Advanced Magnetic Resonance Imaging Modalities for Breast Cancer Diagnosis: An Overview of Recent Findings and Perspectives
Abstract
1. Introduction
2. DWI in the Field of Diagnostic Breast Cancer
3. Comparison of DWI with Other Modalities
4. Different Models in DWI
5. DWI in Treatment Evaluation of Breast Cancer
6. DTI in the Diagnosis of Breast Cancer
7. DTI in Treatment Evaluation of Breast Cancer
8. Amide Proton Transfer-Weighted Imaging in Breast Cancer Diagnosis
9. Diffusion Kurtosis Imaging in Breast Cancer Diagnosis
10. Magnetic Resonance Spectroscopy
11. Perspectives (Future Directions)
12. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Khaniabadi, P.M.; Shahbazi-Gahrouei, D.; Aziz, A.A.; Dheyab, M.A.; Khaniabadi, B.M.; Mehrdel, B.; Jameel, M.S. Trastuzumab conjugated porphyrin-superparamagnetic iron oxide nanoparticle: A potential PTT-MRI bimodal agent for herceptin positive breast cancer. Photodiagnosis Photodyn. Ther. 2020, 31, 101896. [Google Scholar] [CrossRef] [PubMed]
- Shahbazi-Gahrouei, D.; Khaniabadi, P.M.; Khaniabadi, B.M.; Shahbazi-Gahrouei, S. Medical imaging modalities using nanoprobes for cancer diagnosis: A literature review on recent findings. J. Res. Med. Sci. Off. J. Isfahan Univ. Med. Sci. 2019, 24, 38. [Google Scholar] [CrossRef] [PubMed]
- Pereira, F.P.A.; Martins, G.; de Oliveira, R.d.V.C. Diffusion magnetic resonance imaging of the breast. Magn. Reson. Imaging Clin. 2011, 19, 95–110. [Google Scholar] [CrossRef] [PubMed]
- Tsougos, I.; Bakosis, M.; Tsivaka, D.; Athanassiou, E.; Fezoulidis, I.; Arvanitis, D.; Vassiou, K. Diagnostic performance of quantitative diffusion tensor imaging for the differentiation of breast lesions at 3 T MRI. Clin. Imaging 2019, 53, 25–31. [Google Scholar] [CrossRef] [PubMed]
- Yabuuchi, H.; Matsuo, Y.; Sunami, S.; Kamitani, T.; Kawanami, S.; Setoguchi, T.; Sakai, S.; Hatakenaka, M.; Kubo, M.; Tokunaga, E. Detection of non-palpable breast cancer in asymptomatic women by using unenhanced diffusion-weighted and T2-weighted MR imaging: Comparison with mammography and dynamic contrast-enhanced MR imaging. Eur. Radiol. 2011, 21, 11–17. [Google Scholar] [CrossRef] [PubMed]
- Shahbazi-Gahrouei, D.; Khaniabadi, P.M.; Shahbazi-Gahrouei, S.; Khorasani, A.; Mahmoudi, F. A literature review on multimodality molecular imaging nanoprobes for cancer detection. Pol. J. Med. Phys. Eng. 2019, 25, 57–68. [Google Scholar] [CrossRef]
- Partridge, S.C.; Nissan, N.; Rahbar, H.; Kitsch, A.E.; Sigmund, E.E. Diffusion-weighted breast MRI: Clinical applications and emerging techniques. J. Magn. Reson. Imaging 2017, 45, 337–355. [Google Scholar] [CrossRef]
- Baliyan, V.; Das, C.J.; Sharma, R.; Gupta, A.K. Diffusion weighted imaging: Technique and applications. World J. Radiol. 2016, 8, 785. [Google Scholar] [CrossRef]
- Malayeri, A.A.; El Khouli, R.H.; Zaheer, A.; Jacobs, M.A.; Corona-Villalobos, C.P.; Kamel, I.R.; Macura, K.J. Principles and applications of diffusion-weighted imaging in cancer detection, staging, and treatment follow-up. Radiographics 2011, 31, 1773–1791. [Google Scholar] [CrossRef]
