Differential Diagnosis of Visual Phenomena Associated with Migraine: Spotlight on Aura and Visual Snow Syndrome
Abstract
1. Introduction
2. Migraine Aura
2.1. Clinical Features of Migraine Aura
Positive visual phenomena
|
Negative visual phenomena
|
Disturbances of visual perception
|
| Symptom | Description | Frequency | Notes and Differential Diagnoses |
|---|---|---|---|
| Visual Snow | Uncountable tiny dots in the entire visual field, unremitting for several months Usually black/grey on white background and grey/white on black background; They can also be transparent, white flashing or coloured | Main symptom (100%) | Without other symptoms and normal evaluation, consider primary visual snow |
| Palinopsia | Continued perception of an object after it is no longer in the visual field:
| 33–85% | Consider retinal afterimages, if they only occur when staring at a high contrast image and are in complementary colour Exclude cortical lesions of the posterior visual pathway [19] |
| Enhanced entopic phenomena |
| 71–86% 41–76% 44–71% 53–57% | Exclude ophthalmic pathology |
| Nyctalopia | Poorer vision in dim light | 44–78% | |
| Photophobia | Continuous hypersensitivity to light | 54–81% | Independent from comorbid migraine or aura Can have similar range of a migraine attack [20] |
| Comorbid conditions | |||
| Tinnitus | 34–75% | Overlap with other perceptual disorders, including similar pathophysiology and frequent migraine comorbidity [21] Tinnitus and migraine may worsen VSS | |
| Migraine | 52–72% | ||
| Other neurological comorbidities |
| / | |
| Psychiatric comorbidities |
| frequent | Treatment should be offered |
2.2. Pathophysiology of Aura
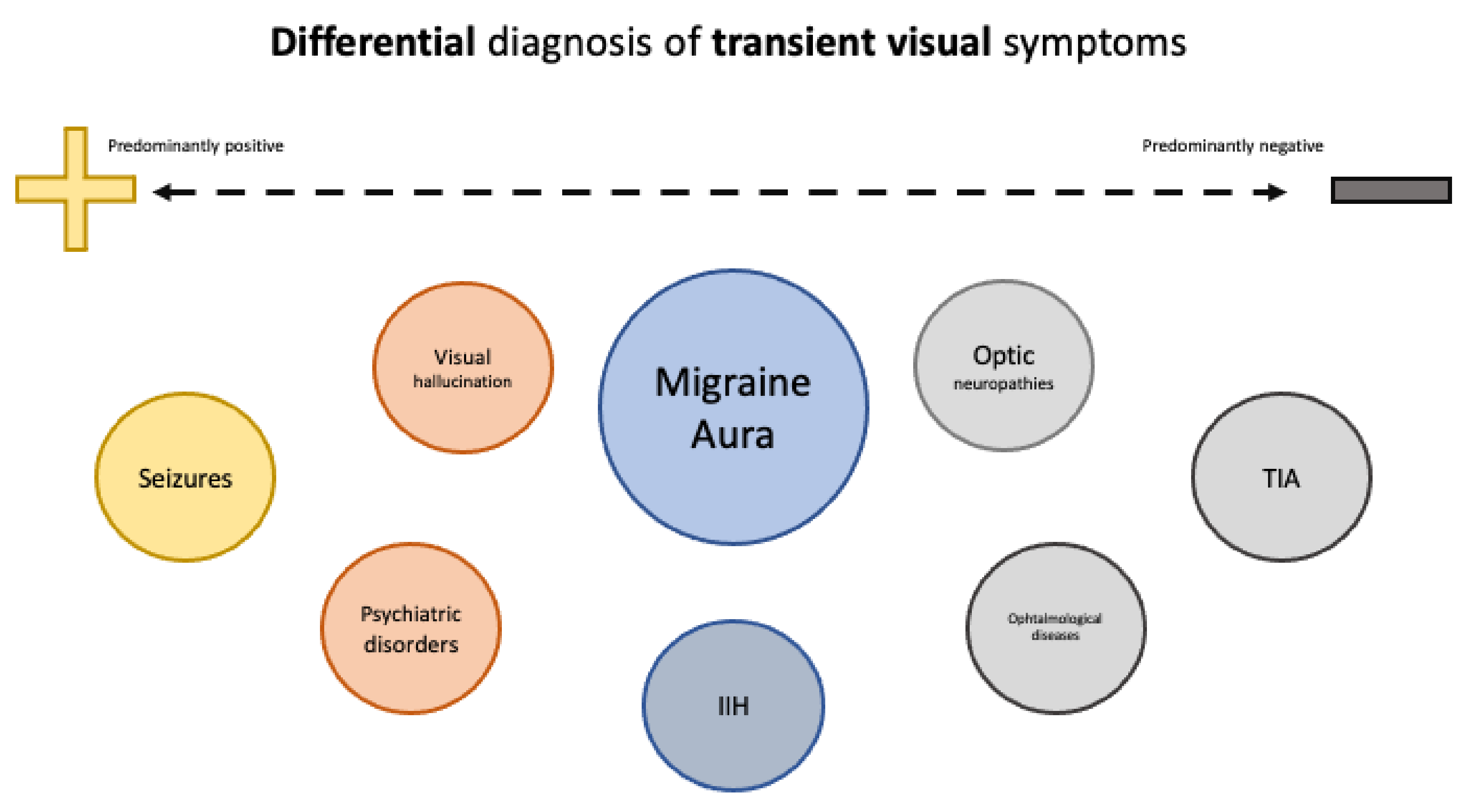
2.3. Differential Diagnosis of Aura
3. Visual Snow Syndrome
3.1. History and Clinical Features of Visual Snow Syndrome
3.2. Pathophysiology of VSS
3.3. Differential Diagnosis of VSS
4. Treatment
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Steiner, T.J.; Stovner, L.J.; Jensen, R.; Uluduz, D.; Katsarava, Z. Lifting The Burden: The Global Campaign against Headache. Migraine remains second among the world’s causes of disability, and first among young women: Findings from GBD2019. J. Headache Pain 2020, 21, 137. [Google Scholar] [CrossRef] [PubMed]
- Ferrari, M.D.; Goadsby, P.J.; Burstein, R.; Kurth, T.; Ayata, C.; Charles, A.; Ashina, M.; van den Maagdenberg, A.; Dodick, D.W. Migraine. Nat. Rev. Dis. Prim. 2022, 8, 2. [Google Scholar] [CrossRef] [PubMed]
- Headache Classification Committee of the International Headache Society (IHS). The International Classification of Headache Disorders, 3rd edition. Cephalalgia 2018, 38, 1–211. [Google Scholar] [CrossRef] [PubMed]
- Eigenbrodt, A.K.; Ashina, H.; Khan, S.; Diener, H.C.; Mitsikostas, D.D.; Sinclair, A.J.; Pozo-Rosich, P.; Martelletti, P.; Ducros, A.; Lanteri-Minet, M.; et al. Diagnosis and management of migraine in ten steps. Nat. Rev. Neurol. 2021, 17, 501–514. [Google Scholar] [CrossRef] [PubMed]
