Desquamative Gingivitis in the Context of Autoimmune Bullous Dermatoses and Lichen Planus—Challenges in the Diagnosis and Treatment
Abstract
1. Introduction
2. Etiopathogenesis
2.1. Genetic Predisposition
2.2. Metabolic Factors
2.3. Neuropsychiatric Factors
2.4. Medication
2.5. Infectious Factors
2.6. The Graft-versus-Host Reaction
2.7. Certain Dental Materials
2.8. Autoimmunity
3. Diagnostic Approach
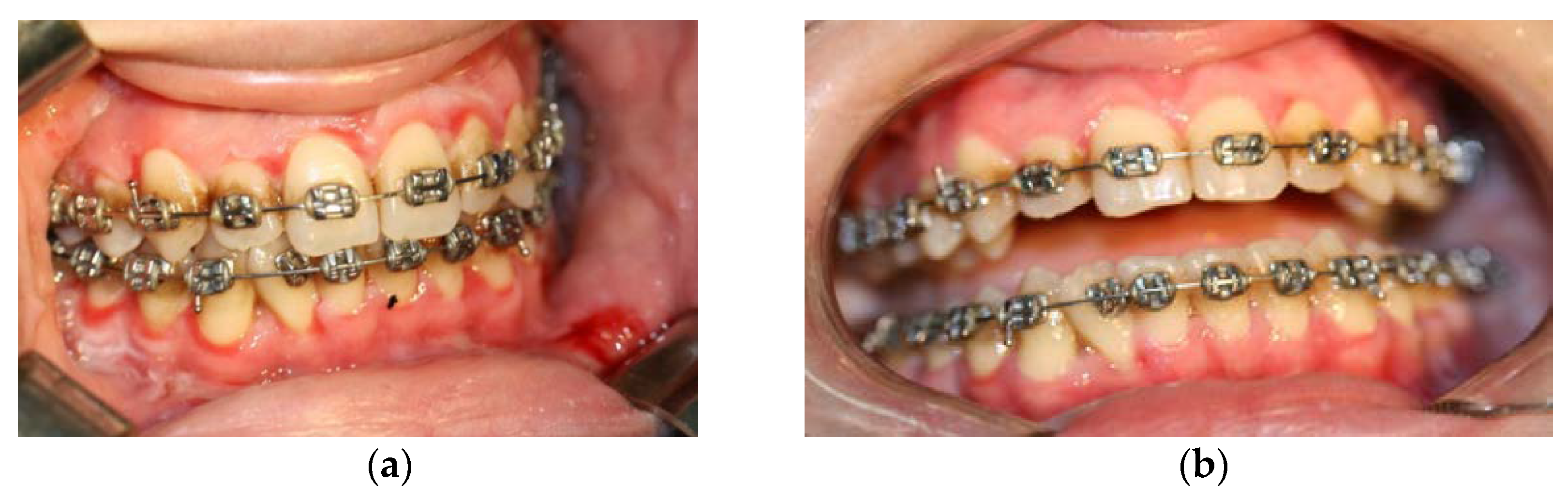
3.1. Clinical Findings
- (1)
- non-plaque gingival erythema;
- (2)
- gingival desquamation;
- (3)
- the presence of other intraoral and sometimes extraoral lesions;
- (4)
- pain, especially when eating spicy foods.
3.2. Patients’ History
3.3. Paraclinical Explorations
