Overall Hemostatic Potential Assay Detects Risk of Progression to Post-Thrombotic Syndrome in Anticoagulated Patients following Deep Vein Thrombosis
Abstract
1. Introduction
2. Materials and Methods
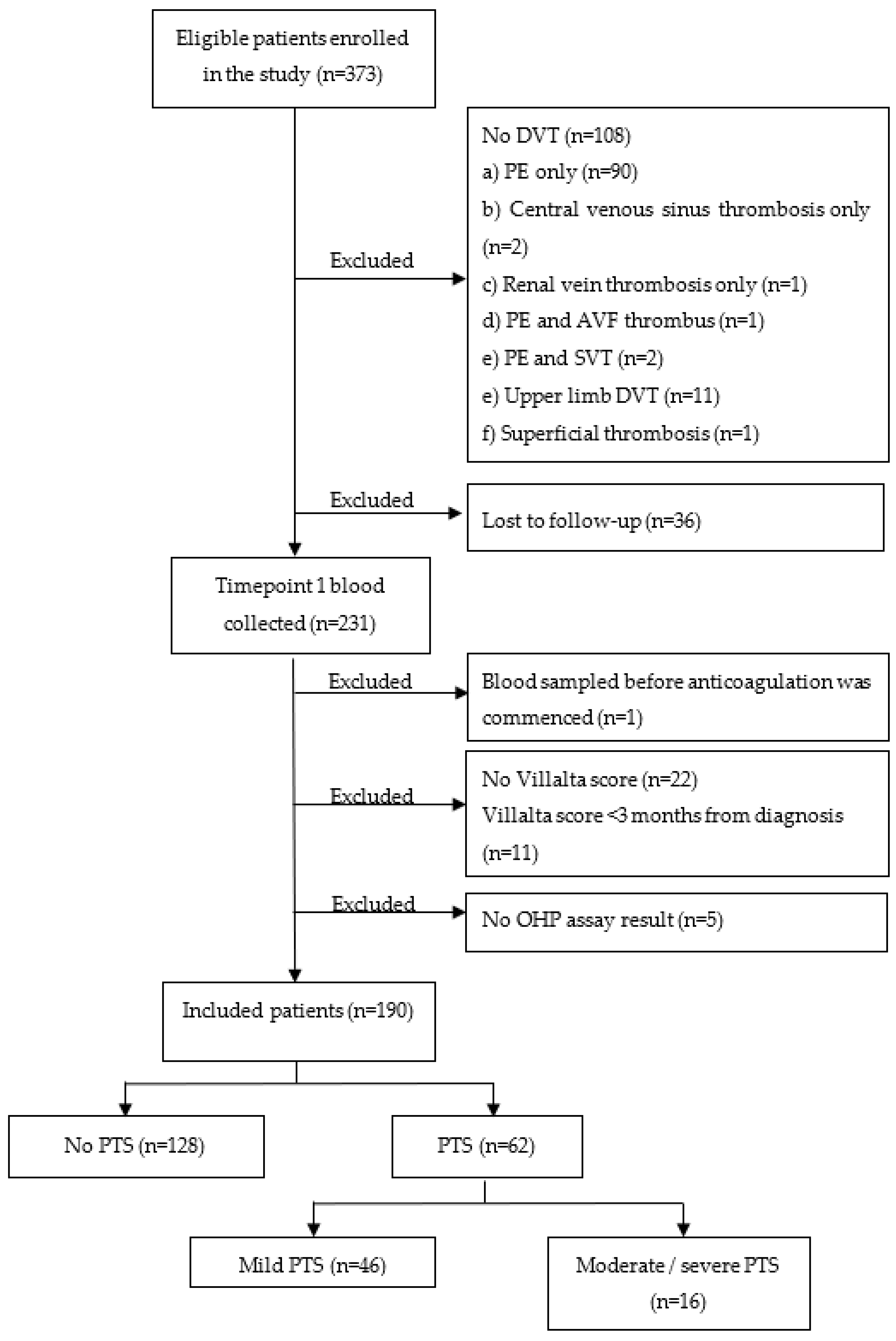
2.1. Study Participants
2.2. Follow Up
2.3. Laboratory Procedure
2.4. Healthy Controls
2.5. Modified Overall Hemostatic Potential (OHP) Assay
2.6. Statistical Analysis
3. Results
| No PTS | PTS | p-Value * | Mild PTS | Moderate/ Severe PTS | p-Value ** | |
|---|---|---|---|---|---|---|
| n | 128 | 62 | 46 | 16 | ||
| Age (years), mean (SD) | 56.6 (14.2) | 57.4 (11.2) | 0.72 | 58.2 (11.1) | 54.9 (11.6) | 0.32 |
| Male | 72 (56.2%) | 30 (48.4%) | 0.31 | 19 (41.3%) | 11 (68.8%) | 0.06 |
| Female | 56 (43.8%) | 32 (51.6%) | 27 (58.7%) | 5 (31.2%) | ||
| Weight (kg) | 92.0 [81.0, 104.0] | 100.0 [85.0, 120.0] | 0.016 | 97.0 [80.0, 116.0] | 110.0 [88.5, 129.0] | 0.07 |
| Unprovoked DVT | 77 (60.2%) | 43 (69.4%) | 0.22 | 30 (65.2%) | 13 (81.2%) | 0.23 |
| Proximal DVT | 56 (43.8%) | 39 (62.9%) | 0.013 | 27 (58.7%) | 12 (75.0%) | 0.24 |
| Previous DVT history | 32 (25.0%) | 14 (22.6%) | 0.72 | 12 (26.1%) | 2 (12.5%) | 0.26 |
| Family history DVT/PE | 22 (17.2%) | 15 (24.2%) | 0.25 | 12 (26.1%) | 3 (18.8%) | 0.56 |
| Malignancy | 10 (7.8%) | 2 (3.2%) | 0.22 | 2 (4.3%) | 0 (0.0%) | 0.40 |
| Smoker | 17 (13.6%) | 13 (21.0%) | 0.20 | 11 (23.9%) | 2 (12.5%) | 0.33 |
| History of varicose veins | 6 (4.7%) | 14 (22.6%) | <0.001 | 11 (23.9%) | 3 (18.8%) | 0.67 |
| Length of follow up (days) | 480.5 [261.0, 1072.0] | 815.0 [368.0, 1127.0] | 0.07 | 820.5 [357.0, 1119.0] | 761.0 [405.5, 1127.5] | 1.00 |
| Anticoagulation | ||||||
| DOAC | 110 (85.9%) | 46 (74.2) | 0.048 | 37 (80.4%) | 9 (56.2%) | 0.06 |
| Warfarin | 14 (10.9%) | 14 (22.6%) | 0.034 | 8 (17.4%) | 6 (37.5%) | 0.10 |
| Enoxaparin | 4 (3.1%) | 2 (3.2%) | 0.97 | 1 (2.2%) | 1 (6.2%) | 0.43 |
| Residual thrombus on repeat imaging | 50 (42.4%) | 26 (43.3%) | 0.90 | 18 (40.9%) | 8 (50.0%) | 0.53 |
| Inherited thrombophilia # | 25 (19.7%) | 10 (16.4%) | 0.59 | 7 (15.6%) | 3 (18.8%) | 0.77 |
| Antiphospholipid syndrome ^ | 3 (2.4%) | 1 (1.6%) | 0.75 | 1 (2.2%) | 0 (0.0%) | 0.55 |
| Recurrent VTE during follow-up | 12 (9.4%) | 6 (9.7%) | 0.95 | 4 (8.7%) | 2 (12.5%) | 0.66 |
| Recurrent ipsilateral leg DVT during follow-up | 5 (3.9%) | 4 (6.5%) | 0.44 | 3 (6.5%) | 1 (6.2%) | 0.97 |
| Healthy Controls (HC) | No PTS | PTS | p-Value HC vs. No PTS | p-Value HC vs. PTS | |
|---|---|---|---|---|---|