- Fohlin, H.; Bekkhus, T.; Sandström, J.; Fornander, T.; Nordenskjöld, B.; Carstensen, J.; Stål, O. RAB6C is an independent prognostic factor of estrogen receptor-positive/progesterone receptor-negative breast cancer. Oncol. Lett. 2020, 19, 52–60. [Google Scholar] [CrossRef]
- Horvat, J.V.; Bernard-Davila, B.; Helbich, T.H.; Zhang, M.; Morris, E.A.; Thakur, S.B.; Ochoa-Albiztegui, R.E.; Leithner, D.; Marino, M.A.; Baltzer, P.A. Diffusion-weighted imaging (DWI) with apparent diffusion coefficient (ADC) mapping as a quantitative imaging biomarker for prediction of immunohistochemical receptor status, proliferation rate, and molecular subtypes of breast cancer. J. Magn. Reson. Imaging 2019, 50, 836–846. [Google Scholar] [CrossRef] [PubMed]
- Suo, S.; Zhang, D.; Cheng, F.; Cao, M.; Hua, J.; Lu, J.; Xu, J. Added value of mean and entropy of apparent diffusion coefficient values for evaluating histologic phenotypes of invasive ductal breast cancer with MR imaging. Eur. Radiol. 2019, 29, 1425–1434. [Google Scholar] [CrossRef] [PubMed]
- Iima, M.; Kataoka, M.; Kanao, S.; Onishi, N.; Kawai, M.; Ohashi, A.; Sakaguchi, R.; Toi, M.; Togashi, K. Intravoxel incoherent motion and quantitative non-Gaussian diffusion MR imaging: Evaluation of the diagnostic and prognostic value of several markers of malignant and benign breast lesions. Radiology 2018, 287, 432–441. [Google Scholar] [CrossRef] [PubMed]
- Springer Jr, C.S. Using 1H2O MR to measure and map sodium pump activity in vivo. J. Magn. Reson. 2018, 291, 110–126. [Google Scholar] [CrossRef] [PubMed]
- Baltzer, P.; Dietzel, M.; Vag, T.; Mieczyslaw, G.; Camara, O.; Kaiser, W. Diffusion weighted imaging-useful in all kinds of lesions? A systematic review. Eur. Radiol 2009, 19, S765–S974. [Google Scholar]
- Dorrius, M.D.; Dijkstra, H.; Oudkerk, M.; Sijens, P.E. Effect of b value and pre-admission of contrast on diagnostic accuracy of 1.5-T breast DWI: A systematic review and meta-analysis. Eur. Radiol. 2014, 24, 2835–2847. [Google Scholar] [CrossRef]
- Benndorf, M.; Schelhorn, J.; Dietzel, M.; Kaiser, W.A.; Baltzer, P.A. Diffusion weighted imaging of liver lesions suspect for metastases: Apparent diffusion coefficient (ADC) values and lesion contrast are independent from Gd-EOB-DTPA administration. Eur. J. Radiol. 2012, 81, e849–e853. [Google Scholar] [CrossRef]
- Leithner, D.; Moy, L.; Morris, E.A.; Marino, M.A.; Helbich, T.H.; Pinker, K. Abbreviated MRI of the breast: Does it provide value? J. Magn. Reson. Imaging 2019, 49, e85–e100. [Google Scholar] [CrossRef]
- Kuhl, C.K.; Schrading, S.; Strobel, K.; Schild, H.H.; Hilgers, R.-D.; Bieling, H.B. Abbreviated breast magnetic resonance imaging (MRI): First postcontrast subtracted images and maximum-intensity projection—a novel approach to breast cancer screening with MRI. J. Clin. Oncol. 2014, 32, 2304–2310. [Google Scholar] [CrossRef]
- Yamada, T.; Kanemaki, Y.; Okamoto, S.; Nakajima, Y. Comparison of detectability of breast cancer by abbreviated breast MRI based on diffusion-weighted images and postcontrast MRI. Jpn. J. Radiol. 2018, 36, 331–339. [Google Scholar] [CrossRef]
- Shin, H.J.; Chae, E.Y.; Choi, W.J.; Ha, S.M.; Park, J.Y.; Shin, K.C.; Cha, J.H.; Kim, H.H. Diagnostic performance of fused diffusion-weighted imaging using unenhanced or postcontrast T1-weighted MR imaging in patients with breast cancer. Medicine 2016, 95, e3502. [Google Scholar] [CrossRef] [PubMed]