4. Associated Autoimmune Bullous Dermatoses
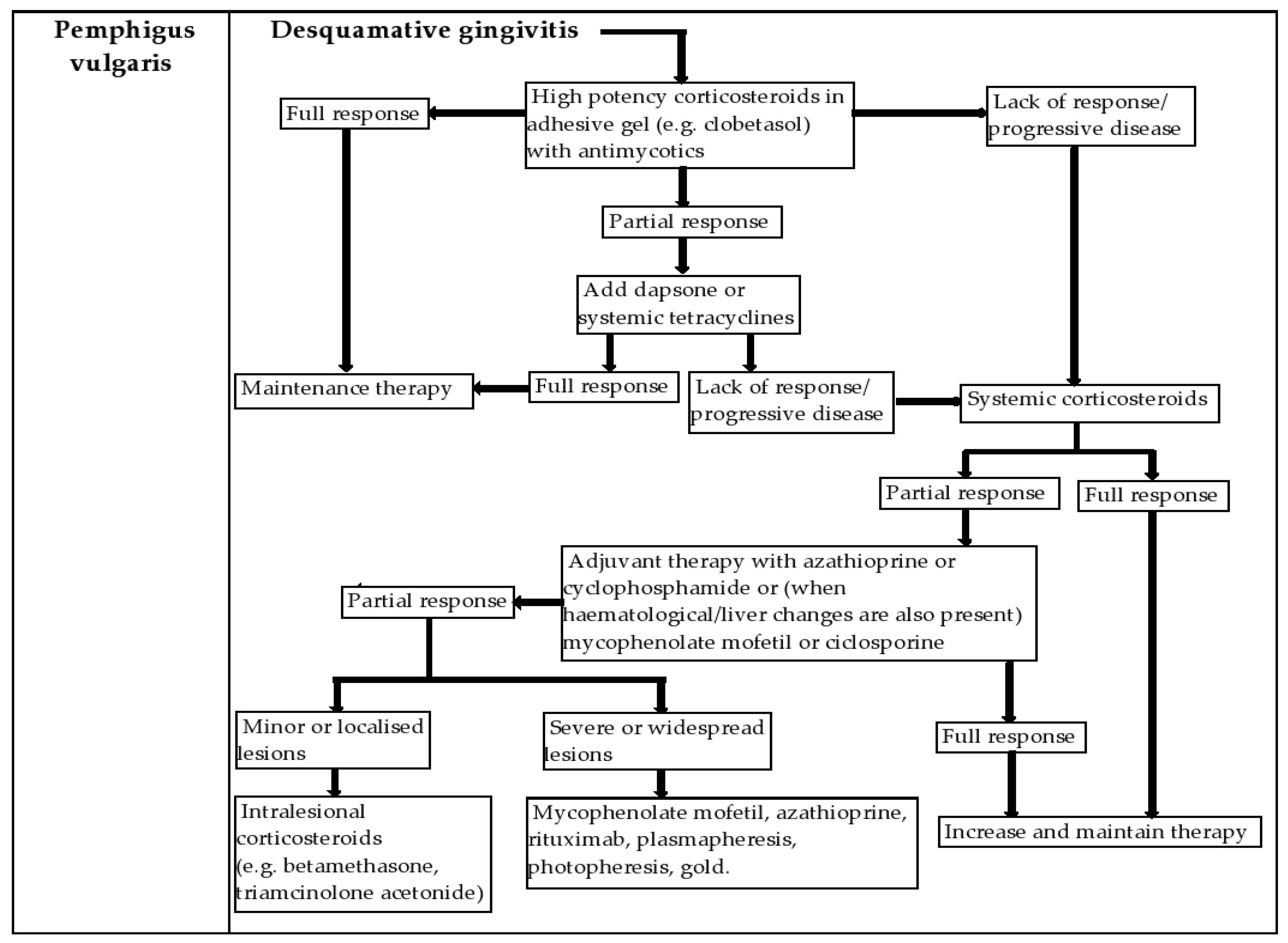
4.1. Pemphigus Vulgaris (PV)
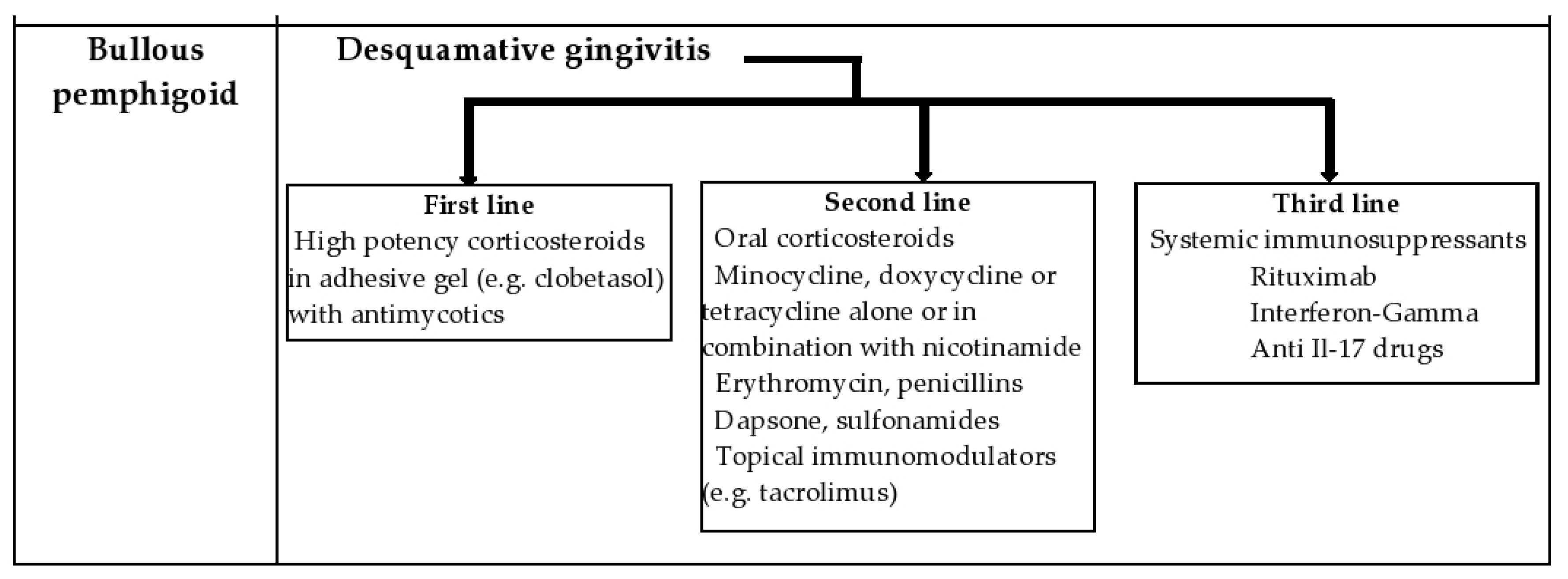
4.2. Bullous Pemphigoid (BP)
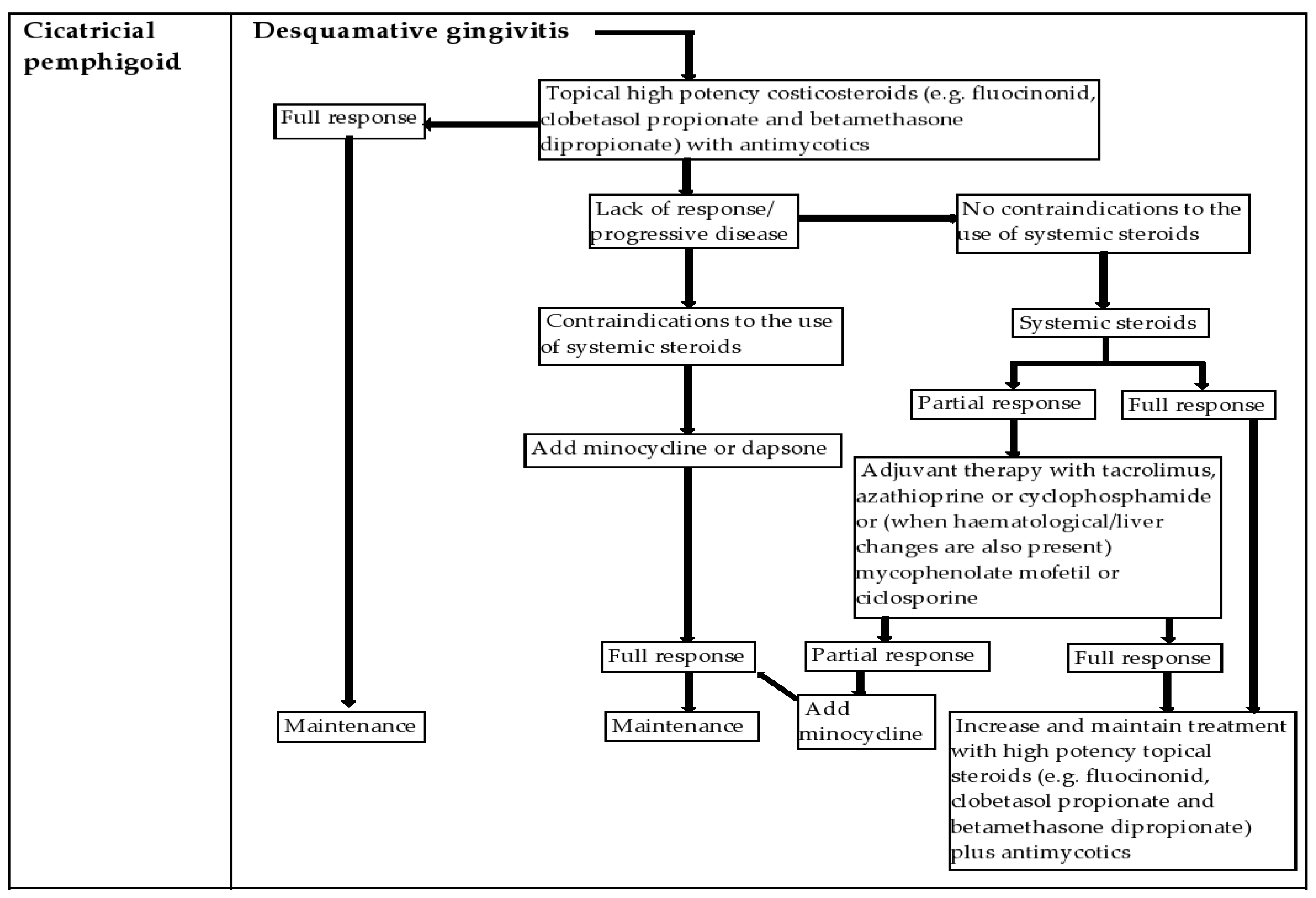
4.3. Cicatricial Pemphigoid (CP)
5. Lichen Planus (LP)
- ⁃
- dental material-induced lichenoid lesions: various types of dental materials, such as amalgam, metals, composite and resin-based, are topographically associated with lichenoid reactions in the oral mucosa. In most cases, these contact allergies are due to a type IV/delayed hypersensitivity reaction. In these situations, a patch test using the suspected materials is useful for establishing the diagnosis [50,77,78,79,80,81,82,83].
