A Critical Analysis of the Robustness of Radiomics to Variations in Segmentation Methods in 18F-PSMA-1007 PET Images of Patients Affected by Prostate Cancer
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Gleason Score
- GS ≤ 6: Signifying tumors with slow growth tendencies that typically do not metastasize to distant organs beyond the prostate (non-metastatic).
- GS = 7: Indicating tumors of intermediate aggressiveness.
- GS between 8 and 10: Corresponding to highly aggressive tumors with a propensity for metastasis.
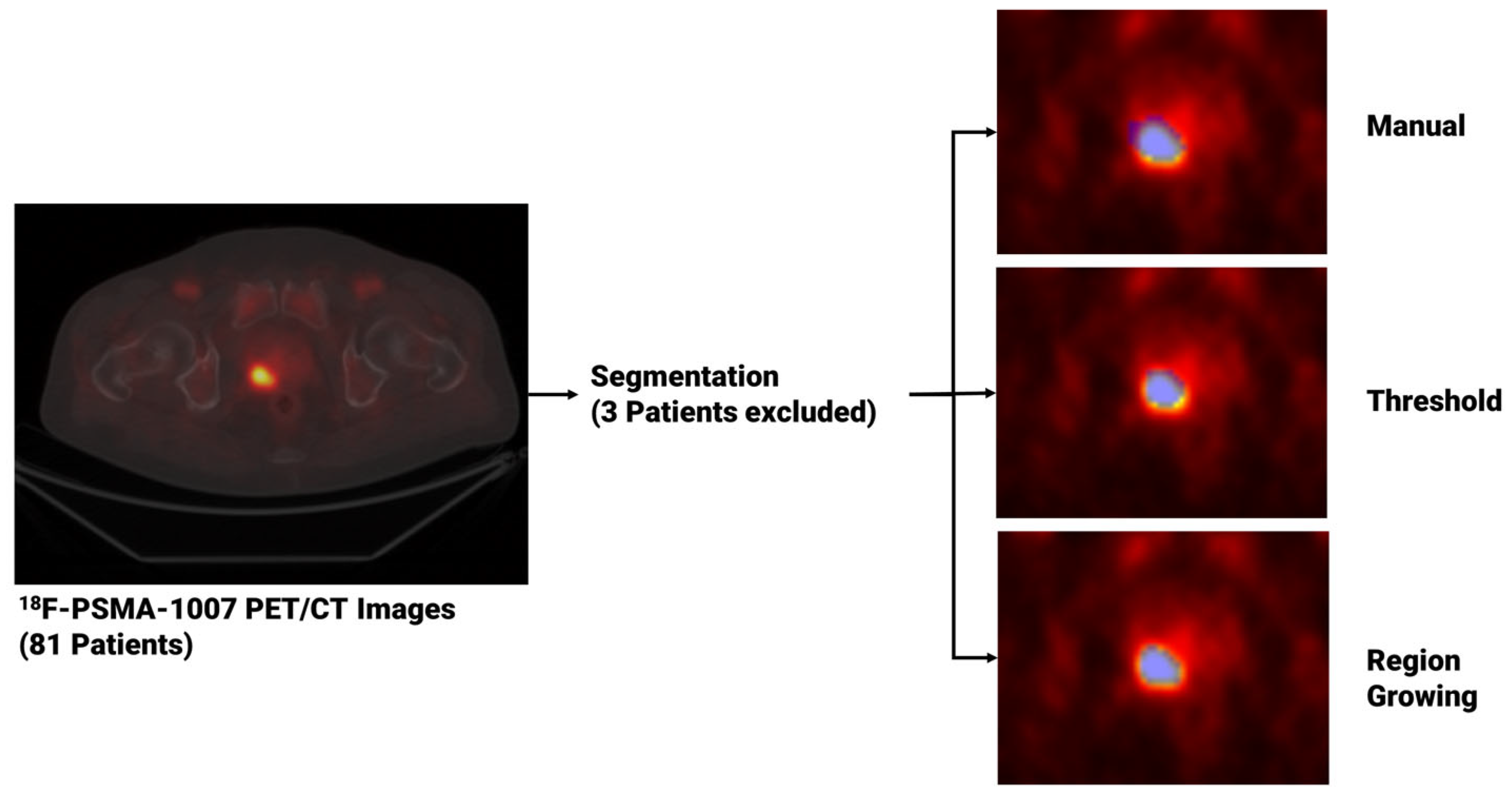
2.3. Segmentation and Segmentation Agreement
2.4. Feature Extraction
2.5. Feature Robustness
2.6. Feature Selection and Machine Learning Methodology
2.7. Statistical Analysis
3. Results