| n | 144 | 128 | 62 | ||
| Age (years) | 42.0 [24.5, 57.0] | 57.0 [46.0, 67.0] | 58.5 [51.0, 65.0] | <0.001 | <0.001 |
| Male | 50 (34.7%) | 72 (56.2%) | 30 (48.4%) | <0.001 | 0.065 |
| Fibrinogen (g/L) | 2.9 [2.5, 3.5] | 3.5 [3.0, 4.2] | 3.9 [3.4, 4.6] | <0.001 | <0.001 |
| D-dimer (mg/L FEU) | 0.2 [0.1, 0.3] | 0.3 [0.3, 0.5] | 0.3 [0.3, 0.5] | <0.001 | <0.001 |
| Factor VIII (%) | 106.0 [86.0, 145.0] | 148.0 [112.5, 190.5] | 162.0 [121.5, 202.5] | <0.001 | <0.001 |
| von-Willebrand antigen (%) | 102.0 [87.0, 142.5] | 148.5 [101.0, 192.0] | 159.5 [118.5, 200.0] | 0.005 | <0.001 |
| OCP (units) | 34.5 [29.0, 43.3] | 38.8 [32.1, 48.1] | 45.8 [39.3, 52.8] | 0.004 | <0.001 |
| OHP (units) | 6.4 [4.8, 9.5] | 9.2 [6.9, 13.2] | 12.8 [8.8, 17.2] | <0.001 | <0.001 |
| OFP (%) | 81.1 [77.5, 84.1] | 75.6 [71.0, 80.5] | 74.1 [64.9, 77.3] | <0.001 | <0.001 |
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Appendix A
| Symptoms and Signs | None | Mild | Moderate | Severe |
|---|---|---|---|---|
| Symptoms | ||||
| Pain | 0 | 1 | 2 | 3 |
| Cramps | 0 | 1 | 2 | 3 |
| Heaviness | 0 | 1 | 2 | 3 |
| Paresthesia | 0 | 1 | 2 | 3 |
| Pruritus | 0 | 1 | 2 | 3 |
| Clinical signs | ||||
| Pretibial edema | 0 | 1 | 2 | 3 |
| Skin induration | 0 | 1 | 2 | 3 |
| Hyperpigmentation | 0 | 1 | 2 | 3 |
| Redness | 0 | 1 | 2 | 3 |
| Venous ectasia | 0 | 1 | 2 | 3 |
| Pain on calf compression | 0 | 1 | 2 | 3 |
| Venous ulceration | Absent | Present |
References
- Prandoni, P.; Kahn, S.R. Post-thrombotic Syndrome: Prevalence, Prognostication and Need for Progress. Br. J. Haematol. 2009, 145, 286–295. [Google Scholar] [CrossRef]
- Rabinovich, A.; Kahn, S.R. How I Treat the Postthrombotic Syndrome. Blood 2018, 131, 2215–2222. [Google Scholar] [CrossRef]
- MacDougall, D.A.; Feliu, A.L.; Boccuzzi, S.J.; Lin, J. Economic Burden of Deep-Vein Thrombosis, Pulmonary Embolism, and Post-Thrombotic Syndrome. Am. J. Health-Syst. Pharm. 2006, 63, S5–S15. [Google Scholar] [CrossRef] [PubMed]
- Kachroo, S.; Boyd, D.; Bookhart, B.K.; LaMori, J.; Schein, J.R.; Rosenberg, D.J.; Reynolds, M.W. Quality of Life and Economic Costs Associated with Postthrombotic Syndrome. Am. J. Health-Syst. Pharm. 2012, 69, 567–572. [Google Scholar] [CrossRef]
- Roberts, L.N.; Patel, R.K.; Donaldson, N.; Bonner, L.; Arya, R. Post-Thrombotic Syndrome Is an Independent Determinant of Health-Related Quality of Life Following Both First Proximal and Distal Deep Vein Thrombosis. Haematologica 2014, 99, e41–e43. [Google Scholar] [CrossRef]
