Complications after Thermal Ablation of Hepatocellular Carcinoma and Liver Metastases: Imaging Findings
Abstract
:1. Introduction
2. Vascular Complications
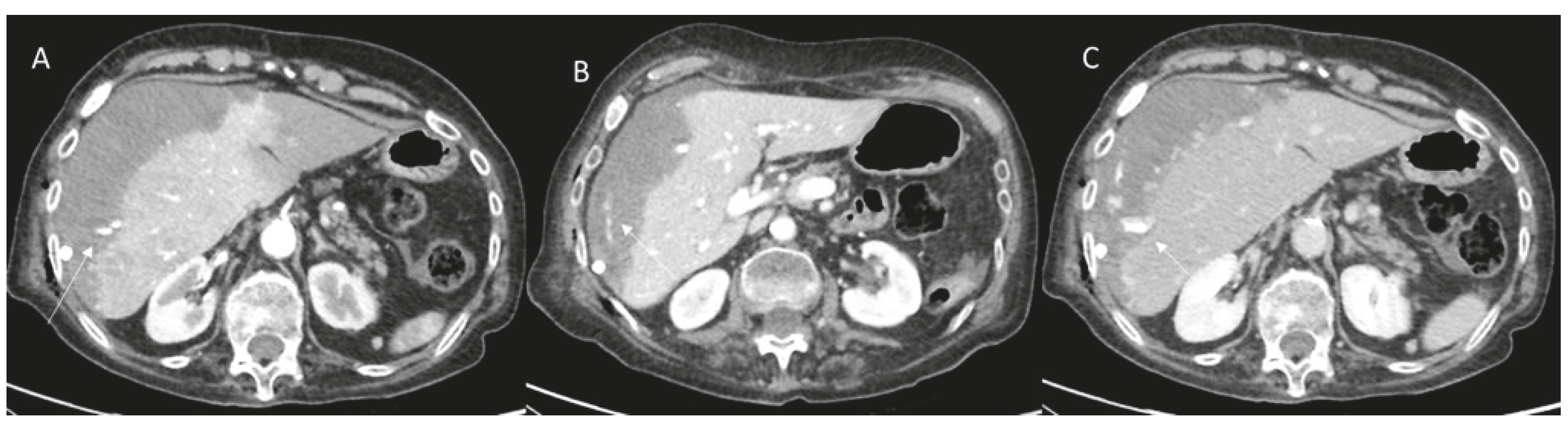
2.1. Bleeding
2.2. Vascular Thromboses
2.3. Pseudoaneurysm
3. Biliary Complications
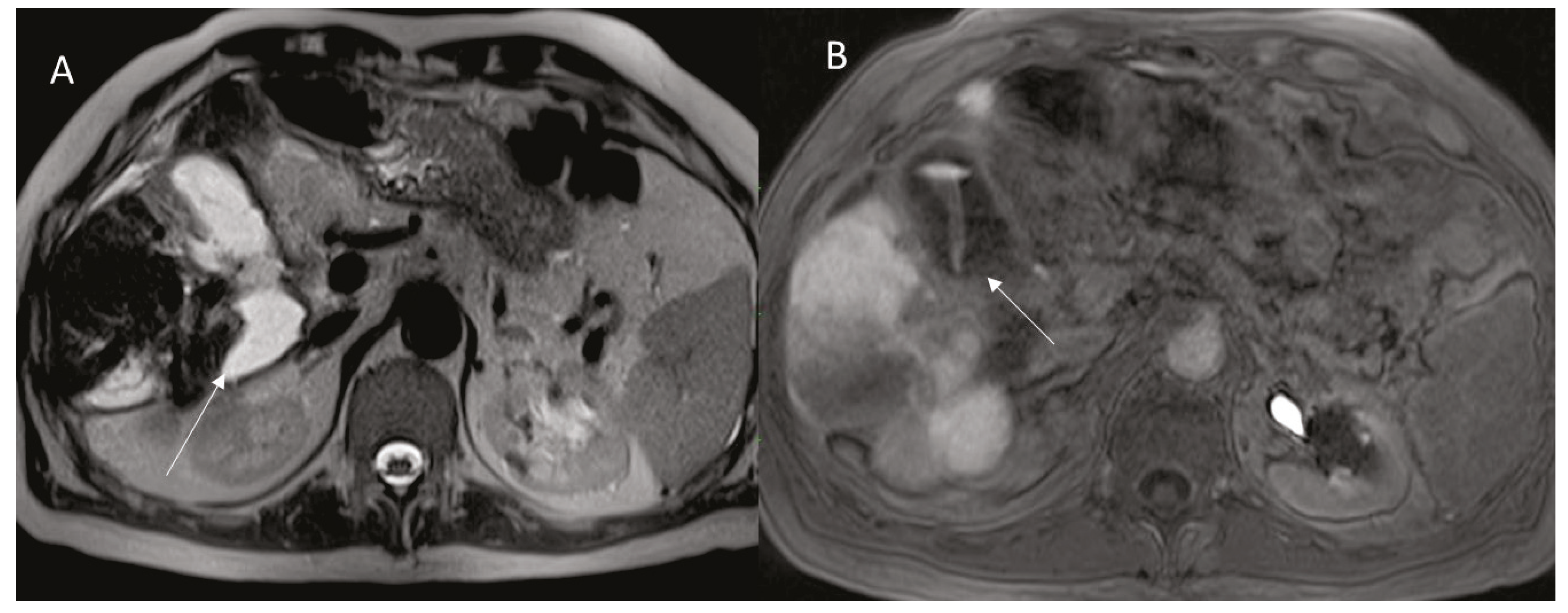
3.1. Ductal Stenosis and Biloma
3.2. Hemobilia
3.3. Cholecystitis
4. Infectious Complications
Abscess
5. Extrahepatic Complications
Gastro Intestinal Tract Injuries
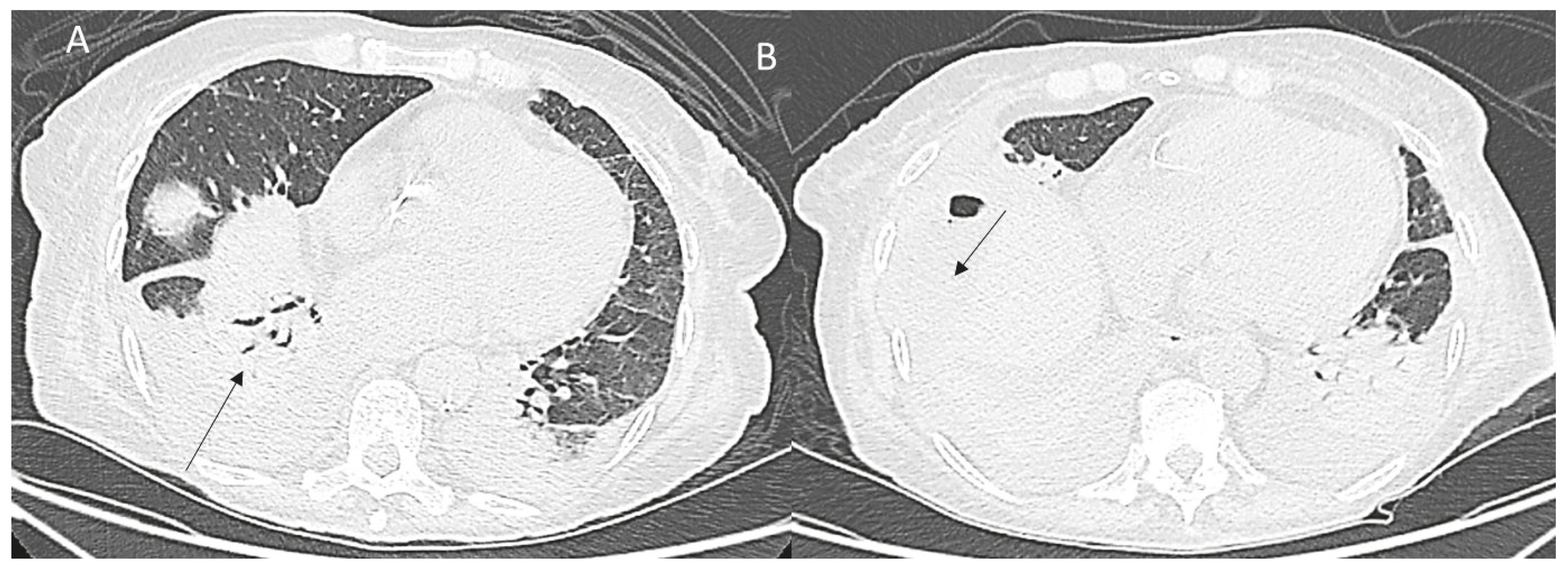
6. Thoracic Complications
7. Tumor Seeding
8. Fistulae
9. Conclusions
Funding
Acknowledgments
Conflicts of Interest
References
- Granata, V.; Grassi, R.; Fusco, R.; Belli, A.; Cutolo, C.; Pradella, S.; Grazzini, G.; La Porta, M.; Brunese, M.C.; De Muzio, F.; et al. Diagnostic evaluation and ablation treatments assessment in hepatocellular carcinoma. Infect. Agents Cancer 2021, 16, 53. [Google Scholar] [CrossRef]
- Nakamura, Y.; Higaki, T.; Honda, Y.; Tatsugami, F.; Tani, C.; Fukumoto, W.; Narita, K.; Kondo, S.; Akagi, M.; Awai, K. Advanced CT techniques for assessing hepatocellular carcinoma. Radiol. Med. 2021, 126, 925–935. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Cao, B.; Bi, X.; Chen, W.; Wang, L.; Du, Z.; Zhang, X.; Yu, X. Evaluation of liver function in patients with chronic hepatitis B using Gd-EOB-DTPA-enhanced T1 mapping at different acquisition time points: A feasibility study. Radiol. Med. 2021, 126, 1149–1158. [Google Scholar] [CrossRef] [PubMed]
- Esposito, A.; Buscarino, V.; Raciti, D.; Casiraghi, E.; Manini, M.; Biondetti, P.; Forzenigo, L. Characterization of liver nodules in patients with chronic liver disease by MRI: Performance of the Liver Imaging Reporting and Data System (LI-RADS v.2018) scale and its comparison with the Likert scale. Radiol. Med. 2020, 125, 15–23. [Google Scholar] [CrossRef] [PubMed]
- Izzo, F.; Granata, V.; Grassi, R.; Fusco, R.; Palaia, R.; Delrio, P.; Carrafiello, G.; Azoulay, D.; Petrillo, A.; Curley, A.S. Radiofrequency Ablation and Microwave Ablation in Liver Tumors: An Update. Oncologist 2019, 24, e990–e1005. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Knavel, E.M.; Brace, C.L. Tumor ablation: Common Modalities and General Practices. Tech. Vasc. Interv. Radiol. 2013, 16, 192–200. [Google Scholar] [CrossRef] [Green Version]
- Brace, C.L. Radiofrequency and Microwave Ablation of the Liver, Lung, Kidney, and Bone: What Are the Differences? Curr. Probl. Diagn. Radiol. 2009, 38, 135–143. [Google Scholar] [CrossRef] [Green Version]
- Granata, V.; Grassi, R.; Fusco, R.; Setola, S.V.; Palaia, R.; Belli, A.; Miele, V.; Brunese, L.; Petrillo, A.; Izzo, F. Assessment of Ablation Therapy in Pancreatic Cancer: The Radiologist’s Challenge. Front. Oncol. 2020, 10, 560952. [Google Scholar] [CrossRef]
- Granata, V.; Fusco, R.; Setola, S.V.; Piccirillo, M.; Leongito, M.; Palaia, R.; Granata, F.; Lastoria, S.; Izzo, F.; Petrillo, A. Early radiological assessment of locally advanced pancreatic cancer treated with electrochemotherapy. World J. Gastroenterol. 2017, 23, 4767–4778. [Google Scholar] [CrossRef]
- Granata, V.; de Lutio di Castelguidone, E.; Fusco, R.; Catalano, O.; Piccirillo, M.; Palaia, R.; Izzo, F.; Gallipoli, A.D.; Petrillo, A. Irreversible electroporation of hepatocellular carcinoma: Preliminary report on the diagnostic accuracy of magnetic resonance, computer tomography, and contrast-enhanced ultrasound in evaluation of the ablated area. Radiol. Med. 2016, 121, 122–131. [Google Scholar] [CrossRef]
- Granata, V.; Grassi, R.; Fusco, R.; Belli, A.; Palaia, R.; Carrafiello, G.; Miele, V.; Petrillo, A.; Izzo, F. Local ablation of pancreatic tumors: State of the art and future perspectives. World J. Gastroenterol. 2021, 27, 3413–3428. [Google Scholar] [CrossRef] [PubMed]
