Author Contributions
Conceptualization, H.W.; methodology, Y.I. and M.E.; validation, T.I. and K.S.; formal analysis, H.W.; investigation, M.S.; resources, A.Y.; data curation, M.T.; writing—original draft preparation, A.Y.; writing—review and editing, M.K. and H.W.; visualization, K.S.-I.; supervision, H.S.; project administration, K.S.; funding acquisition, H.W. All authors have read and agreed to the published version of the manuscript.
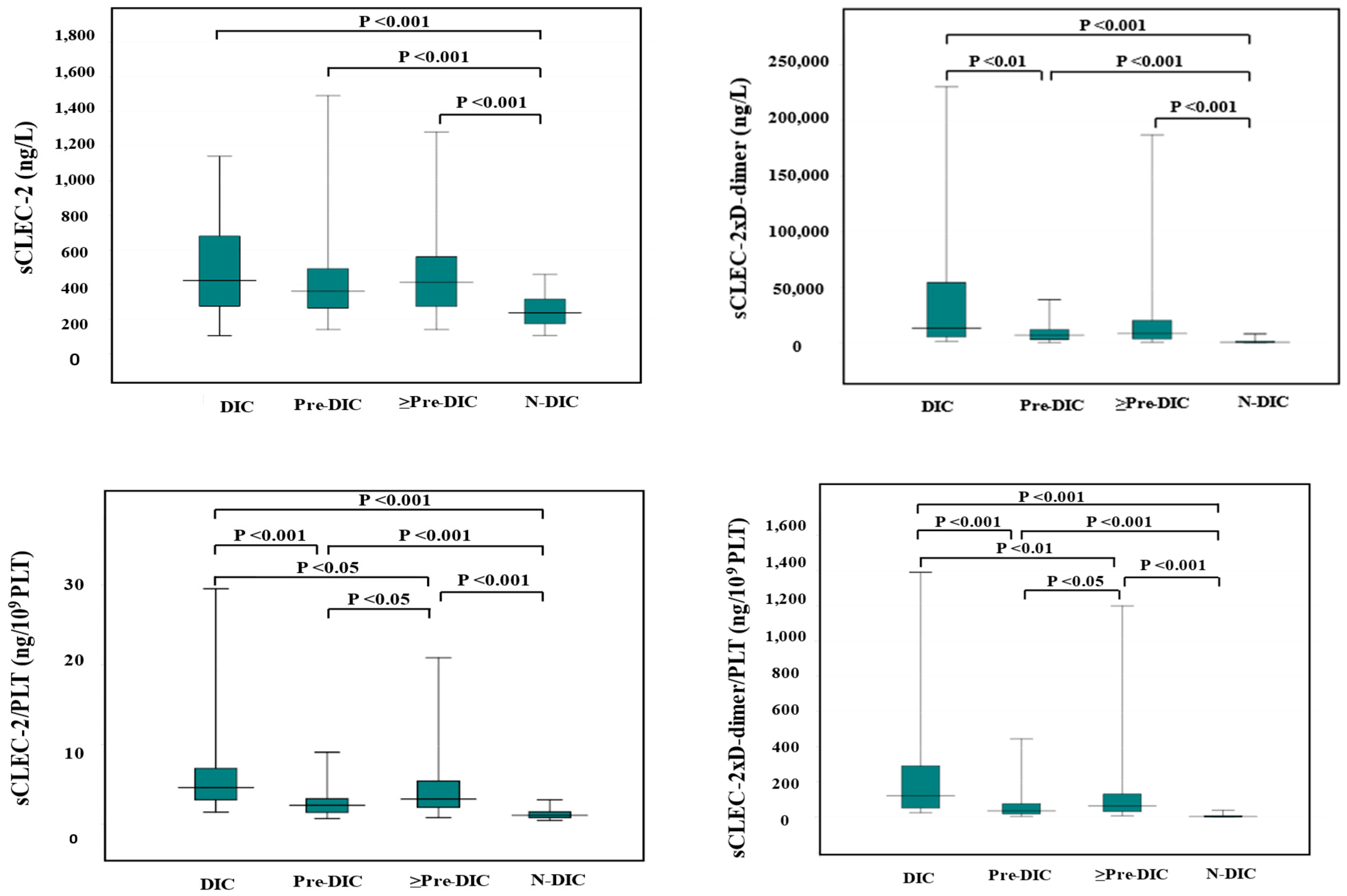
Figure 1.
