P.; Pastina, P.; Falzea, A.C.; Macheda, S.; Caraglia, M.; Luce, A.; et al. Inflammatory Markers and Procalcitonin Predict the Outcome of Metastatic Non-Small-Cell-Lung-Cancer Patients Receiving PD-1/PD-L1 Immune-Checkpoint Blockade. Front. Oncol. 2021, 11, 684110. [Google Scholar] [CrossRef]
- Nardone, V.; Giannicola, R.; Giannarelli, D.; Saladino, R.E.; Azzarello, D.; Romeo, C.; Bianco, G.; Rizzo, M.R.; Di Meo, I.; Nesci, A.; et al. Distinctive Role of the Systemic Inflammatory Profile in Non-Small-Cell Lung Cancer Younger and Elderly Patients Treated with a PD-1 Immune Checkpoint Blockade: A Real-World Retrospective Multi-Institutional Analysis. Life 2021, 11, 1235. [Google Scholar] [CrossRef]
- Nardone, V.; Tini, P.; Pastina, P.; Botta, C.; Reginelli, A.; Carbone, S.F.; Giannicola, R.; Calabrese, G.; Tebala, C.; Guida, C.; et al. Radiomics predicts survival of patients with advanced non-small cell lung cancer undergoing PD-1 blockade using Nivolumab. Oncol. Lett. 2020, 19, 1559–1566. [Google Scholar] [CrossRef]
- Reginelli, A.; Belfiore, M.P.; Monti, R.; Cozzolino, I.; Costa, M.; Vicidomini, G.; Grassi, R.; Morgillo, F.; Urraro, F.; Nardone, V.; et al. The texture analysis as a predictive method in the assessment of the cytological specimen of CT-guided FNAC of the lung cancer. Med. Oncol. (Northwood Lond. Engl.) 2020, 37, 54. [Google Scholar] [CrossRef] [PubMed]
- Giannicola, R.; D’Arrigo, G.; Botta, C.; Agostino, R.; Del Medico, P.; Falzea, A.C.; Barbieri, V.; Staropoli, N.; Del Giudice, T.; Pastina, P.; et al. Early blood rise in auto-antibodies to nuclear and smooth muscle antigens is predictive of prolonged survival and autoimmunity in metastatic-non-small cell lung cancer patients treated with PD-1 immune-check point blockade by nivolumab. Mol. Clin. Oncol. 2019, 11, 81–90. [Google Scholar] [CrossRef] [PubMed]
- Correale, P.; Saladino, R.E.; Nardone, V.; Giannicola, R.; Agostino, R.; Pirtoli, L.; Caraglia, M.; Botta, C.; Tagliaferri, P. Could PD-1/PDL1 immune checkpoints be linked to HLA signature? Immunotherapy 2019, 11, 1523–1526. [Google Scholar] [CrossRef] [PubMed]
- Pacifico, P.; Colciago, R.R.; De Felice, F.; Boldrini, L.; Salvestrini, V.; Nardone, V.; Desideri, I.; Greco, C.; Arcangeli, S. A critical review on oligometastatic disease: A radiation oncologist’s perspective. Med. Oncol. (Northwood Lond. Engl.) 2022, 39, 181. [Google Scholar] [CrossRef]
- Stamatis, G.; Muller, S.; Weinreich, G.; Schwarz, B.; Eberhardt, W.; Pottgen, C.; Aigner, C. Significantly favourable outcome for patients with non-small-cell lung cancer stage IIIA/IIIB and single-station persistent N2 (skip or additionally N1) disease after multimodality treatment. Eur. J. Cardio-Thorac. Surg. Off. J. Eur. Assoc. Cardio-Thorac. Surg. 2021, 61, 269–276. [Google Scholar] [CrossRef]
- Taylor, S.; Yorke, J.; Tsim, S.; Navani, N.; Baldwin, D.; Woolhouse, I.; Edwards, J.; Grundy, S.; Robson, J.; Rhodes, S.; et al. Impact on quality of life from multimodality treatment for lung cancer: A randomised controlled feasibility trial of surgery versus no surgery as part of multimodality treatment in potentially resectable stage III-N2 NSCLC (the PIONEER trial). BMJ Open Respir. Res. 2021, 8, e000846. [Google Scholar] [CrossRef] [PubMed]