- ⁃
- drug-induced lichenoid damage: certain drugs, such as beta-blockers, non-steroidal anti-inflammatory drugs, antihypertensive agents (e.g., angiotensin converting enzyme inhibitors), dapsone, diuretics, oral hypoglycemic agents, gold salts and penicillamine have been reported to induce oral lichenoid damage. They often involve the lip and have a symmetrical distribution. An associated skin rash may suggest a drug-related injury [81,82,83,84].
- ⁃
- erythema multiforme (EM): a reaction of hypersensitivity that occurs with damage to the skin or mucous membranes. Mucosal manifestations are common and have been reported in 25% to 70% of patients with EM. The oral mucosa is most commonly involved. The lesions begin as areas of erythema with edema that progress to erythematous plaques with bullous and erosive lesions, with pseudomembrane formation. Mucosal damage often occurs in combination with skin damage, which can provide a clue for diagnosis when typical and atypical targetoid lesions are found, often in an acral distribution. Isolated mucosal lesions may make diagnosis more difficult [81,82,83].
- ⁃
- graft-versus-host disease: a multi-organ disease that occurs most frequently in patients who have undergone allogeneic stem cell transplantation. In the acute form, oral damage usually occurs in patients with more severe erosive skin disease. In chronic cases, oral lesions are usually similar to those of OLP, with white, reticulated, sometimes eroded plaques involving the oral mucosa, gums, and lips. Other mucosal membranes commonly affected include genital and eye mucosae [81,83,84].
- ⁃
- cicatricial pemphigoid: a disease phenotype comprising several autoimmune bullous disorders characterized by subepithelial bullae and erosions with scarring of mucous membranes, skin or both and linear IgG deposition. The most commonly affected site of the oral mucosa is the gums, followed by the jugal mucosa and palate, and the most common presentation is DG. Oral damage usually presents as erythematous spots that progress to blisters, erosions and pseudomembranous lesions. Healing takes place slowly and often the visible scars in the oral mucosa are not obvious; white reticulated plaques similar to Wickham striae may be residual signs after healing of the erosions [66,81,82].
- ⁃
- complex aphthosis/Behcet’s disease: it is a condition that occurs frequently on the non-keratinized mucosa. The disease often develops during adolescence or young adulthood and improves over time, with the appearance of reduced and milder outbreaks. Clinically, canker sores occur in the form of well-defined round or oval ulcers, often with a pseudomembranous base, on the oral and labial mucosa, dorsal and ventral surfaces of the tongue, soft palate and oropharynx [66,81,83].
- ⁃
- pemphigus vulgaris: oral damage is usually the onset of pemphigus vulgaris and often precedes skin manifestations by several months. Irregular erosions and painful ulcerations are usually seen along the oral and labial mucosa, followed by the palate and tongue. These erosions develop on the mucosa with a healthy appearance, without erythema or surrounding edema. Intact blisters are rarely seen because they are fragile and prone to early rupture. The Nikolsky sign can be seen near an active lesion. Ulcers are persistent and do not heal spontaneously [66,82].
- ⁃
- acquired bullous epidermolysis (EBA): damage to the mucosa may be manifested with erythema and erosions during the inflammatory phase and may progress over time to the loss of the lingual papillae and the healing of areas of the oral mucosa with the development of strictures. The distinguishing features of EBA are its refractory nature and poor response to treatment [82,83].
6. Management of the Patient with DG in the Dental Office and New Treatment Options
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Tofan, E.C.; Părlătescu, I.; Ţovaru, Ş.; Nicolae, C.; Preda, A.S.; Funieru, C. Desquamative gingivitis-A clinicopathological review. Curr. Health Sci. J. 2018, 44, 331–336. [Google Scholar]
- Paul, G.T. Desquamative gingivitis: Does the gingiva tell you more than what meets the eye? A comprehensive review. J. Adv. Clin. Res. Insights 2019, 6, 48–52. [Google Scholar] [CrossRef]
- Leao, J.C.; Ingafou, M.; Khan, A.; Scully, C.; Porter, S. Desquamative gingivitis: Retrospective analysis of disease associations of a large cohort. Oral Dis. 2008, 14, 556–560. [Google Scholar] [CrossRef] [PubMed]
- Ramesh, A.; Bhat, R.M.; Madhumita, M.; Jaganathan, P. Desquamative gingivitis in dermatological disorders. Indian J. Dermatol. Venereol. Leprol. 2021, 87, 446–451. [Google Scholar] [CrossRef]
- Pande, A.; Rai, P.; Shetty, D.; Dharmadhikari, S. Oral lichenoid reaction to dental amalgam and its association with desquamative gingivitis “a diagnostic conundrum”: A case report. Int. J. Oral Care Res. 2021, 9, 42. [Google Scholar]
- Martu, M.-A.; Maftei, G.-A.; Luchian, I.; Stefanescu, O.M.; Scutariu, M.M.; Solomon, S.M. The Effect of Acknowledged and Novel Anti-Rheumatic Therapies on Periodontal Tissues—A Narrative Review. Pharmaceuticals 2021, 14, 1209. [Google Scholar] [CrossRef]