- Bickelhaupt, S.; Laun, F.B.; Tesdorff, J.; Lederer, W.; Daniel, H.; Stieber, A.; Delorme, S.; Schlemmer, H.-P. Fast and noninvasive characterization of suspicious lesions detected at breast cancer X-ray screening: Capability of diffusion-weighted MR imaging with MIPs. Radiology 2016, 278, 689–697. [Google Scholar] [CrossRef] [PubMed]
- Rajagopalan, V.; Jiang, Z.; Yue, G.H.; Radic, J.S.; Pioro, E.P.; Wylie, G.R.; Das, A. A Basic Introduction to Diffusion Tensor Imaging Mathematics and Image Processing Steps. Brain Disord 2017, 6, 2. [Google Scholar] [CrossRef]
- Wang, Y.; Zhang, X.-P.; Li, Y.-L.; Li, X.-T.; Hu, Y.; Cui, Y.; Sun, Y.-S.; Zhang, X.-Y. Optimization of the parameters for diffusion tensor magnetic resonance imaging data acquisition for breast fiber tractography at 1.5 T. Clin. Breast Cancer 2014, 14, 61–67. [Google Scholar] [CrossRef] [PubMed]
- Koay, C.G.; Chang, L.-C.; Carew, J.D.; Pierpaoli, C.; Basser, P.J. A unifying theoretical and algorithmic framework for least squares methods of estimation in diffusion tensor imaging. J. Magn. Reson. 2006, 182, 115–125. [Google Scholar] [CrossRef] [PubMed]
- Le Bihan, D.; Poupon, C.; Amadon, A.; Lethimonnier, F. Artifacts and pitfalls in diffusion MRI. J. Magn. Reson. Imaging Off. J. Int. Soc. Magn. Reson. Med. 2006, 24, 478–488. [Google Scholar] [CrossRef]
- Bogner, W.; Gruber, S.; Pinker, K.; Grabner, G.; Stadlbauer, A.; Weber, M.; Moser, E.; Helbich, T.H.; Trattnig, S. Diffusion-weighted MR for differentiation of breast lesions at 3.0 T: How does selection of diffusion protocols affect diagnosis? Radiology 2009, 253, 341–351. [Google Scholar] [CrossRef]
- Iima, M.; Honda, M.; Sigmund, E.E.; Ohno Kishimoto, A.; Kataoka, M.; Togashi, K. Diffusion MRI of the breast: Current status and future directions. J. Magn. Reson. Imaging 2020, 52, 70–90. [Google Scholar] [CrossRef]
- Sinha, S.; Lucas-Quesada, F.A.; Sinha, U.; DeBruhl, N.; Bassett, L.W. In vivo diffusion-weighted MRI of the breast: Potential for lesion characterization. J. Magn. Reson. Imaging: Off. J. Int. Soc. Magn. Reson. Med. 2002, 15, 693–704. [Google Scholar] [CrossRef]
- Geijer, B.; Sundgren, P.; Lindgren, A.; Brockstedt, S.; Ståhlberg, F.; Holtås, S. The value of b required to avoid T2 shine-through from old lacunar infarcts in diffusion-weighted imaging. Neuroradiology 2001, 43, 511–517. [Google Scholar] [CrossRef]
- Ohlmeyer, S.; Laun, F.B.; Bickelhaupt, S.; Palm, T.; Janka, R.; Weiland, E.; Uder, M.; Wenkel, E. Ultra-High b-Value Diffusion-Weighted Imaging-Based Abbreviated Protocols for Breast Cancer Detection. Investig. Radiol. 2021, 56, 629–636. [Google Scholar] [CrossRef] [PubMed]
- Amornsiripanitch, N.; Bickelhaupt, S.; Shin, H.J.; Dang, M.; Rahbar, H.; Pinker, K.; Partridge, S.C. Diffusion-weighted MRI for unenhanced breast cancer screening. Radiology 2019, 293, 504–520. [Google Scholar] [CrossRef]
- Baltzer, P.; Mann, R.M.; Iima, M.; Sigmund, E.E.; Clauser, P.; Gilbert, F.J.; Martincich, L.; Partridge, S.C.; Patterson, A.; Pinker, K. Diffusion-weighted imaging of the breast—a consensus and mission statement from the EUSOBI International Breast Diffusion-Weighted Imaging working group. Eur. Radiol. 2020, 30, 1436–1450. [Google Scholar] [CrossRef]