- Glatzer, M.; Leskow, P.; Caparrotti, F.; Elicin, O.; Furrer, M.; Gambazzi, F.; Dutly, A.; Gelpke, H.; Guckenberger, M.; Heuberger, J.; et al. Stage III N2 non-small cell lung cancer treatment: Decision-making among surgeons and radiation oncologists. Transl. Lung Cancer Res. 2021, 10, 1960–1968. [Google Scholar] [CrossRef]
- Evers, J.; de Jaeger, K.; Hendriks, L.E.L.; van der Sangen, M.; Terhaard, C.; Siesling, S.; De Ruysscher, D.; Struikmans, H.; Aarts, M.J. Trends and variations in treatment of stage I-III non-small cell lung cancer from 2008 to 2018: A nationwide population-based study from the Netherlands. Lung Cancer (Amst. Neth.) 2021, 155, 103–113. [Google Scholar] [CrossRef]
- Nardone, V.; Correale, P.; Guida, C. The Optimal Choice of Local Therapy for Stage IIIA-N2 NSCLC: Is Radiotherapy Inferior to Surgery? Ann. Thorac. Surg. 2019, 108, 961–962. [Google Scholar] [CrossRef]
- Milgrom, S.A.; Varghese, B.; Gladish, G.W.; Choi, A.D.; Dong, W.; Patel, Z.S.; Chung, C.C.; Rao, A.; Pinnix, C.C.; Gunther, J.R.; et al. Coronary Artery Dose-Volume Parameters Predict Risk of Calcification After Radiation Therapy. J. Cardiovasc. Imaging 2019, 27, 268–279. [Google Scholar] [CrossRef] [PubMed]
- Tohidinezhad, F.; Pennetta, F.; van Loon, J.; Dekker, A.; de Ruysscher, D.; Traverso, A. Prediction models for treatment-induced cardiac toxicity in patients with non-small-cell lung cancer: A systematic review and meta-analysis. Clin. Transl. Radiat. Oncol. 2022, 33, 134–144. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Gao, W.; Ning, Y.; Zou, X.; Zhang, W.; Zeng, L.; Liu, J. Cardiovascular Toxicity With PD-1/PD-L1 Inhibitors in Cancer Patients: A Systematic Review and Meta-Analysis. Front. Immunol. 2022, 13, 908173. [Google Scholar] [CrossRef] [PubMed]
- Liang, Z.; He, Y.; Hu, X. Cardio-Oncology: Mechanisms, Drug Combinations, and Reverse Cardio-Oncology. Int. J. Mol. Sci. 2022, 23, 10617. [Google Scholar] [CrossRef] [PubMed]
- Agatston, A.S.; Janowitz, W.R.; Hildner, F.J.; Zusmer, N.R.; Viamonte, M., Jr.; Detrano, R. Quantification of coronary artery calcium using ultrafast computed tomography. J. Am. Coll. Cardiol. 1990, 15, 827–832. [Google Scholar] [CrossRef] [PubMed]
- Sandfort, V.; Bluemke, D.A. CT calcium scoring. History, current status and outlook. Diagn. Interv. Imaging 2017, 98, 3–10. [Google Scholar] [CrossRef] [PubMed]
- Nioche, C.; Orlhac, F.; Boughdad, S.; Reuze, S.; Goya-Outi, J.; Robert, C.; Pellot-Barakat, C.; Soussan, M.; Frouin, F.; Buvat, I. LIFEx: A Freeware for Radiomic Feature Calculation in Multimodality Imaging to Accelerate Advances in the Characterization of Tumor Heterogeneity. Cancer Res. 2018, 78, 4786–4789. [Google Scholar] [CrossRef]
- Faivre-Finn, C.; Vicente, D.; Kurata, T.; Planchard, D.; Paz-Ares, L.; Vansteenkiste, J.F.; Spigel, D.R.; Garassino, M.C.; Reck, M.; Senan, S.; et al. Four-Year Survival With Durvalumab After Chemoradiotherapy in Stage III NSCLC-an Update From the PACIFIC Trial. J. Thorac. Oncol. Off. Publ. Int. Assoc. Study Lung Cancer 2021, 16, 860–867. [Google Scholar] [CrossRef]