- Kahn, S.R.; Hirsch, A.; Shrier, I. Effect of Postthrombotic Syndrome on Health-Related Quality of Life After Deep Venous Thrombosis. Arch. Intern. Med. 2002, 162, 1144–1148. [Google Scholar] [CrossRef] [PubMed]
- Johnson, B.F.; Manzo, R.A.; Bergelin, R.O.; Strandness, D.E. Relationship between Changes in the Deep Venous System and the Development of the Postthrombotic Syndrome after an Acute Episode of Lower Limb Deep Vein Thrombosis: A One- to Six-Year Follow-Up. J. Vasc. Surg. 1995, 21, 307–313. [Google Scholar] [CrossRef] [PubMed]
- Kurstjens, R.L.M.; Wolf, M.A.F.; Konijn, H.W.; Toonder, I.M.; Nelemans, P.J.; Graaf, R.; Wittens, C.H.A. Intravenous Pressure Changes in Patients with Postthrombotic Deep Venous Obstruction: Results Using a Treadmill Stress Test. J. Thromb. Haemost. 2016, 14, 1163–1170. [Google Scholar] [CrossRef] [PubMed]
- Kreidy, R. Pathophysiology of Post-Thrombotic Syndrome: The Effect of Recurrent Venous Thrombosis and Inherited Thrombophilia. ISRN Vasc. Med. 2011, 2011, 513503. [Google Scholar] [CrossRef]
- Deatrick, K.B.; Elfline, M.; Baker, N.; Luke, C.E.; Blackburn, S.; Stabler, C.; Wakefield, T.W.; Henke, P.K. Postthrombotic Vein Wall Remodeling: Preliminary Observations. J. Vasc. Surg. 2011, 53, 139–146. [Google Scholar] [CrossRef]
- Henke, P.K.; Varma, M.R.; Deatrick, K.B.; Dewyer, N.A.; Drewyer, N.A.; Lynch, E.M.; Moore, A.J.; Dubay, D.A.; Sukheepod, P.; Pearce, C.G.; et al. Neutrophils Modulate Post-Thrombotic Vein Wall Remodeling but Not Thrombus Neovascularization. Thromb. Haemost. 2006, 95, 272–281. [Google Scholar] [CrossRef] [PubMed]
- Bouman, A.C.; Smits, J.J.M.; Cate, H.T.; Cate-Hoek, A.J.T. Markers of Coagulation, Fibrinolysis and Inflammation in Relation to Post-thrombotic Syndrome. J. Thromb. Haemost. 2012, 10, 1532–1538. [Google Scholar] [CrossRef] [PubMed]
- Bittar, L.F.; da Silva, L.Q.; de Andrade Orsi, F.L.; Zapponi, K.C.S.; de Moraes Mazetto, B.; de Paula, E.V.; de Lima Montalvão, S.A.; Annichino-Bizzacchi, J.M. Increased Inflammation and Endothelial Markers in Patients with Late Severe Post-Thrombotic Syndrome. PLoS ONE 2020, 15, e0227150. [Google Scholar] [CrossRef] [PubMed]
- Rabinovich, A.; Cohen, J.M.; Cushman, M.; Kahn, S.R.; Investigators, B.; Anderson, D.R.; Chagnon, I.; Demers, C.; Desmarais, S.; Ginsberg, J.S.; et al. Association between Inflammation Biomarkers, Anatomic Extent of Deep Venous Thrombosis, and Venous Symptoms after Deep Venous Thrombosis. J. Vasc. Surg. Venous Lymphat. Disord. 2015, 3, 347–353.e1. [Google Scholar] [CrossRef] [PubMed]
