Clinical Model for the Prediction of Severe Liver Fibrosis in Adult Patients with Type II Diabetes Mellitus
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Population
2.2. Anthropometry and Risk Factor Measurements
2.3. Laboratory Tests and Metabolic Syndrome
2.4. Liver Fibrosis Assessment Using Point Shear-Wave Elastography
2.5. Liver Fibrosis Assessment Using Serum Markers
2.6. Liver Fibrosis Definition
2.7. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Targher, G. Non-alcoholic fatty liver disease, the metabolic syndrome and the risk of cardiovascular disease: The plot thickens. Diabet. Med. 2007, 24, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Adams, L.A.; Waters, O.R.; Knuiman, M.W.; Elliott, R.R.; Olynyk, J.K. NAFLD as a Risk Factor for the Development of Diabetes and the Metabolic Syndrome: An Eleven-Year Follow-up Study. Am. J. Gastroenterol. 2009, 104, 861–867. [Google Scholar] [CrossRef] [PubMed]
- Eslam, M.; Sanyal, A.J.; George, J.; International Consensus Panel. MAFLD: A Consensus-Driven Proposed Nomenclature for Metabolic Associated Fatty Liver Disease. Gastroenterology 2020, 158, 1999–2014.e1991. [Google Scholar] [CrossRef] [PubMed]
- Dascalu, A.M.; Stoian, A.P.; Cherecheanu, A.P.; Serban, D.; Costea, D.O.; Tudosie, M.S.; Stana, D.; Tanasescu, D.; Sabau, A.D.; Gangura, G.A.; et al. Outcomes of Diabetic Retinopathy Post-Bariatric Surgery in Patients with Type 2 Diabetes Mellitus. J. Clin. Med. 2021, 10, 3736. [Google Scholar] [CrossRef]
- Jakubiak, G.K.; Osadnik, K.; Lejawa, M.; Osadnik, T.; Goławski, M.; Lewandowski, P.; Pawlas, N. “Obesity and Insulin Resistance” Is the Component of the Metabolic Syndrome Most Strongly Associated with Oxidative Stress. Antioxidants 2021, 11, 79. [Google Scholar] [CrossRef]
- Regufe, V.M.G.; Pinto, C.; Perez, P. Metabolic syndrome in type 2 diabetic patients: A review of current evidence. Porto Biomed. J. 2020, 5, e101. [Google Scholar] [CrossRef]
- Tolman, K.G.; Fonseca, V.; Dalpiaz, A.; Tan, M.H. Spectrum of Liver Disease in Type 2 Diabetes and Management of Patients With Diabetes and Liver Disease. Diabetes Care 2007, 30, 734–743. [Google Scholar] [CrossRef] [Green Version]
- Pocha, C.; Xie, C. Hepatocellular carcinoma in alcoholic and non-alcoholic fatty liver disease-one of a kind or two different enemies? Transl. Gastroenterol. Hepatol. 2019, 4, 72. [Google Scholar] [CrossRef]