3.1. Clinical Data
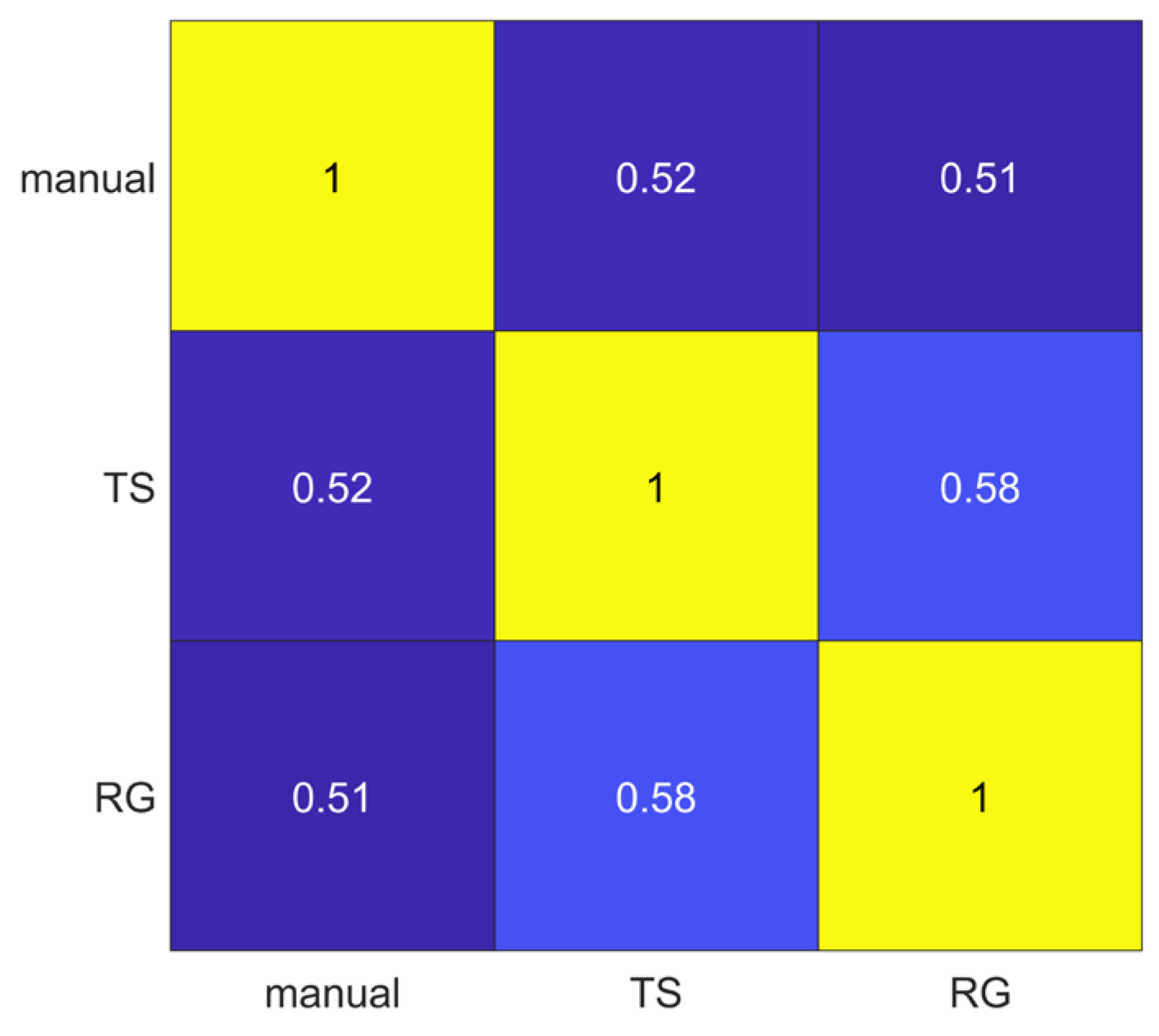
3.2. Segmentation Agreement
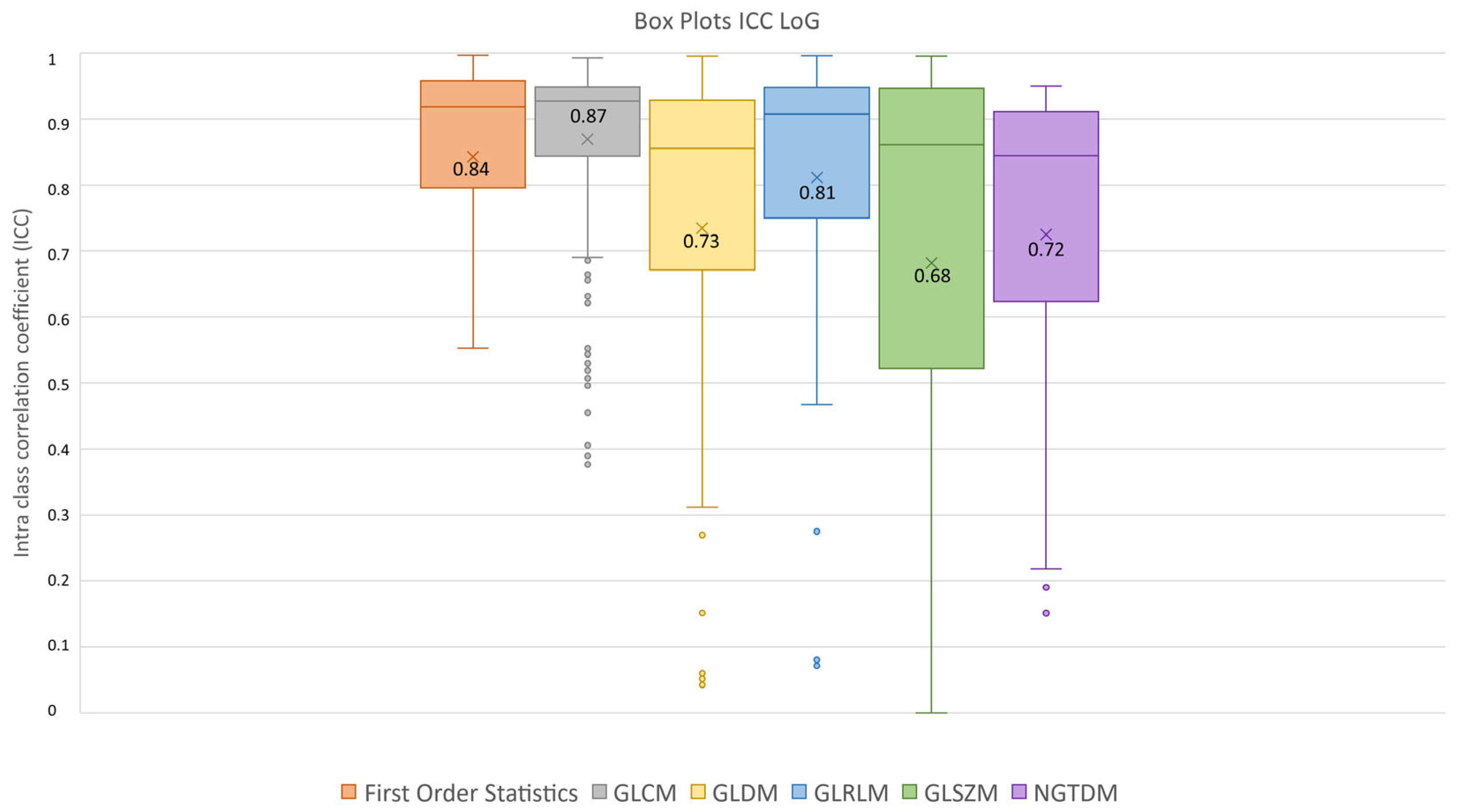
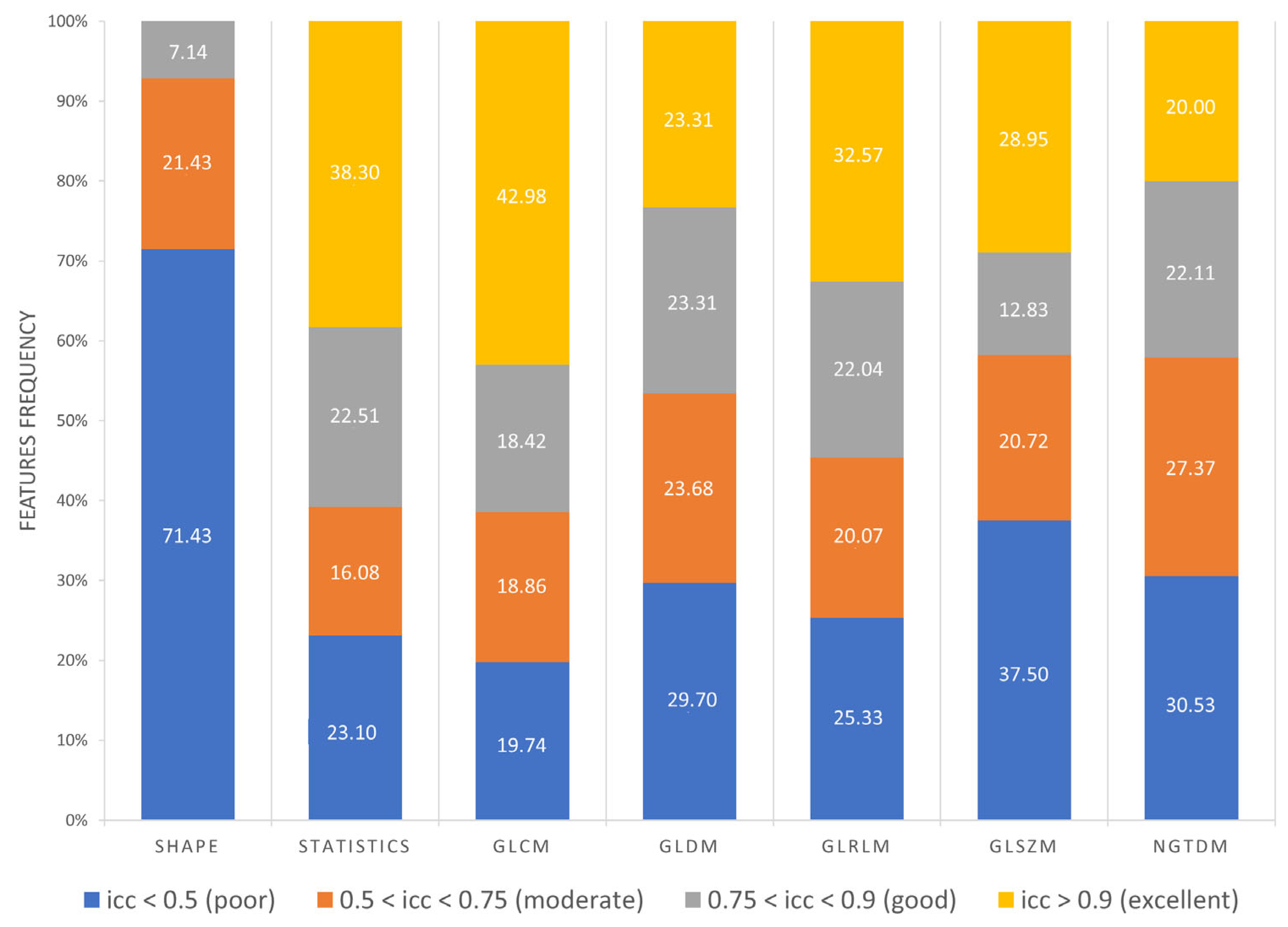
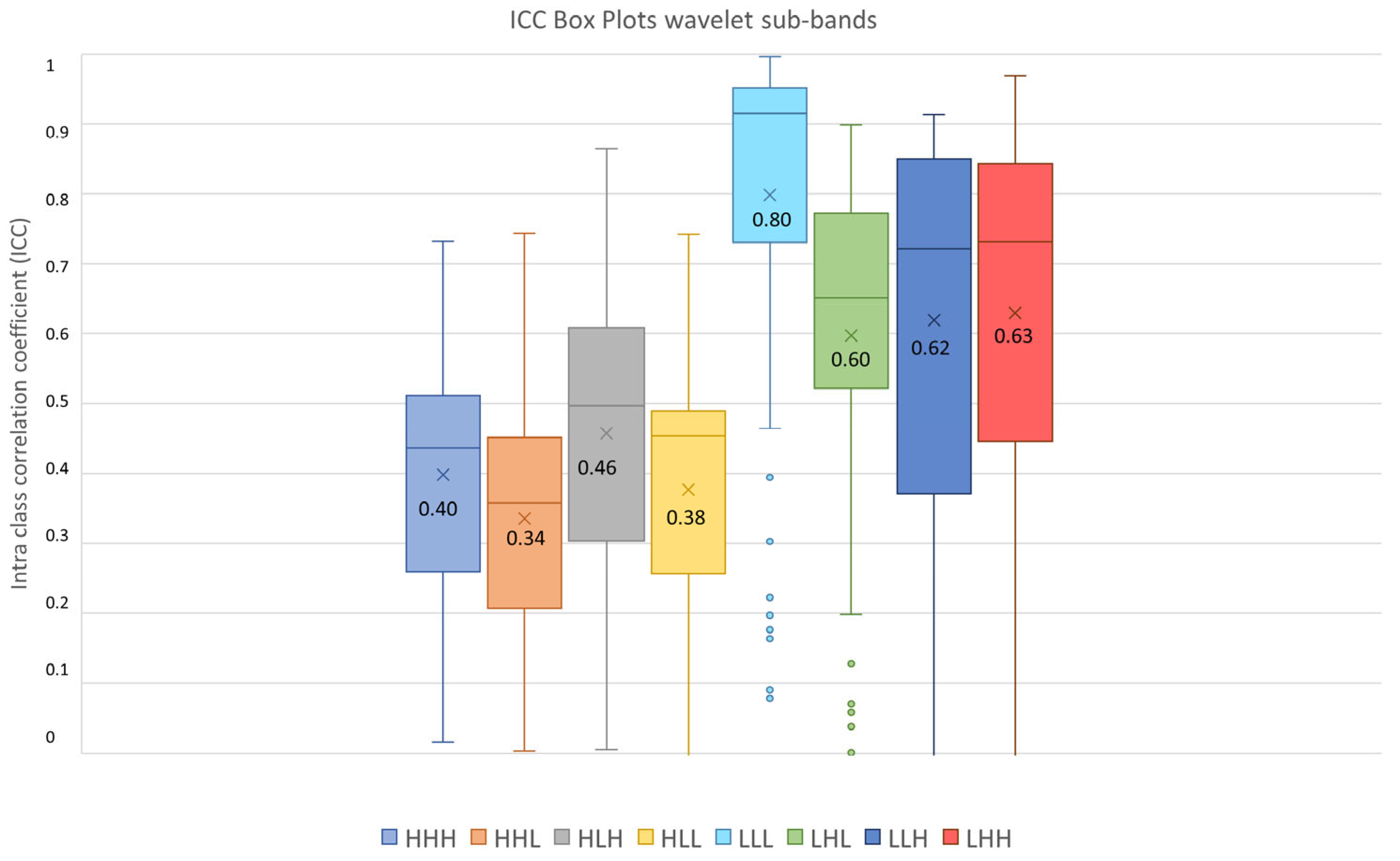
3.3. Feature Robustness Results
- Excellent reproducibility (ICC > 0.9) was reached by 51.18% LoG features, 48.60% original features and only 9.01% wavelet features.