- Leuci, S.; Ruoppo, E.; Adamo, D.; Calabria, E.; Mignogna, M.D. Oral autoimmune vesicobullous diseases: Classification, clinical presentations, molecular mechanisms, diagnostic algorithms, and management. Periodontology 2000 2019, 80, 77–88. [Google Scholar] [CrossRef] [PubMed]
- Lo Russo, L.; Fierro, G.; Guiglia, R.; Compilato, D.; Testa, N.F.; Lo Muzio, L.; Campisi, G. Epidemiology of desquamative gingivitis: Evaluation of 125 patients and review of the literature. Int. J. Dermatol. 2009, 48, 1049–1052. [Google Scholar] [CrossRef] [PubMed]
- Teodorescu, A.C.; Martu, I.; Teslaru, S.; Kappenberg-Nitescu, D.C.; Goriuc, A.; Luchian, I.; Martu, M.A.; Solomon, S.M.; Martu, S. Assessment of Salivary Levels of RANKL and OPG in Aggressive versus Chronic Periodontitis. J. Immunol. Res. 2019, 2019, 6195258. [Google Scholar] [CrossRef] [PubMed]
- Martu, M.A.; Solomon, S.M.; Toma, V.; Maftei, G.A.; Iovan, A.; Gamen, A.; Hurjui, L.; Rezus, E.; Foia, L.; Forna, N.C. The importance of cytokines in periodontal disease and rheumatoid arthritis. Review. Rom. J. Oral Rehabil. 2019, 11, 220–240. [Google Scholar]
- Budimir, J.; Lugović Mihić, L.; Šitum, M.; Bulat, V.; Peršić, S.; Tomljanović-Veselski, M. Oral lesions in patients with pemphigus vulgaris and bullous pemphigoid. Acta Clin. Croat. 2008, 47, 18. [Google Scholar]
- Misra, S.R.; Baskaran, P. Desquamative Gingivitis in Cicatricial Pemphigoid: A Diagnostic Paradox! Indian J. Forensic Med. Toxicol. 2020, 14, 8286–8290. [Google Scholar]
- Popa, C.; Filioreanu, A.M.; Stelea, C.; Alexandru Maftei, G.A.; Popescu, E. Prevalence of oral lesions modulated by patients age: The young versus the elderly. Rom. J. Oral Rehabil. 2018, 10, 50–56. [Google Scholar]
- Martu, M.A.; Maftei, G.A.; Luchian, I.; Popa, C.; Filioreanu, A.M.; Tatarciuc, D.; Nichitean, G.; Hurjui, L.-L.; Foia, L.-G. Wound healing of periodontal and oral tissues: Part II—Patho-phisiological conditions and metabolic diseases. Rom. J. Oral Rehabil. 2020, 12, 30–40. [Google Scholar]
- Ricketts, D.N.J.; Morgan, C.L.; McGregor, J.M.; Morgan, P.R. Kindler syndrome: A rare cause of desquamative lesions of the gingiva. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endodontology 1997, 84, 488–491. [Google Scholar] [CrossRef]
- Antonelli, E.; Bassotti, G.; Tramontana, M.; Hansel, K.; Stingeni, L.; Ardizzone, S.; Genovese, G.; Marzano, A.V.; Maconi, G. Dermatological Manifestations in Inflammatory Bowel Diseases. J. Clin. Med. 2021, 10, 364. [Google Scholar] [CrossRef] [PubMed]
- Paradowska-Stolarz, A.M. Oral manifestations of COVID-19: Brief review. Dent. Med. Probl. 2021, 58, 123–126. [Google Scholar] [CrossRef]
- Zhang, X.; Gu, H.; Xie, S.; Su, Y. Periodontitis in patients with psoriasis: A systematic review and meta-analysis. Oral Dis. 2022, 28, 33–43. [Google Scholar] [CrossRef]
- Cao, T.; Shao, S.; Fang, H.; Li, B.; Wang, G. Role of regulatory immune cells and molecules in autoimmune bullous dermatoses. Front. Immunol. 2019, 10, 1746. [Google Scholar] [CrossRef]
- Martu, M.A.; Maftei, G.A.; Sufaru, I.G.; Jelihovschi, I.; Luchian, I.; Hurjui, L.; Martu, I.; Pasarin, L. COVID-19 and Periodontal Disease-Ethiopathogenic and Clinical Implications. Rom. J. Oral Rehab. 2020, 12, 116–124. [Google Scholar]
- Shaqman, M.; Hamdan, A.; Karadsheh, O.; Sawair, F.; Hassona, Y. Desquamative gingivitis: A challenging diagnosis for clinicians. Br. Dent. J. 2020, 229, 26–30. [Google Scholar] [CrossRef] [PubMed]
- Légeret, C.; Furlano, R. Oral ulcers in children-a clinical narrative overview. Ital. J. Pediatrics 2021, 47, 144. [Google Scholar] [CrossRef] [PubMed]
- Lo Russo, L.; Gallo, C.; Pellegrino, G.; Lo Muzio, L.; Pizzo, G.; Campisi, G.; Di Fede, O. Periodontal clinical and microbiological data in desquamative gingivitis patients. Clin. Oral Investig. 2014, 18, 917–925. [Google Scholar] [CrossRef] [PubMed]
- Ertugrul, A.S.; Dursun, R.; Dundar, N.; Avunduk, M.C.; Hakki, S.S. MMP-1, MMP-9, and TIMP-1 levels in oral lichen planus patients with gingivitis or periodontitis. Arch. Oral Biol. 2013, 58, 843–852. [Google Scholar] [CrossRef] [PubMed]
- Ertugrul, A.S.; Arslan, U.; Dursun, R.; Sezgin Hakki, S. Periodontopathogen profile of healthy and oral lichen planus patients with gingivitis or periodontitis. Int. J. Oral Sci. 2013, 5, 92–97. [Google Scholar] [CrossRef]
- Jascholt, I.; Lai, O.; Zillikens, D.; Kasperkiewicz, M. Periodontitis in oral pemphigus and pemphigoid: A systematic review of published studies. J. Am. Acad. Dermatol. 2017, 76, 975–978. [Google Scholar] [CrossRef]
- Haverman, T.M.; Raber-Durlacher, J.E.; Raghoebar, I.I.; Rademacher, W.M.H.; Rozema, F.R.; Hazenberg, M.D.; Epstein, J.B.; Treister, N.S. Oral chronic graft-versus-host disease: What the general dental practitioner needs to know. J. Am. Dent. Assoc. 2020, 151, 846–856. [Google Scholar] [CrossRef]
- Taraboanta, I.; Stoleriu, S.; Nica, I.; Georgescu, A.; Gamen, A.C.; Maftei, G.A.; Andrian, S. Roughness variation of a nonhybrid composite resin submitted to acid and abrasive challenges. Int. J. Med. Dent. 2020, 24, 182–187. [Google Scholar]