- Granata, V.; Fusco, R.; Barretta, M.L.; Picone, C.; Avallone, A.; Belli, A.; Patrone, R.; Ferrante, M.; Cozzi, D.; Grassi, R.; et al. Radiomics in hepatic metastasis by colorectal cancer. Infect. Agents Cancer 2021, 16, 39. [Google Scholar] [CrossRef] [PubMed]
- Granata, V.; Bicchierai, G.; Fusco, R.; Cozzi, D.; Grazzini, G.; Danti, G.; De Muzio, F.; Maggialetti, N.; Smorchkova, O.; D’Elia, M.; et al. Diagnostic protocols in oncology: Workup and treatment planning. Part 2: Abbreviated MR protocol. Eur. Rev. Med. Pharmacol. Sci. 2021, 25, 6499–6528. [Google Scholar] [CrossRef] [PubMed]
- Granata, V.; Fusco, R.; Petrillo, A. Additional Considerations on Use of Abbreviated Liver MRI in Patients with Colorectal Liver Metastases. AJR Am. J. Roentgenol. 2021, 217, W1. [Google Scholar] [CrossRef] [PubMed]
- Granata, V.; Fusco, R.; Avallone, A.; Catalano, O.; Piccirillo, M.; Palaia, R.; Nasti, G.; Petrillo, A.; Izzo, F. A radiologist’s point of view in the presurgical and intraoperative setting of colorectal liver metastases. Future Oncol. 2018, 14, 2189–2206. [Google Scholar] [CrossRef] [PubMed]
- Fiorentino, A.; Gregucci, F.; Bonaparte, I.; Vitulano, N.; Surgo, A.; Mazzola, R.; Di Monaco, A.; Carbonara, R.; Alongi, F.; Langialonga, T.; et al. Stereotactic Ablative radiation therapy (SABR) for cardiac arrhythmia: A new therapeutic option? Radiol. Med. 2021, 126, 155–162. [Google Scholar] [CrossRef] [PubMed]
- Fersino, S.; Borghesi, S.; Jereczek-Fossa, B.A.; Arcangeli, S.; Mortellaro, G.; Magrini, S.M.; Alongi, F.; Uro-Oncology Study Group of Italian Association of Radiotherapy and Clinical Oncology (AIRO). PROACTA: A survey on the actual attitude of the Italian radiation oncologists in the management and prescription of hormonal therapy in prostate cancer patients. Radiol. Med. 2021, 126, 460–465. [Google Scholar] [CrossRef]
- Arrigoni, F.; Bianchi, G.; Formiconi, F.; Palumbo, P.; Zugaro, L.; Gravina, G.L.; Barile, A.; Masciocchi, C. CT-guided cryoablation for management of bone metastases: A single center experience and review of the literature. Radiol. Med. 2022, 127, 199–205. [Google Scholar] [CrossRef]
- Goldberg, S.N.; Charboneau, J.W.; Dodd, G.D., 3rd; Dupuy, D.; Gervais, D.A.; Gillams, A.R.; Kane, R.A.; Lee, F.T.; Livraghi, T.; McGahan, J.P.; et al. Image-guided tumor ablation: Proposal for standardization of terms and reporting criteria. Radiology 2003, 228, 335–345. [Google Scholar] [CrossRef]
- Ahmed, M.; Solbiati, L.; Brace, C.L.; Breen, D.J.; Callstrom, M.R.; Charboneau, J.W.; Chen, M.-H.; Choi, B.I.; De Baère, T.; Dodd, G.D., 3rd; et al. Image-guided tumor ablation: Standardization of terminology and reporting criteria—A 10-year update. Radiology 2014, 273, 241–260. [Google Scholar] [CrossRef]
- Granata, V.; Grassi, R.; Fusco, R.; Setola, S.V.; Belli, A.; Ottaiano, A.; Nasti, G.; La Porta, M.; Danti, G.; Cappabianca, S.; et al. Intrahepatic cholangiocarcinoma and its differential diagnosis at MRI: How radiologist should assess MR features. Radiol. Med. 2021, 126, 1584–1600. [Google Scholar] [CrossRef] [PubMed]
- Arrigoni, F.; Bruno, F.; Gianneramo, C.; Palumbo, P.; Zugaro, L.; Zoccali, C.; Barile, A.; Masciocchi, C. Evolution of the imaging features of osteoid osteoma treated with RFA or MRgFUS during a long-term follow-up: A pictorial review with clinical correlations. Radiol. Med. 2020, 125, 578–584. [Google Scholar] [CrossRef] [PubMed]
- Ippolito, D.; Giandola, T.; Maino, C.; Pecorelli, A.; Capodaglio, C.; Ragusi, M.; Porta, M.; Gandola, D.; Masetto, A.; Drago, S.; et al. Acute pulmonary embolism in hospitalized patients with SARS-CoV-2-related pneumonia: Multicentric experience from Italian endemic area. Radiol Med. 2021, 126, 669–678. [Google Scholar] [CrossRef] [PubMed]
- Mathy, R.M.; Fritz, F.; Mayer, P.; Klauss, M.; Grenacher, L.; Stiller, W.; Kauczor, H.U.; Skornitzke, S. Iodine concentration and tissue attenuation in dual-energy contrast-enhanced CT as a potential quantitative parameter in early detection of local pancreatic carcinoma recurrence after surgical resection. Eur. J. Radiol. 2021, 143, 109944. [Google Scholar] [CrossRef]