- Good reproducibility (0.75 < ICC < 0.9) was reached by 14.02% original features, 22.47% LoG features and 17.07% wavelet features.
- Moderate reproducibility (0.5 < ICC < 0.75) was reached by 18.69% original features, 15.38% LoG features, 26.08% wavelet features.
- Poor reproducibility (ICC < 0.5) was reached by 18.69% original features, 10.97% LoG features, 47.85 % wavelet features.

3.4. Feature Selection and Machine Learning Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Siegel, R.L.; Miller, K.D.; Wagle, N.S.; Jemal, A. Cancer Statistics, 2023. CA Cancer J. Clin. 2023, 73, 17–48. [Google Scholar] [CrossRef] [PubMed]
- Sharma, S.; Zapatero-Rodríguez, J.; O’Kennedy, R. Prostate Cancer Diagnostics: Clinical Challenges and the Ongoing Need for Disruptive and Effective Diagnostic Tools. Biotechnol. Adv. 2017, 35, 135–149. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, H.U.; El-Shater Bosaily, A.; Brown, L.C.; Gabe, R.; Kaplan, R.; Parmar, M.K.; Collaco-Moraes, Y.; Ward, K.; Hindley, R.G.; Freeman, A.; et al. Diagnostic Accuracy of Multi-Parametric MRI and TRUS Biopsy in Prostate Cancer (PROMIS): A Paired Validating Confirmatory Study. Lancet 2017, 389, 815–822. [Google Scholar] [CrossRef] [PubMed]
- Moe, A.; Hayne, D. Transrectal Ultrasound Biopsy of the Prostate: Does It Still Have a Role in Prostate Cancer Diagnosis? Transl. Androl. Urol. 2020, 9, 3018–3024. [Google Scholar] [CrossRef] [PubMed]
- Prostate Cancer—DIAGNOSTIC EVALUATION—Uroweb. Available online: https://uroweb.org/guidelines/prostate-cancer/chapter/diagnostic-evaluation (accessed on 9 October 2023).
- Maurer, T.; Eiber, M.; Schwaiger, M.; Gschwend, J.E. Current Use of PSMA–PET in Prostate Cancer Management. Nat. Rev. Urol. 2016, 13, 226–235. [Google Scholar] [CrossRef] [PubMed]
- Han, S.; Woo, S.; Kim, Y.J.; Suh, C.H. Impact of 68Ga-PSMA PET on the Management of Patients with Prostate Cancer: A Systematic Review and Meta-Analysis. Eur. Urol. 2018, 74, 179–190. [Google Scholar] [CrossRef]
- Perera, M.; Papa, N.; Roberts, M.; Williams, M.; Udovicich, C.; Vela, I.; Christidis, D.; Bolton, D.; Hofman, M.S.; Lawrentschuk, N.; et al. Gallium-68 Prostate-Specific Membrane Antigen Positron Emission Tomography in Advanced Prostate Cancer—Updated Diagnostic Utility, Sensitivity, Specificity, and Distribution of Prostate-Specific Membrane Antigen-Avid Lesions: A Systematic Review and Meta-Analysis. Eur. Urol. 2020, 77, 403–417. [Google Scholar] [CrossRef]
- Giesel, F.L.; Hadaschik, B.; Cardinale, J.; Radtke, J.; Vinsensia, M.; Lehnert, W.; Kesch, C.; Tolstov, Y.; Singer, S.; Grabe, N.; et al. F-18 Labelled PSMA-1007: Biodistribution, Radiation Dosimetry and Histopathological Validation of Tumor Lesions in Prostate Cancer Patients. Eur. J. Nucl. Med. Mol. Imaging 2017, 44, 678–688. [Google Scholar] [CrossRef]
- Banna, G.L.; Anile, G.; Russo, G.; Vigneri, P.; Castaing, M.; Nicolosi, M.; Strano, S.; Gieri, S.; Spina, R.; Patanè, D.; et al. Predictive and Prognostic Value of Early Disease Progression by PET Evaluation in Advanced Non-Small Cell Lung Cancer. Oncology 2017, 92, 39–47. [Google Scholar] [CrossRef]
- Liu, J.; Li, S.; Lin, H.; Pang, P.; Luo, P.; Fan, B.; Yu, J. Development of MRI-Based Radiomics Predictive Model for Classifying Endometrial Lesions. Sci. Rep. 2023, 13, 1590. [Google Scholar] [CrossRef] [PubMed]
- Torrisi, S.E.; Palmucci, S.; Stefano, A.; Russo, G.; Torcitto, A.G.; Falsaperla, D.; Gioè, M.; Pavone, M.; Vancheri, A.; Sambataro, G.; et al. Assessment of Survival in Patients with Idiopathic Pulmonary Fibrosis Using Quantitative HRCT Indexes. Multidiscip. Respir. Med. 2018, 13, 43. [Google Scholar] [CrossRef]
- Liberini, V.; Laudicella, R.; Balma, M.; Nicolotti, D.G.; Buschiazzo, A.; Grimaldi, S.; Lorenzon, L.; Bianchi, A.; Peano, S.; Bartolotta, T.V.; et al. Radiomics and Artificial Intelligence in Prostate Cancer: New Tools for Molecular Hybrid Imaging and Theragnostics. Eur. Radiol. Exp. 2022, 6, 27. [Google Scholar] [CrossRef] [PubMed]
- Vernuccio, F.; Arnone, F.; Cannella, R.; Verro, B.; Comelli, A.; Agnello, F.; Stefano, A.; Gargano, R.; Rodolico, V.; Salvaggio, G.; et al. Diagnostic Performance of Qualitative and Radiomics Approach to Parotid Gland Tumors: Which Is the Added Benefit of Texture Analysis? Br. J. Radiol. 2021, 94, 20210340. [Google Scholar] [CrossRef] [PubMed]
- Alongi, P.; Sardina, D.S.; Coppola, R.; Scalisi, S.; Puglisi, V.; Arnone, A.; Raimondo, G.D.; Munerati, E.; Alaimo, V.; Midiri, F.; et al. 18F-Florbetaben PET/CT to Assess Alzheimer’s Disease: A New Analysis Method for Regional Amyloid Quantification. J. Neuroimaging 2019, 29, 383–393. [Google Scholar] [CrossRef] [PubMed]
