An Unfavorable Outcome of M. chimaera Infection in Patient with Silicosis
Abstract
:1. Introduction
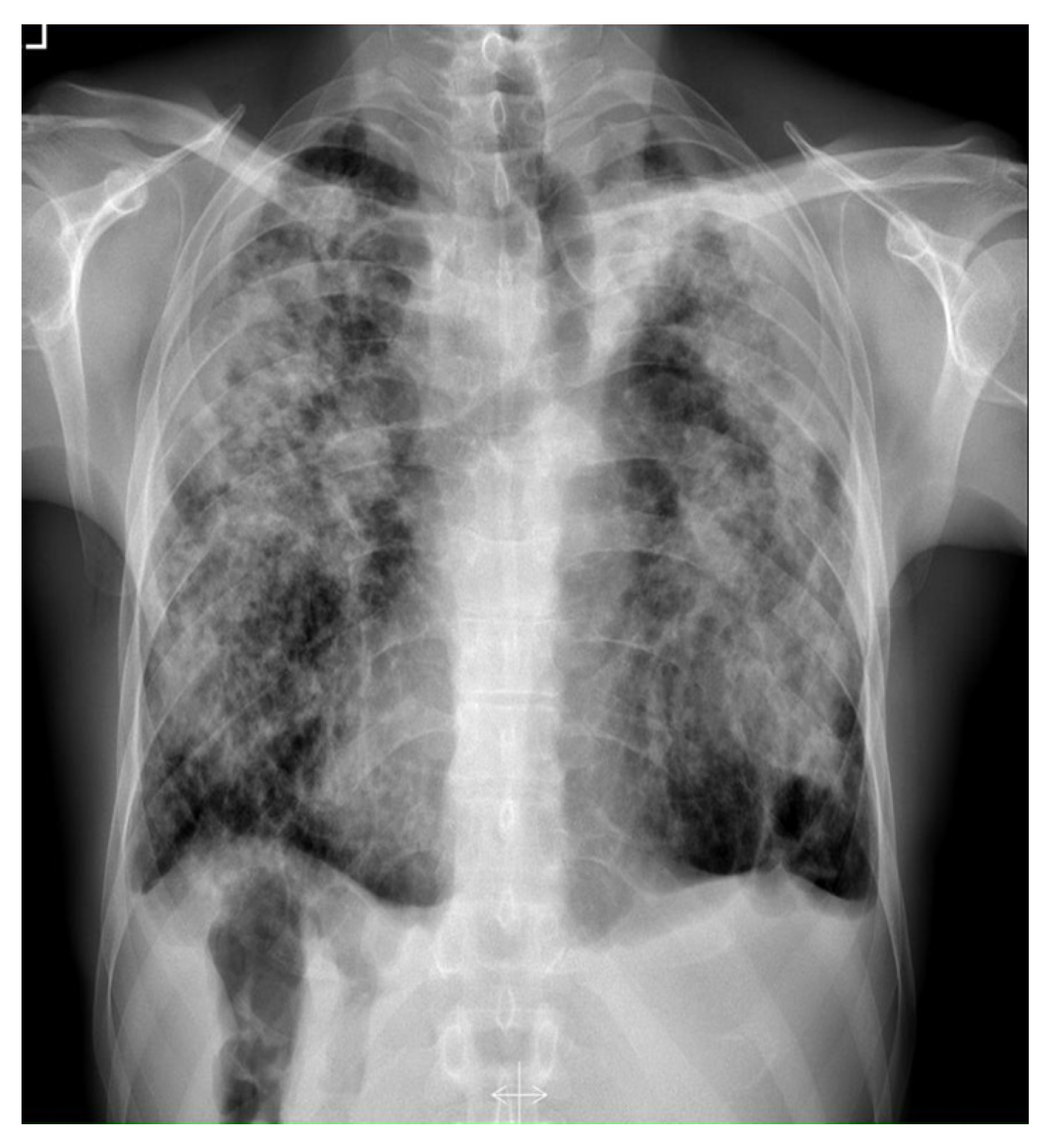
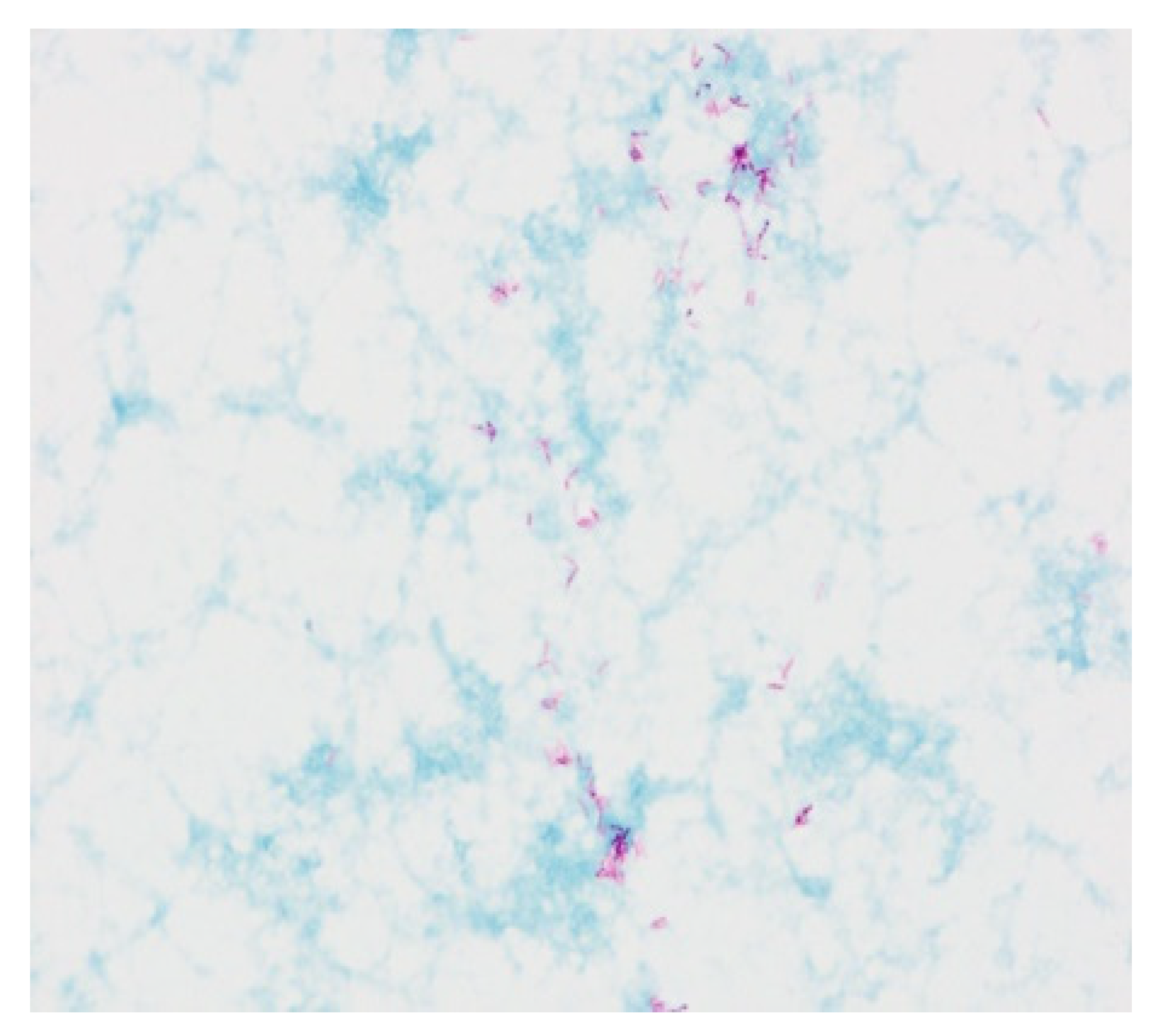
2. Case Report
3. Discussion
4. Conclusions
Author Contributions
Funding
Institution Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Riccardi, N.; Monticelli, J.; Antonello, R.M.; Gabrielli, M.; Ferrarese, M.; Codecasa, L.; Di Bella, S.; Giacobbe, D.R.; Luzzati, R. Mycobacterium chimaera infections: An update. J. Infect. Chemother. 2020, 26, 199–205. [Google Scholar] [CrossRef] [PubMed]
- Miskoff, J.A.; Chaudhri, M. Mycobacterium Chimaera: A Rare Presentation. Cureus 2018, 10, e2750. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- De Melo Carvalho, R.; Nunes, A.L.; Sa, R.; Ramos, I.; Valente, C.; Saraiva da Cunha, J. Mycobacterium chimaera Disseminated Infection. J. Med. Cases. 2020, 11, 35–36. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zabost, A.T.; Szturmowicz, M.; Brzezińska, S.A.; Klatt, M.D.; Augustynowicz-Kopeć, E.M. Mycobacterium chimaera as an Underestimated Cause of NTM Lung Diseases in Patients Hospitalized in Pulmonary Wards. Pol. J. Microbiol. 2021, 70, 315–320. [Google Scholar] [CrossRef] [PubMed]
- Mlika, M.; Adigun, R.; Bhutta, B.S. Silicosis; StatPearls Publishing: Treasure Island, FL, USA, 2022. [Google Scholar]
- Barboza, C.E.; Winter, D.H.; Seiscento, M.; Santos Ude, P.; Terra Filho, M. Tuberculosis and silicosis: Epidemiology, diagnosis and chemoprophylaxis. J. Bras. Pneumol. 2008, 34, 959–966. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kwiatkowska, S.; Augustynowicz-Kopeć, E.; Korzeniewska-Koseła, M.; Filipczak, D.; Gruszczyński, P.; Zabost, A.; Klatt, M.; Sadkowska-Todys, M. Nontuberculous mycobacteria strains isolated from patients between 2013 and 2017 in Poland. Our data with respect to the global trends. Adv. Respir. Med. 2018, 86, 291–298. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Scriven, J.E.; Scobie, A.; Verlander, N.Q.; Houston, A.; Collyns, T.; Cajic, V.; Kon, O.M.; Mitchell, T.; Rahama, O.; Robinson, A.; et al. Mycobacterium chimaera infection following cardiac surgery in the United Kingdom: Clinical features and outcome of the first 30 cases. Clin. Microbiol. Infect. 2018, 24, 1164–1170. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hall, H.; Cosgrove, C.; Houston, A.; Macallan, D.C.; Aul, R. Diagnostic challenges in Mycobacteria chimaera infection. QJM Int. J. Med. 2018, 111, 501–502. [Google Scholar] [CrossRef] [PubMed]