- Iima, M.; Partridge, S.C.; Le Bihan, D. Six DWI questions you always wanted to know but were afraid to ask: Clinical relevance for breast diffusion MRI. Eur. Radiol. 2020, 30, 2561–2570. [Google Scholar] [CrossRef] [PubMed]
- Mori, M.; Fujioka, T.; Katsuta, L.; Yashima, Y.; Nomura, K.; Yamaga, E.; Hosoya, T.; Oda, G.; Nakagawa, T.; Kubota, K.; et al. Clinical usefulness of the fast protocol of breast diffusion-weighted imaging using 3T magnetic resonance imaging with a 16-channel breast coil. Clin. Imaging 2021, 78, 217–222. [Google Scholar] [CrossRef]
- Huang, S.Y.; Seethamraju, R.T.; Patel, P.; Hahn, P.F.; Kirsch, J.E.; Guimaraes, A.R. Body MR imaging: Artifacts, k-Space, and solutions. Radiographics 2015, 35, 1439–1460. [Google Scholar] [CrossRef] [PubMed]
- Liney, G.P.; Holloway, L.; Al Harthi, T.; Sidhom, M.; Moses, D.; Juresic, E.; Rai, R.; Manton, D.J. Quantitative evaluation of diffusion-weighted imaging techniques for the purposes of radiotherapy planning in the prostate. Br. J. Radiol. 2015, 88, 20150034. [Google Scholar] [CrossRef]
- Gatidis, S.; Graf, H.; Weiß, J.; Stemmer, A.; Kiefer, B.; Nikolaou, K.; Notohamiprodjo, M.; Martirosian, P. Diffusion-weighted echo planar MR imaging of the neck at 3 T using integrated shimming: Comparison of MR sequence techniques for reducing artifacts caused by magnetic-field inhomogeneities. Magn. Reson. Mater. Phys. Biol. Med. 2017, 30, 57–63. [Google Scholar] [CrossRef]
- Peng, Y.; Li, Z.; Tang, H.; Wang, Y.; Hu, X.; Shen, Y.; Hu, D. Comparison of reduced field-of-view diffusion-weighted imaging (DWI) and conventional DWI techniques in the assessment of rectal carcinoma at 3.0 T: Image quality and histological T staging. J. Magn. Reson. Imaging 2018, 47, 967–975. [Google Scholar] [CrossRef]
- Baxter, G.C.; Patterson, A.J.; Woitek, R.; Allajbeu, I.; Graves, M.J.; Gilbert, F. Improving the image quality of DWI in breast cancer: Comparison of multi-shot DWI using multiplexed sensitivity encoding to conventional single-shot echo-planar imaging DWI. Br. J. Radiol. 2020, 93, 20200427. [Google Scholar] [CrossRef]
- Park, J.Y.; Shin, H.J.; Shin, K.C.; Sung, Y.S.; Choi, W.J.; Chae, E.Y.; Cha, J.H.; Kim, H.H. Comparison of readout segmented echo planar imaging (EPI) and EPI with reduced field-of-view diffusion-weighted imaging at 3T in patients with breast cancer. J. Magn. Reson. Imaging 2015, 42, 1679–1688. [Google Scholar] [CrossRef] [PubMed]
- Kuhl, C.K.; Schrading, S.; Leutner, C.C.; Morakkabati-Spitz, N.; Wardelmann, E.; Fimmers, R.; Kuhn, W.; Schild, H.H. Mammography, breast ultrasound, and magnetic resonance imaging for surveillance of women at high familial risk for breast cancer. J. Clin. Oncol. 2005, 23, 8469–8476. [Google Scholar] [CrossRef] [PubMed]
- Lord, S.; Lei, W.; Craft, P.; Cawson, J.; Morris, I.; Walleser, S.; Griffiths, A.; Parker, S.; Houssami, N. A systematic review of the effectiveness of magnetic resonance imaging (MRI) as an addition to mammography and ultrasound in screening young women at high risk of breast cancer. Eur. J. Cancer 2007, 43, 1905–1917. [Google Scholar] [CrossRef] [PubMed]