- Queiroz, L.P.; Friedman, D.I.; Rapoport, A.M.; Purdy, R.A. Characteristics of migraine visual aura in Southern Brazil and Northern USA. Cephalalgia 2011, 31, 1652–1658. [Google Scholar] [CrossRef]
- Schankin, C.J.; Viana, M.; Goadsby, P.J. Persistent and Repetitive Visual Disturbances in Migraine: A Review. Headache 2017, 57, 1–16. [Google Scholar] [CrossRef]
- Viana, M.; Tronvik, E.A.; Do, T.P.; Zecca, C.; Hougaard, A. Clinical features of visual migraine aura: A systematic review. J. Headache Pain 2019, 20, 64. [Google Scholar] [CrossRef]
- Lykke Thomsen, L.; Kirchmann Eriksen, M.; Faerch Romer, S.; Andersen, I.; Ostergaard, E.; Keiding, N.; Olesen, J.; Russell, M.B. An epidemiological survey of hemiplegic migraine. Cephalalgia 2002, 22, 361–375. [Google Scholar] [CrossRef]
- Tsao, Y.-C.; Wang, Y.-F.; Fuh, J.-L.; Chen, W.-T.; Lai, K.-L.; Liu, H.-Y.; Wang, S.-J.; Chen, S.-P. Non-aura visual disturbance with high visual aura rating scale scores has stronger association with migraine chronification than typical aura. Cephalalgia 2022, 42, 1487–1497. [Google Scholar] [CrossRef]
- Puledda, F.; Ffytche, D.H.; O’Daly, O.; Goadsby, P.J. Imaging the Visual Network in the Migraine Spectrum. Front. Neurol. 2019, 10, 1325. [Google Scholar] [CrossRef]
- Queiroz, L.P.; Rapoport, A.M.; Weeks, R.E.; Sheftell, F.D.; Siegel, S.E.; Baskin, S.M. Characteristics of migraine visual aura. Headache 1997, 37, 137–141. [Google Scholar] [CrossRef]
- Mastria, G.; Mancini, V.; Cesare, M.D.; Puma, M.; Alessiani, M.; Petolicchio, B.; Viganò, A.; Piero, V.D. Prevalence and characteristics of Alice in Wonderland Syndrome in adult migraineurs: Perspectives from a tertiary referral headache unit. Cephalalgia 2021, 41, 515–524. [Google Scholar] [CrossRef]
- Podoll, K.; Robinson, D. Corona phenomenon as visual aura symptom in migraine. Cephalalgia 2001, 21, 712–717. [Google Scholar] [CrossRef]
- Schott, G.D. Exploring the visual hallucinations of migraine aura: The tacit contribution of illustration. Brain 2007, 130, 1690–1703. [Google Scholar] [CrossRef]
- Hansen, J.M.; Goadsby, P.J.; Charles, A.C. Variability of clinical features in attacks of migraine with aura. Cephalalgia 2016, 36, 216–224. [Google Scholar] [CrossRef]
- Russell, M.B.; Iversen, H.K.; Olesen, J. Improved description of the migraine aura by a diagnostic aura diary. Cephalalgia 1994, 14, 107–117. [Google Scholar] [CrossRef]
- Eriksen, M.K.; Thomsen, L.L.; Andersen, I.; Nazim, F.; Olesen, J. Clinical characteristics of 362 patients with familial migraine with aura. Cephalalgia 2004, 24, 564–575. [Google Scholar] [CrossRef]
- Foroozan, R.; Cutrer, F.M. Transient Neurologic Dysfunction in Migraine. Neurol. Clin. 2019, 37, 673–694. [Google Scholar] [CrossRef]
- Schimansky, S.; Bennetto, L.; Harrison, R. Palinopsia. Pr. Neurol. 2022, 22, 392–395. [Google Scholar] [CrossRef]