sCLEC-2, sCLEC-2xD-dimer, sCLEC-2/PLT, and sCLEC-2xD-dimer/PLT in DIC, pre-DIC, ≥Pre-DIC and N-DIC. DIC, disseminated intravascular coagulation; PLT, platelet count; sCLEC-2, soluble C-type lectin-like receptor 2; ≥Pre-DIC, DIC + pre-DIC; N-DIC, non-DIC.
Figure 1.
sCLEC-2, sCLEC-2xD-dimer, sCLEC-2/PLT, and sCLEC-2xD-dimer/PLT in DIC, pre-DIC, ≥Pre-DIC and N-DIC. DIC, disseminated intravascular coagulation; PLT, platelet count; sCLEC-2, soluble C-type lectin-like receptor 2; ≥Pre-DIC, DIC + pre-DIC; N-DIC, non-DIC.
Figure 2.
The ROC analysis of sCLEC-2, sCLEC-2xD-dimer, sCLEC-2/PLT, and sCLEC-2xD-dimer/PLT for DIC vs. N-DIC. ROC, receiver operating characteristic; DIC, disseminated intravascular coagulation; PLT, platelet count; sCLEC-2, soluble C-type lectin-like receptor 2; N-DIC, non-DIC. sCLEC-2 (□); sCLEC-2xD-dimer (▽); sCLEC-2/PLT (△); sCLEC-2xD-dimer/PLT (●).
Figure 2.
The ROC analysis of sCLEC-2, sCLEC-2xD-dimer, sCLEC-2/PLT, and sCLEC-2xD-dimer/PLT for DIC vs. N-DIC. ROC, receiver operating characteristic; DIC, disseminated intravascular coagulation; PLT, platelet count; sCLEC-2, soluble C-type lectin-like receptor 2; N-DIC, non-DIC. sCLEC-2 (□); sCLEC-2xD-dimer (▽); sCLEC-2/PLT (△); sCLEC-2xD-dimer/PLT (●).
Figure 3.
The ROC analysis of sCLEC-2, sCLEC-2xD-dimer, sCLEC-2/PLT, and sCLEC-2xD-dimer/PLT for ≥Pre-DIC vs. N-DIC. ROC, receiver operating characteristic; DIC, disseminated intravascular coagulation; PLT, platelet count; sCLEC-2, soluble C-type lectin-like receptor 2; ≥Pre-DIC, DIC + pre-DIC; N-DIC, non-DIC; sCLEC-2 (□); sCLEC-2xD-dimer (▽); sCLEC-2/PLT (△); sCLEC-2xD-dimer/PLT (●).
Figure 3.
The ROC analysis of sCLEC-2, sCLEC-2xD-dimer, sCLEC-2/PLT, and sCLEC-2xD-dimer/PLT for ≥Pre-DIC vs. N-DIC. ROC, receiver operating characteristic; DIC, disseminated intravascular coagulation; PLT, platelet count; sCLEC-2, soluble C-type lectin-like receptor 2; ≥Pre-DIC, DIC + pre-DIC; N-DIC, non-DIC; sCLEC-2 (□); sCLEC-2xD-dimer (▽); sCLEC-2/PLT (△); sCLEC-2xD-dimer/PLT (●).
Figure 4.
sCLEC-2, sCLEC-2xD-dimer, sCLEC-2/PLT, and sCLEC-2xD-dimer/PLT in non-survivors and survivors. PLT, platelet count; sCLEC-2, soluble C-type lectin-like receptor 2.