- Bonelli, L.A.; Calabrese, M.; Belli, P.; Corcione, S.; Losio, C.; Montemezzi, S.; Pediconi, F.; Petrillo, A.; Zuiani, C.; Camera, L.; et al. MRI versus Mammography plus Ultrasound in Women at Intermediate Breast Cancer Risk: Study Design and Protocol of the MRIB Multicenter, Randomized, Controlled Trial. Diagnostics 2021, 11, 1635. [Google Scholar] [CrossRef]
- Gelardi, F.; Ragaini, E.M.; Sollini, M.; Bernardi, D.; Chiti, A. Contrast-Enhanced Mammography versus Breast Magnetic Resonance Imaging: A Systematic Review and Meta-Analysis. Diagnostics 2022, 12, 1890. [Google Scholar] [CrossRef]
- Zhang, H.; Zhang, X.Y.; Wang, Y. Value of magnetic resonance diffusion combined with perfusion imaging techniques for diagnosing potentially malignant breast lesions. World J. Clin. Cases 2022, 10, 6021–6031. [Google Scholar] [CrossRef]
- Lehman, C.D.; Isaacs, C.; Schnall, M.D.; Pisano, E.D.; Ascher, S.M.; Weatherall, P.T.; Bluemke, D.A.; Bowen, D.J.; Marcom, P.K.; Armstrong, D.K. Cancer yield of mammography, MR, and US in high-risk women: Prospective multi-institution breast cancer screening study. Radiology 2007, 244, 381–388. [Google Scholar] [CrossRef]
- Woodhams, R.; Matsunaga, K.; Iwabuchi, K.; Kan, S.; Hata, H.; Kuranami, M.; Watanabe, M.; Hayakawa, K. Diffusion-weighted imaging of malignant breast tumors: The usefulness of apparent diffusion coefficient (ADC) value and ADC map for the detection of malignant breast tumors and evaluation of cancer extension. J. Comput. Assist. Tomogr. 2005, 29, 644–649. [Google Scholar] [CrossRef]
- Yabuuchi, H.; Matsuo, Y.; Okafuji, T.; Kamitani, T.; Soeda, H.; Setoguchi, T.; Sakai, S.; Hatakenaka, M.; Kubo, M.; Sadanaga, N. Enhanced mass on contrast-enhanced breast MR imaging: Lesion characterization using combination of dynamic contrast-enhanced and diffusion-weighted MR images. J. Magn. Reson. Imaging Off. J. Int. Soc. Magn. Reson. Med. 2008, 28, 1157–1165. [Google Scholar] [CrossRef]
- Kuhl, C.K.; Keulers, A.; Strobel, K.; Schneider, H.; Gaisa, N.; Schrading, S. Not all false positive diagnoses are equal: On the prognostic implications of false-positive diagnoses made in breast MRI versus in mammography/digital tomosynthesis screening. Breast Cancer Res. 2018, 20, 1–9. [Google Scholar] [CrossRef]
- Zhang, M.; Horvat, J.V.; Bernard-Davila, B.; Marino, M.A.; Leithner, D.; Ochoa-Albiztegui, R.E.; Helbich, T.H.; Morris, E.A.; Thakur, S.; Pinker, K. Multiparametric MRI model with dynamic contrast-enhanced and diffusion-weighted imaging enables breast cancer diagnosis with high accuracy. J. Magn. Reson. Imaging 2019, 49, 864–874. [Google Scholar] [CrossRef] [PubMed]
- Egnell, L.; Vidić, I.; Jerome, N.P.; Bofin, A.M.; Bathen, T.F.; Goa, P.E. Stromal collagen content in breast tumors correlates with in vivo diffusion-weighted imaging: A comparison of multi b-Value DWI with histologic specimen from benign and malignant breast lesions. J. Magn. Reson. Imaging 2020, 51, 1868–1878. [Google Scholar] [CrossRef] [PubMed]