- Eren, O.E.; Ruscheweyh, R.; Straube, A.; Schankin, C.J. Quantification of photophobia in visual snow syndrome: A case-control study. Cephalalgia 2020, 40, 393–398. [Google Scholar] [CrossRef]
- Klein, A.; Schankin, C.J. Visual snow syndrome, the spectrum of perceptual disorders, and migraine as a common risk factor: A narrative review. Headache 2021, 61, 1306–1313. [Google Scholar] [CrossRef] [PubMed]
- Hansen, J.M.; Baca, S.M.; Vanvalkenburgh, P.; Charles, A. Distinctive anatomical and physiological features of migraine aura revealed by 18 years of recording. Brain 2013, 136, 3589–3595. [Google Scholar] [CrossRef]
- Viana, M.; Sances, G.; Linde, M.; Nappi, G.; Khaliq, F.; Goadsby, P.J.; Tassorelli, C. Prolonged migraine aura: New insights from a prospective diary-aided study. J. Headache Pain 2018, 19, 77. [Google Scholar] [CrossRef] [PubMed]
- Viana, M.; Linde, M.; Sances, G.; Ghiotto, N.; Guaschino, E.; Allena, M.; Terrazzino, S.; Nappi, G.; Goadsby, P.J.; Tassorelli, C. Migraine aura symptoms: Duration, succession and temporal relationship to headache. Cephalalgia 2016, 36, 413–421. [Google Scholar] [CrossRef] [PubMed]
- Goadsby, P.J.; Holland, P.R.; Martins-Oliveira, M.; Hoffmann, J.; Schankin, C.; Akerman, S. Pathophysiology of Migraine: A Disorder of Sensory Processing. Physiol. Rev. 2017, 97, 553–622. [Google Scholar] [CrossRef]
- Leao, A.A. Further observations on the spreading depression of activity in the cerebral cortex. J. Neurophysiol. 1947, 10, 409–414. [Google Scholar] [CrossRef]
- Leão, A.A.P. Spreading depression of activity in the cerebral cortex. J. Neurophysiol. 1944, 7, 359–390. [Google Scholar] [CrossRef]
- Hayne, D.P.; Martin, P.R. Relating Photophobia, Visual Aura, and Visual Triggers of Headache and Migraine. Headache 2019, 59, 430–442. [Google Scholar] [CrossRef]
- Recober, A. Pathophysiology of Migraine. Continuum 2021, 27, 586–596. [Google Scholar] [CrossRef]
- Tarnutzer, A.A.; Lee, S.H.; Robinson, K.A.; Wang, Z.; Edlow, J.A.; Newman-Toker, D.E. ED misdiagnosis of cerebrovascular events in the era of modern neuroimaging: A meta-analysis. Neurology 2017, 88, 1468–1477. [Google Scholar] [CrossRef]
- Mitsikostas, D.D.; Ashina, M.; Craven, A.; Diener, H.C.; Goadsby, P.J.; Ferrari, M.D.; Lampl, C.; Paemeleire, K.; Pascual, J.; Siva, A.; et al. European Headache Federation consensus on technical investigation for primary headache disorders. J. Headache Pain 2015, 17, 5. [Google Scholar] [CrossRef]
- Angus-Leppan, H. Migraine: Mimics, borderlands and chameleons. Pr. Neurol. 2013, 13, 308–318. [Google Scholar] [CrossRef]
- Petrusic, I.; Viana, M.; Zecca, C.; Zidverc-Trajkovic, J. Dysphasia and Other Higher Cortical Dysfunctions During the Migraine Aura-a Systematic Review of Literature. Curr. Pain Headache Rep. 2020, 24, 3. [Google Scholar] [CrossRef]