- Karmazanovsky, G.; Gruzdev, I.; Tikhonova, V.; Kondratyev, E.; Revishvili, A. Computed tomography-based radiomics approach in pancreatic tumors characterization. Radiol. Med. 2021, 126, 1388–1395. [Google Scholar] [CrossRef]
- Sun, J.; Li, H.; Gao, J.; Li, J.; Li, M.; Zhou, Z.; Peng, Y. Performance evaluation of a deep learning image reconstruction (DLIR) algorithm in “double low” chest CTA in children: A feasibility study. Radiol. Med. 2021, 126, 1181–1188. [Google Scholar] [CrossRef]
- Palmisano, A.; Darvizeh, F.; Cundari, G.; Rovere, G.; Ferrandino, G.; Nicoletti, V.; Cilia, F.; De Vizio, S.; Palumbo, R.; Esposito, A.; et al. Advanced cardiac imaging in athlete’s heart: Unravelling the grey zone between physiologic adaptation and pathology. Radiol. Med. 2021, 126, 1518–1531. [Google Scholar] [CrossRef]
- Stein, S.I.; Soliman, M.M.; Sparapani, J.; Doustaly, R.; Cobb, B.W.; Malhotra, A.; Charalel, R.; May, B.J.; Lee, K.S.; Madoff, D.C.; et al. Conventional Hepatic Volumetry May Lead to Inaccurate Segmental Yttrium-90 Radiation Dosimetry. Cardiovasc. Interv. Radiol. 2021, 44, 1973–1985. [Google Scholar] [CrossRef]
- Fanelli, F.; Cannavale, A.; Chisci, E.; Citone, M.; Falcone, G.M.; Michelagnoli, S.; Miele, V. Direct percutaneous embolization of aneurysm sac: A safe and effective procedure to treat post-EVAR type II endoleaks. Radiol. Med. 2020, 126, 258–263. [Google Scholar] [CrossRef]
- Battaglia, V.; Cervelli, R. Liver investigations: Updating on US technique and contrast-enhanced ultrasound (CEUS). Eur. J. Radiol. 2017, 96, 65–73. [Google Scholar] [CrossRef]
- Faccia, M.; Garcovich, M.; Ainora, M.E.; Riccardi, L.; Pompili, M.; Gasbarrini, A.; Zocco, M.A. Contrast-Enhanced Ultrasound for Monitoring Treatment Response in Different Stages of Hepatocellular Carcinoma. Cancers 2022, 14, 481. [Google Scholar] [CrossRef] [PubMed]
- Ossola, C.; Curti, M.; Calvi, M.; Tack, S.; Mazzoni, S.; Genesio, L.; Venturini, M.; Genovese, E.A. Role of ultrasound and magnetic resonance imaging in the prognosis and classification of muscle injuries in professional football players: Correlation between imaging and return to sport time. Radiol. Med. 2021, 126, 1460–1467. [Google Scholar] [CrossRef] [PubMed]
- Soyer Güldoğan, E.; Ergun, O.; Taşkın Türkmenoğlu, T.; Yılmaz, K.B.; Akdağ, T.; Özbal Güneş, S.; Durmaz, H.A.; Hekimoğlu, B. The impact of TI-RADS in detecting thyroid malignancies: A prospective study. Radiol. Med. 2021, 126, 1335–1344. [Google Scholar] [CrossRef] [PubMed]
- Celletti, I.; Fresilli, D.; De Vito, C.; Bononi, M.; Cardaccio, S.; Cozzolino, A.; Durante, C.; Grani, G.; Grimaldi, G.; Isidori, A.M.; et al. TIRADS, SRE and SWE in INDETERMINATE thyroid nodule characterization: Which has better diagnostic performance? Radiol. Med. 2021, 126, 1189–1200. [Google Scholar] [CrossRef]
- Rosa, F.; Martinetti, C.; Veirana, M.A.; Attieh, A.; Trisoglio, A.; Sabattini, R.; Gandolfo, N.; Gastaldo, A. How embryology knowledge can help radiologists in the differential diagnosis of canal of Nuck pathologies. Radiol. Med. 2021, 126, 910–924. [Google Scholar] [CrossRef]
- Wang, X.; Liang, P.; Yu, J.; Yao, J.D.; Fan, F.Y.; Yu, X.; Cheng, Z.G.; Han, Z.Y.; Liu, F.Y.; Dou, J.P. Contrast-enhanced ultrasound features predict the prognosis of percutaneous microwave ablation of intrahepatic cholangiocarcinoma. Br. J. Radiol. 2022, 95, 20211379. [Google Scholar] [CrossRef]
- Bai, L.; Wang, X.; Shi, S.; Gao, J.; Li, X.; Wang, Y.; Jiang, M.; Zheng, C.; Liu, H. Evaluation of 3D-CEUS in the Recurrence of Liver Cancer after Radiofrequency Ablation. J. Health Eng. 2021, 2021, 3123553. [Google Scholar] [CrossRef]
- Danti, G.; Flammia, F.; Matteuzzi, B.; Cozzi, D.; Berti, V.; Grazzini, G.; Pradella, S.; Recchia, L.; Brunese, L.; Miele, V. Gastrointestinal neuroendocrine neoplasms (GI-NENs): Hot topics in morphological, functional, and prognostic imaging. Radiol. Med. 2021, 126, 1497–1507. [Google Scholar] [CrossRef]
