Left Ventricle Outflow Tract Velocity-Time Index and Right Ventricle to Left Ventricle Ratio as Predictors for in Hospital Outcome in Intermediate-Risk Pulmonary Embolism
Abstract
:1. Introduction
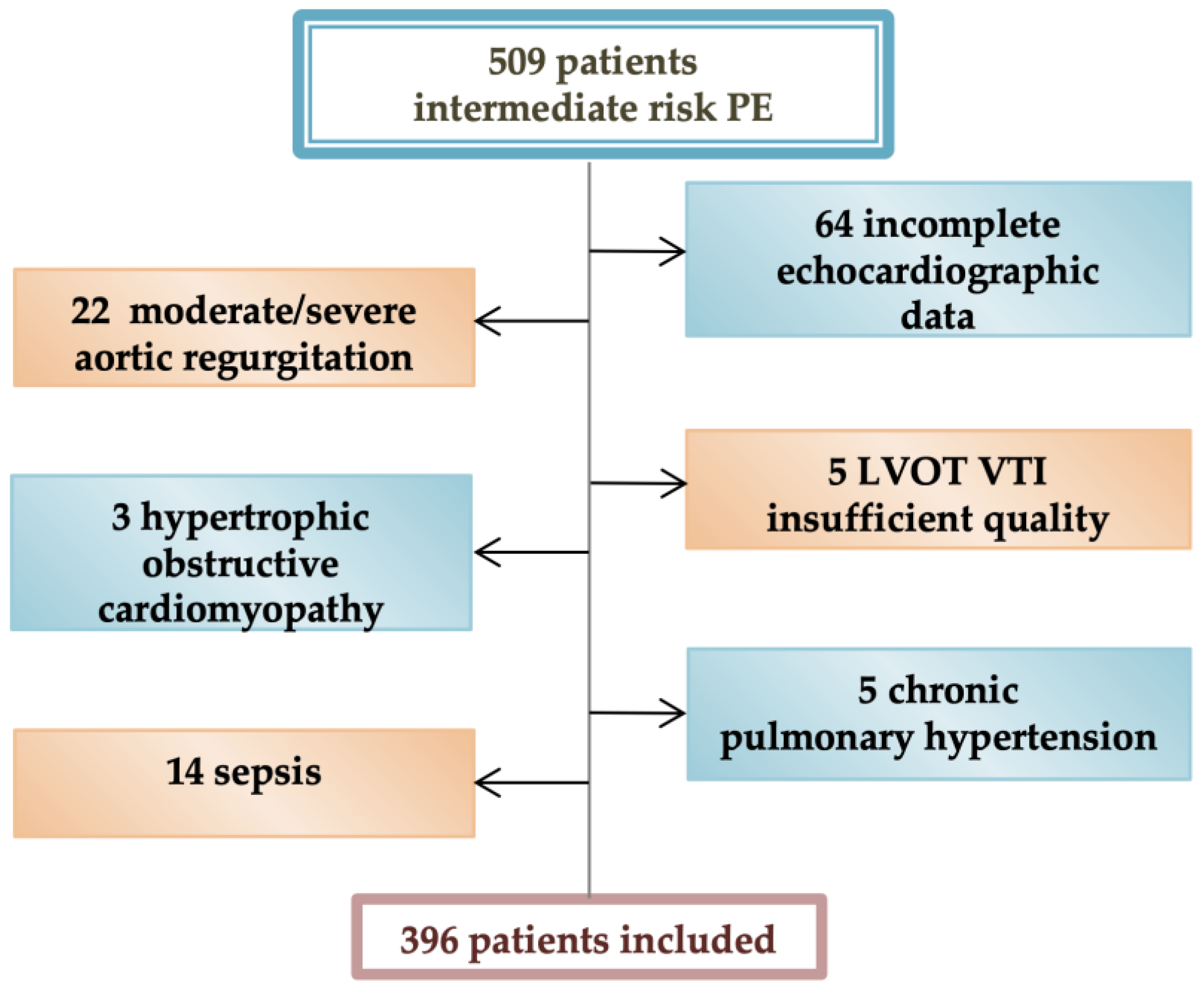
2. Materials and Methods
2.1. Study Design
2.2. Methodology
2.3. Statistical Analysis
3. Results
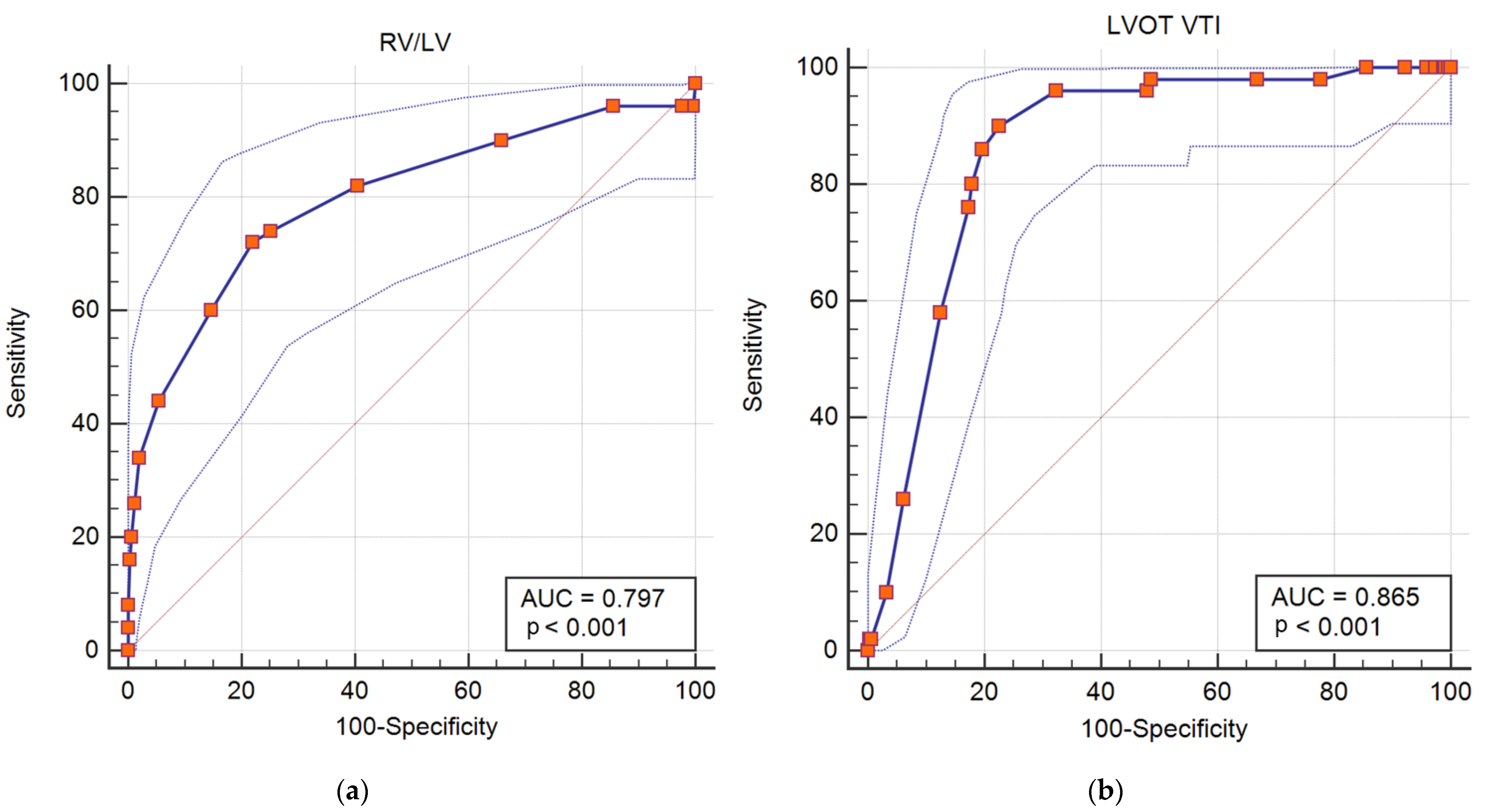
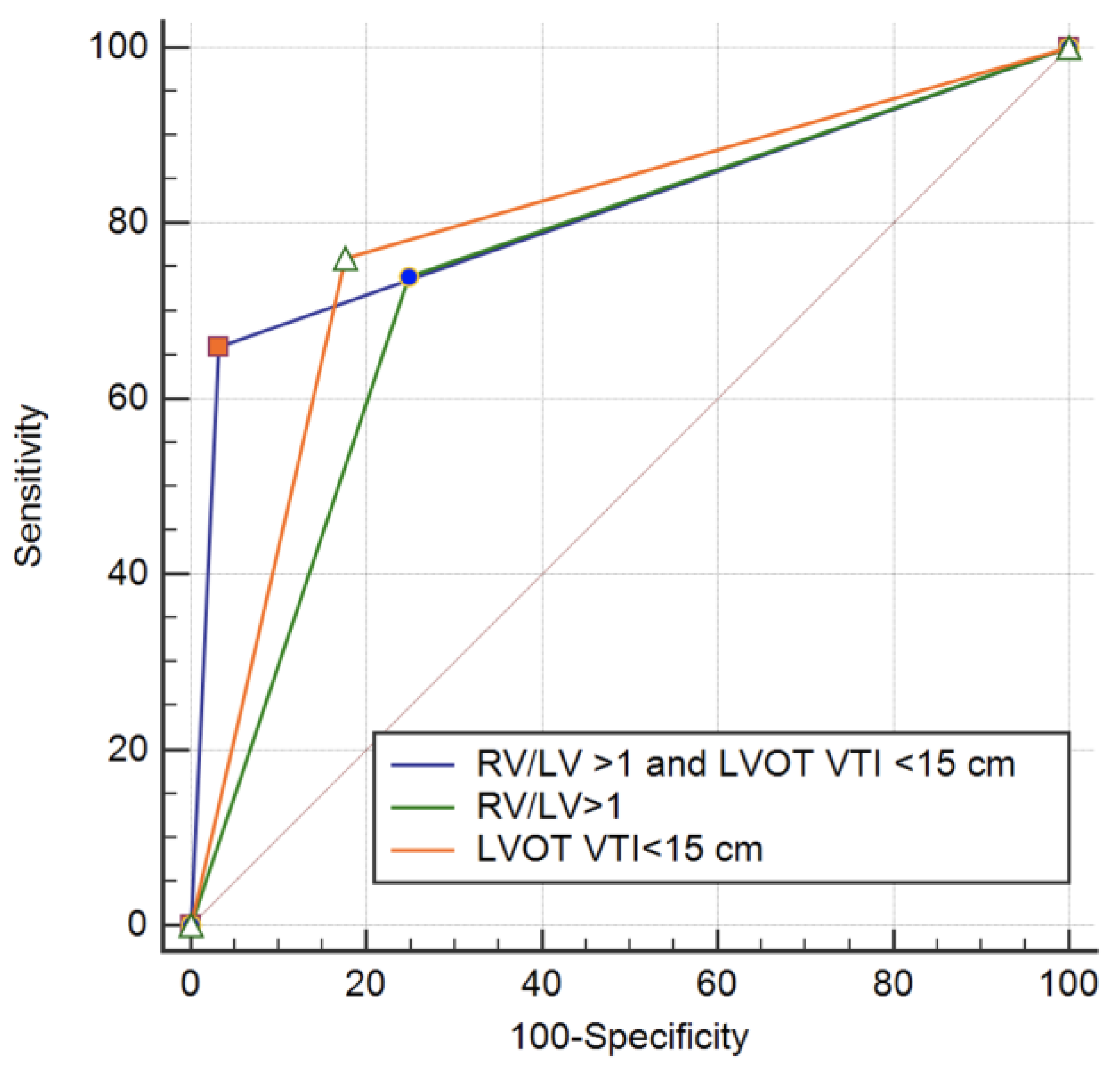
3.1. Predictors of in Hospital Adverse Outcomes: Death, Resuscitated Cardiac Arrest, Hemodynamic Instability and Need of Reperfusion Therapy
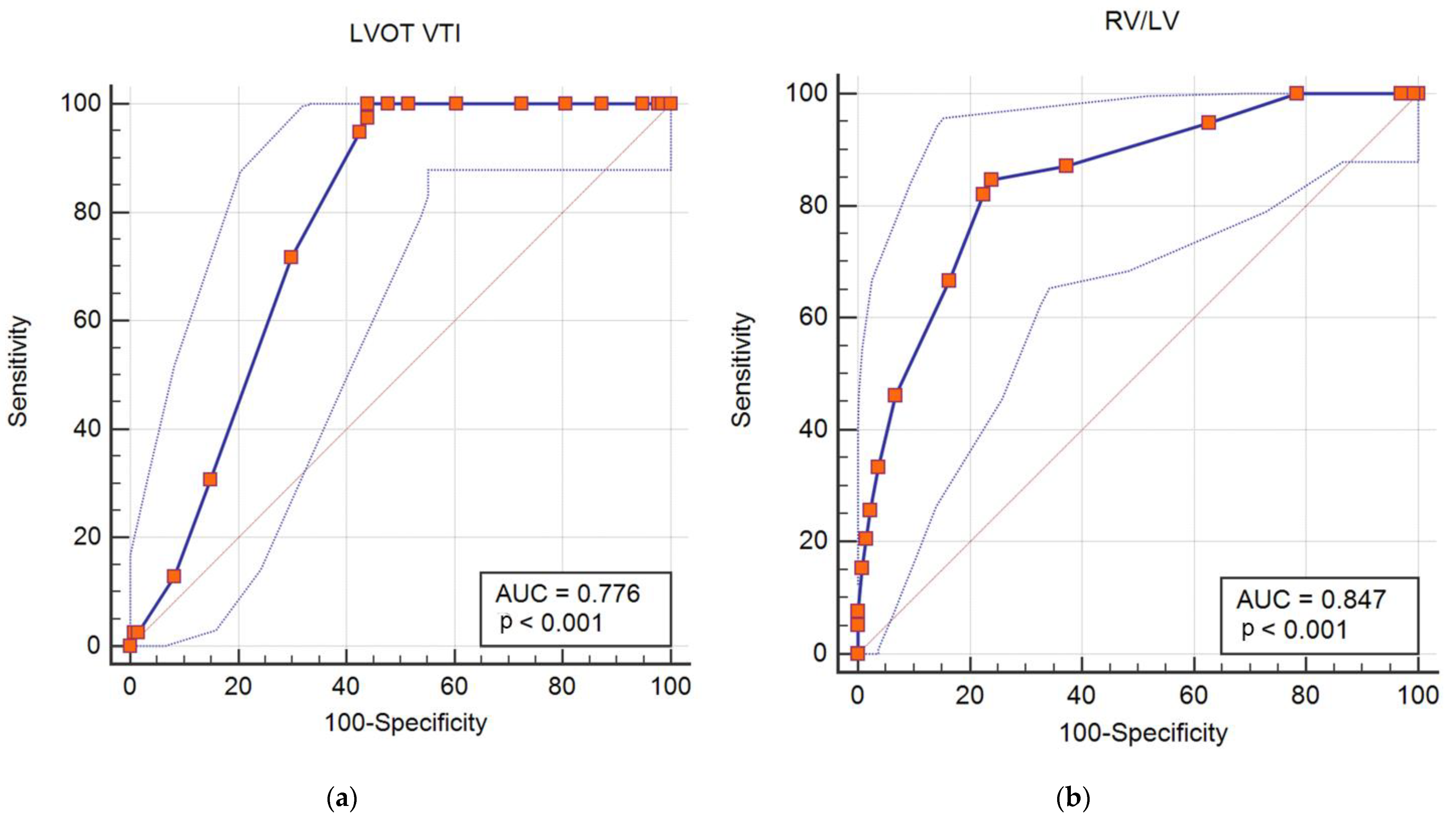
3.2. Predictors of in Hospital Death and Resuscitated Cardiac Arrest
3.3. Predictors of in Hospital Complications: Hemodynamic Instability or Need of Rescue Thrombolysis
3.4. LVOT VTI and RV/LV Ratio in Intermediate-High Risk Patients
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Keller, K.; Hobohm, L.; Ebner, M.; Kresoja, K.P.; Munzel, T.; Konstantinides, S.V.; Lankeit, M. Trends in thrombolytic treatment and outcomes of acute pulmonary embolism in Germany. Eur. Heart J. 2020, 41, 522–529. [Google Scholar] [CrossRef] [Green Version]
- De Miguel-Diez, J.; Jimenez-Garcia, R.; Jimenez, D.; Monreal, M.; Guijarro, R.; Otero, R.; Hernandez-Barrera, V.; Trujillo-Santos, J.; Lopez de Andres, A.; Carrasco-Garrido, P. Trends in hospital admissions for pulmonary embolism in Spain from 2002 to 2011. Eur. Respir. J. 2014, 44, 942–950. [Google Scholar] [CrossRef] [Green Version]
- Dentali, F.; Ageno, W.; Pomero, F.; Fenoglio, L.; Squizzato, A.; Bonzini, M. Time trends and case fatality rate of in-hospital treated pulmonary embolism during 11 years of observation in Northwestern Italy. Thromb. Haemost. 2016, 115, 399–405. [Google Scholar]
- Lehnert, P.; Lange, T.; Moller, C.H.; Olsen, P.S.; Carlsen, J. Acute pulmonary embolism in a national Danish cohort: Increasing incidence and decreasing mortality. Thromb. Haemost. 2018, 118, 539–546. [Google Scholar] [CrossRef]
- Pruszczyk, P.; Skowronska, M.; Ciurzynski, M.; Kurnica, K.; Lankeit, M.; Konstantinidis, S. Assessment of pulmonary embolism severity and the risk of early death. Pol. Arch. Intern. Med. 2021, 131, 16134. [Google Scholar] [CrossRef]
- Konstantinides, S.V.; Meyer, G.; Becattini, C.; Bueno, H.; Geersing, G.J.; Harjola, V.P.; Huisman, M.V.; Humbert, M.; Jennings, C.S.; Jimenez, D.; et al. 2019 ESC Guidelines for the diagnosis and management of acute pulmonary embolism developed in collaboration with the European Respiratory Society (ERS). Eur. Heart J. 2020, 41, 543–603. [Google Scholar] [CrossRef]