- Nardone, V.; Reginelli, A.; De Marco, G.; Di Pietro, T.; Grassi, R.; Corte, C.M.D.; Fasano, M.; Ciammella, P.; Vicidomini, G.; Morgillo, F.; et al. The role of coronary artery calcification score in lung cancer patients. Eur. J. Radiol. 2022, 147, 110140. [Google Scholar] [CrossRef]
- Reginelli, A.; Nardone, V.; Giacobbe, G.; Belfiore, M.P.; Grassi, R.; Schettino, F.; Del Canto, M.; Grassi, R.; Cappabianca, S. Radiomics as a New Frontier of Imaging for Cancer Prognosis: A Narrative Review. Diagnostics 2021, 11, 1796. [Google Scholar] [CrossRef]
- Wei, T.; Cheng, Y. The cardiac toxicity of radiotherapy—A review of characteristics, mechanisms, diagnosis, and prevention. Int. J. Radiat. Biol. 2021, 97, 1333–1340. [Google Scholar] [CrossRef]
- Banfill, K.; Giuliani, M.; Aznar, M.; Franks, K.; McWilliam, A.; Schmitt, M.; Sun, F.; Vozenin, M.C.; Faivre Finn, C. Cardiac Toxicity of Thoracic Radiotherapy: Existing Evidence and Future Directions. J. Thorac. Oncol. Off. Publ. Int. Assoc. Study Lung Cancer 2021, 16, 216–227. [Google Scholar] [CrossRef] [PubMed]
- Moey, M.Y.Y.; Tomdio, A.N.; McCallen, J.D.; Vaughan, L.M.; O’Brien, K.; Naqash, A.R.; Cherry, C.; Walker, P.R.; Carabello, B.A. Characterization of Immune Checkpoint Inhibitor-Related Cardiotoxicity in Lung Cancer Patients From a Rural Setting. JACC. CardioOncology 2020, 2, 491–502. [Google Scholar] [CrossRef] [PubMed]
- Arangalage, D.; Degrauwe, N.; Michielin, O.; Monney, P.; Özdemir, B.C. Pathophysiology, diagnosis and management of cardiac toxicity induced by immune checkpoint inhibitors and BRAF and MEK inhibitors. Cancer Treat. Rev. 2021, 100, 102282. [Google Scholar] [CrossRef]
- Gernaat, S.A.; Išgum, I.; de Vos, B.D.; Takx, R.A.; Young-Afat, D.A.; Rijnberg, N.; Grobbee, D.E.; van der Graaf, Y.; de Jong, P.A.; Leiner, T.; et al. Automatic Coronary Artery Calcium Scoring on Radiotherapy Planning CT Scans of Breast Cancer Patients: Reproducibility and Association with Traditional Cardiovascular Risk Factors. PLoS ONE 2016, 11, e0167925. [Google Scholar] [CrossRef]
- Andersen, R.; Wethal, T.; Günther, A.; Fosså, A.; Edvardsen, T.; Fosså, S.D.; Kjekshus, J. Relation of coronary artery calcium score to premature coronary artery disease in survivors >15 years of Hodgkin’s lymphoma. Am. J. Cardiol. 2010, 105, 149–152. [Google Scholar] [CrossRef]
- Herrmann, B.L.; Severing, M.; Schmermund, A.; Berg, C.; Budde, T.; Erbel, R.; Mann, K. Impact of disease duration on coronary calcification in patients with acromegaly. Exp. Clin. Endocrinol. Diabetes Off. J. Ger. Soc. Endocrinol. Ger. Diabetes Assoc. 2009, 117, 417–422. [Google Scholar] [CrossRef] [PubMed]
- Cappabianca, S.; Belfiore, M.P.; Reginelli, A.; Capasso, R.; Del Prete, A.; Petrillo, M.; Mascia, P.; Rossi, C.; Romano, F.; De Filippo, M.; et al. Mandatory Reporting of Coronary Artery Calcifications Incidentally Noted on Chest Multi-Detector Computed Tomography: A Multicentre Experience. Curr. Vasc. Pharmacol. 2019, 17, 92–98. [Google Scholar] [CrossRef] [PubMed]