- Natanti, A.; Palpacelli, M.; Valsecchi, M.; Tagliabracci, A.; Pesaresi, M. Mycobacterium chimaera: A report of 2 new cases and literature review. Int. J. Legal. Med. 2021, 135, 2667–2679. [Google Scholar] [CrossRef] [PubMed]
- Ganatra, S.; Sharma, A.; D’Agostino, R.; Gage, T.; Kinnunen, P. Mycobacterium chimaera Mimicking Sarcoidosis. Methodist. Debakey Cardiovasc. J. 2018, 14, 301–302. [Google Scholar] [CrossRef] [PubMed]
- Rosero, C.I.; Shams, W.E. Mycobacterium chimaera infection masquerading as a lung mass in a healthcare worker. IDCases 2019, 15, e00526. [Google Scholar] [CrossRef] [PubMed]
- Chand, M.; Lamagni, T.; Kranzer, K.; Hedge, J.; Moore, G.; Parks, S.; Collins, S.; del Ojo Elias, C.; Ahmed, N.; Brown, T.; et al. Insidious risk of severe Mycobacterial chimera infection in patients after cardiac surgery. Clin. Infect. Dis. 2017, 64, 335–342. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kohler, P.; Kuster, S.P.; Bloemberg, G.; Schulthess, B.; Frank, M.; Tanner, F.C.; Rössle, M.; Böni, C.; Falk, V.; Wilhelm, M.J.; et al. Healthcare-associated heart valve prosthesis, aortic vascular grafting, and disseminated Mycobacterium chimaera infections after open heart surgery. Eur. Heart J. 2015, 36, 2745–2753. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sax, H.; Bloemberg, G.; Hasse, B.; Sommerstein, R.; Kohler, P.; Achermann, Y.; Rössle, M.; Falk, V.; Kuster, S.P.; Böttger, E.C.; et al. Prolonged epidemic of Mycobacterium chimaera infection after open chest surgery. Clin. Infect. Dis. 2015, 61, 67–75. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Griffith, D.E.; Aksamit, T.; Brown-Elliott, B.A.; Catanzaro, A.; Daley, C.; Gordin, F.; Holland, S.M.; Horsburgh, R.; Huitt, G.; Iademarco, M.F.; et al. ATS Mycobacterial Diseases Subcommittee; American Thoracic Society; Infectious Disease Society of America. An official ATS/IDSA statement: Diagnosis, treatment, and prevention of nontuberculous mycobacterial diseases. Am. J. Respir. Crit. Care Med. 2007, 175, 367–416. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Oren, A.; Garrity, G.M. List of new names and new combinations previously effectively, but not validly, published. Int. J. Syst. Evol. Microbiol. 2018, 68, 3379–3393. [Google Scholar] [CrossRef] [PubMed]
- Lecorche, E.; Haenn, S.; Mougari, F.; Kumanski, S.; Veziris, N.; Benmansour, H.; Raskine, L.; Moulin, L.; Cambau, E.; CNR-MyRMA. Comparison of methods available for identification of Mycobacterium chimaera. Clin. Microbiol. Infect. 2018, 24, 409–413. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Buchanan, R.; Agarwal, A.; Mathai, E.; Cherian, B.P. Mycobacterium chimaera: A novel pathogen with potential risk to cardiac surgical patients. Natl. Med. J. India 2020, 33, 284–287. [Google Scholar] [CrossRef] [PubMed]
- Daley, C.L.; Iaccarino, J.M.; Lange, C.; Cambau, E.; Wallace, R.J., Jr.; Andrejak, C.; Böttger, E.C.; Brozek, J.; Griffith, D.E.; Guglielmetti, L.; et al. Treatment of Nontuberculous Mycobacterial Pulmonary Disease: An Official ATS/ERS/ESCMID/IDSA Clinical Practice Guideline. Clin. Infect. Dis. 2020, 71, 905–913. [Google Scholar] [CrossRef] [PubMed]
- Mok, S.; Hannan, M.M.; Nölke, L.; Stapleton, P.; O’Sullivan, N.; Murphy, P.; McLaughlin, A.M.; McNamara, E.; Fitzgibbon, M.M.; Rogers, T.R. Antimicrobial Susceptibility of Clinical and Environmental Mycobacterium chimaera Isolates. Antimicrob. Agents Chemother. 2019, 63, e00755-19. [Google Scholar] [CrossRef] [PubMed] [Green Version]




| Pred | Act1 | %Pred | SR | ||
|---|---|---|---|---|---|
| Date Time | 15-02-02 08:57:42 | ||||
| FEV 1% VC MAX FEV 1% FVC | [%] [%] | 79.83 81.76 | 84.26 84.26 | 105.5 103.1 | 0.62 0.42 |
| VC MAX FVC | [L] [L] | 4.63 7.79 | 2.14 2.14 | 46.2 44.7 | −4.47 −4.28 |
| FEV 1 MMEF 75/25 PEF | [L] [L/s] [L/s] | 3.87 4.25 8.89 | 1.80 1.72 5.76 | 46.6 40.4 64.9 | −4.70 −2.44 −2.59 |
| FET V backextrapolation ex | [s] [L] | 7.16 0.12 | |||
| R tot ITGV RV TLC RV % TLC | [KPa*s/L] [L] [L] [L] [%] | 0.30 3.28 1.91 6.58 29.95 | 0.28 2.47 1.70 3.84 44.27 | 93.7 75.3 88.9 58.3 147.8 | −1.35 −0.52 −3.93 2.63 |