- Nepi, V.; Pasini, G.; Bini, F.; Marinozzi, F.; Russo, G.; Stefano, A. MRI-Based Radiomics Analysis for Identification of Features Correlated with the Expanded Disability Status Scale of Multiple Sclerosis Patients. In Image Analysis and Processing. ICIAP 2022 Workshops; Springer: Cham, Switzerland, 2022; Volume 13373, pp. 362–373. [Google Scholar] [CrossRef]
- Zang, S.; Ai, S.; Yang, R.; Zhang, P.; Wu, W.; Zhao, Z.; Ni, Y.; Zhang, Q.; Sun, H.; Guo, H.; et al. Development and Validation of 68Ga-PSMA-11 PET/CT-Based Radiomics Model to Detect Primary Prostate Cancer. EJNMMI Res. 2022, 12, 63. [Google Scholar] [CrossRef]
- Zamboglou, C.; Bettermann, A.S.; Gratzke, C.; Mix, M.; Ruf, J.; Kiefer, S.; Jilg, C.A.; Benndorf, M.; Spohn, S.; Fassbender, T.F.; et al. Uncovering the Invisible—Prevalence, Characteristics, and Radiomics Feature–Based Detection of Visually Undetectable Intraprostatic Tumor Lesions in 68GaPSMA-11 PET Images of Patients with Primary Prostate Cancer. Eur. J. Nucl. Med. Mol. Imaging 2021, 48, 1987–1997. [Google Scholar] [CrossRef]
- Papp, L.; Spielvogel, C.P.; Grubmüller, B.; Grahovac, M.; Krajnc, D.; Ecsedi, B.; Sareshgi, R.A.M.; Mohamad, D.; Hamboeck, M.; Rausch, I.; et al. Supervised Machine Learning Enables Non-Invasive Lesion Characterization in Primary Prostate Cancer with [68Ga]Ga-PSMA-11 PET/MRI. Eur. J. Nucl. Med. Mol. Imaging 2021, 48, 1795–1805. [Google Scholar] [CrossRef] [PubMed]
- Yao, F.; Bian, S.; Zhu, D.; Yuan, Y.; Pan, K.; Pan, Z.; Feng, X.; Tang, K.; Yang, Y. Machine Learning-Based Radiomics for Multiple Primary Prostate Cancer Biological Characteristics Prediction with 18F-PSMA-1007 PET: Comparison among Different Volume Segmentation Thresholds. Radiol. Medica 2022, 127, 1170–1178. [Google Scholar] [CrossRef] [PubMed]
- Acar, E.; Leblebici, A.; Ellidokuz, B.E.; Başbinar, Y.; Kaya, G.Ç. Full Paper: Machine Learning for Differentiating Metastatic and Completely Responded Sclerotic Bone Lesion in Prostate Cancer: A Retrospective Radiomics Study. Br. J. Radiol. 2019, 92, 20190286. [Google Scholar] [CrossRef]
- Hinzpeter, R.; Baumann, L.; Guggenberger, R.; Huellner, M.; Alkadhi, H.; Baessler, B. Radiomics for Detecting Prostate Cancer Bone Metastases Invisible in CT: A Proof-of-Concept Study. Eur. Radiol. 2022, 32, 1823–1832. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Li, Y.; Zheng, A.; Gao, J.; Yuan, W.; Shen, C.; Bai, L.; Duan, X. Evaluation of a Radiomics Nomogram Derived from Fluoride-18 PSMA-1007 PET/CT for Risk Stratification in Newly Diagnosed Prostate Cancer. Front. Oncol. 2022, 12, 1018833. [Google Scholar] [CrossRef]
- Zwanenburg, A.; Vallières, M.; Abdalah, M.A.; Aerts, H.J.W.L.; Andrearczyk, V.; Apte, A.; Ashrafinia, S.; Bakas, S.; Beukinga, R.J.; Boellaard, R.; et al. The Image Biomarker Standardization Initiative: Standardized Quantitative Radiomics for High-Throughput Image-Based Phenotyping. Radiology 2020, 295, 328–338. [Google Scholar] [CrossRef]
- Pasini, G.; Stefano, A.; Russo, G.; Comelli, A.; Marinozzi, F.; Bini, F. Phenotyping the Histopathological Subtypes of Non-Small-Cell Lung Carcinoma: How Beneficial Is Radiomics? Diagnostics 2023, 13, 1167. [Google Scholar] [CrossRef] [PubMed]
- Poirot, M.G.; Caan, M.W.A.; Ruhe, H.G.; Bjørnerud, A.; Groote, I.; Reneman, L.; Marquering, H.A. Robustness of Radiomics to Variations in Segmentation Methods in Multimodal Brain MRI. Sci. Rep. 2022, 12, 16712. [Google Scholar] [CrossRef] [PubMed]
- Haarburger, C.; Müller-Franzes, G.; Weninger, L.; Kuhl, C.; Truhn, D.; Merhof, D. Radiomics Feature Reproducibility under Inter-Rater Variability in Segmentations of CT Images. Sci. Rep. 2020, 10, 12688. [Google Scholar] [CrossRef] [PubMed]
- Granzier, R.W.Y.; Verbakel, N.M.H.; Ibrahim, A.; van Timmeren, J.E.; van Nijnatten, T.J.A.; Leijenaar, R.T.H.; Lobbes, M.B.I.; Smidt, M.L.; Woodruff, H.C. MRI-Based Radiomics in Breast Cancer: Feature Robustness with Respect to Inter-Observer Segmentation Variability. Sci. Rep. 2020, 10, 14163. [Google Scholar] [CrossRef] [PubMed]
- Laudicella, R.; Spataro, A.; Crocè, L.; Giacoppo, G.; Romano, D.; Davì, V.; Lopes, M.; Librando, M.; Nicocia, A.; Rappazzo, A.; et al. Preliminary Findings of the Role of FAPi in Prostate Cancer Theranostics. Diagnostics 2023, 13, 1175. [Google Scholar] [CrossRef]
- Prostate Cancer: Stages and Grades|Cancer.Net. Available online: https://www.cancer.net/cancer-types/prostate-cancer/stages-and-grades (accessed on 23 November 2023).