- Tjessem, K.H.; Bosse, G.; Fosså, K.; Reinertsen, K.V.; Fosså, S.D.; Johansen, S.; Fosså, A. Coronary calcium score in 12-year breast cancer survivors after adjuvant radiotherapy with low to moderate heart exposure—Relationship to cardiac radiation dose and cardiovascular risk factors. Radiother. Oncol. J. Eur. Soc. Ther. Radiol. Oncol. 2015, 114, 328–334. [Google Scholar] [CrossRef]
- Gal, R.; van Velzen, S.G.M.; Hooning, M.J.; Emaus, M.J.; van der Leij, F.; Gregorowitsch, M.L.; Blezer, E.L.A.; Gernaat, S.A.M.; Lessmann, N.; Sattler, M.G.A.; et al. Identification of Risk of Cardiovascular Disease by Automatic Quantification of Coronary Artery Calcifications on Radiotherapy Planning CT Scans in Patients With Breast Cancer. JAMA Oncol. 2021, 7, 1024–1032. [Google Scholar] [CrossRef]
- Miki, T.; Miyauchi, S.; Miyoshi, T.; Yoshida, M.; Ichikawa, K.; Soh, J.; Nakamura, K.; Kiura, K.; Kanazawa, S.; Toyooka, S.; et al. Chemoradiation therapy for non-small cell lung cancer exacerbates thoracic aortic calcification determined by computed tomography. Heart Vessel. 2020, 35, 1401–1408. [Google Scholar] [CrossRef]
- Abravan, A.; van Herk, M.; Brewster, F.; Faivre-Finn, C.; McWilliam, A.; Vasquez Osorio, E.M. Predictive value of vascular calcification identified in 4D planning CT of lung cancer patients treated with stereotactic body radiation therapy. Phys. Med. PM Int. J. Devoted Appl. Phys. Med. Biol. Off. J. Ital. Assoc. Biomed. Phys. (AIFB) 2020, 78, 173–178. [Google Scholar] [CrossRef]
- Wang, K.; Malkin, H.E.; Patchett, N.D.; Pearlstein, K.A.; Heiling, H.M.; McCabe, S.D.; Deal, A.M.; Mavroidis, P.; Oakey, M.; Fenoli, J.; et al. Coronary Artery Calcifications and Cardiac Risk after Radiotherapy for Stage III Lung Cancer. Int. J. Radiat. Oncol. Biol. Phys. 2021, 112, 188–196. [Google Scholar] [CrossRef] [PubMed]
- Han, D.; Lee, J.H.; Hartaigh, B.; Min, J.K. Role of computed tomography screening for detection of coronary artery disease. Clin. Imaging 2016, 40, 307–310. [Google Scholar] [CrossRef] [PubMed]
- Rosmini, S.; Aggarwal, A.; Chen, D.H.; Conibear, J.; Davies, C.L.; Dey, A.K.; Edwards, P.; Guha, A.; Ghosh, A.K. Cardiac computed tomography in cardio-oncology: An update on recent clinical applications. Eur. Heart J. Cardiovasc. Imaging 2021, 22, 397–405. [Google Scholar] [CrossRef]
- Chao, H.; Shan, H.; Homayounieh, F.; Singh, R.; Khera, R.D.; Guo, H.; Su, T.; Wang, G.; Kalra, M.K.; Yan, P. Deep learning predicts cardiovascular disease risks from lung cancer screening low dose computed tomography. Nat. Commun. 2021, 12, 2963. [Google Scholar] [CrossRef] [PubMed]
- Semrau, S.; Klautke, G.; Fietkau, R. Baseline cardiopulmonary function as an independent prognostic factor for survival of inoperable non-small-cell lung cancer after concurrent chemoradiotherapy: A single-center analysis of 161 cases. Int. J. Radiat. Oncol. Biol. Phys. 2011, 79, 96–104. [Google Scholar] [CrossRef]
- Ren, M.; Ma, Y.; Wei, M.; Ning, Y.; Liu, H.; Shi, X.; Yao, Y.; Guo, F. Atrial Cardiomyopathy Predicts Worse Outcome in Patients With Lung Cancer. Front. Cardiovasc. Med. 2022, 9, 932044. [Google Scholar] [CrossRef]