- Ratziu, V.; Charlotte, F.; Heurtier, A.; Gombert, S.; Giral, P.; Bruckert, E.; Grimaldi, A.; Capron, F.; Poynard, T. Sampling Variability of Liver Biopsy in Nonalcoholic Fatty Liver Disease. Gastroenterology 2005, 128, 1898–1906. [Google Scholar] [CrossRef]
- Ratziu, V.; Bugianesi, E.; Dixon, J.; Fassio, E.; Ekstedt, M.; Charlotte, F.; Kechagias, S.; Poynard, T.; Olsson, R. Histological progression of non-alcoholic fatty liver disease: A critical reassessment based on liver sampling variability. Aliment. Pharmacol. Ther. 2007, 26, 821–830. [Google Scholar] [CrossRef]
- Xiao, G.; Zhu, S.; Xiao, X.; Yan, L.; Yang, J.; Wu, G. Comparison of laboratory tests, ultrasound, or magnetic resonance elastography to detect fibrosis in patients with nonalcoholic fatty liver disease: A meta-analysis. Hepatology 2017, 66, 1486–1501. [Google Scholar] [CrossRef] [Green Version]
- Dhyani, M.; Anvari, A.; Samir, A.E. Ultrasound elastography: Liver. Abdom Imaging 2015, 40, 698–708. [Google Scholar] [CrossRef]
- Lin, Y.; Li, H.; Jin, C.; Wang, H.; Jiang, B. The diagnostic accuracy of liver fibrosis in non-viral liver diseases using acoustic radiation force impulse elastography: A systematic review and meta-analysis. PLoS ONE 2020, 15, e0227358. [Google Scholar] [CrossRef] [Green Version]
- Barr, R.G.; Wilson, S.R.; Rubens, D.; Garcia-Tsao, G.; Ferraioli, G. Update to the Society of Radiologists in Ultrasound Liver Elastography Consensus Statement. Radiology 2020, 296, 263–274. [Google Scholar] [CrossRef]
- Palmeri, M.L.; Wang, M.H.; Rouze, N.C.; Abdelmalek, M.F.; Guy, C.D.; Moser, B.; Diehl, A.M.; Nightingale, K.R. Noninvasive evaluation of hepatic fibrosis using acoustic radiation force-based shear stiffness in patients with nonalcoholic fatty liver disease. J. Hepatol. 2011, 55, 666–672. [Google Scholar] [CrossRef] [Green Version]
- Guha, I.N.; Parkes, J.; Roderick, P.; Chattopadhyay, D.; Cross, R.; Harris, S.; Kaye, P.; Burt, A.D.; Ryder, S.D.; Aithal, G.P.; et al. Noninvasive markers of fibrosis in nonalcoholic fatty liver disease: Validating the European Liver Fibrosis Panel and exploring simple markers. Hepatology 2008, 47, 455–460. [Google Scholar] [CrossRef]
- Unsal, I.O.; Calapkulu, M.; Sencar, M.E.; Cakal, B.; Ozbek, M. Evaluation of NAFLD fibrosis, FIB-4 and APRI score in diabetic patients receiving exenatide treatment for non-alcoholic fatty liver disease. Sci. Rep. 2022, 12, 283. [Google Scholar] [CrossRef]