- Jiménez, D.; de Miguel-Díez, J.; Guijarro, R.; Trujillo-Santos, J.; Otero, R.; Barba, R.; Muriel, A.; Meyer, G.; Yusen, R.D.; Monreal, M.; et al. Trends in the management and outcomes of acute pulmonary embolism: Analysis from the RIETE Registry. J. Am. Coll. Cardiol. 2016, 67, 162–170. [Google Scholar] [CrossRef] [Green Version]
- Beckman, M.G.; Hooper, W.C.; Critchley, S.E.; Ortel, T.L. Venous thromboembolism: A public health concern. Am. J. Prev. Med. 2010, 38, S495–S501. [Google Scholar] [CrossRef]
- Alotaibi, G.S.; Wu, C.; Senthilselvan, A.; McMurtry, M.S. Secular trends in incidence and mortality of acute venous thromboembolism: The AB-VTE population-based study. Am. J. Med. 2016, 129, 879. [Google Scholar] [CrossRef]
- Kahn, S.R.; Hirsch, A.M.; Akaberi, A.; Hernandez, P.; Anderson, D.R.; Wells, P.S.; Rodger, M.A.; Solymoss, S.; Kovacs, M.J.; Rudski, L.; et al. Functional and exercise limitations after a first episode of pulmonary embolism: Results of the ELOPE prospective cohort study. Chest 2017, 151, 1058–1068. [Google Scholar] [CrossRef]
- Bloomer, T.L.; El-Hayek, G.E.; McDaniel, M.C.; Sandvall, B.C.; Liberman, H.A.; Devireddy, C.M.; Kumar, G.; Fong, P.P.; Jaber, W.A. Safety of catheter-directed thrombolysis for massive and sub massive pulmonary embolism: Results of a multicentre registry and meta-analysis. Catheter. Cardiovasc. Interv. 2017, 89, 754–760. [Google Scholar] [CrossRef]
- Babes, E.E.; Bustea, C.; Behl, T.; Abdel-Daim, M.M.; Nechifor, A.C.; Stoicescu, M.; Brisc, C.M.; Moisi, M.; Gitea, D.; Iovanovici, D.C.; et al. Acute coronary syndromes in diabetic patients, outcome, revascularization, and antithrombotic therapy. Biomed. Pharmacother. 2022, 148, 112772. [Google Scholar] [CrossRef]
- Chen, Y.L.; Wright, C.; Pietropaoli, A.P.; Elbadawi, A.; Delehanty, J.; Barrus, B.; Gosev, I.; Trawick, D.; Patel, D.; Cameron, S.J. Right ventricular dysfunction is superior and sufficient for risk stratification by a pulmonary embolism response team. J. Thromb. Thrombolysis 2020, 49, 34–41. [Google Scholar] [CrossRef]
- Becattini, C.; Maraziti, G.; Vinson, D.R.; Ng, A.C.C.; den Exter, P.L.; Côté, B.; Vanni, S.; Doukky, R.; Khemasuwan, K.; Weekes, A.J.; et al. Right ventricle assessment in patients with pulmonary embolism at low risk for death based on clinical models: An individual patient data meta-analysis. Eur. Heart J. 2021, 42, 3190–3199. [Google Scholar] [CrossRef]
- Piazza, G.; Goldhaber, S.Z. Management of sub massive pulmonary embolism. Circulation 2010, 122, 1124–1129. [Google Scholar] [CrossRef] [Green Version]