- Meissner, M.H.; Zierler, B.K.; Bergelin, R.O.; Chandler, W.L.; Strandness, D.E. Coagulation, Fibrinolysis, and Recanalization after Acute Deep Venous Thrombosis. J. Vasc. Surg. 2002, 35, 278–285. [Google Scholar] [CrossRef] [PubMed]
- Baldwin, J.F.; Sood, V.; Elfline, M.A.; Luke, C.E.; Dewyer, N.A.; Diaz, J.A.; Myers, D.D.; Wakefield, T.; Henke, P.K. The Role of Urokinase Plasminogen Activator and Plasmin Activator Inhibitor-1 on Vein Wall Remodeling in Experimental Deep Vein Thrombosis. J. Vasc. Surg. 2012, 56, 1089–1097. [Google Scholar] [CrossRef]
- Siudut, J.; Grela, M.; Wypasek, E.; Plens, K.; Undas, A. Reduced Plasma Fibrin Clot Permeability and Susceptibility to Lysis Are Associated with Increased Risk of Postthrombotic Syndrome. J. Thromb. Haemost. 2016, 14, 784–793. [Google Scholar] [CrossRef]
- Mrozinska, S.; Cieslik, J.; Broniatowska, E.; Undas, A. Elevated Leptin and Decreased Adiponectin Independently Predict the Post-Thrombotic Syndrome in Obese and Non-Obese Patients. Sci. Rep. 2018, 8, 6938. [Google Scholar] [CrossRef]
- Wang, J.; Lim, H.Y.; Brook, R.; Lai, J.; Nandurkar, H.; Ho, P. Overall Haemostatic Potential (OHP) Assay Can Risk Stratify for Venous Thromboembolism Recurrence in Anticoagulated Patients. J. Thromb. Thrombolys. 2022; Online ahead of print. [Google Scholar] [CrossRef]
- Soosainathan, A.; Moore, H.M.; Gohel, M.S.; Davies, A.H. Scoring Systems for the Post-Thrombotic Syndrome. J. Vasc. Surg. 2013, 57, 254–261. [Google Scholar] [CrossRef]
- Utne, K.K.; Ghanima, W.; Foyn, S.; Kahn, S.; Sandset, P.M.; Wik, H.S. Development and Validation of a Tool for Patient Reporting of Symptoms and Signs of the Post-Thrombotic Syndrome. Thromb. Haemost. 2016, 115, 361–367. [Google Scholar] [CrossRef] [PubMed]
- Lim, H.Y.; Lui, B.; Tacey, M.; Kwok, A.; Varadarajan, S.; Donnan, G.; Nandurkar, H.; Ho, P. Global Coagulation Assays in Patients with Diabetes Mellitus. Res. Pract. Thromb. Haemost. 2021, 5, e12611. [Google Scholar] [CrossRef] [PubMed]
- Curnow, J.L.; Morel-Kopp, M.-C.; Roddie, C.; Aboud, M.; Ward, C.M. Reduced Fibrinolysis and Increased Fibrin Generation Can Be Detected in Hypercoagulable Patients Using the Overall Hemostatic Potential Assay. J. Thromb. Haemost. 2007, 5, 528–534. [Google Scholar] [CrossRef] [PubMed]
- Miyakis, S.; Lockshin, M.D.; Atsumi, T.; Branch, D.W.; Brey, R.L.; Cervera, R.; Derksen, R.H.W.M.; Groot, P.G.D.; Koike, T.; Meroni, P.L.; et al. International Consensus Statement on an Update of the Classification Criteria for Definite Antiphospholipid Syndrome (APS). J. Thromb. Haemost. 2006, 4, 295–306. [Google Scholar] [CrossRef]