- Hoffmann, J.; Mollan, S.P.; Paemeleire, K.; Lampl, C.; Jensen, R.H.; Sinclair, A.J. European Headache Federation guideline on idiopathic intracranial hypertension. J. Headache Pain 2018, 19, 93. [Google Scholar] [CrossRef]
- Liu, G.T.; Schatz, N.J.; Galetta, S.L.; Volpe, N.J.; Skobieranda, F.; Kosmorsky, G.S. Persistent positive visual phenomena in migraine. Neurology 1995, 45, 664–668. [Google Scholar] [CrossRef]
- Jager, H.R.; Giffin, N.J.; Goadsby, P.J. Diffusion- and perfusion-weighted MR imaging in persistent migrainous visual disturbances. Cephalalgia 2005, 25, 323–332. [Google Scholar] [CrossRef]
- Schankin, C.J.; Goadsby, P.J. Visual snow—Persistent positive visual phenomenon distinct from migraine aura. Curr. Pain Headache Rep. 2015, 19, 23. [Google Scholar] [CrossRef]
- Schankin, C.J.; Maniyar, F.H.; Digre, K.B.; Goadsby, P.J. ’Visual snow’—A disorder distinct from persistent migraine aura. Brain 2014, 137, 1419–1428. [Google Scholar] [CrossRef]
- White, O.B.; Clough, M.; McKendrick, A.M.; Fielding, J. Visual Snow: Visual Misperception. J. Neuro-Ophthalmol. Off. J. N. Am. Neuro-Ophthalmol. Soc. 2018, 38, 514–521. [Google Scholar] [CrossRef]
- Schankin, C.J.; Maniyar, F.H.; Sprenger, T.; Chou, D.E.; Eller, M.; Goadsby, P.J. The relation between migraine, typical migraine aura and "visual snow". Headache 2014, 54, 957–966. [Google Scholar] [CrossRef]
- Kondziella, D.; Olsen, M.H.; Dreier, J.P. Prevalence of visual snow syndrome in the UK. Eur. J. Neurol. 2020, 27, 764–772. [Google Scholar] [CrossRef] [PubMed]
- Schankin, C.J.; Puledda, F.; Goadsby, P.J. Visual snow syndrome: Is it normal or a disorder—and what to do with patients? Eur. J. Neurol. 2020, 27, 2393–2395. [Google Scholar] [CrossRef] [PubMed]
- Puledda, F.; Schankin, C.; Goadsby, P.J. Visual snow syndrome: A clinical and phenotypical description of 1100 cases. Neurology 2020, 94, e564–e574. [Google Scholar] [CrossRef] [PubMed]
- Graber, M.; Scutelnic, A.; Klein, A.; Puledda, F.; Goadsby, P.J.; Schankin, C.J. Natural course of visual snow syndrome: A long-term follow-up study. Brain Commun. 2022, 4, fcac230. [Google Scholar] [CrossRef] [PubMed]
- Mehta, D.G.; Garza, I.; Robertson, C.E. Two hundred and forty-eight cases of visual snow: A review of potential inciting events and contributing comorbidities. Cephalalgia 2021, 41, 1015–1026. [Google Scholar] [CrossRef]
- Hodak, J.; Fischer, U.; Bassetti, C.L.A.; Schankin, C.J. Episodic Visual Snow Associated With Migraine Attacks. JAMA Neurol. 2020, 77, 392–393. [Google Scholar] [CrossRef]
- Puledda, F.; Villar-Martínez, M.D.; Goadsby, P.J. Case Report: Transformation of Visual Snow Syndrome From Episodic to Chronic Associated With Acute Cerebellar Infarct. Front. Neurol. 2022, 13, 811490. [Google Scholar] [CrossRef]
- Puledda, F.; Vandenbussche, N.; Moreno-Ajona, D.; Eren, O.; Schankin, C.; Goadsby, P.J. Evaluation of treatment response and symptom progression in 400 patients with visual snow syndrome. Br. J. Ophthalmol. 2022, 106, 1318–1324. [Google Scholar] [CrossRef]