- Pruszczyk, P.; Goliszek, S.; Lichodziejewska, B.; Kostrubiec, M.; Ciurzyński, M.; Kurnicka, K.; Dzikowska-Diduch, O.; Palczewski, P.; Wyzgal, A. Prognostic value of echocardiography in normotensive patients with acute pulmonary embolism. JACC Cardiovasc. Imaging 2014, 7, 553–560. [Google Scholar] [CrossRef] [Green Version]
- Sanchez, O.; Trinquart, L.; Colombet, I.; Durieux, P.; Huisman, M.V.; Chatellier, G.; Meyer, G. Prognostic value of right ventricular dysfunction in patients with hemodynamically stable pulmonary embolism: A systematic review. Eur. Heart J. 2008, 29, 1569–1577. [Google Scholar] [CrossRef] [Green Version]
- Cho, J.H.; Sridharan, G.K.; Kim, S.H.; Kaw, R.; Abburi, T.; Irfan, A.; Kocheril, A.G. Right ventricular dysfunction as an echocardiographic prognostic factor in hemodynamically stable patients with acute pulmonary embolism: A meta-analysis. BMC Cardiovasc. Disord. 2014, 14, 64. [Google Scholar] [CrossRef]
- Lobo, J.L.; Holley, A.; Taoism, V.; Moores, L.; Oribe, M.; Barrón, M.; Otero, R.; Nauffal, D.; Valle, R.; Monreal, M.; et al. Prognostic significance of tricuspid annular displacement in normotensive patients with acute symptomatic pulmonary embolism. J. Thromb. Haemost. 2014, 12, 1020–1027. [Google Scholar] [CrossRef]
- McConnell, M.; Solomon, S.D.; Rayan, M.E.; Come, P.C.; Goldhaber, S.Z.; Leeet, R.T. Regional right ventricular dysfunction detected by echocardiography in acute pulmonary embolism. Am. J. Cardiol. 1996, 78, 469–473. [Google Scholar] [CrossRef]
- Ten Wolde, M.; Sohne, M.; Quak, E.; Mac Gilavry, M.R.; Bulller, H.R. Prognostic value of echocardiographically assessed right ventricular dysfunction in patients with pulmonary embolism. Arch. Intern. Med. 2004, 164, 1685–1689. [Google Scholar] [CrossRef] [Green Version]
- Coutance, G.; Cauderlier, E.; Ehtisham, J.; Hamon, M.; Hamon, M. The prognostic value of markers of right ventricular dysfunction in pulmonary embolism: A meta-analysis. Crit. Care 2011, 15, R103. [Google Scholar] [CrossRef] [Green Version]
- Alerhand, S.; Sundaram, T.; Gottlieb, M. What are the echocardiographic findings of acute right ventricular strain that suggest pulmonary embolism? Anaesth. Crit. Care Pain Med. 2021, 40, 100852. [Google Scholar] [CrossRef]
- Dahhan, T.; Siddiqui, I.; Tapson, V.F.; Velazquez, E.J.; Sun, S.; Davenport, C.A.; Samad, Z.; Rajagopal, S. Clinical and echocardiographic predictors of mortality in acute pulmonary embolism. Cardiovasc. Ultrasound 2016, 14, 44. [Google Scholar] [CrossRef] [Green Version]