- Adams, L.A.; Chan, W.-K. Noninvasive Tests in the Assessment of NASH and NAFLD Fibrosis: Now and Into the Future. Semin. Liver Dis. 2020, 40, 331–338. [Google Scholar] [CrossRef]
- Ellis, E.L.; Mann, D.A. Clinical evidence for the regression of liver fibrosis. J. Hepatol. 2012, 56, 1171–1180. [Google Scholar] [CrossRef] [Green Version]
- Zoubek, M.E.; Trautwein, C.; Strnad, P. Reversal of liver fibrosis: From fiction to reality. Best Pract. Res. Clin. Gastroenterol. 2017, 31, 129–141. [Google Scholar] [CrossRef]
- American Diabetes Association Professional Practice Committee. 2. Classification and Diagnosis of Diabetes: Standards of Medical Care in Diabetes-2022. Diabetes Care 2022, 45 (Suppl. S1), S17–S38. [Google Scholar] [CrossRef]
- Bush, K.; Kivlahan, D.R.; McDonell, M.B.; Fihn, S.D.; Bradley, K.A. The AUDIT Alcohol Consumption Questions (AUDIT-C). An Effective Brief Screening Test for Problem Drinking. Ambulatory Care Quality Improvement Project (ACQUIP). Alcohol Use Disorders Identification Test. Arch. Intern. Med. 1998, 158, 1789–1795. [Google Scholar] [CrossRef] [Green Version]
- Unger, T.; Borghi, C.; Charchar, F.; Khan, N.A.; Poulter, N.R.; Prabhakaran, D.; Ramirez, A.; Schlaich, M.; Stergiou, G.S.; Tomaszewski, M.; et al. 2020 International Society of Hypertension global hypertension practice guidelines. J. Hypertens. 2020, 38, 982–1004. [Google Scholar] [CrossRef]
- Matthews, D.R.; Hosker, J.P.; Rudenski, A.S.; Naylor, B.A.; Treacher, D.F.; Turner, R.C. Homeostasis model assessment: Insulin resistance and beta-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia 1985, 28, 412–419. [Google Scholar] [CrossRef] [Green Version]
- Huang, P.L. A comprehensive definition for metabolic syndrome. Dis. Model. Mech. 2009, 2, 231–237. [Google Scholar] [CrossRef] [Green Version]
- Ferraioli, G.; Monteiro, L.B.S. Ultrasound-based techniques for the diagnosis of liver steatosis. World J. Gastroenterol. 2019, 25, 6053–6062. [Google Scholar] [CrossRef]
- Cheah, M.C.; McCullough, A.J.; Goh, G.B.-B. Current Modalities of Fibrosis Assessment in Non-alcoholic Fatty Liver Disease. J. Clin. Transl. Hepatol. 2017, 5, 261–271. [Google Scholar] [CrossRef] [Green Version]
- Petta, S.; Eslam, M.; Valenti, L.; Bugianesi, E.; Barbara, M.; Cammà, C.; Porzio, M.; Rosso, C.; Fargion, S.; George, J.; et al. Metabolic syndrome and severity of fibrosis in nonalcoholic fatty liver disease: An age-dependent risk profiling study. Liver Int. 2017, 37, 1389–1396. [Google Scholar] [CrossRef] [PubMed]