- Leibfarth, S.; Winter, R.M.; Lyng, H.; Zips, D.; Thorwarth, D. Potentials and challenges of diffusion-weighted magnetic resonance imaging in radiotherapy. Clin. Transl. Radiat. Oncol. 2018, 13, 29–37. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Wang, K.; Li, X.; Zhang, J.; Ding, J.; Spuhler, K.; Duong, T.; Liang, C.; Huang, C. Breast lesion characterization using whole-lesion histogram analysis with stretched-exponential diffusion model. J. Magn. Reson. Imaging 2018, 47, 1701–1710. [Google Scholar] [CrossRef]
- Vidić, I.; Egnell, L.; Jerome, N.P.; White, N.S.; Karunamuni, R.; Rakow-Penner, R.; Dale, A.M.; Bathen, T.F.; Goa, P.E. Modeling the diffusion-weighted imaging signal for breast lesions in the b = 200 to 3000 s/mm2 range: Quality of fit and classification accuracy for different representations. Magn. Reson. Med. 2020, 84, 1011–1023. [Google Scholar] [CrossRef]
- Bedair, R.; Priest, A.N.; Patterson, A.J.; McLean, M.A.; Graves, M.J.; Manavaki, R.; Gill, A.B.; Abeyakoon, O.; Griffiths, J.R.; Gilbert, F.J. Assessment of early treatment response to neoadjuvant chemotherapy in breast cancer using non-mono-exponential diffusion models: A feasibility study comparing the baseline and mid-treatment MRI examinations. Eur. Radiol. 2017, 27, 2726–2736. [Google Scholar] [CrossRef] [PubMed]
- Suo, S.; Cheng, F.; Cao, M.; Kang, J.; Wang, M.; Hua, J.; Hua, X.; Li, L.; Lu, Q.; Liu, J. Multiparametric diffusion-weighted imaging in breast lesions: Association with pathologic diagnosis and prognostic factors. J. Magn. Reson. Imaging 2017, 46, 740–750. [Google Scholar] [CrossRef] [PubMed]
- Camps-Herrero, J. Diffusion-weighted imaging of the breast: Current status as an imaging biomarker and future role. BJR| Open 2019, 1, 20180049. [Google Scholar] [CrossRef]
- Patterson, D.M.; Padhani, A.R.; Collins, D.J. Technology insight: Water diffusion MRI—A potential new biomarker of response to cancer therapy. Nat. Clin. Pract. Oncol. 2008, 5, 220–233. [Google Scholar] [CrossRef]
- Noij, D.P.; Pouwels, P.J.; Ljumanovic, R.; Knol, D.L.; Doornaert, P.; de Bree, R.; Castelijns, J.A.; de Graaf, P. Predictive value of diffusion-weighted imaging without and with including contrast-enhanced magnetic resonance imaging in image analysis of head and neck squamous cell carcinoma. Eur. J. Radiol. 2015, 84, 108–116. [Google Scholar] [CrossRef]
- Liu, H.; Zhan, H.; Zhang, Y.; He, G.; Wang, H.; Zhang, Q.; Zheng, L. Comparison of BSGI and MRI as Approaches to Evaluating Residual Tumor Status after Neoadjuvant Chemotherapy in Chinese Women with Breast Cancer. Diagnostics 2021, 11, 1846. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.; Kang, B.J.; Park, G.E.; Kim, S.H. The Usefulness of Magnetic Resonance Imaging (MRI) for the Detection of Local Recurrence after Mastectomy with Reconstructive Surgery in Breast Cancer Patients. Diagnostics 2022, 12, 2203. [Google Scholar] [CrossRef] [PubMed]
- Turan, U.; Aygun, M.; Duman, B.B.; Kelle, A.P.; Cavus, Y.; Tas, Z.A.; Dirim, A.B.; Irkorucu, O. Efficacy of US, MRI, and F-18 FDG-PET/CT for Detecting Axillary Lymph Node Metastasis after Neoadjuvant Chemotherapy in Breast Cancer Patients. Diagnostics 2021, 11, 2361. [Google Scholar] [CrossRef]
- Choi, J.H.; Lim, H.I.; Lee, S.K.; Kim, W.W.; Kim, S.M.; Cho, E.; Ko, E.Y.; Han, B.K.; Park, Y.H.; Ahn, J.S. The role of PET CT to evaluate the response to neoadjuvant chemotherapy in advanced breast cancer: Comparison with ultrasonography and magnetic resonance imaging. J. Surg. Oncol. 2010, 102, 392–397. [Google Scholar] [CrossRef] [PubMed]