- Burgos, L.M.; Scatularo, C.E.; Cigalini, I.M.; Jauregui, J.C.; Bernal, M.I.; Bonorino, J.M.; Thierer, J.; Zaidel, E.J. The addition of echocardiographic parameters to PESI score improves mortality prediction in patients with acute pulmonary embolism: PESI-Echo score. Eur. Heart J. Acute Cardiovasc. Care 2021, 10, 250–257. [Google Scholar] [CrossRef]
- Rodrigues, A.C.T.; Cordovil, A.; Monaco, C.G.; Guimares, L.A.B.; de Oliveira, W.A.A.; Fischer, C.H.; de Lira-Pilho, E.B.; Vieira, M.L.C.; Morhy, S.S. Assessing prognosis of pulmonary embolism using tissue-Doppler echocardiography and brain natriuretic peptide. Einstein 2013, 11, 338–344. [Google Scholar] [CrossRef]
- Yuriditsky, E.; Mitchell, O.J.; Sibley, R.A.; Xia, Y.; Sista, A.K.; Zhong, J.; Moore, W.H.; Amoroso, N.E.; Goldenberg, R.M.; Smith, D.E.; et al. Low left ventricular outflow tract velocity time integral is associated with poor outcomes in acute pulmonary embolism. Vasc. Med. 2020, 25, 133–140. [Google Scholar] [CrossRef]
- Tan, C.; Rubenson, D.; Srivastava, A.; Mohan, R.; Smith, M.R.; Billick, K.; Bardarian, S.; Heywood, J.T. Left ventricular outflow tract velocity time integral outperforms ejection fraction and Doppler-derived cardiac output for predicting outcomes in a select advanced heart failure cohort. Cardiovasc. Ultrasound 2017, 15, 18. [Google Scholar] [CrossRef]
- Betcher, J.; Majkrzac, A.; Cranford, J.; Kessler, R.; Theyyunni, N.; Huang, R. Feasibility study of advanced focused cardiac measurements within the emergency department. Crit. Ultrasound J. 2018, 10, 10. [Google Scholar] [CrossRef]
- Blanco, P. Rationale for using the velocity–time integral and the minute distance for assessing the stroke volume and cardiac output in point-of-care settings. Ultrasound J. 2020, 12, 21. [Google Scholar] [CrossRef] [Green Version]
- Chinen, D.; Fujino, M.; Anzai, T.; Kitakaze, M.; Goto, Y.; Ishihara, M.; Shimizu, W.; Yokoyama, H.; Yasuda, S.; Ogawa, H. Left ventricular outflow tract velocity time integral correlates with low cardiac output syndrome in patients with acute decompensated heart failure. Eur. Heart J. 2013, 34 (Suppl. 1), P4249. [Google Scholar] [CrossRef] [Green Version]
- IBM Corp. IBM SPSS Statistics for Windows; Version 25.0; IBM Corp.: Armonk, NY, USA, 2017. [Google Scholar]
- Dean, A.G.; Sullivan, K.M.; Soe, M.M. OpenEpi: Open Source Epidemiologic Statistics for Public Health, Version. 2013. Available online: http://www.openepi.com/Menu/OE_Menu.htm (accessed on 18 March 2022).