- Seo, J.A. Metabolic Syndrome: A Warning Sign of Liver Fibrosis. J. Obes. Metab. Syndr. 2022, 30, 1. [Google Scholar] [CrossRef] [PubMed]
- Kashyap, S.R.; Diab, D.L.; Baker, A.R.; Yerian, L.; Bajaj, H.; Gray-McGuire, C.; Schauer, P.R.; Gupta, M.; Feldstein, A.E.; Hazen, S.L.; et al. Triglyceride levels and not adipokine concentrations are closely related to severity of nonalcoholic fatty liver disease in an obesity surgery cohort. Obesity 2009, 17, 1696–1701. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bedogni, G.; Bellentani, S.; Miglioli, L.; Masutti, F.; Passalacqua, M.; Castiglione, A.; Tiribelli, C. The Fatty Liver Index: A simple and accurate predictor of hepatic steatosis in the general population. BMC Gastroenterol. 2006, 6, 33. [Google Scholar] [CrossRef] [Green Version]
- Bazick, J.; Donithan, M.; Neuschwander-Tetri, B.A.; Kleiner, D.; Brunt, E.M.; Wilson, L.; Doo, E.; Lavine, J.; Tonascia, J.; Loomba, R. Clinical Model for NASH and Advanced Fibrosis in Adult Patients With Diabetes and NAFLD: Guidelines for Referral in NAFLD. Diabetes Care 2015, 38, 1347–1355. [Google Scholar] [CrossRef] [Green Version]
- Haring, R.; Wallaschofski, H.; Nauck, M.; Dörr, M.; Baumeister, S.E.; Völzke, H. Ultrasonographic hepatic steatosis increases prediction of mortality risk from elevated serum gamma-glutamyl transpeptidase levels. Hepatology 2009, 50, 1403–1411. [Google Scholar] [CrossRef]
- Tilg, H.; Moschen, A.R.; Roden, M. NAFLD and diabetes mellitus. Nat. Rev. Gastroenterol. Hepatol. 2017, 14, 32–42. [Google Scholar] [CrossRef]
- Lee, J.; Vali, Y.; Boursier, J.; Spijker, R.; Anstee, Q.M.; Bossuyt, P.M.; Zafarmand, M.H. Prognostic accuracy of FIB-4, NAFLD fibrosis score and APRI for NAFLD-related events: A systematic review. Liver Int. 2021, 41, 261–270. [Google Scholar] [CrossRef]
- D’Onofrio, M.; Crosara, S.; De Robertis, R.; Canestrini, S.; Demozzi, E.; Gallotti, A.; Mucelli, R.P. Acoustic radiation force impulse of the liver. World J. Gastroenterol. 2013, 19, 4841–4849. [Google Scholar] [CrossRef]
- Friedrich-Rust, M.; Nierhoff, J.; Lupsor-Platon, M.; Sporea, I.; Fierbinteanu-Braticevici, C.; Strobel, D.; Takahashi, H.; Yoneda, M.; Suda, T.; Zeuzem, S.; et al. Performance of Acoustic Radiation Force Impulse imaging for the staging of liver fibrosis: A pooled meta-analysis. J. Viral Hepat. 2012, 19, e212–e219. [Google Scholar] [CrossRef]