- Pasini, G.; Bini, F.; Russo, G.; Comelli, A.; Marinozzi, F.; Stefano, A. MatRadiomics: A Novel and Complete Radiomics Framework, from Image Visualization to Predictive Model. J. Imaging 2022, 8, 221. [Google Scholar] [CrossRef]
- Whitaker, R.T. A Level-Set Approach to 3D Reconstruction from Range Data. Int. J. Comput. Vis. 1998, 29, 203–231. [Google Scholar] [CrossRef]
- Chan, T.F.; Vese, L.A. Active Contours without Edges. IEEE Trans. Image Process. 2001, 10, 266–277. [Google Scholar] [CrossRef] [PubMed]
- Foster, B.; Bagci, U.; Mansoor, A.; Xu, Z.; Mollura, D.J. A Review on Segmentation of Positron Emission Tomography Images. Comput. Biol. Med. 2014, 50, 76–96. [Google Scholar] [CrossRef] [PubMed]
- Schinagl, D.A.X.; Vogel, W.V.; Hoffmann, A.L.; van Dalen, J.A.; Oyen, W.J.; Kaanders, J.H.A.M. Comparison of Five Segmentation Tools for 18F-Fluoro-Deoxy-Glucose–Positron Emission Tomography–Based Target Volume Definition in Head and Neck Cancer. Int. J. Radiat. Oncol. Biol. Phys. 2007, 69, 1282–1289. [Google Scholar] [CrossRef] [PubMed]
- Jaccard, P. The Distribution of the Flora in the Alpine Zone. New Phytol. 1912, 11, 37–50. [Google Scholar] [CrossRef]
- Van Griethuysen, J.J.M.; Fedorov, A.; Parmar, C.; Hosny, A.; Aucoin, N.; Narayan, V.; Beets-Tan, R.G.H.; Fillion-Robin, J.C.; Pieper, S.; Aerts, H.J.W.L. Computational Radiomics System to Decode the Radiographic Phenotype. Cancer Res. 2017, 77, e104–e107. [Google Scholar] [CrossRef] [PubMed]
- Haralick, R.M.; Dinstein, I.; Shanmugam, K. Textural Features for Image Classification. IEEE Trans. Syst. Man Cybern. 1973, SMC-3, 610–621. [Google Scholar] [CrossRef]
- Galloway, M.M. Texture Analysis Using Gray Level Run Lengths. Comput. Graph. Image Process. 1975, 4, 172–179. [Google Scholar] [CrossRef]
- Thibault, G.; Angulo, J.; Meyer, F. Advanced Statistical Matrices for Texture Characterization: Application to Cell Classification. IEEE Trans. Biomed. Eng. 2014, 61, 630–637. [Google Scholar] [CrossRef]
- Amadasun, M.; King, R. Texural Features Corresponding to Texural Properties. IEEE Trans. Syst. Man Cybern. 1989, 19, 1264–1274. [Google Scholar] [CrossRef]
- Sun, C.; Wee, W.G. Neighboring Gray Level Dependence Matrix for Texture Classification. Comput. Vis. Graph. Image Process 1983, 23, 341–352. [Google Scholar] [CrossRef]
- Koo, T.K.; Li, M.Y. A Guideline of Selecting and Reporting Intraclass Correlation Coefficients for Reliability Research. J. Chiropr. Med. 2016, 15, 155–163. [Google Scholar] [CrossRef]
- McGraw, K.O.; Wong, S.P. Forming Inferences about Some Intraclass Correlation Coefficients. Psychol. Methods 1996, 1, 30–46. [Google Scholar] [CrossRef]
- Barone, S.; Cannella, R.; Comelli, A.; Pellegrino, A.; Salvaggio, G.; Stefano, A.; Vernuccio, F. Hybrid Descriptive-Inferential Method for Key Feature Selection in Prostate Cancer Radiomics. Appl. Stoch. Models Bus. Ind. 2021, 37, 961–972. [Google Scholar] [CrossRef]
- Lilliefors, H.W. On the Kolmogorov-Smirnov Test for the Exponential Distribution with Mean Unknown. J. Am. Stat. Assoc. 1969, 64, 387–389. [Google Scholar] [CrossRef]
- Lilliefors, H.W. On the Kolmogorov-Smirnov Test for Normality with Mean and Variance Unknown. J. Am. Stat. Assoc. 1967, 62, 399–402. [Google Scholar] [CrossRef]
- Brown, M.B.; Forsythe, A.B. Robust Tests for the Equality of Variances. J. Am. Stat. Assoc. 1974, 69, 364–367. [Google Scholar] [CrossRef]
- Kruskal, W.H.; Wallis, W.A. Use of Ranks in One-Criterion Variance Analysis. J. Am. Stat. Assoc. 1952, 47, 583–621. [Google Scholar] [CrossRef]
- Wilcoxon, F. Individual Comparisons by Ranking Methods. Biom. Bull. 1945, 1, 80. [Google Scholar] [CrossRef]
- Friedman, M. The Use of Ranks to Avoid the Assumption of Normality Implicit in the Analysis of Variance. J. Am. Stat. Assoc. 1937, 32, 675–701. [Google Scholar] [CrossRef]
- Friedman, M. Errata. A Correction. J. Am. Stat. Assoc. 1939, 34, 109. [Google Scholar] [CrossRef]
- Šidák, Z. Rectangular Confidence Regions for the Means of Multivariate Normal Distributions. J. Am. Stat. Assoc. 1967, 62, 626–633. [Google Scholar] [CrossRef]
- DiCiccio, T.J.; Efron, B. Bootstrap Confidence Intervals. Stat. Sci. 1996, 11, 189–212. [Google Scholar] [CrossRef]
- R2023b—Aggiornamenti Alle Famiglie Di Prodotti MATLAB e Simulink—MATLAB & Simulink. Available online: https://it.mathworks.com/products/new_products/latest_features.html (accessed on 24 November 2023).