- Barco, S.; Mahmoudpour, S.; Planquette, B.; Sanchez, O.; Konstantinides, S.V.; Meyer, G. Prognostic value of right ventricular dysfunction or elevated cardiac biomarkers in patients with low-risk pulmonary embolism: A systematic review and meta-analysis. Eur. Heart J. 2019, 40, 902–910. [Google Scholar] [CrossRef] [Green Version]
- Meinel, F.G.; Nance, J.V., Jr.; Schoepf, U.J.; Hoffmann, V.S.; Thierfelder, K.M.; Costello, P.; Goldhaber, S.Z.; Bamberg, F. Predictive value of computed tomography in acute pulmonary embolism Systematic review and meta-analysis. Am. J. Med. 2015, 128, 747–759. [Google Scholar] [CrossRef]
- Meyer, G.; Vicaut, E.; Danays, T.; Agnelli, G.; Becattini, C.; Beyer-Westendorf, J.; Blumhki, E.; Bouvaist, H.; Brenner, B.; Couturaud, F.; et al. Fibrinolysis for patients with intermediate-risk pulmonary embolism. N. Engl. J. Med. 2014, 370, 1402–1411. [Google Scholar] [CrossRef]
- Pruszczyk, P.; Kurnicka, K.; Ciurzyński, M.; Hobohm, L.; Thielmann, A.; Sobkowicz, B.; Sawicka, E.; Kostrubiec, M.; Ptaszyńska-Kopczyńska, K.; Dzikowska-Diduch, O.; et al. Defining right ventricular dysfunction by echocardiography in normotensive patients with pulmonary embolism. Pol. Arch. Intern. Med. 2020, 130, 741–747. [Google Scholar] [CrossRef]
- Cho, S.-u.; Cho, Y.-d.; Choi, S.-h.; Yoon, Y.-h.; Park, J.-h.; Park, S.-j.; Lee, E.-s. Assessing the severity of pulmonary embolism among patients in the emergency department: Utility of RV/LV diameter ratio. PLoS ONE 2020, 15, e0242340. [Google Scholar] [CrossRef]
- Khemasuwan, D.; Yingchoncharoen, T.; Tunsupon, P.; Kusunose, K.; Moghekar, A.; Klein, A.; Tonelli, A.R. Right ventricular echocardiographic parameters are associated with mortality after acute pulmonary embolism. J. Am. Soc. Echocardiogr. 2015, 28, 355–362. [Google Scholar] [CrossRef]
- McLean, A.S. Echocardiography in shock management. Crit. Care 2016, 20, 275. [Google Scholar] [CrossRef] [Green Version]
- Blanco, P.; Aguiar, F.M.; Blaivas, M. Rapid Ultrasound in Shock (RUSH) Velocity-Time Integral: A proposal to expand the RUSH protocol. J. Ultrasound Med. 2015, 24, 1691–1700. [Google Scholar] [CrossRef]
- Bergenzaun, L.; Gudmundsson, P.; Öhlin, H.; During, J.; Ersson, A.; Ihrman, L.; Willenheimer, R.; Chew, M.S. Assessing left ventricular systolic function in shock: Evaluation of echocardiographic parameters in intensive care. Crit. Care 2011, 15, R200. [Google Scholar] [CrossRef] [Green Version]
- Dinh, V.A.; Ko, H.S.; Rao, R.; Bansal, R.C.; Smith, D.D.; Kim, T.E.; Nguyen, H.B. Measuring cardiac index with a focused cardiac ultrasound examination in the ED. Am. J. Emerg. Med. 2012, 30, 1845–1851. [Google Scholar] [CrossRef]
- McGregor, D.; Sharma, S.; Gupta, S.; Ahmad, S.; Godec, T.; Harris, T. Emergency department non-invasive cardiac output study (EDNICO): A feasibility and repeatability study. Scand. J. Trauma Resusc. Emerg. Med. 2019, 27, 30. [Google Scholar] [CrossRef]
- Villavicencio, C.; Leache, J.; Marin, J.; Oliva, I.; Rodriguez, A.; Bodí, M.; Soni, N.J. Basic critical care echocardiography training of intensivists allows reproducible and reliable measurements of cardiac output. Ultrasound J. 2019, 11, 5. [Google Scholar] [CrossRef]
- Prosperi-Porta, G.; Solverson, K.; Fine, N.; Humphreys, C.J.; Ferland, A.; Weatherald, J. Echocardiography-Derived Stroke Volume Index Is Associated with Adverse In-Hospital Outcomes in Intermediate-Risk Acute Pulmonary Embolism: A Retrospective Cohort Study. Chest 2020, 158, 1132–1142. [Google Scholar] [CrossRef]