- Fierbinteanu-Braticevici, C.; Andronescu, D.; Usvat, R.; Cretoiu, D.; Baicus, C.; Marinoschi, G. Acoustic radiation force imaging sonoelastography for noninvasive staging of liver fibrosis. World J. Gastroenterol. 2009, 15, 5525–5532. [Google Scholar] [CrossRef]
- Bota, S.; Sporea, I.; Sirli, R.; Popescu, A.; Danila, M.; Jurchis, A.; Gradinaru-Tascau, O. Factors associated with the impossibility to obtain reliable liver stiffness measurements by means of Acoustic Radiation Force Impulse (ARFI) elastography—Analysis of a cohort of 1031 subjects. Eur. J. Radiol. 2014, 83, 268–272. [Google Scholar] [CrossRef]
- Yoo, J.; Lee, J.M.; Joo, I.; Yoon, J.H. Assessment of liver fibrosis using 2-dimensional shear wave elastography: A prospective study of intra- and inter-observer repeatability and comparison with point shear wave elastography. Ultrasonography 2020, 39, 52–59. [Google Scholar] [CrossRef] [Green Version]
- Heyens, L.J.M.; Busschots, D.; Koek, G.H.; Robaeys, G.; Francque, S. Liver Fibrosis in Non-alcoholic Fatty Liver Disease: From Liver Biopsy to Non-invasive Biomarkers in Diagnosis and Treatment. Front. Med. 2021, 8, 615978. [Google Scholar] [CrossRef]
- Lomonaco, R.; Leiva, E.G.; Bril, F.; Shrestha, S.; Mansour, L.; Budd, J.; Romero, J.P.; Schmidt, S.; Chang, K.-L.; Samraj, G.; et al. Advanced Liver Fibrosis Is Common in Patients with Type 2 Diabetes Followed in the Outpatient Setting: The Need for Systematic Screening. Diabetes Care 2021, 44, 399–406. [Google Scholar] [CrossRef]
- Puchakayala, B.K.; Verma, S.; Kanwar, P.; Hart, J.; Sanivarapu, R.R.; Mohanty, S.R. Histopathological differences utilizing the nonalcoholic fatty liver disease activity score criteria in diabetic (type 2 diabetes mellitus) and non-diabetic patients with nonalcoholic fatty liver disease. World J. Hepatol. 2015, 7, 2610–2618. [Google Scholar] [CrossRef] [Green Version]
- Younossi, Z.M.; Golabi, P.; de Avila, L.; Paik, J.M.; Srishord, M.; Fukui, N.; Qiu, Y.; Burns, L.; Afendy, A.; Nader, F. The global epidemiology of NAFLD and NASH in patients with type 2 diabetes: A systematic review and meta-analysis. J. Hepatol. 2019, 71, 793–801. [Google Scholar] [CrossRef] [PubMed]
- Younossi, Z.M.; Yilmaz, Y.; Yu, M.L.; Wai-Sun Wong, V.; Fernandez, M.C.; Isakov, V.A.; Duseja, A.K.; Mendez-Sanchez, N.; Eguchi, Y.; Bugianesi, E.; et al. Clinical and Patient-Reported Outcomes from Patients with Nonalcoholic Fatty Liver Disease Across the World: Data From the Global Non-Alcoholic Steatohepatitis (NASH)/ Non-Alcoholic Fatty Liver Disease (NAFLD) Registry. Clin. Gastroenterol. Hepatol. 2021. [Google Scholar] [CrossRef] [PubMed]