- Saschenbrecker, S.; Karl, I.; Komorowski, L.; Probst, C.; Dähnrich, C.; Fechner, K. Serological diagnosis of autoimmune bullous skin diseases. Front. Immunol. 2019, 10, 1974. [Google Scholar] [CrossRef]
- Mathew, M.; Nayak, V.S.; Ramaiah, S.K.; Sethuraman, S. Desquamative gingivitis: A definite indicator for mucocutaneous disorders. BMJ Case Rep. CP 2022, 15, e247857. [Google Scholar] [CrossRef]
- Alkahtani, Z.M.; Arunachalam, R.; Bapat, R.A.; Thangavelu, A.; Jegatheeswaran, A.G. Idiopathic desquamative gingivitis. J. Pharm. Bioallied Sci. 2021, 13 (Suppl. 1), S865. [Google Scholar]
- Suresh, L.; Neiders, M.E. Definitive and differential diagnosis of desquamative gingivitis through direct immunofluorescence studies. J. Periodontol. 2012, 83, 1270s1278. [Google Scholar] [CrossRef] [PubMed]
- Endo, H.; Rees, T.D.; Allen, E.P.; Kuyama, K.; Aoki, S.; Yamamoto, H.; Ito, T. A stab-and-roll biopsy technique to maintain gingival epithelium for desquamative gingivitis. J. Periodontol. 2014, 85, 802–809. [Google Scholar] [CrossRef]
- Sano, S.M.; Quarracino, M.C.; Aguas, S.C.; González, E.J.; Harada, L.; Krupitzki, H.; Mordoh, A. Sensitivity of direct immunofluorescence in oral diseases. Study of 125 cases. Med. Oral Patol. Oral Y Cir. Bucal 2008, 13, E287–E291. [Google Scholar]
- Mutasim, D.F.; Adams, B.B. Immunofluorescence in dermatology. J. Am. Acad. Dermatol. 2001, 45, 803–824. [Google Scholar] [CrossRef]
- Maderal, A.D.; Salisbury III, P.L.; Jorizzo, J.L. Desquamative gingivitis: Diagnosis and treatment. J. Am. Acad. Dermatol. 2018, 78, 851–861. [Google Scholar] [CrossRef] [PubMed]
- Brazao-Silva, M.T.; Cabral, L.N.; Pinheiro, T.N. Proposal of a minimally invasive approach diagnosing desquamative gingivitis-associated diseases. J. Indian Soc. Periodontol. 2021, 25, 83. [Google Scholar] [PubMed]
- Oliveira, L.A.; Freitas, P.L.; Ferreira, N.H.; Assunção, J.N.R.; Teixeira, V.P.; De Almeida, R.C.; Loureiro, V. Desquamative Gingivitis: Challenges in the Diagnosis. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. 2018, 126, e127. [Google Scholar] [CrossRef]
- Panzarella, V.; Bartolone, A.; Coniglio, R.; Rodolico, V.; Maniscalco, L.; Capocasale, G.; Iurato Carbone, M.; Campisi, G. Diagnostic Concordance between Optical Coherence Tomography and Histological Investigations for Immune-Mediated Desquamative Gingivitis: Observational Study. Int. J. Environ. Res. Public Health 2021, 18, 9095. [Google Scholar] [CrossRef]
- Panzarella, V.; Bartolone, A.; Rodolico, V.; Capocasale, G.; Maniscalco, L.; Matranga, D.; Di Fede, O.; Campisi, G. Immune-Mediated Desquamative Gingivitis and Optical Coherence Tomography Diagnostic Patterns: Clinical Implication from a Systematic Review. Diagnostics 2021, 11, 1453. [Google Scholar] [CrossRef]
- Rashid, H.; Lamberts, A.; Diercks, G.F.; Pas, H.H.; Meijer, J.M.; Bolling, M.C.; Horváth, B. Oral lesions in autoimmune bullous diseases: An overview of clinical characteristics and diagnostic algorithm. Am. J. Clin. Dermatol. 2019, 20, 847–861. [Google Scholar] [CrossRef]
- Pollmann, R.; Schmidt, T.; Eming, R.; Hertl, M. Pemphigus: A comprehensive review on pathogenesis, clinical presentation and novel therapeutic approaches. Clin. Rev. Allergy Immunol. 2018, 54, 1–25. [Google Scholar] [CrossRef]
- Huang, Y.; Jedličková, H.; Cai, Y.; Rehman, A.; Gammon, L.; Ahmad, U.S.; Uttagomol, J.; Parkinson, E.K.; Fortune, F.; Wan, H. Oxidative Stress-Mediated YAP Dysregulation Contributes to the Pathogenesis of Pemphigus Vulgaris. Front. Immunol. 2021, 12, 1253. [Google Scholar] [CrossRef] [PubMed]
- Rehman, A.; Huang, Y.; Wan, H. Evolving Mechanisms in the Pathophysiology of Pemphigus Vulgaris: A Review Emphasizing the Role of Desmoglein 3 in Regulating p53 and the Yes-Associated Protein. Life 2021, 11, 621. [Google Scholar] [CrossRef] [PubMed]
- Nili, A.; Salehi Farid, A.; Asgari, M.; Tavakolpour, S.; Mahmoudi, H.; Daneshpazhooh, M. Current status and prospects for the diagnosis of pemphigus vulgaris. Expert Rev. Clin. Immunol. 2021, 17, 819–834. [Google Scholar] [CrossRef] [PubMed]
- Dal Prá, K.J.; de Assis Tristão, S.D.S.S.; Franco, J.B.; Matias, D.T.; Carrillo, C.M.; de Melo Peres, M.P.S.; Ribas, P.F. Oral management of pemphigus vulgaris in the intensive care unit. Spec. Care Dent. 2020, 40, 280–284. [Google Scholar] [CrossRef] [PubMed]
- Gandolfo, S.; Scully, C.; Carrozzo, M. Oral Medicine, Edit; Churchill Livingstone Elsevier: London, UK, 2010. [Google Scholar]
- Lee, M.S.; Yeh, Y.C.; Tu, Y.K.; Chan, T.C. Network meta-analysis–based comparison of first-line steroid-sparing adjuvants in the treatment of pemphigus vulgaris and pemphigus foliaceus. J. Am. Acad. Dermatol. 2021, 85, 176–186. [Google Scholar] [CrossRef]
- Kridin, K. Emerging treatment options for the management of pemphigus vulgaris. Ther. Clin. Risk Manag. 2018, 14, 757. [Google Scholar] [CrossRef]
- Maderal, A.D.; Salisbury III, P.L.; Jorizzo, J.L. Desquamative gingivitis: Clinical findings and diseases. J. Am. Acad. Dermatol. 2018, 78, 839–848. [Google Scholar] [CrossRef]