- Chu, W.; Jin, W.; Liu, D.; Wang, J.; Geng, C.; Chen, L.; Huang, X. Diffusion-weighted imaging in identifying breast cancer pathological response to neoadjuvant chemotherapy: A meta-analysis. Oncotarget 2018, 9, 7088. [Google Scholar] [CrossRef]
- El Ameen, N.F.; Abdel Gawad, E.A.; Abdel Ghany, H.S. Diffusion-weighted imaging versus dynamic contrast-enhanced MRI: A new horizon for characterisation of suspicious breast lesions. Clin. Radiol. 2021, 76, 80.e81–80.e88. [Google Scholar] [CrossRef]
- van der Hoogt, K.J.J.; Schipper, R.J.; Winter-Warnars, G.A.; Ter Beek, L.C.; Loo, C.E.; Mann, R.M.; Beets-Tan, R.G.H. Factors affecting the value of diffusion-weighted imaging for identifying breast cancer patients with pathological complete response on neoadjuvant systemic therapy: A systematic review. Insights Into Imaging 2021, 12, 187. [Google Scholar] [CrossRef]
- Lai, H.-W.; Chen, C.-J.; Lin, Y.-J.; Chen, S.-L.; Wu, H.-K.; Wu, Y.-T.; Kuo, S.-J.; Chen, S.-T.; Chen, D.-R. Does Breast Magnetic Resonance Imaging Combined with Conventional Imaging Modalities Decrease the Rates of Surgical Margin Involvement and Reoperation?: A Case–Control Comparative Analysis. Medicine 2016, 95, e3810. [Google Scholar] [CrossRef]
- Hashem, L.M.B.; Sawy, Y.A.E.; Kamal, R.M.; Ahmed, S.M.; Elmesidy, D.S. The additive role of dynamic contrast-enhanced and diffusion-weighted MR imaging in preoperative staging of breast cancer. Egypt. J. Radiol. Nucl. Med. 2021, 52, 36. [Google Scholar] [CrossRef]
- Ergul, N.; Kadioglu, H.; Yildiz, S.; Yucel, S.B.; Gucin, Z.; Erdogan, E.B.; Aydin, M.; Muslumanoglu, M. Assessment of multifocality and axillary nodal involvement in early-stage breast cancer patients using 18F-FDG PET/CT compared to contrast-enhanced and diffusion-weighted magnetic resonance imaging and sentinel node biopsy. Acta Radiol. 2015, 56, 917–923. [Google Scholar] [CrossRef]
- Guo, Y.; Cai, Y.Q.; Cai, Z.L.; Gao, Y.G.; An, N.Y.; Ma, L.; Mahankali, S.; Gao, J.H. Differentiation of clinically benign and malignant breast lesions using diffusion-weighted imaging. J. Magn. Reson. Imaging Off. J. Int. Soc. Magn. Reson. Med. 2002, 16, 172–178. [Google Scholar] [CrossRef] [PubMed]
- Chhetri, A.; Li, X.; Rispoli, J.V. Current and emerging magnetic resonance-based techniques for breast cancer. Front. Med. 2020, 7, 175. [Google Scholar] [CrossRef] [PubMed]
- Le Bihan, D.; Mangin, J.F.; Poupon, C.; Clark, C.A.; Pappata, S.; Molko, N.; Chabriat, H. Diffusion tensor imaging: Concepts and applications. J. Magn. Reson. Imaging Off. J. Int. Soc. Magn. Reson. Med. 2001, 13, 534–546. [Google Scholar] [CrossRef]
- Plana, M.N.; Carreira, C.; Muriel, A.; Chiva, M.; Abraira, V.; Emparanza, J.I.; Bonfill, X.; Zamora, J. Magnetic resonance imaging in the preoperative assessment of patients with primary breast cancer: Systematic review of diagnostic accuracy and meta-analysis. Eur. Radiol. 2012, 22, 26–38. [Google Scholar] [CrossRef] [PubMed]
- Scaranelo, A.M.; Degani, H.; Grobgeld, D.; Talbot, N.; Bodolai, K.; Furman-Haran, E. Effect of IV Administration of a Gadolinium-Based Contrast Agent on Breast Diffusion-Tensor Imaging. Am. J. Roentgenol. 2020, 215, 1030–1036. [Google Scholar] [CrossRef] [PubMed]