- Rydzek, J.; Gąsior, Z.T.; Dąbek, J.; Wojnar, J.; Skrzypek, M. Assessment of risk factors for mortality in patients with cardiovascular disease and a history of treatment for malignancy. Kardiol. Pol. 2015, 73, 730–739. [Google Scholar] [CrossRef]

| Clinical Parameters | |
|---|---|
| Sex Male Female | |
| 118 (68.2%) 55 (31.8%) | |
| Age 18–50 51–65 66–75 ≥76 | |
| 10 (5.8%) 75 (43.3%) 80 (46.2%) 8 (4.7%) | |
| Smoke exposure Unknown Non smoker Previous smoker Actual smoker | |
| 6 (3.5%) 17 (9.8%) 69 (39.9%) 81 (46.8%) | |
| Stage of disease I/II III A III B III C IV | |
| 7 (4%) 54 (31.2%) 62 (35.8%) 8 (4.6%) 42 (24.4%) |
| Cardiological Characteristics | |
|---|---|
| CAD Score None Mild Moderate Severe | |
| 40 (23.1%) 17 (9.8%) 18 (10.4%) 98 (56.6%) | |
| Diabetes Yes No | |
| 26 (15%) 147 (85%) | |
| Dyslipidemia Yes No | |
| 38 (21.9%) 135 (78.1%) | |
| Previous ischemic heart disease Yes No | |
| 28 (16.2%) 145 (83.8%) | |
| TIA or stroke Yes No | |
| 8 (4.6%) 165 (95.4%) | |
| Hypertension Yes No | |
| 97 (56%) 76 (44%) | |
| Sartan use Yes No | |
| 34 (19.6%) 139 (80.4%) | |
| Anticoagulant use Yes No | |
| 40 (23.1%) 133 (76.9%) | |
| Beta blocker use Yes No | |
| 36 (20.8%) 137 (79.2%) | |
| Calcium channel blocker use Yes No | |
| 28 (16.2%) 145 (83.8%) | |
| Diabetes drug use Yes No | |
| 16 (9.2%) 157 (90.8%) | |
| Diuretic use Yes No | |
| 26 (15%) 147 (85%) | |
| Cardiac ejection fraction ≤60% >60% | |
| 111 (64%) 62 (36%) |
| Cox Regression Analysis | |||
|---|---|---|---|
| Parameter | p-Value | Β | OR (95% CI) |
| Stage of disease | 0.016 | 0.238 | 1.26 (1.05–1.53) |
| CAD score | 0.014 | 0.443 | 1.56 (1.04–1.83) |
| Cardiac ejection fraction | 0.011 | −0.767 | 0.46 (0.25–0.84) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nardone, V.; Reginelli, A.; De Marco, G.; Natale, G.; Patanè, V.; De Chiara, M.; Buono, M.; Russo, G.M.; Monti, R.; Balestrucci, G.; et al. Role of Cardiac Biomarkers in Non-Small Cell Lung Cancer Patients. Diagnostics 2023, 13, 400. https://doi.org/10.3390/diagnostics13030400
Nardone V, Reginelli A, De Marco G, Natale G, Patanè V, De Chiara M, Buono M, Russo GM, Monti R, Balestrucci G, et al. Role of Cardiac Biomarkers in Non-Small Cell Lung Cancer Patients. Diagnostics. 2023; 13(3):400. https://doi.org/10.3390/diagnostics13030400
Chicago/Turabian StyleNardone, Valerio, Alfonso Reginelli, Giuseppina De Marco, Giovanni Natale, Vittorio Patanè, Marco De Chiara, Mauro Buono, Gaetano Maria Russo, Riccardo Monti, Giovanni Balestrucci, and et al. 2023. "Role of Cardiac Biomarkers in Non-Small Cell Lung Cancer Patients" Diagnostics 13, no. 3: 400. https://doi.org/10.3390/diagnostics13030400
APA StyleNardone, V., Reginelli, A., De Marco, G., Natale, G., Patanè, V., De Chiara, M., Buono, M., Russo, G. M., Monti, R., Balestrucci, G., Salvarezza, M., Di Guida, G., D’Ippolito, E., Sangiovanni, A., Grassi, R., D’Onofrio, I., Belfiore, M. P., Cimmino, G., Della Corte, C. M., ... Cappabianca, S. (2023). Role of Cardiac Biomarkers in Non-Small Cell Lung Cancer Patients. Diagnostics, 13(3), 400. https://doi.org/10.3390/diagnostics13030400