- Genovese, G.; Di Zenzo, G.; Cozzani, E.; Berti, E.; Cugno, M.; Marzano, A.V. New insights into the pathogenesis of bullous pemphigoid: 2019 update. Front. Immunol. 2019, 10, 1506. [Google Scholar] [CrossRef]
- Cirillo, N.; Prime, S.S. A Scoping Review of the Role of Metalloproteinases in the Pathogenesis of Autoimmune Pemphigus and Pemphigoid. Biomolecules 2021, 11, 1506. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Zhao, W.; Jin, H.; Li, L. Risk Factors for Mucosal Involvement in Bullous Pemphigoid and the Possible Mechanism: A Review. Front. Med. 2021, 8, 680871. [Google Scholar] [CrossRef]
- Kridin, K.; Bergman, R. Assessment of the prevalence of mucosal involvement in bullous pemphigoid. JAMA Dermatol. 2019, 155, 166–171. [Google Scholar] [CrossRef]
- Chuah, S.Y.; Tan, S.H.; Chua, S.H.; Tang, M.B.; Lim, Y.L.; Neoh, C.Y.; Tan, A.W. A retrospective review of the therapeutic response with remission in patients with newly diagnosed bullous pemphigoid. Australas. J. Dermatol. 2014, 55, 149–151. [Google Scholar] [CrossRef] [PubMed]
- Di Lernia, V.; Casanova, D.M.; Goldust, M.; Ricci, C. Pemphigus vulgaris and bullous pemphigoid: Update on diagnosis and treatment. Dermatol. Pract. Concept. 2020, 10, e2020050. [Google Scholar] [CrossRef] [PubMed]
- Ruggiero, A.; Megna, M.; Villani, A.; Comune, R.; Fabbrocini, G.; di Vico, F. Strategies to Improve Outcomes of Bullous Pemphigoid: A Comprehensive Review of Clinical Presentations, Diagnosis, and Patients’ Assessment. Clin. Cosmet. Investig. Dermatol. 2022, 15, 661. [Google Scholar] [CrossRef]
- Patel, P.M.; Jones, V.A.; Murray, T.N.; Amber, K.T. A review comparing international guidelines for the management of bullous pemphigoid, pemphigoid gestationis, mucous membrane pemphigoid, and epidermolysis bullosa acquisita. Am. J. Clin. Dermatol. 2020, 21, 557–565. [Google Scholar] [CrossRef] [PubMed]
- Afarideh, M.; Borucki, R.; Werth, V.P. A Review of the Immunologic Pathways Involved in Bullous Pemphigoid and Novel Therapeutic Targets. J. Clin. Med. 2022, 11, 2856. [Google Scholar] [CrossRef]
- Endo, H.; Rees, T.D.; Niwa, H.; Kuyama, K.; Oshima, M.; Serizawa, T.; Tanaka, S.; Iijima, M.; Komiya, M.; Ito, T. Desquamative gingivitis: Early sign of mucous membrane pemphigoid and pemphigus vulgaris. In Periodontology and Dental Implantology; IntechOpen: Vienna, Austria, 2018. [Google Scholar]
- Besagar, S.; London, A.O.; Jairam, M.P.; Trief, D.; Dagi Glass, L.R. Chronic Early-Stage Ocular Cicatricial Pemphigoid. Ophthalmic Plast. Reconstr. Surg. 2021, 37, e209–e213. [Google Scholar] [CrossRef]
- Chou, H.C.; Wu, Y.H.; Chiang, C.P.; Yang, J.H. Cicatricial pemphigoid presenting as desquamative gingivitis. J. Dent. Sci. 2020, 15, 110. [Google Scholar] [CrossRef]
- Giannetti, L.; Murri Dello Diago, A. Therapy of autoimmune mouth bullous disease: 2020 review. Dermatol. Ther. 2021, 34, e14376. [Google Scholar] [CrossRef] [PubMed]
- Saeed, S.; Hasan, S.; Panigrahi, R.; Panda, S.; Ajiz, S.T. Desquamative Gingivitis as the Sole Manifestation in Oral Lichen Planus: Report of Case and Brief Literature Review. Med. -Leg. Update 2020, 20, 3119. [Google Scholar]
- Bolognia, J.; Jorizzo, J.L.; Schaffer, J.V. Dermatology, 4th ed.; Elsevier Saunders: Amsterdam, The Netherlands, 2017; Chapter 17; pp. 1223–1224. [Google Scholar]
- Ion, D.I.; Setterfield, J.F. Oral Lichen Planus. Prim. Dent. J. 2016, 5, 40–44. [Google Scholar] [CrossRef] [PubMed]
- Alajbeg, I.; Challacombe, S.J.; Holmstrup, P.; Jontell, M. Red and White Lesions of the Oral Mucosa. Burket’s Oral Med. 2021, 85–138. [Google Scholar] [CrossRef]
- Kurago, Z.B. Etiology and Pathogenesis of Oral Lichen Planus: An Overview. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. 2016, 122, 72–80. [Google Scholar] [CrossRef]
- Saleh, W.; Youssef, J.M.; Ata, F.; Anees, M.M.; Cha, S.; Katz, J. Risk co-factors inducing malignant transformation of oral lichen planus: A literature review of clinical studies. J. Clin. Adv. Dent. 2021, 5, 005–011. [Google Scholar]
- Nogueira, P.A.; Carneiro, S.; Ramos-e-Silva, M. Oral Lichen Planus: An update on its pathogenesis. Int. J. Dermatol. 2015, 54, 1005–1010. [Google Scholar] [CrossRef]
- Balighi, K.; Shams-Davatchi, C.; Ghobadi, S.; Daneshpajooh, M.; Lajevardi, V.; Mahmoodi, H.; Aryanian, Z.; Teymourpour, A.; Seirafi, R.; Beigmohammadi, F. Retrospective study of gingival involvement in pemphigus: A difficult to treat phenomenon. Dermatol. Ther. 2022, 35, e15475. [Google Scholar] [CrossRef]
- Weston, G.; Payette, M. Update on lichen planus and its clinical variants. Int. J. Womens Dermatol. 2015, 1, 140–149. [Google Scholar] [CrossRef]
- Scully, C.; Lodi, G. EAOM Handbook, 1st ed. 2020, pp. 179–186. Available online: https://eaom.eu/wp-content/uploads/2020/08/EAOM-Handbook.pdf (accessed on 17 July 2022).
- Fabiana, M.C. Desquamative gingivitis as a clinical sign of oral lichen planus: Review of literature. J. Dent. Health Oral. Disord. Ther. 2018, 9, 443–445. [Google Scholar] [CrossRef][Green Version]