- Gersztenkorn, D.; Lee, A.G. Palinopsia revamped: A systematic review of the literature. Surv. Ophthalmol. 2015, 60, 1–35. [Google Scholar] [CrossRef]
- Solly, E.J.; Clough, M.; Foletta, P.; White, O.B.; Fielding, J. The Psychiatric Symptomology of Visual Snow Syndrome. Front. Neurol. 2021, 12, 703006. [Google Scholar] [CrossRef]
- Metzler, A.I.; Robertson, C.E. Visual Snow Syndrome: Proposed Criteria, Clinical Implications, and Pathophysiology. Curr. Neurol. Neurosci. Rep. 2018, 18, 52. [Google Scholar] [CrossRef]
- Sampatakakis, S.N.; Lymperopoulos, L.; Mavridis, T.; Karagiorgis, G.; Papadopoulos, C.; Deligianni, C.I.; Mitsikostas, D.D. Visual snow: A systematic review and a case series. Cephalalgia 2022, 42, 1409–1419. [Google Scholar] [CrossRef]
- Viana, M.; Puledda, F.; Goadsby, P.J. Visual snow syndrome: A comparison between an Italian and British population. Eur. J. Neurol. 2020, 27, 2099–2101. [Google Scholar] [CrossRef]
- Lauschke, J.L.; Plant, G.T.; Fraser, C.L. Visual snow: A thalamocortical dysrhythmia of the visual pathway? J. Clin. Neurosci. Off. J. Neurosurg. Soc. Australas. 2016, 28, 123–127. [Google Scholar] [CrossRef]
- Puledda, F.; Schankin, C.; Digre, K.; Goadsby, P.J. Visual snow syndrome: What we know so far. Curr. Opin. Neurol. 2018, 31, 52–58. [Google Scholar] [CrossRef]
- Boulloche, N.; Denuelle, M.; Payoux, P.; Fabre, N.; Trotter, Y.; Geraud, G. Photophobia in migraine: An interictal PET study of cortical hyperexcitability and its modulation by pain. J. Neurol. Neurosurg. Psychiatry 2010, 81, 978–984. [Google Scholar] [CrossRef]
- Silvestro, M.; Tessitore, A.; Di Nardo, F.; Scotto di Clemente, F.; Trojsi, F.; Cirillo, M.; Esposito, F.; Tedeschi, G.; Russo, A. Functional connectivity changes in complex migraine aura: Beyond the visual network. Eur. J. Neurol. 2022, 29, 295–304. [Google Scholar] [CrossRef]
- Shibata, M.; Tsutsumi, K.; Iwabuchi, Y.; Kameyama, M.; Takizawa, T.; Nakahara, T.; Fujiwara, H.; Jinzaki, M.; Nakahara, J.; Dodick, D.W. [(123)I]-IMP single-photon emission computed tomography imaging in visual snow syndrome: A case series. Cephalalgia 2020, 40, 1671–1675. [Google Scholar] [CrossRef]
- Puledda, F.; O’Daly, O.; Schankin, C.; Ffytche, D.; Williams, S.C.; Goadsby, P.J. Disrupted connectivity within visual, attentional and salience networks in the visual snow syndrome. Hum. Brain Mapp. 2021, 42, 2032–2044. [Google Scholar] [CrossRef]
- Puledda, F.; Schankin, C.J.; O’Daly, O.; Ffytche, D.; Eren, O.; Karsan, N.; Williams, S.C.R.; Zelaya, F.; Goadsby, P.J. Localised increase in regional cerebral perfusion in patients with visual snow syndrome: A pseudo-continuous arterial spin labelling study. J. Neurol. Neurosurg. Psychiatry 2021, 92, 918–926. [Google Scholar] [CrossRef]