- Falsetti, L.; Marra, A.M.; Zaccone, V.; Sampaolesi, M.; Riccomi, F.; Giovenali, L.; Guerrieri, E.; Viticchi, G.; D’Agostino, A.; Gentili, T.; et al. Echocardiographic predictors of mortality in intermediate-risk pulmonary embolism. Intern. Emerg. Med. 2022. [Google Scholar] [CrossRef]
- Kamran, H.; Hariri, E.H.; Iskandar, J.P.; Sahai, A.; Haddadin, I.; Harb, S.C.; Campbell, J.; Tefera, L.; Delehanty, J.M.; Heresi, G.A.; et al. Simultaneous Pulmonary Artery Pressure and Left Ventricle Stroke Volume Assessment Predicts Adverse Events in Patients with Pulmonary Embolism. J. Am. Heart Assoc. 2021, 10, e019849. [Google Scholar] [CrossRef]
- Antoine, D.; Chuich, T.; Mylvaganam, R.; Malaisrie, C.; Freed, B.; Cuttica, M.; Schimmel, D., Jr. Relationship of left ventricular outflow tract velocity time integral to treatment strategy in submassive and massive pulmonary embolism. Pulm. Circ. 2020, 10, 1–7. [Google Scholar] [CrossRef]
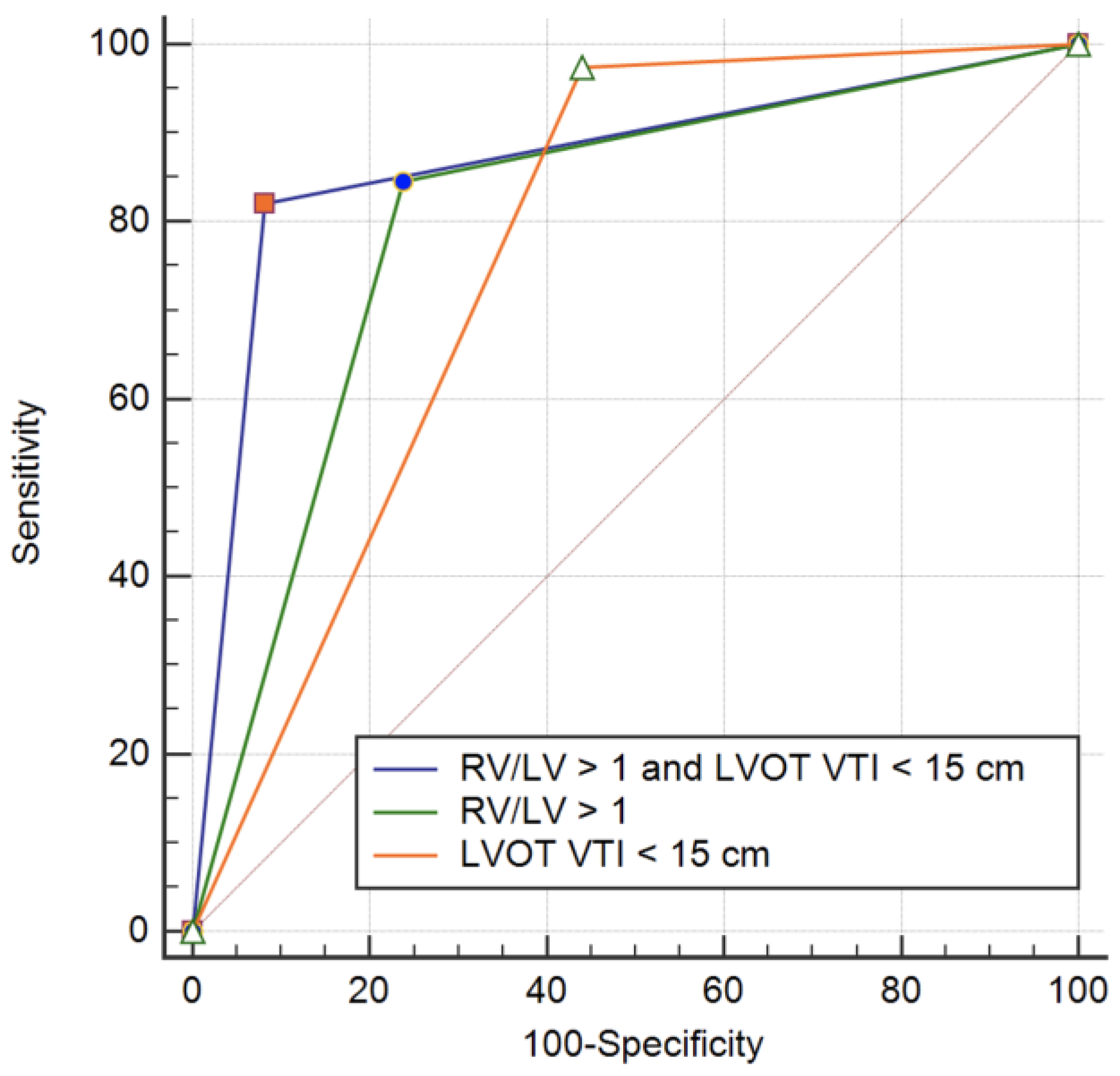
| Death, Resuscitated Cardiac Arrest, Hemodynamic Instability and Need of Reperfusion Therapy | p | |||
|---|---|---|---|---|
| Adverse Outcome | Total (396) | Yes (50) | No (346) | |
| Age (years) | 73.58 ± 11.221 | 71.22 ± 13.27 | 73.92 ± 10.87 | 0.09 |
| Sex (F) | 202/396 (51%) | 27/50 (54%) | 175/346 (50.57%) | 0.120 |
| Smoking | 134/396 (33.83%) | 15/50 (30%) | 119/346 (34.39%) | 0.26 |
| History of cancer | 67/396 (16.92%) | 8/50 (16%) | 59/346 (17.05%) | 0.628 |
| History of cardiovascular disease | 302/396 (76.3%) | 31/50 (62%) | 271/346 (78.32%) | 0.01 * |
| Heart failure | 132/396 (33.33%) | 14/50 (28%) | 118/346 (34.10%) | 0.19 |
| Coronary artery disease | 86/396 (21.71%) | 8/50 (16%) | 78/346 (22.54%) | 0.14 |
| Valvular heart disease | 114/396 (28.79%) | 10/50 (20%) | 104/346 (30.06%) | 0.07 |
| Hypertension | 210/396 (53.03%) | 20/50 (40%) | 190/346 (54.91%) | 0.02 * |
| History of pulmonary disease | 106/396 (26.8%) | 13/50 (26%) | 93/346 (26.88%) | 0.896 |
| Obstructive pulmonary disease | 79/396 (19.94%) | 10/50 (20%) | 69/346 (19.94%) | 0.49 |
| Restrictive pulmonary disease | 19/396 (4.80%) | 4/50 (8%) | 15/346 (4.33%) | 0.12 |
| Combined obstructive and restrictive pulmonary disease | 8/396 (2.02%) | 2/50 (4%) | 6/346 (1.73%) | 0.14 |
| Diabetes | 82/396 (20.7%) | 13/50 (26%) | 69/346 (19.94%) | 0.324 |
| History of stroke | 41/396 (10.35%) | 5/50 (10%) | 36/346 (10.40%) | 0.46 |
| History of recent surgery | 23/396 (5.80%) | 3/50 (6%) | 20/346 (5.78%) | 0.47 |
| History of recent orthopedic surgery or major trauma | 20/396 (5.05%) | 4/50 (8%) | 16/346 (4.62%) | 0.15 |
| Previous DVT/PE | 54/396 (13.63%) | 10/50 (20%) | 44/346 (12.72%) | 0.03 * |
| BMI (kg/m²) | 30.25 ± 6.73 | 32.87 ± 6.06 | 27.62 ± 6.69 | 0.12 |
| Systolic BP (mm Hg) | 126.85 ± 22.234 | 121.73 ± 21.94 | 128.25 ± 22.17 | 0.07 |
| HR | 91.571 ± 19.14 | 94.142 ± 19.86 | 89 ± 18.42 | 0.368 |
| DVT | 69/396 (17.42%) | 8/50 (16%) | 61/346 (17.63%) | 0.649 |
| Cholesterol (mg/dL) | 232.6 ± 66.61 | 251.40 ± 32.88 | 213.8 ± 89.55 | 0.06 |
| NT-proBNP (pg/mL) | 5267.354 ± 6991.84 | 7307.27 ± 9350.97 | 4975 ± 6583.97 | 0.187 |
| Hs-Troponin (pg/mL) | 306.66 ± 579.82 | 711.054 ± 596.66 | 193.32 ± 523.62 | <0.001 * |
| Death, Resuscitated Cardiac Arrest, Hemodynamic Instability and Need of Reperfusion Therapy | p | |||
|---|---|---|---|---|
| Adverse Outcome | Total (396) | Yes (50) | No (346) | |
| TAPSE < 16 mm | 116/396 (29.29%) | 23/50 (46%) | 93/346 (26.88%) | 0.022 * |
| TAPSE (mm) | 20.35 ± 6.52 | 17.68 ± 5.93 | 20.76 ± 6.51 | 0.001 * |
| Paradoxical movement or flattening of IVS | 113/396 (28.54%) | 20/50 (40%) | 93/346 (26.88%) | 0.057 |