- Rhim, H.; Yoon, K.H.; Lee, J.M.; Cho, Y.; Cho, J.S.; Kim, S.H.; Lee, W.J.; Lim, H.K.; Nam, G.J.; Han, S.S.; et al. Major complications after radio-frequency thermal ablation of hepatic tumors: Spectrum of imaging findings. RadioGraphics 2003, 23, 123–134. [Google Scholar] [CrossRef]
- Kwon, H.J.; Kim, P.N.; Byun, J.H.; Kim, K.W.; Won, H.J.; Shin, Y.M.; Lee, M.G. Various complications of percutaneous radiofrequency ablation for hepatic tumors: Radiologic findings and technical tips. Acta Radiol. 2014, 55, 1082–1092. [Google Scholar] [CrossRef]
- Sugihara, T.; Koda, M.; Tokunaga, S.; Matono, T.; Nagahara, T.; Ueki, M.; Murawaki, Y.; Kaminou, T. Contrast-enhanced ultrasonography revealed active thoracic bleeding. J. Med. Ultrason. 2010, 37, 143–145. [Google Scholar] [CrossRef] [PubMed]
- Catalano, O.; Sandomenico, F.; Raso, M.M.; Siani, A. Real-time, contrast-enhanced sonography: A new tool for detecting active bleeding. J. Trauma Inj. Infect. Crit. Care 2005, 59, 933–939. [Google Scholar] [CrossRef] [PubMed]
- Marano, R.; Pontone, G.; Agricola, E.; Alushi, B.; Bartorelli, A.; Cameli, M.; Carrabba, N.; Esposito, A.; Faletti, R.; Francone, M.; et al. Recommendations in pre-procedural imaging assessment for TAVI intervention: SIC-SIRM position paper part 2 (CT and MR angiography, standard medical reporting, future perspectives). Radiol. Med. 2022, 127, 277–293. [Google Scholar] [CrossRef] [PubMed]
- Masselli, G.; Almberger, M.; Tortora, A.; Capoccia, L.; Dolciami, M.; D’Aprile, M.R.; Valentini, C.; Avventurieri, G.; Bracci, S.; Ricci, P. Role of CT angiography in detecting acute pulmonary embolism associated with COVID-19 pneumonia. Radiol. Med. 2021, 126, 1553–1560. [Google Scholar] [CrossRef]
- Mirabile, A.; Lucarelli, N.M.; Sollazzo, E.P.; Ianora, A.A.S.; Sardaro, A.; Mirabile, G.; Lorusso, F.; Racanelli, V.; Maggialetti, N.; Scardapane, A. CT pulmonary angiography appropriateness in a single emergency department: Does the use of revised Geneva score matter? Radiol. Med. 2021, 126, 1544–1552. [Google Scholar] [CrossRef]
- Cozzi, D.; Moroni, C.; Cavigli, E.; Bindi, A.; Caviglioli, C.; Nazerian, P.; Vanni, S.; Miele, V.; Bartolucci, M. Prognostic value of CT pulmonary angiography parameters in acute pulmonary embolism. Radiol. Med. 2021, 126, 1030–1036. [Google Scholar] [CrossRef]
- De Cecco, C.N.; Buffa, V.; Fedeli, S.; Luzietti, M.; Vallone, A.; Ruopoli, R.; Miele, V.; Rengo, M.; Maurizi Enrici, M.; Fina, P.; et al. Preliminary experience with abdominal dual-energy CT (DECT): True versus virtual nonenhanced images of the liver. Radiol. Med. 2010, 115, 1258–1266. [Google Scholar] [CrossRef]
- Rawashdeh, M.A.; Saade, C. Radiation dose reduction considerations and imaging patterns of ground glass opacities in coronavirus: Risk of over exposure in computed tomography. Radiol. Med. 2020, 126, 380–387. [Google Scholar] [CrossRef]
- Trinci, M.; Cirimele, V.; Cozzi, D.; Galluzzo, M.; Miele, V. Diagnostic accuracy of pneumo-CT-cystography in the detection of bladder rupture in patients with blunt pelvic trauma. Radiol. Med. 2020, 125, 907–917. [Google Scholar] [CrossRef]
- Ferorelli, D.; Donno, F.; de Giorgio, G.; Mele, F.; Favia, M.; Riefoli, F.; Andresciani, S.; Melodia, R.; Zotti, F.; Dell’Erba, A. Head CT scan in emergency room: Is it still abused? Quantification and causes analysis of overprescription in an Italian Emergency Department. Radiol. Med. 2020, 125, 595–599. [Google Scholar] [CrossRef]
- Masjedi, H.; Zare, M.H.; Keshavarz Siahpoush, N.; Razavi-Ratki, S.K.; Alavi, F.; Shabani, M. European trends in radiology: Investigating factors affecting the number of examinations and the effective dose. Radiol. Med. 2020, 125, 296–305. [Google Scholar] [CrossRef] [PubMed]