- Schankin, C.J.; Maniyar, F.H.; Chou, D.E.; Eller, M.; Sprenger, T.; Goadsby, P.J. Structural and functional footprint of visual snow syndrome. Brain 2020, 143, 1106–1113. [Google Scholar] [CrossRef] [PubMed]
- Puledda, F.; Bruchhage, M.; O’Daly, O.; Ffytche, D.; Williams, S.C.R.; Goadsby, P.J. Occipital cortex and cerebellum gray matter changes in visual snow syndrome. Neurology 2020, 95, e1792–e1799. [Google Scholar] [CrossRef] [PubMed]
- Puledda, F.; Ffytche, D.; Lythgoe, D.J.; O’Daly, O.; Schankin, C.; Williams, S.C.R.; Goadsby, P.J. Insular and occipital changes in visual snow syndrome: A BOLD fMRI and MRS study. Ann. Clin. Transl. Neurol. 2020, 7, 296–306. [Google Scholar] [CrossRef] [PubMed]
- Eren, O.; Rauschel, V.; Ruscheweyh, R.; Straube, A.; Schankin, C.J. Evidence of dysfunction in the visual association cortex in visual snow syndrome. Ann. Neurol. 2018, 84, 946–949. [Google Scholar] [CrossRef] [PubMed]
- McKendrick, A.M.; Chan, Y.M.; Tien, M.; Millist, L.; Clough, M.; Mack, H.; Fielding, J.; White, O.B. Behavioral measures of cortical hyperexcitability assessed in people who experience visual snow. Neurology 2017, 88, 1243–1249. [Google Scholar] [CrossRef]
- Solly, E.J.; Clough, M.; McKendrick, A.M.; Foletta, P.; White, O.B.; Fielding, J. Ocular motor measures of visual processing changes in visual snow syndrome. Neurology 2020, 95, e1784–e1791. [Google Scholar] [CrossRef]
- Foletta, P.J.; Clough, M.; McKendrick, A.M.; Solly, E.J.; White, O.B.; Fielding, J. Delayed Onset of Inhibition of Return in Visual Snow Syndrome. Front. Neurol. 2021, 12, 738599. [Google Scholar] [CrossRef]
- Hang, C.; Leishangthem, L.; Yan, Y. Not All Cases of Visual Snows are Benign: Mimics of Visual Snow Syndrome. Neuropsychiatr. Dis. Treat. 2021, 17, 3293–3300. [Google Scholar] [CrossRef]
- Vaphiades, M.S.; Grondines, B.; Cooper, K.; Gratton, S.; Doyle, J. Diagnostic Evaluation of Visual Snow. Front. Neurol. 2021, 12, 743608. [Google Scholar] [CrossRef]
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, 5th ed.; American Psychiatric Association: Arlington, VA, USA, 2013. [Google Scholar]
- Martinotti, G.; Santacroce, R.; Pettorruso, M.; Montemitro, C.; Spano, M.C.; Lorusso, M.; di Giannantonio, M.; Lerner, A.G. Hallucinogen Persisting Perception Disorder: Etiology, Clinical Features, and Therapeutic Perspectives. Brain Sci. 2018, 8, 47. [Google Scholar] [CrossRef]
- van Dongen, R.M.; Alderliefste, G.J.; Onderwater, G.L.J.; Ferrari, M.D.; Terwindt, G.M. Migraine prevalence in visual snow with prior illicit drug use (hallucinogen persisting perception disorder) versus without. Eur. J. Neurol. 2021, 28, 2631–2638. [Google Scholar] [CrossRef]
- Antonaci, F.; Dumitrache, C.; De Cillis, I.; Allena, M. A review of current European treatment guidelines for migraine. J. Headache Pain 2010, 11, 13–19. [Google Scholar] [CrossRef]
- Ailani, J.; Burch, R.C.; Robbins, M.S.; Board of Directors of the American Headache, S. The American Headache Society Consensus Statement: Update on integrating new migraine treatments into clinical practice. Headache 2021, 61, 1021–1039. [Google Scholar] [CrossRef]