| PSAP (mmHg) | 40.38 ± 12.202 | 43.66 ± 14.52 | 39.99 ± 11.808 | 0.129 |
| Mc Connell sign | 124/396 (31.31%) | 19/50 (38%) | 105/346 (30.34%) | 0.283 |
| RV/LV ≥ 1 | 123/396 (31.1%) | 36/50 (72%) | 87/346 (25.14%) | <0.001 * |
| RV/LV | 0.93 ± 0.31 | 1.28 ± 0.45 | 0.884 ± 0.26 | <0.001 * |
| LVOT VTI (cm) | 18.87 ± 4.38 | 13.88 ± 2.58 | 19.60 ± 4.10 | <0.001 * |
| LVOT VTI ≤ 15 cm | 99/396 (25%) | 38/50 (76%) | 61/346 (17.63%) | <0.001 * |
| LVEF (%) | 47.60 ± 8.13 | 48.25 ± 8.86 | 47.51 ± 8.06 | 0.610 |
| IVC (mm) | 21.94 ± 8.07 | 23.18 ± 7.97 | 20.25 ± 8.44 | 0.451 |
| Death or Resuscitated Cardiac Arrest | Yes (23) | No (373) | p |
|---|---|---|---|
| Age (years) | 73.00 ± 15.97 | 73.61 ± 10.890 | 0.799 |
| Sex (F) | 13/23 (56.52%) | 189/373 (50.67%) | 0.001 * |
| Smoking | 7/23 (30.43%) | 127/373 (34%) | 0.36 |
| History of cancer | 4/23 (17.39%) | 63/373 (16.89%) | 0.928 |
| History of cardiovascular disease | 14/23 (60.87%) | 288/373 (77.21%) | 0.797 |
| Heart failure | 8/23 (34.78%) | 124/373 (33.24%) | 0.43 |
| Coronary artery disease | 5/23 (21.74%) | 81/373 (21.72%) | 0.49 |
| Valvular heart disease | 6/23 (26.08%) | 108/373 (28.95%) | 0.38 |
| Hypertension | 11/23 (47.82%) | 199/373 (53.35%) | 0.3 |
| History of pulmonary disease | 5/23 (21.74%) | 101/373 (27.08%) | 0.676 |
| Obstructive pulmonary disease | 3/23 (13.04%) | 76/373 (20.38%) | 0.41 |
| Restrictive pulmonary disease | 1/23 (4.35%) | 18/373 (4.83%) | 0.45 |
| Combined obstructive and restrictive pulmonary disease | 1/23 (4.35%) | 7/373 (1.88%) | 0.20 |
| Diabetes | 6/23 (26.09%) | 76/373 (20.38%) | 0.513 |
| History of stroke | 4/23 (17.39%) | 37/373 (9.92%) | 0.12 |
| History of recent surgery | 1/23 (4.34%) | 22/373 (5.90%) | 0.37 |
| History of recent orthopedic surgery or major trauma | 2/23 (8.69%) | 18/373 (4.83%) | 0.20 |
| Previous DVT/PE | 4/23 (17.39%) | 50/373 (13.40%) | 0.13 |
| BMI (kg/m²) | 33.00 ± 5.87 | 28.60 ± 6.96 | 0.20 |
| Systolic BP (mmHg) | 115.83 ± 16.88 | 128.18 ± 22.4 | 0.002 * |
| HR | 96.2 ± 10.59 | 87.5 ± 16.46 | 0.521 |
| DVT | 1/23 (4.34%) | 68/373 (18.23%) | 0.08/0.007 * |
| Cholesterol (mg/dL) | 249 ± 45.17 | 225.57 ± 76.05 | 0.56 |
| NT-proBNP (pg/mL) | 11,029.0 ± 11,349.29 | 4883.24 ± 6488.53 | 0.01 * |
| Hs-Troponin (pg/mL) | 814.70 ± 718.55 | 238.475 ± 525.151 | <0.001 * |
| Death or Resuscitated Cardiac Arrest | Yes (23) | No (373) | p |
|---|---|---|---|
| TAPSE < 16 mm | 9/23 (39.13%) | 107/373 (28.68%) | 0.431 |
| TAPSE (mm) | 19.17 ± 6.27 | 20.43 ± 6.53 | 0.36 |
| Paradoxical movement or flattening of IVS | 11/23 (47.82%) | 102/373 (27.34%) | 0.036 * |
| PSAP (mmHg) | 45.94 ± 16.213 | 39.98 ± 11.808 | 0.059 |
| McConnel sign | 10/23 (43.47%) | 114/373 (30.56%) | 0.07 |
| RV/LV ≥ 1 | 15/23 (65.22%) | 108/373 (28.95%) | 0.001 * |
| RV/LV | 1.2 ± 0.44 | 0.92 ± 0.303 | <0.001 * |
| LVOT VTI (cm) | 13.65 ± 2.75 | 19.19 ± 4.25 | <0.001 * |
| LVOT VTI ≤ 15 cm | 20/23 (86.96%) | 79/373 (21.18%) | <0.001 * |
| LVEF (%) | 47.93 ± 11.472 | 47.59 ± 7.96 | 0.879 |
| IVC (mm) | 24.12 ± 6.75 | 20.36 ± 8.90 | 0.33 |
| Hemodynamic Instability or Reperfusion Therapy | Yes (27) | No (369) | p |
|---|---|---|---|
| Age (years) | 69.70 ± 10.550 | 73.86 ± 11.229 | 0.064 |
| Sex (F) | 13/27 (48.15%) | 189/369 (51.22%) | 0.618 |
| Smoking | 8/279 (9.63%) | 126/369 (34.14%) | 0.31 |
| History of cancer | 4/27 (14.81%) | 63/369 (17.07%) | 0.463 |
| History of cardiovascular disease | 17/27 (62.96%) | 285/369 (77.24%) | 0.095 |
| Heart failure | 6/27 (22.22%) | 126/369 (31.81%) | 0.22 |