- Prashanth, M.; Ganesh, H.K.; Vima, M.V.; John, M.; Bandgar, T.; Joshi, S.R.; Shah, S.R.; Rathi, P.M.; Joshi, A.S.; Thakkar, H.; et al. Prevalence of nonalcoholic fatty liver disease in patients with type 2 diabetes mellitus. J. Assoc. Physicians India 2009, 57, 205–210. [Google Scholar] [PubMed]
- Kim, K.; Oh, T.J.; Cho, H.C.; Lee, Y.K.; Ahn, C.H.; Koo, B.K.; Moon, J.H.; Choi, S.H.; Jang, H.C. Liver fibrosis indices are related to diabetic peripheral neuropathy in individuals with type 2 diabetes. Sci. Rep. 2021, 11, 24372. [Google Scholar] [CrossRef] [PubMed]
- Serban, D.; Papanas, N.; Dascalu, A.M.; Stana, D.; Nicolae, V.A.; Vancea, G.; Badiu, C.D.; Tanasescu, D.; Tudor, C.; Balasescu, S.A.; et al. Diabetic Retinopathy in Patients with Diabetic Foot Ulcer: A Systematic Review. Int. J. Low. Extrem. Wounds 2021, 20, 98–103. [Google Scholar] [CrossRef]
- Huang, J.; Li, R.; Liu, N.; Yi, N.; Zheng, H.; Zhang, Q.; Zhou, L.; Zhou, L.; Hu, R.; Lu, B. Liver fibrosis is independently associated with diabetic peripheral neuropathy in type 2 diabetes mellitus. J. Diabetes Investig. 2021, 12, 2019–2027. [Google Scholar] [CrossRef]
- Singh, S.; Allen, A.M.; Wang, Z.; Prokop, L.J.; Murad, M.H.; Loomba, R. Fibrosis Progression in Nonalcoholic Fatty Liver vs Nonalcoholic Steatohepatitis: A Systematic Review and Meta-analysis of Paired-Biopsy Studies. Clin. Gastroenterol. Hepatol. 2015, 13, 643–654.e9. [Google Scholar] [CrossRef] [Green Version]
- Bica, C.; Sandu, C.; Suceveanu, A.I.; Sarbu, E.; Stoica, R.A.; Gherghiceanu, F.; Bohiltea, R.E.; Stefan, S.D.; Stoian, A.P. Non-alcoholic fatty liver disease: A major challenge in type 2 diabetes mellitus (Review). Exp. Ther. Med. 2020, 20, 2387–2391. [Google Scholar] [CrossRef]

| Parameters | Value |
|---|---|
| Female (%) | 56.25 |
| Male (%) | 43.75 |
| Age (years) | 49.39 ± 8.19 |
| Waist circumference (cm) | 92.28 ± 10.08 |
| BMI (kg/m2) | 29.33 ± 2.51 |
| Hypertension (%) | 73.85 |
| MS (%) | 74 |
| Cholesterol (mg/dL) | 227.42 ± 65.2 |
| HDL-C (mg/dL) | 36.21 ± 6.05 |
| LDL-C (mg/dL) | 106.3 ± 47.2 |
| Serum triglycerides (mg/dL) | 195.65 ± 58.91 |
| AST (U/L) | 83.98 ± 18.11 |
| ALT (U/L) | 86.41 ± 30.41 |
| GGT (U/L) | 107.02 ± 52.17 |
| Albumin (g/dL) | 3.7 ± 0.58 |