- Rabinovich, A.; Cohen, J.M.; Kahn, S.R. The Predictive Value of Markers of Fibrinolysis and Endothelial Dysfunction in the Post Thrombotic Syndrome. Thromb. Haemost. 2014, 111, 1031–1040. [Google Scholar] [CrossRef]
- Sartori, M.; Favaretto, E.; Cini, M.; Legnani, C.; Palareti, G.; Cosmi, B. D-Dimer, FVIII and Thrombotic Burden in the Acute Phase of Deep Vein Thrombosis in Relation to the Risk of Post-Thrombotic Syndrome. Thromb. Res. 2014, 134, 320–325. [Google Scholar] [CrossRef]
- Galanaud, J.P.; Holcroft, C.A.; Rodger, M.A.; Kovacs, M.J.; Betancourt, M.T.; Wells, P.S.; Anderson, D.R.; Chagnon, I.; Gal, G.; Solymoss, S.; et al. Predictors of Post-thrombotic Syndrome in a Population with a First Deep Vein Thrombosis and No Primary Venous Insufficiency. J. Thromb. Haemost. 2013, 11, 474–480. [Google Scholar] [CrossRef]
- Song, M.; Graubard, B.I.; Rabkin, C.S.; Engels, E.A. Neutrophil-to-Lymphocyte Ratio and Mortality in the United States General Population. Sci. Rep. 2021, 11, 464. [Google Scholar] [CrossRef]
- Mureșan, A.V.; Hălmaciu, I.; Arbănași, E.M.; Kaller, R.; Arbănași, E.M.; Budișcă, O.A.; Melinte, R.M.; Vunvulea, V.; Filep, R.C.; Mărginean, L.; et al. Prognostic Nutritional Index, Controlling Nutritional Status (CONUT) Score, and Inflammatory Biomarkers as Predictors of Deep Vein Thrombosis, Acute Pulmonary Embolism, and Mortality in COVID-19 Patients. Diagnostics 2022, 12, 2757. [Google Scholar] [CrossRef]
- Efros, O.; Halevi, T.B.; Meisel, E.; Soffer, S.; Barda, N.; Cohen, O.; Kenet, G.; Lubetsky, A. The Prognostic Role of Neutrophil-to-Lymphocyte Ratio in Patients Hospitalized with Acute Pulmonary Embolism. J. Clin. Med. 2021, 10, 4058. [Google Scholar] [CrossRef]
- Arbănași, E.M.; Mureșan, A.V.; Coșarcă, C.M.; Kaller, R.; Bud, T.I.; Hosu, I.; Voidăzan, S.T.; Arbănași, E.M.; Russu, E. Neutrophil-to-Lymphocyte Ratio and Platelet-to-Lymphocyte Ratio Impact on Predicting Outcomes in Patients with Acute Limb Ischemia. Life 2022, 12, 822. [Google Scholar] [CrossRef] [PubMed]
- Melinte, R.M.; Arbănași, E.M.; Blesneac, A.; Zolog, D.N.; Kaller, R.; Mureșan, A.V.; Arbănași, E.M.; Melinte, I.M.; Niculescu, R.; Russu, E. Inflammatory Biomarkers as Prognostic Factors of Acute Deep Vein Thrombosis Following the Total Knee Arthroplasty. Medicina 2022, 58, 1502. [Google Scholar] [CrossRef] [PubMed]