| DLCOc SB DLCOcNA VIN VA | [mmol/min/kPa] [mmol/min/kPa/L] [L] [L] | 10.26 1.56 4.63 6.43 | 5.57 1.81 2.02 3.08 | 54.3 116.0 43.6 47.9 | −3.34 −4.68 |
| Clinical symptoms (any of the following) | pulmonary—including but not limited to: cough, sputum, hemoptysis |
| systemic—including but not limited to: fever, weight loss, sweats | |
| Radiologic presentation (any of the following) | X-ray: nodular or cavitary lesions |
| HRCT: bronchiectasis and nodular opacities | |
| Microbiologic tests (any of the following) | positive culture results from at least two separate sputum samples |
| positive culture result from at least one bronchial wash or lavage | |
| histopathological features of mycobacterial disease (granulomas or AFB) and positive culture for NTM OR histopathological features of mycobacterial disease and one or more cultures positive for NTM from sputum or bronchial washings | |
| AND: | Exclusion of other diseases |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Łyżwa, E.; Siemion-Szcześniak, I.; Sobiecka, M.; Lewandowska, K.; Zimna, K.; Bartosiewicz, M.; Jakubowska, L.; Augustynowicz-Kopeć, E.; Tomkowski, W. An Unfavorable Outcome of M. chimaera Infection in Patient with Silicosis. Diagnostics 2022, 12, 1826. https://doi.org/10.3390/diagnostics12081826
Łyżwa E, Siemion-Szcześniak I, Sobiecka M, Lewandowska K, Zimna K, Bartosiewicz M, Jakubowska L, Augustynowicz-Kopeć E, Tomkowski W. An Unfavorable Outcome of M. chimaera Infection in Patient with Silicosis. Diagnostics. 2022; 12(8):1826. https://doi.org/10.3390/diagnostics12081826
Chicago/Turabian StyleŁyżwa, Ewa, Izabela Siemion-Szcześniak, Małgorzata Sobiecka, Katarzyna Lewandowska, Katarzyna Zimna, Małgorzata Bartosiewicz, Lilia Jakubowska, Ewa Augustynowicz-Kopeć, and Witold Tomkowski. 2022. "An Unfavorable Outcome of M. chimaera Infection in Patient with Silicosis" Diagnostics 12, no. 8: 1826. https://doi.org/10.3390/diagnostics12081826
APA StyleŁyżwa, E., Siemion-Szcześniak, I., Sobiecka, M., Lewandowska, K., Zimna, K., Bartosiewicz, M., Jakubowska, L., Augustynowicz-Kopeć, E., & Tomkowski, W. (2022). An Unfavorable Outcome of M. chimaera Infection in Patient with Silicosis. Diagnostics, 12(8), 1826. https://doi.org/10.3390/diagnostics12081826