- Odell, E.W. Cawson’s Essentials of Oral Pathology and Oral Medicine, 9th ed.; Elsevier Ltd.: Amsterdam, The Netherlands, 2017; Volume 16, pp. 262–263. [Google Scholar]
- Da Silva Costa, R.; Armelin, A.M.L.; Fernandes, K.G.C.; Boer, N.P.; Simonato, L.E. Líquen Plano Oral Diagnóstico e Conduta. In Proceedings of the IX Jornada Odontológica da Universidade Brazil, Sao Paulo, Brazil, 21–25 August 2017. [Google Scholar]
- Buajeeb, W.; Okuma, N.; Thanakun, S.; Laothumthut, T. Direct Immunofluorescence in Oral Lichen Planus. J. Clin. Diagn. Res. 2015, 9, ZC34–ZC37. [Google Scholar] [CrossRef] [PubMed]
- Belgasem, K.A.; Ali, A.A. Oral lichen planus-Review on etiopathogenesis and management. IJ Clin. Img. Med. Rew. 2022, 1, 1016. [Google Scholar]
- Filioreanu, A.M.; Popa, C.; Maftei, G.A.; Parlatescu, I.; Nicolae, C.L.; Popescu, E. Migratory stomatitis–case presentation. Rom. J. Oral Rehabil. 2018, 10, 54–59. [Google Scholar]
- Chiang, C.P.; Yu-Fong Chang, J.; Wang, Y.P.; Wu, Y.H.; Lu, S.Y.; Sun, A. Oral lichen planus—Differential diagnoses, serum autoantibodies, hematinic deficiencies, and management. J. Formos. Med. Assoc. 2018, 117, 756–765. [Google Scholar] [CrossRef]
- Shirasuna, K. Oral lichen planus: Malignant potential and diagnosis. Oral Sci. Int. 2014, 11, 1–7. [Google Scholar] [CrossRef]
- Alrashdan, M.S.; Cirillo, N.; McCullough, M. Oral lichen planus (Review and update). Arch. Dermatol. Res. 2016, 308, 539–551. [Google Scholar] [CrossRef]
- Karagoz, G.; Bektas-Kayhan, K.; Unur, M. Desquamative gingivitis (Review). J. Istanb. Univ. Fac. Dent. 2016, 50, 54–60. [Google Scholar] [CrossRef]
- Rotaru, D.; Chisnoiu, R.; Picos, A.M.; Picos, A.; Chisnoiu, A. Treatment trends in oral lichen planus and oral lichenoid lesions (Review). Exp. Ther. Med. 2020, 20, 198. [Google Scholar] [CrossRef]
- Shavit, E.; Hagen, K.; Shear, N. Oral lichen planus: A novel staging and algorithmic approach and all that is essential to know. F1000Res. 2020, 9, F1000 Faculty Rev-206. [Google Scholar] [CrossRef]
- Gall, R.; Navarro-Fernandez, I.N. Lichen Planus Erosive Form; StatPearls Publishing: Treasure Island, FL, USA, 2021. [Google Scholar]
- Shim, Y. Dental Implants in Patients with Gingival Oral Lichen Planus. JOMP 2019, 44, 77–82. [Google Scholar] [CrossRef]
- Górski, B. Dental Implant Treatment in Patients Suffering from Oral Lichen Planus: A Narrative Review. Int. J. Environ. Res. Public Health 2022, 19, 8397. [Google Scholar] [CrossRef]
- Hr, S.; Shenoy, N. Menopausal Desquamative Gingivitis—A Diagnostic Challenge. J. Evol. Med. Dent. Sci. 2020, 9, 2927–2930. [Google Scholar] [CrossRef]
- Cabras, M.; Gambino, A.; Broccoletti, R.; Arduino, P.G. Desquamative gingivitis: A systematic review of possible treatments. J. Biol. Regul. Homeost. Agents 2019, 33, 637–642. [Google Scholar] [PubMed]
- Garcia-Pola, M.J.; Rodriguez-Lopez, S.; Fernanz-Vigil, A.; Bagan, L.; Garcia-Martin, J.M. Oral hygiene instructions and professional control as part of the treatment of desquamative gingivitis. Systematic review. Med. Oral Patol. Oral Y Cir. Bucal 2019, 24, e136. [Google Scholar] [CrossRef] [PubMed]
- Cafaro, A.; Albanese, G.; Arduino, P.G.; Mario, C.; Massolini, G.; Mozzati, M.; Broccoletti, R. Effect of low-level laser irradiation on unresponsive oral lichen planus: Early preliminary results in 13 patients. Photomed. Laser Surg. 2010, 28, S-99. [Google Scholar] [CrossRef] [PubMed]
- Mocanu, R.C.; Martu, M.-A.; Luchian, I.; Sufaru, I.G.; Maftei, G.A.; Ioanid, N.; Martu, S.; Tatarciuc, M. Microbiologic Profiles of Patients with Dental Prosthetic Treatment and Periodontitis before and after Photoactivation Therapy—Randomized Clinical Trial. Microorganisms 2021, 9, 713. [Google Scholar] [CrossRef]
- Kalhori, K.A.; Vahdatinia, F.; Jamalpour, M.R.; Vescovi, P.; Fornaini, C.; Merigo, E.; Fekrazad, R. Photobiomodulation in oral medicine. Photobiomodulation Photomed. Laser Surg. 2019, 37, 837–861. [Google Scholar] [CrossRef]
- De Carvalho, M.M.; Hidalgo, M.A.R.; Scarel-Caminaga, R.M.; Ribeiro Junior, N.V.; Sperandio, F.F.; Pigossi, S.C.; de Carli, M.L. Photobiomodulation of gingival lesions resulting from autoimmune diseases: Systematic review and meta-analysis. Clin. Oral Investig. 2022, 26, 3949–3964. [Google Scholar] [CrossRef]
- Bellocchi, C.; Carandina, A.; Montinaro, B.; Targetti, E.; Furlan, L.; Rodrigues, G.D.; Tobaldini, E.; Montano, N. The Interplay between Autonomic Nervous System and Inflammation across Systemic Autoimmune Diseases. Int. J. Mol. Sci. 2022, 23, 2449. [Google Scholar] [CrossRef]
- Patel, P.M.; Jones, V.A.; Kridin, K.; Strong, R.A.; Yale, M.; Amber, K.T. Assessing the risk factors for anxiety, depression and post-traumatic stress disorder through the International Pemphigus and Pemphigoid Foundation. J. Eur. Acad. Dermatol. Venereol. 2022, 36, e139–e141. [Google Scholar] [CrossRef]
- Calabria, E.; Adamo, D.; Leuci, S.; Pecoraro, G.; Coppola, N.; Aria, M.; Mignogna, M.D. The health-related quality of life and psychological profile in patients with oropharyngeal Pemphigus Vulgaris in complete clinical remission: A case-control study. J. Oral Pathol. Med. 2021, 50, 510–519. [Google Scholar] [CrossRef] [PubMed]