- Hansen, J.M.; Charles, A. Differences in treatment response between migraine with aura and migraine without aura: Lessons from clinical practice and RCTs. J. Headache Pain 2019, 20, 96. [Google Scholar] [CrossRef]
- Burch, R. Preventive Migraine Treatment. Continuum 2021, 27, 613–632. [Google Scholar] [CrossRef]
- Buch, D.; Chabriat, H. Lamotrigine in the Prevention of Migraine With Aura: A Narrative Review. Headache 2019, 59, 1187–1197. [Google Scholar] [CrossRef]
- Unal-Cevik, I.; Yildiz, F.G. Visual Snow in Migraine With Aura: Further Characterization by Brain Imaging, Electrophysiology, and Treatment--Case Report. Headache 2015, 55, 1436–1441. [Google Scholar] [CrossRef]
- Eren, O.; Schankin, C.J. Insights into pathophysiology and treatment of visual snow syndrome: A systematic review. Prog. Brain Res. 2020, 255, 311–326. [Google Scholar] [CrossRef]
- Grey, V.; Klobusiakova, P.; Minks, E. Can repetitive transcranial magnetic stimulation of the visual cortex ameliorate the state of patients with visual snow? Bratisl. Lek. Listy 2020, 121, 395–399. [Google Scholar] [CrossRef]
- Grande, M.; Lattanzio, L.; Buard, I.; McKendrick, A.M.; Chan, Y.M.; Pelak, V.S. A Study Protocol for an Open-Label Feasibility Treatment Trial of Visual Snow Syndrome With Transcranial Magnetic Stimulation. Front. Neurol. 2021, 12, 724081. [Google Scholar] [CrossRef]
- Evers, S.; Holle-Lee, D.; Schankin, C.J.; Kull, P.; Raffaelli, B. Visual snow syndrome is probably not mediated by CGRP: A case series. Cephalalgia 2022, 42, 1294–1298. [Google Scholar] [CrossRef] [PubMed]


| Ophtalmologic |
|---|
|
| Neurologic |
|
| Recreational drugs |
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Barral, E.; Martins Silva, E.; García-Azorín, D.; Viana, M.; Puledda, F. Differential Diagnosis of Visual Phenomena Associated with Migraine: Spotlight on Aura and Visual Snow Syndrome. Diagnostics 2023, 13, 252. https://doi.org/10.3390/diagnostics13020252
Barral E, Martins Silva E, García-Azorín D, Viana M, Puledda F. Differential Diagnosis of Visual Phenomena Associated with Migraine: Spotlight on Aura and Visual Snow Syndrome. Diagnostics. 2023; 13(2):252. https://doi.org/10.3390/diagnostics13020252
Chicago/Turabian StyleBarral, Eliseo, Elisa Martins Silva, David García-Azorín, Michele Viana, and Francesca Puledda. 2023. "Differential Diagnosis of Visual Phenomena Associated with Migraine: Spotlight on Aura and Visual Snow Syndrome" Diagnostics 13, no. 2: 252. https://doi.org/10.3390/diagnostics13020252
APA StyleBarral, E., Martins Silva, E., García-Azorín, D., Viana, M., & Puledda, F. (2023). Differential Diagnosis of Visual Phenomena Associated with Migraine: Spotlight on Aura and Visual Snow Syndrome. Diagnostics, 13(2), 252. https://doi.org/10.3390/diagnostics13020252