- Akahane, M.; Koga, H.; Kato, N.; Yamada, H.; Uozumi, K.; Tateishi, R.; Teratani, T.; Shiina, S.; Ohtomo, K. Complications of percutaneous radiofrequency ablation for hepato-cellular carcinoma: Imaging spectrum and management. RadioGraphics 2005, 25, S57–S68. [Google Scholar] [CrossRef] [PubMed]
- Lu, D.S.; Raman, S.S.; Vodopich, D.J.; Wang, M.; Sayre, J.; Lassman, C. Effect of vessel size on creation of hepatic radiofrequency lesions in pig: Assessment of the “heat sink” effect. AJR Am. J. Roentgenol. 2002, 178, 47–51. [Google Scholar] [CrossRef] [PubMed]
- Novick, S.L.; Fishman, E.K. Portal vein thrombosis: Spectrum of helical CT and CT angiographic findings. Abdom. Imaging 1998, 23, 505–510. [Google Scholar] [CrossRef]
- Kim, B.S.; Kim, T.K.; Kim, J.S.; Lee, M.G.; Kim, J.H.; Kim, K.W.; Sung, K.B.; Kim, P.N.; Ha, H.K.; Lee, S.G.; et al. Hepatic venous congestion after living donor liver transplantation with right lobe graft: Two-phase CT findings. Radiology 2004, 232, 173–180. [Google Scholar] [CrossRef]
- Shindo, D.; Yamamoto, A.; Amano, R.; Kimura, K.; Yamazoe, S.; Shimono, T.; Miki, Y. The findings of the contrast-enhanced CT and risk factors for hepatic infarction after pancreatoduodenectomy. Jpn. J. Radiol. 2020, 38, 547–552. [Google Scholar] [CrossRef]
- Sivakumar, V. ‘Washing machine’ and ‘yin-yang’ sign in popliteal pseudoaneurysm. QJM Int. J. Med. 2021, 114, 212. [Google Scholar] [CrossRef]
- Kim, Y.S.; Rhim, H.; Lim, H.K. Imaging after radiofrequency ablation of hepatic tumors. Semin. Ultrasound CT MRI 2009, 30, 49–66. [Google Scholar] [CrossRef]
- Balfour, J.; Ewing, A. Hepatic Biloma; StatPearls Publishing: Treasure Island, FL, USA, 2022. [Google Scholar]
- Kim, Y.S.; Rhim, H.; Lim, H.K.; Choi, D.; Lee, M.W.; Park, M.J. Coagulation necrosis induced by radiofrequency ablation in the liver: Histopathologic and radiologic review of usual to extremely rare changes. RadioGraphics 2011, 31, 377–390. [Google Scholar] [CrossRef]
- Chang, I.S.; Rhim, H.; Kim, S.H.; Kim, Y.S.; Choi, D.; Park, Y.; Lim, H.K. Biloma formation after radiofrequency ablation of hepatocellular carcinoma: Incidence, imaging features, and clinical significance. AJR Am. J. Roentgenol. 2010, 195, 1131–1136. [Google Scholar] [CrossRef]
- Goto, E.; Tateishi, R.; Shiina, S.; Masuzaki, R.; Enooku, K.; Sato, T.; Ohki, T.; Kondo, Y.; Goto, T.; Yoshida, H.; et al. Hemorrhagic complications of percutaneous radiofrequency ablation for liver tumors. J. Clin. Gastroenterol. 2010, 44, 374–380. [Google Scholar] [CrossRef] [PubMed]
- Cathcart, S.; Birk, J.W.; Tadros, M.; Schuster, M. Hemobilia: An Uncommon but Notable Cause of Upper Gastrointestinal Bleeding. J. Clin. Gastroenterol. 2017, 51, 796–804. [Google Scholar] [CrossRef] [PubMed]
- Francica, G.; Meloni, M.F.; Riccardi, L.; Giangregorio, F.; Caturelli, E.; Terracciano, F.; de Sio, I. Role of Contrast-Enhanced Ultrasound in the Detection of Complications After Ultrasound-Guided Liver Interventional Procedures. J. Ultrasound Med. 2020, 40, 1665–1673. [Google Scholar] [CrossRef] [PubMed]
- Feng, W.; Yue, D.; Zaiming, L.; Zhaoyu, L.; Xiangxuan, Z.; Wei, L.; Qiyong, G. Iatrogenic hemobilia: Imaging features and management with transcatheter arterial embolization in 30 patients. Diagn. Interv. Radiol. 2016, 22, 371–377. [Google Scholar] [CrossRef]
- Chopra, S.; Dodd, G.D., 3rd; Chanin, M.P.; Chintapalli, K.N. Radiofrequency ablation of hepatic tumors adjacent to the gallbladder:Feasibility and safety. AJR Am. J. Roentgenol. 2003, 180, 697–701. [Google Scholar] [CrossRef]
- Yamamoto, T.; Kubo, S.; Hirohashi, K.; Tanaka, S.; Uenishi, T.; Ogawa, M.; Sakabe, K.; Hai, S.; Yamamoto, S.; Shuto, T.; et al. Secondary hemocholecyst after radiofrequency ablation therapy for hepatocellular carcinoma. J. Gastroenterol. 2003, 38, 399–403. [Google Scholar] [CrossRef]
- Sainani, N.I.; Gervais, D.A.; Mueller, P.R.; Arellano, R.S. Imaging after percutaneous radiofrequency ablation of hepatic tumors: Part 1, Normal findings. AJR Am. J. Roentgenol. 2013, 200, 184–193. [Google Scholar] [CrossRef]