Figure 4.
sCLEC-2, sCLEC-2xD-dimer, sCLEC-2/PLT, and sCLEC-2xD-dimer/PLT in non-survivors and survivors. PLT, platelet count; sCLEC-2, soluble C-type lectin-like receptor 2.
Figure 5.
ROC analysis of sCLEC-2, sCLEC-2xD-dimer, sCLEC-2/PLT, and sCLEC-2xD-dimer/PLT for non-survivors vs. survivors. ROC, receiver operating characteristic; DIC, disseminated intravascular coagulation; PLT, platelet count; sCLEC-2 (□), soluble C-type lectin-like receptor 2. sCLEC-2xD-dimer (▽); sCLEC-2/PLT (△); sCLEC-2xD-dimer/PLT (●).
Figure 5.
ROC analysis of sCLEC-2, sCLEC-2xD-dimer, sCLEC-2/PLT, and sCLEC-2xD-dimer/PLT for non-survivors vs. survivors. ROC, receiver operating characteristic; DIC, disseminated intravascular coagulation; PLT, platelet count; sCLEC-2 (□), soluble C-type lectin-like receptor 2. sCLEC-2xD-dimer (▽); sCLEC-2/PLT (△); sCLEC-2xD-dimer/PLT (●).
Table 1.
The subject study protocol (2019-K9) was approved by the Human Ethics Review Committee of Mie Prefectural General Medical Center, and informed consent was obtained from each participant. This study was carried out in accordance with the principles of the Declaration of Helsinki.
Table 1.
The subject study protocol (2019-K9) was approved by the Human Ethics Review Committee of Mie Prefectural General Medical Center, and informed consent was obtained from each participant. This study was carried out in accordance with the principles of the Declaration of Helsinki.
| | Solid Cancer | HM | AA | Trauma | Infection | CPA | UCS |
|---|
| n | 27 | 11 | 37 | 41 | 215 | 25 | 74 |
| Age | 73.0 | 73.0 | 74.0 | 69.0 | 77.0 | 81.0 | 57.5 |
| (years old) | (69.0–79.8) | (58.0–83.0) | (67.0–78.0) | (42.0–80.3) | (61.0–83.0) | (68.8–87.3) | (48.0–73.0) |
| Sex (F:M) | 12:15 | 4:7 | 17:20 | 20:21 | 99:116 | 8:17 | 39:35 |
| Death (Mortality) | 8 (29.6%) | 0 (0%) | 3 (8.1%) | 4 (9.8%) | 25 (11.6%) | 22 (88%) | 0 (0%) |
| APTT (sec) | 30.5 | 30.0 | 29.5 | 28.0 | 33.0 *** | 57.0 *** | 29.0 |
| (27.0–35.0) | (27.0–35.8) | (28.0–34.5) | (26.0–34.0) | (29.0–39.0) | (44.3–76.0) | (27.0–32.0) |
| PT-INR | 1.07 *** | 0.97 | 1.02 *** | 0.96 | 1.13 *** | 1.62 *** | 0.96 |
| (1.02–1.21) | (0.94–1.26) | (0.97–1.18) | (0.92–1.05) | (1.03–1.24) | (1.21–1.97) | (0.92–1.00) |
| PLT (×109/L) | 226 | 191 | 160 *** | 227 | 191 *** | 114 *** | 23.4 |
| (137–309) | (132–258) | (119–200) | (166–275) | (125–253) | (85–178) | (188–275) |
| DIC score | 2.0 *** | 1.0 *** | 3.0 *** | 1.0 *** | 2.0 *** | 7.0 *** | 0 |
| (1.0–4.8) | (1.0–3.8) | (1.0–4.0) | (1.0–4.0) | (1.0–4.0) | (5.0–8.0) | (0–0) |
| FDP | 4.4 *** | 4.6 ** | 3.7 *** | 8.5 *** | 5.6 *** | 67.6 *** | 0.7 |
| (μg/mL) | (2.8–28.3) | (1.2–17.9) | (0.6–7.5) | (3.3–37.8) | (2.4–14.5) | (22.2–452.8) | (0.3–1.0) |
| D-dimer (μg/mL) | 3.8 *** | 4.1 ** | 6.8 *** | 7.6 *** | 4.4 *** | 15.7 *** | 0.6 |
| (1.9–16.6) | (0.7–11.9) | (2.6–11.6) | (2.4–18.0) | (1.6–9.7) | (7.6–46.4) | (0.4–1.5) |
| SF | 6.7 *** | 5.8 | 8.1 *** | 6.8 *** | 9.6 *** | 12.7 *** | 2.1 |
| (μg/mL) | (2.2–13.4) | (3.0–21.1) | (1.6–12.7) | (1.1–9.1) | (2.0–15.0) | (2.1–25.5) | (0.5–4.7) |
| sCLEC2 | 260 ** | 278 * | 247 *** | 245 *** | 258 ** | 441 *** | 193 |
| (ng/L) | (172–321) | (167–364) | (174–323) | (178–328) | (195–335) | (310–748) | (143–242) |
Table 2.