- Metwally, D.; Fawzy, M.; ElKalioby, M.; Hegazy, R.; Abdel Hay, R.; Abd-Elreheem, H.; Abolmagd, S.; Elgarib, N. Assessment of the quality of life, prevalence of depression, and the level of interleukin 6 in patients with pemphigus vulgaris. Acta Dermatovenerol. Croat. 2020, 28, 57–62. [Google Scholar]
- Lee, J.; Seiffert-Sinha, K.; Attwood, K.; Sinha, A.A. A retrospective study of patient-reported data of bullous pemphigoid and mucous membrane pemphigoid from a US-based registry. Front. Immunol. 2019, 10, 2219. [Google Scholar] [CrossRef] [PubMed]
- Daume, L.; Kreis, C.; Bohner, L.; Jung, S.; Kleinheinz, J. Clinical characteristics of oral lichen planus and its causal context with dental restorative materials and oral health-related quality of life. BMC Oral Health 2021, 21, 262. [Google Scholar] [CrossRef] [PubMed]
- Krain, R.L.; Kushner, C.J.; Tarazi, M.; Gaffney, R.G.; Yeguez, A.C.; Zamalin, D.E.; Pearson, D.R.; Feng, R.; Payne, A.S.; Werth, V.P. Assessing the correlation between disease severity indices and quality of life measurement tools in pemphigus. Front. Immunol. 2019, 10, 2571. [Google Scholar] [CrossRef]
- Lopez-Jornet, P.; Zavattaro, E.; Mozaffari, H.R.; Ramezani, M.; Sadeghi, M. Evaluation of the Salivary Level of Cortisol in Patients with Oral Lichen Planus: A Meta-Analysis. Medicina 2019, 55, 213. [Google Scholar] [CrossRef]
| Causes of Desquamative Gingivitis | |
|---|---|
| Autoimmune diseases | Oral lichen planus ↑ |
| Cicatricial pemphigoid ↑ | |
| Mucous membrane pemphigoid ↑ | |
| Bullous pemphigoid | |
| Pemphigus vulgaris ↑ | |
| Paraneoplastic pemphigus | |
| Epidermolysis bullosa acquisita ↓ | |
| Lupus erythematosus | |
| Linear IgA disease | |
| Chronic ulcerative stomatitis | |
| Psoriasis ↓ | |
| Other systemic diseases | Erythema multiforme |
| Graft-versus-host disease | |
| Toxic epidermal necrolysis ↓ | |
| Pyostomatitis vegetans ↓ | |
| Irritant or allergic contact dermatitis- caused by | Mouthwash |
| Toothpaste | |
| Dental materials | |
| Medication | |
| Infections | Bacterial |
| Viral | |
| Fungal | |
| Hormonal imbalance | |
| Drug abuse | |
| Idiopathic | |
| Autoimmune Bullous Dermatoses | Target Antigen |
|---|---|
| Pemphigus vulgaris | Desmoglein 1, desmoglein 3, desmocollin 3 |
| Pemphigus foliaceus | Desmoglein 1 |
| Paraneoplastic pemphigus | Desmoplakin I (250 kD), bullous pemphigoid antigen I (230 kD), desmoplakin II (210 kD), envoplakin (210 kD), periplakin (190 kD), plectin (500 kD), and a 170 kD protein |
| Cicatricial pemphigoid | Laminin V/VI, integrin, collagen, Alpha-1 (XVII) collagen chain |
| Bullous pemphigoid | Alpha-1 (XVII) collagen chain (formerly known as BP180/BPAG2) and dystonine (formerly known as BP230/BPAG1) |
| Autoimmune Bullous Dermatoses | Differential Diagnosis |
|---|---|
| Pemphigus vulgaris | Bullous pemphigoid |
| Erythema multiforme | |
| Linear IgA dermatosis | |
| Pemphigus erythematosus | |
| Pemphigus foliaceus | |
| Paraneoplastic pemphigus | |
| Drug-induced pemphigus | |
| Cicatricial pemphigoid | |
| Bullous pemphigoid | Cicatricial pemphigoid |
| Herpetiform dermatitis | |
| Drug-induced bullous dermatitis | |
| Erythema multiforme | |
| Linear IgA dermatosis | |
| Bullous epidermolysis | |
| Cicatricial pemphigoid | Bullous pemphigoid |
| Epidermolysis bullosa | |
| Linear IgA dermatosis | |
| Pemphigus vulgaris | |
| Erosive lichen planus |
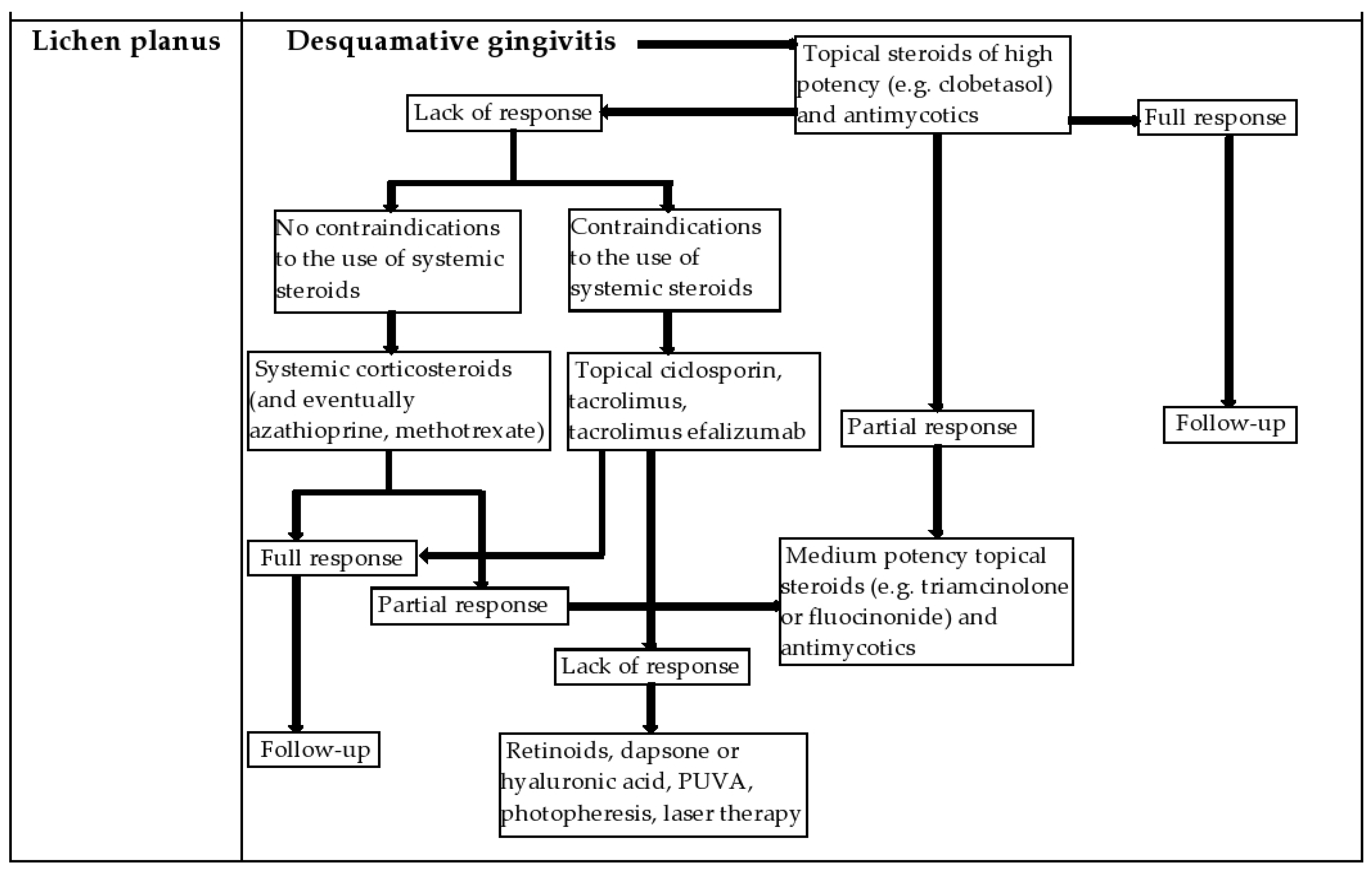
| Management of the Patient with Desquamative Gingivitis in the Dental Office: | |
|---|---|
|
|
| |
| |
| |
| |
| |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sciuca, A.M.; Toader, M.P.; Stelea, C.G.; Maftei, G.A.; Ciurcanu, O.E.; Stefanescu, O.M.; Onofrei, B.-A.; Popa, C. Desquamative Gingivitis in the Context of Autoimmune Bullous Dermatoses and Lichen Planus—Challenges in the Diagnosis and Treatment. Diagnostics 2022, 12, 1754. https://doi.org/10.3390/diagnostics12071754
Sciuca AM, Toader MP, Stelea CG, Maftei GA, Ciurcanu OE, Stefanescu OM, Onofrei B-A, Popa C. Desquamative Gingivitis in the Context of Autoimmune Bullous Dermatoses and Lichen Planus—Challenges in the Diagnosis and Treatment. Diagnostics. 2022; 12(7):1754. https://doi.org/10.3390/diagnostics12071754
Chicago/Turabian StyleSciuca, Ana Maria, Mihaela Paula Toader, Carmen Gabriela Stelea, George Alexandru Maftei, Oana Elena Ciurcanu, Ovidiu Mihail Stefanescu, Bianca-Andreea Onofrei, and Cristina Popa. 2022. "Desquamative Gingivitis in the Context of Autoimmune Bullous Dermatoses and Lichen Planus—Challenges in the Diagnosis and Treatment" Diagnostics 12, no. 7: 1754. https://doi.org/10.3390/diagnostics12071754
APA StyleSciuca, A. M., Toader, M. P., Stelea, C. G., Maftei, G. A., Ciurcanu, O. E., Stefanescu, O. M., Onofrei, B.-A., & Popa, C. (2022). Desquamative Gingivitis in the Context of Autoimmune Bullous Dermatoses and Lichen Planus—Challenges in the Diagnosis and Treatment. Diagnostics, 12(7), 1754. https://doi.org/10.3390/diagnostics12071754