| INR | 1.03 ± 0.14 |
| Duration of T2DM (years) | 7.7 (3.4–11.5) |
| Insulin use (%) | 62.25 |
| Serum glucose (g/dL) | 114.45 ± 19.88 |
| HbA1c (%) | 6.55 (5.1–9.2) |
| HOMA-IR | 3.06 ± 0.54 |
| NFS | 0.29 (−1.5–2.4) |
| FIB-4 | 2.74 (0.66–9.41) |
| APRI | 1.02 ± 0.43 |
| pSWE (m/s) | 1.3 ± 0.45 |
| Variables | F1/F2 | F3/F4 | p-Value * |
|---|---|---|---|
| Sex | |||
| Female (%) | 48.43 | 7.82 | 0.021 |
| Male (%) | 32.82 | 10.93 | 0.032 |
| Age (years) | 48.66 ± 8.19 | 55.41 ± 6.85 | <0.001 |
| Waist circumference (cm) | 91.96 ± 9.51 | 94.66 ± 12.64 | 0.053 |
| BMI (kg/m2) | 28.11 ± 2.45 | 30.22 ± 2.77 | 0.02 |
| Hypertension (%) | 78.3 | 69.4 | 0.087 |
| MS (%) | 68.30 | 79.7 | <0.001 |
| Cholesterol (mg/dl) | 228.76 ± 67.75 | 221.6 ± 54.92 | 0.473 |
| HDL-C (mg/dl) | 37 ± 6.03 | 32 ± 5.09 | 0.341 |
| LDL-C (mg/dl) | 105.4 ± 45.6 | 109.2 ± 48.5 | 0.654 |
| Serum triglycerides (mg/dl) | 194.49 ± 60.7 | 202.58 ± 54.92 | 0.032 |
| AST (U/L) | 82.15 ± 18.14 | 91.94 ± 16.3 | 0.021 |
| ALT (U/L) | 85.09 ± 31.9 | 92.14 ± 23.13 | 0.047 |
| GGT (U/L) | 99.36 ± 48.76 | 140.02 ± 57.58 | <0.001 |
| Albumin (g/dl) | 3.75 ± 0.56 | 3.46 ± 0.59 | 0.054 |
| INR | 1.03 ± 0.14 | 0.14 ± 0.12 | 0.847 |
| Duration of T2DM (years) | 7.1 ± 3.4 | 8.4 ± 4.1 | 0.547 |
| Insulin use (%) | 64.2 | 60.3 | 0.352 |
| Serum glucose (g/dl) | 112.21 ± 18.66 | 124.18 ± 22.83 | 0.046 |
| HbA1c (%) | 6.2(5.1–8.2) | 7.1(5.9–9.2) | 0.032 |
| HOMA-IR | 2.99 ± 0.42 | 3.38 ± 0.85 | 0.025 |
| NFS | 0.01 (−1.5–1.8) | 1.48 (0.74–2.4) | <0.001 |
| FIB-4 | 2.3 ± 0.74 | 4.68 ± 1.95 | 0.021 |
| APRI | 0.87 ± 0.2 | 1.69 ± 0.5 | 0.015 |
| pSWE (m/s) | 0.87 ± 0.22 | 2.04 ± 0.26 | <0.001 |
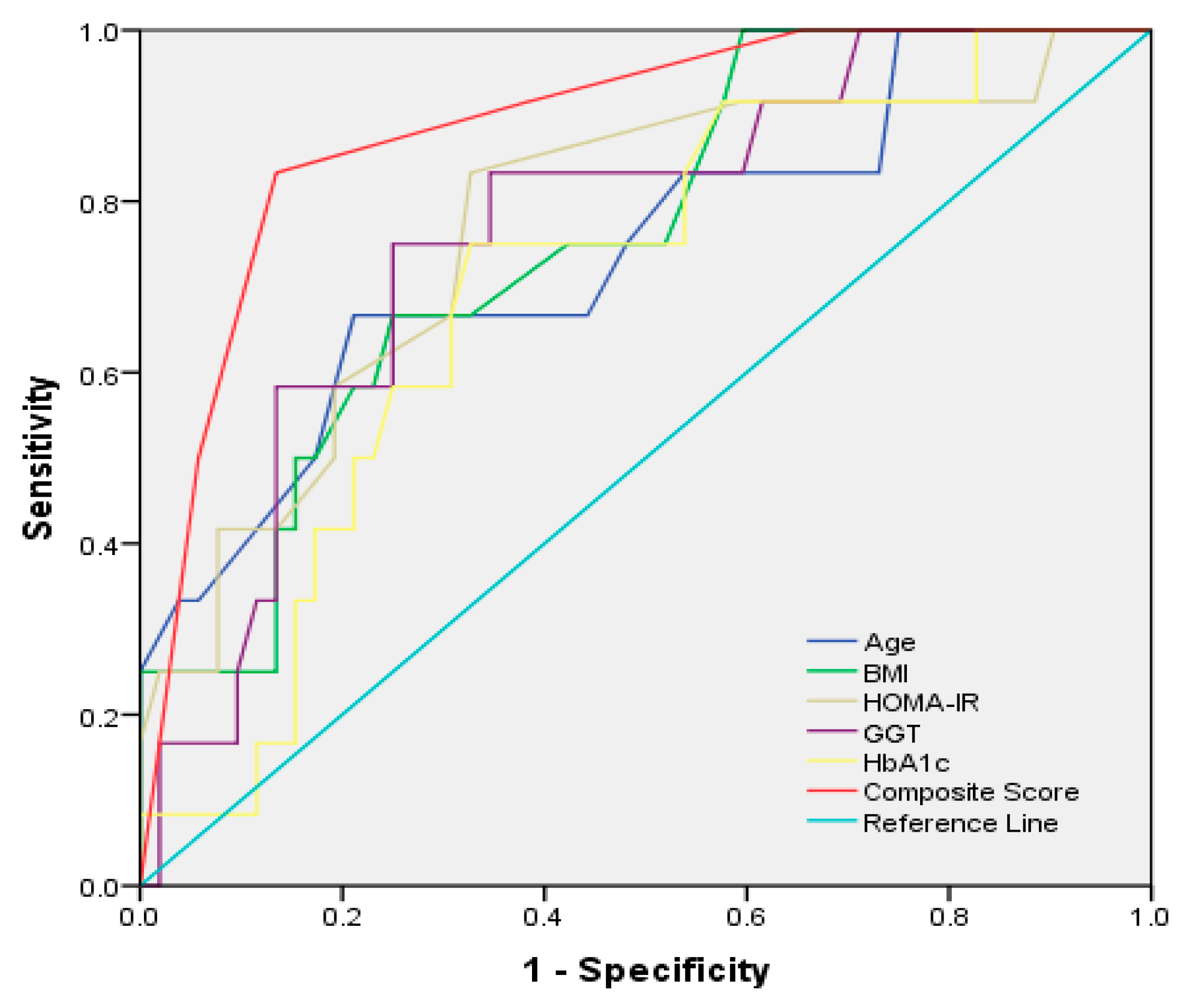
| Variables | Cut-Off Value | AUROC (95% CI) | Std. Error | Sensibility (95% CI) | Specificity (95%CI) | PPV (95% CI) | NPV (95%CI) | p-Value * |
|---|---|---|---|---|---|---|---|---|
| GGT, U/L | 113 | 0.76 (0.627–0.907) | 0.72 | 75.00% | 75.00% | 88.00% | 72.00% | 0.004 |
| (62.7–87.3%) | (61.3–85.5%) | (67.7–97.1%) | (60.6–82.3%) | |||||
| Age, years | 55 | 0.74 (0.578–0.908) | 0.84 | 58.30% | 80.80% | 89.00% | 61.30% | 0.009 |
| (40.2–71.4%) | (62.6–95.8%) | (68.4–98.3%) | (45.6–76.1%) | |||||
| BMI, kg/m2 | 30.1 | 0.75 (0.617–0.897) | 0.71 | 67.60% | 75.00% | 93.00% | 63.40% | 0.006 |
| (49.2–78.8%) | (60.9–85.6%) | (78.4–97.9%) | (46.2–77.7% | |||||
| HOMA-IR, | 3.3 | 0.77 (0.616–0.924) | 0.78 | 66.70% | 88.80% | 95.00% | 78.00% | 0.004 |