- Kuplay, H.; Erdoğan, S.B.; Bastopcu, M.; Arslanhan, G.; Baykan, D.B.; Orhan, G. The Neutrophil-Lymphocyte Ratio and the Platelet-Lymphocyte Ratio Correlate with Thrombus Burden in Deep Venous Thrombosis. J. Vasc. Surg. Venous Lymphat. Disord. 2020, 8, 360–364. [Google Scholar] [CrossRef] [PubMed]
- Grimnes, G.; Horvei, L.D.; Tichelaar, V.; Brækkan, S.K.; Hansen, J.-B. Neutrophil to Lymphocyte Ratio and Future Risk of Venous Thromboembolism and Mortality: The Tromsø Study. Haematologica 2016, 101, e401–e404. [Google Scholar] [CrossRef] [PubMed]
- Karathanos, C.; Nana, P.; Spanos, K.; Kouvelos, G.; Brotis, A.; Matsagas, M.; Giannoukas, A. Efficacy of Rivaroxaban in the Prevention of Post-Thrombotic Syndrome: A Systematic Review and Meta-Analysis. J. Vasc. Surg. Venous Lymphat. Disord. 2021, 9, 1568–1576.e1. [Google Scholar] [CrossRef] [PubMed]
- Terry, C.M.; He, Y.; Cheung, A.K. Rivaroxaban Improves Patency and Decreases Inflammation in a Mouse Model of Catheter Thrombosis. Thromb. Res. 2016, 144, 106–112. [Google Scholar] [CrossRef] [PubMed]
- Dongen, C.J.J.; Prandoni, P.; Frulla, M.; Marchiori, A.; Prins, M.H.; Hutten, B.A. Relation between Quality of Anticoagulant Treatment and the Development of the Postthrombotic Syndrome. J. Thromb. Haemost. 2005, 3, 939–942. [Google Scholar] [CrossRef]
- Rabinovich, A.; Ducruet, T.; Kahn, S.R.; Investigators, S.T. Development of a Clinical Prediction Model for the Postthrombotic Syndrome in a Prospective Cohort of Patients with Proximal Deep Vein Thrombosis. J. Thromb. Haemost. 2018, 16, 262–270. [Google Scholar] [CrossRef]

| No PTS | PTS | p-Value * | Mild PTS | Moderate/Severe PTS | p-Value ** | |
|---|---|---|---|---|---|---|
| n | 128 | 62 | 46 | 16 | ||
| Days from DVT diagnosis to blood sample | 90.0 [46.0, 112.0] | 80.0 [55.0, 105.0] | 0.76 | 82.0 [63.0, 105.0] | 67.0 [50.0, 108.0] | 0.38 |
| Hemoglobin (g/L) | 146.0 [136.0, 155.0] | 144.0 [135.0, 154.0] | 0.44 | 140.0 [135.0, 154.0] | 148.5 [140.0, 154.5] | 0.25 |
| Neutrophils (×109/L) | 3.9 (3.3, 5.2] | 4.4 [3.5, 5.4] | 0.23 | 4.2 [3.5, 5.4] | 4.8 [3.9, 5.8] | 0.25 |
| Lymphocytes (×109/L) | 2.2 [1.8, 2.8] | 1.8 [1.5, 2.3] | 0.007 | 1.8 [1.6, 2.3] | 1.7 [1.4, 2.3] | 0.65 |
| Neutrophil/lymphocyte ratio | 1.9 [1.4, 2.4] | 2.3 [1.6, 3.0] | 0.007 | 2.2 [1.5, 3.0] | 2.3 [1.8, 3.9] | 0.55 |
| Platelets (×109/L) | 256.0 [226.0, 316.5] | 259.0 [217.0, 311.0] | 0.66 | 255.0 [231.0, 296.0] | 272.0 [214.5, 337.5] | 0.48 |
| Platelet/lymphocyte ratio | 120.5 [96.3, 148.4] | 141.8 [110.0, 184.2] | 0.07 | 138.7 [109.5, 164.4] | 150.1 [114.5, 201.6] | 0.43 |