| Coronary artery disease | 3/27 (11.11%) | 83/369 (22.49%) | 0.14 |
| Valvular heart disease | 4/27 (14.81%) | 110/369 (29.81%) | 0.10 |
| Hypertension | 9/27 (33.33%) | 201/369 (54.47%) | 0.08 |
| History of pulmonary disease | 8/27 (29.63%) | 98/369 (26.56%) | 0.729 |
| Obstructive pulmonary disease | 5/27 (18.52%) | 74/369 (20.05%) | 0.42 |
| Restrictive pulmonary disease | 2/27 (7.41%) | 17/369 (4.61%) | 0.27 |
| Combined obstructive and restrictive pulmonary disease | 1/27 (3.70%) | 7/369 (1.89%) | 0.25 |
| Diabetes | 7/27 (25.93%) | 75/369 (20.33%) | 0.489 |
| History of stroke | 1/27 (3.70%) | 40/369 (10.84%) | 0.12 |
| History of recent surgery | 2/27 (7.41%) | 21/369 (5.69%) | 0.35 |
| History of recent orthopedic surgery or major trauma | 2/27 (7.41%) | 18/369 (4.87%) | 0.28 |
| Previous DVT/PE | 6/27 (22.22%) | 48/369 (13%) | 0.08 |
| BMI (kg/m²) | 255 ± 14.14 | 227 ± 74.14 | 0.34 |
| Systolic BP (mm Hg) | 127.69 ± 24.382 | 126.74 ± 22.006 | 0.837 |
| HR | 96.2 ± 10.59 | 87.5 ± 16.46 | 0.521 |
| DVT | 7/27 (25.93%) | 62/369(16.80%) | 0.19 |
| Cholesterol (mg/dL) | 255 ± 15.14 | 227 ± 74.14 | 0.15 |
| NT-proBNP (pg/mL) | 3585 ± 5083 | 5377 ± 7102 | 0.459 |
| Hs-Troponin (pg/mL) | 588.02 ± 400.018 | 272. 67 ± 590.27 | 0.007 * |
| Hemodynamic Instability or Reperfusion Therapy | Yes (27) | No (369) | p |
|---|---|---|---|
| TAPSE < 16 mm | 14/27 (51.85%) | 102/369 (27.64%) | 0.013 * |
| TAPSE (mm) | 16.40 ± 5.43 | 20.66 ± 6.50 | 0.001 * |
| Paradoxical movement or flattening of IVS | 9/27 (33.33%) | 104/369 (28.18%) | 0.57 |
| PSAP (mm Hg) | 40.85 ± 12.164 | 40.35 ± 12.23 | 0.889 |
| Mc Connell sign | 9/27 (33.33%) | 115/369 (31.17%) | 0.82 |
| RV/LV ≥ 1 | 22/27 (81.48%) | 101/369 (27.37%) | <0.001 * |
| RV/LV | 1.34 ± 0.45 | 0.90 ± 0.29 | <0.001 * |
| LVOT VTI (cm) | 13.96 ± 2.38 | 19.23 ± 4.27 | <0.001 * |
| LVOT VTI ≤ 15 cm | 18/27 (66.66%) | 81/369 (21.95%) | <0.001 * |
| LVEF (%) | 48.45 ± 7.015 | 47.54 ± 8.21 | 0.612 |
| IVC (mm) | 20.66 ± 12.05 | 22.19 ± 7.65 | 0.77 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Babes, E.E.; Stoicescu, M.; Bungau, S.G.; Uivarosan, D.; Tit, D.M.; Toma, M.M.; Bungau, A.F.; Bustea, C. Left Ventricle Outflow Tract Velocity-Time Index and Right Ventricle to Left Ventricle Ratio as Predictors for in Hospital Outcome in Intermediate-Risk Pulmonary Embolism. Diagnostics 2022, 12, 1226. https://doi.org/10.3390/diagnostics12051226
Babes EE, Stoicescu M, Bungau SG, Uivarosan D, Tit DM, Toma MM, Bungau AF, Bustea C. Left Ventricle Outflow Tract Velocity-Time Index and Right Ventricle to Left Ventricle Ratio as Predictors for in Hospital Outcome in Intermediate-Risk Pulmonary Embolism. Diagnostics. 2022; 12(5):1226. https://doi.org/10.3390/diagnostics12051226
Chicago/Turabian StyleBabes, Elena Emilia, Manuela Stoicescu, Simona Gabriela Bungau, Diana Uivarosan, Delia Mirela Tit, Mirela Marioara Toma, Alexa Florina Bungau, and Cristiana Bustea. 2022. "Left Ventricle Outflow Tract Velocity-Time Index and Right Ventricle to Left Ventricle Ratio as Predictors for in Hospital Outcome in Intermediate-Risk Pulmonary Embolism" Diagnostics 12, no. 5: 1226. https://doi.org/10.3390/diagnostics12051226
APA StyleBabes, E. E., Stoicescu, M., Bungau, S. G., Uivarosan, D., Tit, D. M., Toma, M. M., Bungau, A. F., & Bustea, C. (2022). Left Ventricle Outflow Tract Velocity-Time Index and Right Ventricle to Left Ventricle Ratio as Predictors for in Hospital Outcome in Intermediate-Risk Pulmonary Embolism. Diagnostics, 12(5), 1226. https://doi.org/10.3390/diagnostics12051226