Hemostatic markers in patients with DIC, pre-DIC, or non-DIC.
Table 2.
Hemostatic markers in patients with DIC, pre-DIC, or non-DIC.
| | DIC (n = 38) | Pre-DIC (n = 39) | DIC + Pre-DIC (n= 77) | Non-DIC (n = 222) |
|---|
| FDP (μg/mL) | 70.5 (35.5–435.9) *** | 27.8 (16.7–49.9) *** | 43.2 (22.9–85.1) *** | 3.9 (1.8–7.5) |
| D-dimer (μg/mL) | 27.9 (16.6–74.9) *** | 18.5 (7.9–29.3) *** | 22.1 (11.6–41.1) *** | 3.0 (1.2–6.0) |
| PT-INR | 1.69 (1.38–1.98) *** | 1.21 (1.02–1.40) *** | 1.39 (1.15–1.74) *** | 1.05 (0.97–1.13) |
| PLT (×109/L) | 93 (45–124) *** | 156 (111–241) *** | 119 (85–200) *** | 214 (156–270) |
| SF (μg/mL) | 49.1 (28.3–98.6) *** | 26.5 (15.8–57.2) *** | 35.3 (17.5–69.1) *** | 11.1 (6.9–20.2) |
| APTT (sec) | 57.5 (47.5–78.3) *** | 35.0 (29.5–45.8) *** | 46.0 (33.0–65.0) *** | 30.0 (27.0–34.0) |
| sCLEC2 (ng/L) | 423 (275–679) *** | 361 (264–492) *** | 413 (274–560) *** | 237 (174–317) *** |
Table 3.
The ROC analysis of hemostatic biomarkers for the diagnosis of DIC, pre-DIC, or DIC + pre-DIC vs. non-DIC.
Table 3.
The ROC analysis of hemostatic biomarkers for the diagnosis of DIC, pre-DIC, or DIC + pre-DIC vs. non-DIC.