| Fibrinogen (g/L) | 3.5 [3.0, 4.2] | 3.9 [3.4, 4.6] | 0.06 | 3.7 [3.4, 4.6] | 4.3 [3.8, 5.0] | 0.08 |
| Fibrinogen/lymphocyte ratio | 1.6 [1.2, 2.1] | 1.9 [1.7, 2.8] | 0.001 | 1.9 [1.6, 2.7] | 2.3 [1.7, 3.7] | 0.47 |
| D-dimer (mg/L FEU) | 0.3 [0.3, 0.5] | 0.3 [0.3, 0.5] | 0.73 | 0.3 [0.3, 0.4] | 0.4 [0.3, 0.7] | 0.33 |
| Factor VIII (%) | 148.0 [112.5, 190.5] | 162.0 [121.5, 202.5] | 0.21 | 158.0 [128.0, 198.0] | 166.0 [113.0, 214.0] | 0.76 |
| von-Willebrand antigen (%) | 148.5 (101.0, 192.0] | 159.5 (118.5, 200.0] | 0.51 | 162.0 (122.0, 200.0] | 147.0 (100.0, 200.0] | 0.56 |
| OCP (units) | 38.8 [32.1, 48.1] | 45.8 [39.3, 52.8] | 0.010 | 44.3 [40.5, 55.8] | 49.3 [32.8, 52.5] | 0.71 |
| OHP (units) | 9.2 [6.9, 13.2] | 12.8 [8.8, 17.2] | 0.005 | 12.3 [8.8, 16.8] | 13.7 [9.2, 17.2] | 0.61 |
| OFP (%) | 75.6 [71.0, 80.5] | 74.1 [64.9, 77.3] | 0.050 | 75.2 [64.9, 77.3] | 72.7 [65.7, 76.9] | 0.46 |
| Odds Ratio | Standard Error | p-Value | 95% Confidence Interval of Odds Ratio | |
|---|---|---|---|---|
| Proximal DVT | 2.22 | 0.79 | 0.026 | 1.10–4.47 |
| History of varicose veins | 7.51 | 4.28 | <0.001 | 2.46–22.96 |
| NLR ≥ 2.6 | 2.35 | 0.95 | 0.035 | 1.06–5.18 |
| OHP > 13.0 units | 2.17 | 0.79 | 0.033 | 1.06–4.43 |
| Weight > 108 kg | 2.86 | 1.11 | 0.007 | 1.34–6.11 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
McLeod, B.; Lim, H.Y.; Nandurkar, H.; Ho, P.; Wang, J. Overall Hemostatic Potential Assay Detects Risk of Progression to Post-Thrombotic Syndrome in Anticoagulated Patients following Deep Vein Thrombosis. Diagnostics 2022, 12, 3165. https://doi.org/10.3390/diagnostics12123165
McLeod B, Lim HY, Nandurkar H, Ho P, Wang J. Overall Hemostatic Potential Assay Detects Risk of Progression to Post-Thrombotic Syndrome in Anticoagulated Patients following Deep Vein Thrombosis. Diagnostics. 2022; 12(12):3165. https://doi.org/10.3390/diagnostics12123165
Chicago/Turabian StyleMcLeod, Blake, Hui Yin Lim, Harshal Nandurkar, Prahlad Ho, and Julie Wang. 2022. "Overall Hemostatic Potential Assay Detects Risk of Progression to Post-Thrombotic Syndrome in Anticoagulated Patients following Deep Vein Thrombosis" Diagnostics 12, no. 12: 3165. https://doi.org/10.3390/diagnostics12123165
APA StyleMcLeod, B., Lim, H. Y., Nandurkar, H., Ho, P., & Wang, J. (2022). Overall Hemostatic Potential Assay Detects Risk of Progression to Post-Thrombotic Syndrome in Anticoagulated Patients following Deep Vein Thrombosis. Diagnostics, 12(12), 3165. https://doi.org/10.3390/diagnostics12123165