- Zwanenburg, A. Radiomics in Nuclear Medicine: Robustness, Reproducibility, Standardization, and How to Avoid Data Analysis Traps and Replication Crisis. Eur. J. Nucl. Med. Mol. Imaging 2019, 46, 2638–2655. [Google Scholar] [CrossRef] [PubMed]
- Thulasi Seetha, S.; Garanzini, E.; Tenconi, C.; Marenghi, C.; Avuzzi, B.; Catanzaro, M.; Stagni, S.; Villa, S.; Chiorda, B.N.; Badenchini, F.; et al. Stability of Multi-Parametric Prostate MRI Radiomic Features to Variations in Segmentation. J. Pers. Med. 2023, 13, 1172. [Google Scholar] [CrossRef] [PubMed]
- Scalco, E.; Belfatto, A.; Mastropietro, A.; Rancati, T.; Avuzzi, B.; Messina, A.; Valdagni, R.; Rizzo, G. T2w-MRI Signal Normalization Affects Radiomics Features Reproducibility. Med. Phys. 2020, 47, 1680–1691. [Google Scholar] [CrossRef]
- Parmar, C.; Velazquez, E.R.; Leijenaar, R.; Jermoumi, M.; Carvalho, S.; Mak, R.H.; Mitra, S.; Shankar, B.U.; Kikinis, R.; Haibe-Kains, B.; et al. Robust Radiomics Feature Quantification Using Semiautomatic Volumetric Segmentation. PLoS ONE 2014, 9, e102107. [Google Scholar] [CrossRef] [PubMed]
- Yang, F.; Simpson, G.; Young, L.; Ford, J.; Dogan, N.; Wang, L. Impact of Contouring Variability on Oncological PET Radiomics Features in the Lung. Sci. Rep. 2020, 10, 369. [Google Scholar] [CrossRef] [PubMed]
- Salvaggio, G.; Comelli, A.; Portoghese, M.; Cutaia, G.; Cannella, R.; Vernuccio, F.; Stefano, A.; Dispensa, N.; La Tona, G.; Salvaggio, L.; et al. Deep Learning Network for Segmentation of the Prostate Gland with Median Lobe Enlargement in T2-Weighted MR Images: Comparison with Manual Segmentation Method. Curr. Probl. Diagn. Radiol. 2022, 51, 328–333. [Google Scholar] [CrossRef]
- Lu, L.; Lv, W.; Jiang, J.; Ma, J.; Feng, Q.; Rahmim, A.; Chen, W. Robustness of Radiomic Features in [11C]Choline and [18F]FDG PET/CT Imaging of Nasopharyngeal Carcinoma: Impact of Segmentation and Discretization. Mol. Imaging Biol. 2016, 18, 935–945. [Google Scholar] [CrossRef] [PubMed]
- Pfaehler, E.; Beukinga, R.J.; de Jong, J.R.; Slart, R.H.J.A.; Slump, C.H.; Dierckx, R.A.J.O.; Boellaard, R. Repeatability of 18F-FDG PET Radiomic Features: A Phantom Study to Explore Sensitivity to Image Reconstruction Settings, Noise, and Delineation Method. Med. Phys. 2019, 46, 665–678. [Google Scholar] [CrossRef]
- Keller, H.; Shek, T.; Driscoll, B.; Xu, Y.; Nghiem, B.; Nehmeh, S.; Grkovski, M.; Schmidtlein, C.R.; Budzevich, M.; Balagurunathan, Y.; et al. Noise-Based Image Harmonization Significantly Increases Repeatability and Reproducibility of Radiomics Features in PET Images: A Phantom Study. Tomography 2022, 8, 1113–1128. [Google Scholar] [CrossRef]
- Ibrahim, A.; Primakov, S.; Beuque, M.; Woodruff, H.C.; Halilaj, I.; Wu, G.; Refaee, T.; Granzier, R.; Widaatalla, Y.; Hustinx, R.; et al. Radiomics for Precision Medicine: Current Challenges, Future Prospects, and the Proposal of a New Framework. Methods 2021, 188, 20–29. [Google Scholar] [CrossRef] [PubMed]
- Rai, R.; Holloway, L.C.; Brink, C.; Field, M.; Christiansen, R.L.; Sun, Y.; Barton, M.B.; Liney, G.P. Multicenter Evaluation of MRI-Based Radiomic Features: A Phantom Study. Med. Phys. 2020, 47, 3054–3063. [Google Scholar] [CrossRef] [PubMed]



| Bin width | 0.25 |
| Isotropic Resampling | 2 × 2 × 2 |
| Interpolator | SitkBSpline |
| Wavelet Method | Coif1 |
| Log Sigma | [0.5, 1, 1.5, 2, 2.5, 3, 3.5, 4, 4.5, 5] |
| Normalization | True; Scale = 1 |
| PET/CT Scanner | Patients | Low Grade GS = 3 + 3/3 + 4 | High Grade GS = 4 + 3/4 + 4/4 + 5 |
|---|---|---|---|
| GE | 30 | 20 | 10 |
| Siemens | 51 | 26 | 25 |
| Total | 81 | 46 | 35 |
| PET/CT Scanner | Patients | Low Grade GS = 3 + 3/3 + 4 | High Grade GS = 4 + 3/4 + 4/4 + 5 |
|---|---|---|---|
| GE | 29 | 19 | 10 |
| Siemens | 49 | 24 | 25 |
| Total | 78 | 43 | 35 |
| Image Type | Shape | Statistics | GLCM | GLDM | GLRLM | GLSZM | NGTDM |
|---|---|---|---|---|---|---|---|
| Original | 0.27 | 0.87 | 0.89 | 0.75 | 0.77 | 0.65 | 0.72 |
| LoG | - | 0.84 | 0.87 | 0.73 | 0.81 | 0.68 | 0.72 |
| Wavelet | - | 0.56 | 0.56 | 0.51 | 0.54 | 0.44 | 0.49 |
| Segmentation | Selected Features |
|---|---|
| Manual | wavelet_LLL_firstorder_Minimum |
| TS | wavelet_HHL_glszm_LowGrayLevelZoneEmphasis |
| RG | wavelet_HLH_glszm_LowGrayLevelZoneEmphasis |
| Segmentation | Accuracy | AUC | Sensitivity | Specificity | Precision | Fscore |
|---|---|---|---|---|---|---|
| Manual | 70.64% (70.47–70.81%) | 73.08% (72.69–73.41%) | 51.71% (51.43–52.19%) | 86.05% (86.05–86.05%) | 75.10% (74.99–75.27%) | 61.25% (61.01–61.63%) |
| TS | 70.60% (70.26–70.85%) | 73.40% (73.63–74.38%) | 74.48% (74.29–74.95%) | 67.44% (66.90–67.91%) | 65.07% (64.69–65.42%) | 69.45% (69.21–69.72%) |