| | | DIC | Pre-DIC | DIC + Pre-DIC |
|---|
| FDP | Cutoff value (μg/mL) | 21.8 | 12.5 | 16.0 |
| Sensitivity (specificity) | 91.9% | 84.6% | 86.5% |
| AUC | 0.972 | 0.894 | 0.933 |
| Odds ratio | 132.2 | 30.4 | 42.9 |
| D-dimer | Cutoff value (μg/mL) | 10.4 | 7.0 | 8.3 |
| Sensitivity (specificity) | 90.1% | 79.5% | 84.4% |
| AUC | 0.960 | 0.883 | 0.921 |
| Odds ratio | 77.3 | 15.2 | 30.0 |
| PT-INR | Cutoff value | 1.20 | 1.08 | 1.13 |
| Sensitivity (specificity) | 88.2% | 64.7% | 76.0% |
| AUC | 0.943 | 0.701 | 0.821 |
| Odds ratio | 48.0 | 3.3 | 9.8 |
| PLT | Cutoff value (×109/L) | 137 | 190 | 170 |
| Sensitivity (specificity) | 81.6% | 59.0% | 68.8% |
| AUC | 0.890 | 0.652 | 0.758 |
| Odds ratio | 20.2 | 2.1 | 4.9 |
| SF | Cutoff value (μg/mL) | 24.1 | 16.2 | 19.1 |
| Sensitivity (specificity) | 81.6% | 66.3% | 74.0% |
| AUC | 0.905 | 0.749 | 0.819 |
| Odds ratio | 27.9 | 3.9 | 8.3 |
| APTT | Cutoff value | 35.1 | 31.4 | 33.0 |
| Sensitivity (specificity) | 83.4% | 64.5% | 73.8% |
| AUC | 0.911 | 0.670 | 0.787 |
| Odds ratio | 25.4 | 3.1 | 7.9 |
| sCLEC-2 | Cutoff value (ng/L) | 287 | 284 | 285 |
| Sensitivity (specificity) | 68.4% | 67.6% | 67.6% |
| AUC | 0.801 | 0.748 | 0.774 |
| Odds ratio | 4.7 | 4.2 | 4.4 |
Table 4.
sCLEC-2, sCLEC-2/PLT, sCLEC-2xD-dimer, and sCLEC-2xD-dimer/PLT.
Table 4.
sCLEC-2, sCLEC-2/PLT, sCLEC-2xD-dimer, and sCLEC-2xD-dimer/PLT.
| | DIC | Pre-DIC | DIC + Pre-DIC | Non-DIC |
|---|
| sCLEC-2(ng/L) | 423 (275–679) *** | 361 (264–492) *** | 413 (274–560) *** | 237 (174–317) |
| sCLEC-2/PLT | 4.78 (3.05–7.05) *** | 2.37 (1.47–3.21) *** | 3.16 (2.12–5.60) *** | 1.12 (0.82–1.55) |
| sCLEC-2xD-dimer | 13,198 (5255–54,369) *** | 6923 (2906–11,842) *** | 8533 (3467–20,348) *** | 657 (254–1542) |
| sCLEC-2xD-dimer/PLT | 128 (53.0–509) *** | 35.6 (19.5–761) *** | 65.4 (33.2–141) *** | 3.45 (1.21–7.35) |
Table 5.
The ROC analysis of sCLEC-2, sCLEC2/PLT, sCLEC2xD-dimer, and sCLEC-2xD-dimer/PLT for the diagnosis of DIC, pre-DIC, or DIC + pre-DIC vs. non-DIC.
Table 5.
The ROC analysis of sCLEC-2, sCLEC2/PLT, sCLEC2xD-dimer, and sCLEC-2xD-dimer/PLT for the diagnosis of DIC, pre-DIC, or DIC + pre-DIC vs. non-DIC.
| | | DIC | Pre-DIC | DIC + Pre-DIC |
|---|
| sCLEC-2 | Cutoff value (ng/L) | 287 | 284 | 285 |
| Sensitivity (specificity) | 68.4% | 67.6% | 67.6% |
| AUC | 0.801 | 0.748 | 0.774 |
| Odds ratio | 4.7 | 4.2 | 4.4 |
| sCLEC-2/PLT | Cutoff value | 2.07 | 1.54 | 1.74 |
| Sensitivity (specificity) | 89.6% | 73.9% | 81.8% |
| AUC | 0.970 | 0.822 | 0.895 |
| Odds ratio | 100.9 | 6.4 | 20.5 |
| sCLEC-2xD-dimer | Cutoff value | 2993 | 2252 | 2429 |
| Sensitivity (specificity) | 91.9% | 86.5% | 89.2% |
| AUC | 0.966 | 0.911 | 0.938 |
| Odds ratio | 132.2 | 43.5 | 71.2 |
| sCLEC-2xD-dimer/PLT | Cutoff value | 25.6 | 13.1 | 17.0 |
| Sensitivity (specificity) | 94.7% | 85.6% | 89.6% |
| AUC | 0.993 | 0.929 | 0.961 |
| Odds ratio | 315.0 | 32.7 | 74.6 |
Table 6.