- Cannataci, C.; Cimo’, B.; Mamone, G.; Tuzzolino, F.; D’Amico, M.; Cortis, K.; Maruzzelli, L.; Miraglia, R. Portal vein puncture-related complications during transjugular intrahepatic portosystemic shunt creation: Colapinto needle set vs Rösch-Uchida needle set. Radiol. Med. 2021, 126, 1487–1495. [Google Scholar] [CrossRef]
- Cicero, G.; Ascenti, G.; Albrecht, M.H.; Blandino, A.; Cavallaro, M.; D’Angelo, T.; Carerj, M.L.; Vogl, T.J.; Mazziotti, S. Extra-abdominal dual-energy CT applications: A comprehensive overview. Radiol Med. 2020, 125, 384–397. [Google Scholar] [CrossRef]
- Ledda, R.E.; Milanese, G.; Cademartiri, F.; Maffei, E.; Benedetti, G.; Goldoni, M.; Silva, M.; Sverzellati, N. Association of hepatic steatosis with epicardial fat volume and coronary artery disease in symptomatic patients. Radiol. Med. 2021, 126, 652–660. [Google Scholar] [CrossRef]
- Lombardi, A.F.; Afsahi, A.M.; Gupta, A.; Gholamrezanezhad, A. Severe acute respiratory syndrome (SARS), Middle East respiratory syndrome (MERS), influenza, and COVID-19, beyond the lungs: A review article. Radiol. Med. 2021, 126, 561–569. [Google Scholar] [CrossRef] [PubMed]
- Schullian, P.; Johnston, E.; Laimer, G.; Putzer, D.; Eberle, G.; Amann, A.; Effenberger, M.; Maglione, M.; Freund, M.C.; Loizides, A.; et al. Frequency and risk factors for major complications after stereotactic radiofrequency ablation of liver tumors in 1235 ablation sessions: A 15-year experience. Eur. Radiol. 2021, 31, 3042–3052. [Google Scholar] [CrossRef] [PubMed]
- Powerski, M.; Drewes, R.; Omari, J.; Relja, B.; Surov, A.; Pech, M. Intra-hepatic Abscopal Effect Following Radioembolization of Hepatic Metastases. Cardiovasc. Interv. Radiol. 2020, 43, 1641–1649. [Google Scholar] [CrossRef]
- Schullian, P.; Putzer, D.; Eberle, G.; Laimer, G.; Bale, R. Simultaneous Stereotactic Radiofrequency Ablation of Multiple (≥ 4) Liver Tumors: Feasibility, Safety, and Efficacy. J. Vasc. Interv. Radiol. 2020, 31, 943–952. [Google Scholar] [CrossRef]
- Giurazza, F.; Corvino, F.; Cavaglià, E.; Silvestre, M.; Cangiano, G.; Amodio, F.; De Magistris, G.; Niola, R. Emborrhoid in patients with portal hypertension and chronic hemorrhoidal bleeding: Preliminary results in five cases with a new coiling release fashion “Spaghetti technique”. Radiol. Med. 2020, 125, 1008–1011. [Google Scholar] [CrossRef]
- Aberle, S.; Kenkel, D.; Becker, A.S.; Puippe, G.; Burger, I.; Schaefer, N.; Pfammatter, T. Outpatient Yttrium-90 microsphere radioembolization: Assessment of radiation safety and quantification of post-treatment adverse events causing hospitalization. Radiol. Med. 2020, 125, 971–980. [Google Scholar] [CrossRef]
- Livraghi, T.; Solbiati, L.; Meloni, M.F.; Gazelle, G.S.; Halpern, E.F.; Goldberg, S.N. Treatment of focal liver tumors with percutaneous radio-frequency ablation: Complications encountered in a multicenter Study. Radiology 2003, 226, 441–451. [Google Scholar] [CrossRef]
- Choi, D.; Lim, H.K.; Kim, M.J.; Kim, S.J.; Kim, S.H.; Lee, W.J.; Lim, J.H.; Paik, S.W.; Yoo, B.C.; Choi, M.S.; et al. Liver abscess after percutaneous radiofrequency ablation for hepatocellular carcinomas: Frequency and risk factors. AJR Am. J. Roentgenol. 2005, 184, 1860–1867. [Google Scholar] [CrossRef]
- Malekzadeh, S.; Widmer, L.; Salahshour, F.; Egger, B.; Ronot, M.; Thoeny, H.C. Typical imaging finding of hepatic infections: A pictorial essay. Abdom. Radiol. 2021, 46, 544–561. [Google Scholar] [CrossRef]
- Catalano, O.; Sandomenico, F.; Raso, M.M.; Siani, A. Low mechanical index contrast-enhanced sonographic findings of pyogenic hepatic abscesses. AJR Am. J. Roentgenol. 2004, 182, 447–450. [Google Scholar] [CrossRef]
- Giambelluca, D.; Panzuto, F.; Giambelluca, E.; Midiri, M. The “double target sign” in liver abscess. Abdom. Radiol. 2018, 43, 2885–2886. [Google Scholar] [CrossRef] [PubMed]