| RG | 74.49% (74.10–74.91%) | 79.20% (78.89–79.50%) | 84.38% (83.90–84.86%) | 66.43% (65.81–67.13%) | 67.19% (66.78–67.65%) | 74.80% (74.44–75.19%) |
| Segmentation | Accuracy | AUC | Sensitivity | Specificity | Precision | Fscore |
|---|---|---|---|---|---|---|
| Manual | 70.47% (70–71.11%) | 73% (72.56–73.41%) | 57.05% (56.29–58%) | 81.40% (80.78–82.17%) | 71.43% (70.73–72.45%) | 63.41% (62.72–64.17%) |
| TS | 68.50% (67.78–69.19%) | 72.50% (71.89–73.08%) | 74.19% (73.43–74.86%) | 63.88% (63.02–64.81%) | 62.60% (61.91–63.31%) | 67.89% (67.19–68.51%) |
| RG | 71.84% (74.10–74.91%) | 77.32% (77.06–77.63%) | 85.24% (84.57–85.71%) | 60.93% (60.16–61.63%) | 63.99% (63.57–64.38%) | 73.09% (72.71–73.40%) |
| Segmentation | Accuracy | AUC | Sensitivity | Specificity | Precision | Fscore |
|---|---|---|---|---|---|---|
| Manual | 70.26% (69.65–71.07%) | 67.41% (66.76–67.97%) | 58.76% (57.62–59.71%) | 79.61% (78.76–80.70%) | 70.19% (69.25–71.39%) | 63.92% (63.04–64.77%) |
| TS | 66.37% (65.62–67.14%) | 68.58% (67.84–69.18%) | 67.33% (66.19–68.38%) | 65.58% (64.49–67.21%) | 61.51% (60.61–62.43%) | 64.23% (63.51–65.03%) |
| RG | 71.75% (70.81–72.61%) | 77.86% (77.13–78.58%) | 77.05% (76–78.10%) | 67.44% (66.12–68.84%) | 65.91% (64.89–66.87%) | 71% (70.13–71.82%) |
| Segmentation | Accuracy | AUC | Sensitivity | Specificity | Precision | Fscore |
|---|---|---|---|---|---|---|
| Manual | 67.65% (66.88–68.46%) | 66.77% (65.79–67.61%) | 57.81% (56–59.33%) | 75.66% (74.73–76.59%) | 65.90% (64.93–66.98%) | 61.52% (60.18–62.66%) |
| TS | 63.55% (62.52–64.66%) | 67.40% (66.55–68.12%) | 65.62% (63.60–67.81%) | 61.86% (60.19–63.68%) | 58.40% (57.43–59.62%) | 61.67% (60.29–63.03%) |
| RG | 71.15% (70.43–72.01%) | 73.82% (73.01–74.61%) | 77.05% (75.62–78.48%) | 66.36% (65.27–67.37%) | 65.12% (64.44–65.94%) | 70.54% (69.72–71.46%) |
| Segmentation | Accuracy | AUC | Sensitivity | Specificity | Precision | Fscore |
|---|---|---|---|---|---|---|
| Manual | 63.16% (61.79–64.44%) | 62.91% (61.81–63.88%) | 54.48% (52.27–56.57%) | 70.23% (68.45–71.71%) | 59.87% (58.19–61.35%) | 56.95% (55.09–58.56%) |
| TS | 57.22% (56.11–58.33%) | 62.70% (61.56–63.74%) | 58.19% (56.10–60.38%) | 56.43% (54.88–57.91%) | 52.06% (50.96–53.32%) | 54.87% 53.51–56.30%) |
| RG | 63.72% (62.56–64.74%) | 68.99% (67.88–70.05%) | 64.19% (61.71–65.71%) | 63.33% (61.86–64.81%) | 58.78% (57.52–59.81%) | 61.29% (59.64–62.40%) |
| Segmentation | Accuracy | AUC | Sensitivity | Specificity | Precision | Fscore |
|---|---|---|---|---|---|---|
| Manual | 58.72% (57.48–60.56%) | 56.05% (54.20–57.97%) | 46.19% (43.43–48.95%) | 68.91% (66.82–70.68%) | 54.69% (52.90–57.02%) | 49.90% (47.79–52.48%) |
| TS | 62.14% (60.66–63.50%) | 63.50% (61.91–65.04%) | 66.76% (64.57–69.14%) | 58.37% (56.43–60.54%) | 56.74% (55.34–58.23%) | 61.22% (59.70–62.74%) |
| RG | 65.77% (64.27–66.97%) | 69.24% (67.96–70.33%) | 69.14% (65.90–71.52%) | 63.02% (61.32–64.73%) | 60.35% (58.95–61.55%) | 64.29% (62.18–65.79%) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pasini, G.; Russo, G.; Mantarro, C.; Bini, F.; Richiusa, S.; Morgante, L.; Comelli, A.; Russo, G.I.; Sabini, M.G.; Cosentino, S.; et al. A Critical Analysis of the Robustness of Radiomics to Variations in Segmentation Methods in 18F-PSMA-1007 PET Images of Patients Affected by Prostate Cancer. Diagnostics 2023, 13, 3640. https://doi.org/10.3390/diagnostics13243640
Pasini G, Russo G, Mantarro C, Bini F, Richiusa S, Morgante L, Comelli A, Russo GI, Sabini MG, Cosentino S, et al. A Critical Analysis of the Robustness of Radiomics to Variations in Segmentation Methods in 18F-PSMA-1007 PET Images of Patients Affected by Prostate Cancer. Diagnostics. 2023; 13(24):3640. https://doi.org/10.3390/diagnostics13243640
Chicago/Turabian StylePasini, Giovanni, Giorgio Russo, Cristina Mantarro, Fabiano Bini, Selene Richiusa, Lucrezia Morgante, Albert Comelli, Giorgio Ivan Russo, Maria Gabriella Sabini, Sebastiano Cosentino, and et al. 2023. "A Critical Analysis of the Robustness of Radiomics to Variations in Segmentation Methods in 18F-PSMA-1007 PET Images of Patients Affected by Prostate Cancer" Diagnostics 13, no. 24: 3640. https://doi.org/10.3390/diagnostics13243640
APA StylePasini, G., Russo, G., Mantarro, C., Bini, F., Richiusa, S., Morgante, L., Comelli, A., Russo, G. I., Sabini, M. G., Cosentino, S., Marinozzi, F., Ippolito, M., & Stefano, A. (2023). A Critical Analysis of the Robustness of Radiomics to Variations in Segmentation Methods in 18F-PSMA-1007 PET Images of Patients Affected by Prostate Cancer. Diagnostics, 13(24), 3640. https://doi.org/10.3390/diagnostics13243640