Hemostatic markers in survivors and non-survivors.
Table 6.
Hemostatic markers in survivors and non-survivors.
| | Survivors | Non-Survivors |
|---|
| APTT (sec) | 31.0 (27.0–34.0) *** | 46.0 (33.0–65.0) *** |
| PT-INR | 1.06 (0.97–1.16) *** | 1.40 (1.13–1.87) *** |
| PLT (×109/L) | 200 (138–267) *** | 134 (90–207) *** |
| FDP (μg/mL) | 3.5 (16.0–8.6) *** | 49.9 (16.1–179.4) *** |
| DIC score | 2.0 (1.0–3.0) *** | 6.0 (4.0–8.0) *** |
| D-dimer LG (μg/mL) | 3.8 (1.4–8.3) *** | 16.8 (7.3–33.2) *** |
| SF (μg/mL) | 12.7 (7.5–23.8) *** | 30.2 (14.5–56.2) *** |
| sCLEC2 (ng/L) | 247 (180–328) *** | 365 (261–480) *** |
| sCLEC-2/PLT | 1.25 (0.85–1.77) *** | 2.53 (1.55–5.04) *** |
| sCLEC-2xD-dimer | 771 (280–2254) *** | 4962 (1934–13,624) *** |
| sCLEC-2xD-dimer/PLT | 4.29 (1.24–13.2) *** | 46.3 (15.6–128) *** |
Table 7.
The ROC analysis of hemostatic biomarkers for non-survivors vs. survivors.
Table 7.
The ROC analysis of hemostatic biomarkers for non-survivors vs. survivors.
| FDP | Cutoff value (μg/mL) | 14.7 |
| Sensitivity (specificity) | 76.8% |
| AUC | 0.840 |
| Odds ratio | 11.0 |
| D-dimer | Cutoff value (μg/mL) | 7.9 |
| Sensitivity (specificity) | 74.5% |
| AUC | 0.786 |
| Odds ratio | 8.0 |
| PT-INR | Cutoff value | 1.14 |
| Sensitivity (specificity) | 70.6% |
| AUC | 0.817 |
| Odds ratio | 5.5 |
| PLT | Cutoff value (×109/L) | 171 |
| Sensitivity (specificity) | 63.2% |
| AUC | 0.701 |
| Odds ratio | 2.9 |
| DIC score | Cutoff value (×1010/L) | 3.3 |
| Sensitivity (specificity) | 79.3% |
| AUC | 0.847 |
| Odds ratio | 15.0 |
| SF | Cutoff value (μg/mL) | 18.3 |
| Sensitivity (specificity) | 66.1% |
| AUC | 0.709 |
| Odds ratio | 3.8 |
| APTT | Cutoff value (μg/mL) | 33.5 |
| Sensitivity (specificity) | 72.7% |
| AUC | 0.768 |
| Odds ratio | 6.2 |
| sCLEC2 | Cutoff value (ng/L) | 286 |
| Sensitivity (specificity) | 64.3% |
| AUC | 0.695 |
| Odds ratio | 3.2 |
| sCLEC-2/PLT | Cutoff value | 1.67 |
| Sensitivity (specificity) | 71.4% |
| AUC | 0.781 |
| Odds ratio | 6.3 |
| sCLEC-2xD-dimer | Cutoff value | 2041 |
| Sensitivity (specificity) | 72.8% |
| AUC | 0.798 |
| Odds ratio | 7.3 |
| sCLEC-2xD-dimer/PLT | Cutoff value | 14.4 |
| Sensitivity (specificity) | 76.8% |
| AUC | 0.816 |
| Odds ratio | 11.0 |