- Lim, H.K.; Choi, N.; Lee, W.J.; Kim, S.H.; Lee, S.J.; Jang, H.J.; Lee, J.H.; Lim, J.H.; Choo, I.W. Hepatocellular carcinoma treated with percutaneous radio-frequency ablation: Evaluation with follow-up multiphase helical CT. Radiology 2001, 221, 447–454. [Google Scholar] [CrossRef] [PubMed]
- De Filippo, M.; Puglisi, S.; D’Amuri, F.; Gentili, F.; Paladini, I.; Carrafiello, G.; Maestroni, U.; Del Rio, P.; Ziglioli, F.; Pagnini, F. CT-guided percutaneous drainage of abdominopelvic collections: A pictorial essay. Radiol. Med. 2021, 126, 1561–1570. [Google Scholar] [CrossRef]
- Jeong, Y.S.; Kim, S.H.; Lee, J.M.; Lee, J.Y.; Kim, J.H.; Lee, D.H.; Kang, H.J.; Yoon, C.J.; Han, J.K. Gastrointestinal tract complications after hepatic radiofrequency ablation: CT prediction for major complications. Abdom. Radiol. 2018, 43, 583–592. [Google Scholar] [CrossRef] [PubMed]
- Schmit, G.D.; Kurup, A.N.; Schmitz, J.J.; Atwell, T.D. The “Leverage Technique”: Using Needles to Displace the Stomach during Liver Ablation. J. Vasc. Interv. Radiol. 2016, 27, 1765–1767. [Google Scholar] [CrossRef]
- Head, H.W.; Dodd, G.D., 3rd; Dalrymple, N.C.; Prasad, S.R.; El-Merhi, F.M.; Freckleton, M.W.; Hubbard, L.G. Percutaneous radiofrequency ablation of hepatic tumors against the diaphragm: Frequency of diaphragmatic injury1. Radiology 2007, 243, 877–884. [Google Scholar] [CrossRef] [PubMed]
- Abe, T.; Shinzawa, H.; Wakabayashi, H.; Aoki, M.; Sugahara, K.; Iwaba, A.; Haga, H.; Miyano, S.; Terui, Y.; Mitsuhashi, H.; et al. Value of laparoscopic microwave coagulation therapy for hepatocellular carcinoma in relation to tumor size and location. Endoscopy 2000, 32, 598–603. [Google Scholar] [CrossRef] [PubMed]
- Llovet, J.M.; Vilana, R.; Brú, C.; Bianchi, L.; Salmeron, J.M.; Boix, L.; Ganau, S.; Sala, M.; Pagès, M.; Ayuso, C.; et al. Increased risk of tumor seeding after percutaneous radiofrequency ablation for single hepatocellular carcinoma. Hepatology 2001, 33, 1124–1129. [Google Scholar] [CrossRef]
- Thiemann, M.; Benhidjeb, T.; Anders, S.; Gebauer, B.; Strik, M.W. Hepato-pericardial fistula following radiofrequency ablation (RFA) for liver metastasis: A case report and review of the literature. Langenbeck’s Arch. Surg. 2008, 393, 1013–1016. [Google Scholar] [CrossRef]
- Puijk, R.S.; Ruarus, A.H.; Scheffer, H.J.; Vroomen, L.G.P.H.; van Tilborg, A.A.J.M.; de Vries, J.J.J.; Berger, F.H.; van den Tol, P.M.P.; Meijerink, M.R. Percutaneous Liver Tumour Ablation: Image Guidance, Endpoint Assessment, and Quality Control. Can. Assoc. Radiol. J. 2018, 69, 51–62. [Google Scholar] [CrossRef] [Green Version]
- Izzo, F.; Palaia, R.; Albino, V.; Amore, A.; Di Giacomo, R.; Piccirillo, M.; Leongito, M.; Nasto, A.; Granata, V.; Petrillo, A.; et al. Hepatocellular carcinoma and liver metastases: Clinical data on a new dual-lumen catheter kit for surgical sealant infusion to prevent perihepatic bleeding and dissemination of cancer cells following biopsy and loco-regional treatments. Infect. Agents Cancer 2015, 10, 11. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Granata, V.; Fusco, R.; Piccirillo, M.; Palaia, R.; Petrillo, A.; Lastoria, S.; Izzo, F. Electrochemotherapy in locally advanced pancreatic cancer: Preliminary results. Int. J. Surg. 2015, 18, 230–236. [Google Scholar] [CrossRef] [PubMed]
- Tafuto, S.; von Arx, C.; De Divitiis, C.; Maura, C.T.; Palaia, R.; Albino, V.; Fusco, R.; Membrini, M.; Petrillo, A.; Granata, V.; et al. Electrochemotherapy as a new approach on pancreatic cancer and on liver metastases. Int. J. Surg. 2015, 21, S78–S82. [Google Scholar] [CrossRef]
- Pfannenstiel, A.; Sebek, J.; Fallahi, H.; Beard, W.L.; Ganta, C.K.; Dupuy, D.E.; Prakash, P. Directional Microwave Ablation: Experimental Evaluation of a 2.45-GHz Applicator in Ex Vivo and In Vivo Liver. J. Vasc. Interv. Radiol. 2020, 31, 1170–1177. [Google Scholar] [CrossRef] [PubMed]