| (48.7–79.4%) | (66.3–97.1%) | (81.2–99.3%) | (65.4–90.9%) | |||||
| HbA1c, % | 6.5 | 0.70 (0.548–0.852) | 0.78 | 64.30% | 72.20% | 90.50% | 61.20% | 0.032 |
| (48.3–80.5%) | (59.8–82.6%) | (69.7–99.3%) | (58.9–75.4%) | |||||
| FIB-4 | 1.35 | 0.80 (0.689–0.915) | 0.58 | 82.3% | 65.2% | 74% | 92.1% | <0.001 |
| (62.9–96.6%) | (48.3–78.8%) | (60.7–82.9%) | (78.5–99.9%) | |||||
| APRI | 1 | 0.79 (0.663–0.918) | 0.65 | 72.4% | 87% | 72.3% | 89.6% | 0.002 |
| (58.4–81.9%) | (66.1–97.3%) | (57.9–80.1%) | (67.8–98.1%) | |||||
| NFS | >0.65 | 0.82 (0.67–0.93) | 0.63 | 78.4% | 63.2% | 67.4% | 94.3% | |
| (61.2–89.2%) | (50.1–79.5%) | (52.8–81.3%) | (79.4–99.3%) | <0.001 |
| Univariate Analyses | Multivariate Analyses | ||||
|---|---|---|---|---|---|
| Variables | Cut-Off Values | Or (CI 95%) | p-Value * | OR (CI 95%) | p-Value * |
| GGT, U/L | ≥113 | 1.912 (1.534–2.861) | 0.0479 | 8.993 (2.11–38.311) | 0.003 |
| BMI, kg/m2 | ≥30.1 | 1.544 (1.121–2.128) | 0.0079 | 5.996 (1.548–23.245) | 0.009 |
| Age, years | ≥55 | 1.347 (1.0376–1.589) | 0.0074 | 7.453 (1.889–29.401) | 0.004 |
| HOMA-IR | ≥3.3 | 3.342 (1.874–8.457) | 0.0271 | 5.879 (1.5413–22.431) | 0.009 |
| HbA1c, % | ≥6.5 | 2.6616 (1.7613–3.446) | 0.0051 | 6.851 (1.954–19.547) | 0.002 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Calapod, O.P.; Marin, A.M.; Pantea Stoian, A.; Fierbinteanu-Braticevici, C. Clinical Model for the Prediction of Severe Liver Fibrosis in Adult Patients with Type II Diabetes Mellitus. Diagnostics 2022, 12, 1829. https://doi.org/10.3390/diagnostics12081829
Calapod OP, Marin AM, Pantea Stoian A, Fierbinteanu-Braticevici C. Clinical Model for the Prediction of Severe Liver Fibrosis in Adult Patients with Type II Diabetes Mellitus. Diagnostics. 2022; 12(8):1829. https://doi.org/10.3390/diagnostics12081829
Chicago/Turabian StyleCalapod, Ovidiu Paul, Andreea Maria Marin, Anca Pantea Stoian, and Carmen Fierbinteanu-Braticevici. 2022. "Clinical Model for the Prediction of Severe Liver Fibrosis in Adult Patients with Type II Diabetes Mellitus" Diagnostics 12, no. 8: 1829. https://doi.org/10.3390/diagnostics12081829
APA StyleCalapod, O. P., Marin, A. M., Pantea Stoian, A., & Fierbinteanu-Braticevici, C. (2022). Clinical Model for the Prediction of Severe Liver Fibrosis in Adult Patients with Type II Diabetes Mellitus. Diagnostics, 12(8), 1829. https://doi.org/10.3390/diagnostics12081829