- Vogel, J.A.; van Veldhuisen, E.; Alles, L.K.; Busch, O.R.; Dijk, F.; van Gulik, T.M.; Huijzer, G.M.; Besselink, M.G.; van Lienden, K.P.; Verheij, J. Time-Dependent Impact of Irreversible Electroporation on Pathology and Ablation Size in the Porcine Liver: A 24-Hour Experimental Study. Technol. Cancer Res. Treat. 2019, 18, 1533033819876899. [Google Scholar] [CrossRef] [PubMed]
- Sun, J.; Yang, L.; Zhou, Z.; Zhang, D.; Han, W.; Zhang, Q.; Peng, Y. Performance evaluation of two iterative reconstruction algorithms, MBIR and ASIR, in low radiation dose and low contrast dose abdominal CT in children. Radiol. Med. 2020, 125, 918–925. [Google Scholar] [CrossRef]
- Imajo, K.; Ogawa, Y.; Yoneda, M.; Saito, S.; Nakajima, A. A review of conventional and newer generation microwave ablation systems for hepatocellular carcinoma. J. Med. Ultrason. 2020, 47, 265–277. [Google Scholar] [CrossRef]
- Liebl, M.; Schulze-Hagen, M.; Zimmermann, M.; Pedersoli, F.; Kuhl, C.; Bruners, P.; Isfort, P. Microwave Ablation in the Proximity of Surgical Clips: Is there a Safety Issue? Cardiovasc. Interv. Radiol. 2020, 43, 918–923. [Google Scholar] [CrossRef] [Green Version]
- Cicero, G.; Mazziotti, S.; Silipigni, S.; Blandino, A.; Cantisani, V.; Pergolizzi, S.; D’Angelo, T.; Stagno, A.; Maimone, S.; Squadrito, G.; et al. Dual-energy CT quantification of fractional extracellular space in cirrhotic patients: Comparison between early and delayed equilibrium phases and correlation with oesophageal varices. Radiol. Med. 2021, 126, 761–767. [Google Scholar] [CrossRef]
- Granata, V.; Fusco, R.; Catalano, O.; Piccirillo, M.; De Bellis, M.; Izzo, F.; Petrillo, A. Percutaneous ablation therapy of hepatocellular carcinoma with irreversible electroporation: MRI findings. AJR Am. J. Roentgenol. 2015, 204, 1000–1007. [Google Scholar] [CrossRef]
- Granata, V.; Fusco, R.; Salati, S.; Petrillo, A.; Di Bernardo, E.; Grassi, R.; Palaia, R.; Danti, G.; La Porta, M.; Cadossi, M.; et al. A Systematic Review about Imaging and Histopathological Findings for Detecting and Evaluating Electroporation Based Treatments Response. Int. J. Environ. Res. Public Health 2021, 18, 5592. [Google Scholar] [CrossRef] [PubMed]
- Fang, C.; Cortis, K.; Yusuf, G.T.; Gregory, S.; Lewis, D.; Kane, P.; Peddu, P. Complications from percutaneous microwave ablation of liver tumours: A pictorial review. Br. J. Radiol. 2019, 92, 20180864. [Google Scholar] [CrossRef] [PubMed]



Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
De Muzio, F.; Cutolo, C.; Dell’Aversana, F.; Grassi, F.; Ravo, L.; Ferrante, M.; Danti, G.; Flammia, F.; Simonetti, I.; Palumbo, P.; et al. Complications after Thermal Ablation of Hepatocellular Carcinoma and Liver Metastases: Imaging Findings. Diagnostics 2022, 12, 1151. https://doi.org/10.3390/diagnostics12051151
De Muzio F, Cutolo C, Dell’Aversana F, Grassi F, Ravo L, Ferrante M, Danti G, Flammia F, Simonetti I, Palumbo P, et al. Complications after Thermal Ablation of Hepatocellular Carcinoma and Liver Metastases: Imaging Findings. Diagnostics. 2022; 12(5):1151. https://doi.org/10.3390/diagnostics12051151
Chicago/Turabian StyleDe Muzio, Federica, Carmen Cutolo, Federica Dell’Aversana, Francesca Grassi, Ludovica Ravo, Marilina Ferrante, Ginevra Danti, Federica Flammia, Igino Simonetti, Pierpaolo Palumbo, and et al. 2022. "Complications after Thermal Ablation of Hepatocellular Carcinoma and Liver Metastases: Imaging Findings" Diagnostics 12, no. 5: 1151. https://doi.org/10.3390/diagnostics12051151
APA StyleDe Muzio, F., Cutolo, C., Dell’Aversana, F., Grassi, F., Ravo, L., Ferrante, M., Danti, G., Flammia, F., Simonetti, I., Palumbo, P., Bruno, F., Pierpaoli, L., Fusco, R., Giovagnoni, A., Miele, V., Barile, A., & Granata, V. (2022). Complications after Thermal Ablation of Hepatocellular Carcinoma and Liver Metastases: Imaging Findings. Diagnostics, 12(5), 1151. https://doi.org/10.3390/diagnostics12051151