- Nissan, N.; Allweis, T.; Menes, T.; Brodsky, A.; Paluch-Shimon, S.; Haas, I.; Golan, O.; Miller, Y.; Barlev, H.; Carmon, E. Breast MRI during lactation: Effects on tumor conspicuity using dynamic contrast-enhanced (DCE) in comparison with diffusion tensor imaging (DTI) parametric maps. Eur. Radiol. 2020, 30, 767–777. [Google Scholar] [CrossRef]
- Abdelhady, D.; Abdelbary, A.; Afifi, A.H.; Abdelhamid, A.E.; Hassan, H.H.M. Diffusion tensor imaging on 3-T MRI breast: Diagnostic performance in comparison to diffusion-weighted imaging. Egypt. J. Radiol. Nucl. Med. 2021, 52, 98. [Google Scholar] [CrossRef]
- Onaygil, C.; Kaya, H.; Ugurlu, M.U.; Aribal, E. Diagnostic performance of diffusion tensor imaging parameters in breast cancer and correlation with the prognostic factors. J. Magn. Reson. Imaging: JMRI 2017, 45, 660–672. [Google Scholar] [CrossRef]
- Furman-Haran, E.; Nissan, N.; Ricart-Selma, V.; Martinez-Rubio, C.; Degani, H.; Camps-Herrero, J. Quantitative evaluation of breast cancer response to neoadjuvant chemotherapy by diffusion tensor imaging: Initial results. J. Magn. Reson. Imaging 2018, 47, 1080–1090. [Google Scholar] [CrossRef]
- Mao, C.; Jiang, W.; Huang, J.; Wang, M.; Yan, X.; Yang, Z.; Wang, D.; Zhang, X.; Shen, J. Quantitative Parameters of Diffusion Spectrum Imaging: HER2 Status Prediction in Patients with Breast Cancer. Front. Oncol. 2022, 12, 817070. [Google Scholar] [CrossRef]
- Cho, E.; Baek, H.J.; Szczepankiewicz, F.; An, H.J.; Jung, E.J.; Lee, H.J.; Lee, J.; Gho, S.M. Clinical experience of tensor-valued diffusion encoding for microstructure imaging by diffusional variance decomposition in patients with breast cancer. Quant. Imaging Med. Surg. 2022, 12, 2002–2017. [Google Scholar] [CrossRef] [PubMed]
- Wilmes, L.J.; Li, W.; Shin, H.J.; Newitt, D.C.; Proctor, E.; Harnish, R.; Hylton, N.M. Diffusion tensor imaging for assessment of response to neoadjuvant chemotherapy in patients with breast cancer. Tomography 2016, 2, 438. [Google Scholar] [CrossRef] [PubMed]
- Menning, S.; de Ruiter, M.B.; Veltman, D.J.; Boogerd, W.; Oldenburg, H.S.; Reneman, L.; Schagen, S.B. Changes in brain white matter integrity after systemic treatment for breast cancer: A prospective longitudinal study. Brain Imaging Behav. 2018, 12, 324–334. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.; Payen, J.-F.; Wilson, D.A.; Traystman, R.J.; van Zijl, P. Using the amide proton signals of intracellular proteins and peptides to detect pH effects in MRI. Nat. Med. 2003, 9, 1085–1090. [Google Scholar] [CrossRef]
- Zaric, O.; Farr, A.; Rodriguez, E.P.; Mlynarik, V.; Bogner, W.; Gruber, S.; Asseryanis, E.; Singer, C.F.; Trattnig, S. 7T CEST MRI: A potential imaging tool for the assessment of tumor grade and cell proliferation in breast cancer. Magn. Reson. Imaging 2019, 59, 77–87. [Google Scholar] [CrossRef]
- Crescenzi, R.; Donahue, P.M.; Mahany, H.; Lants, S.K.; Donahue, M.J. CEST MRI quantification procedures for breast cancer treatment-related lymphedema therapy evaluation. Magn. Reson. Med. 2020, 83, 1760–1773. [Google Scholar] [CrossRef]
- Someya, Y.; Iima, M.; Imai, H.; Yoshizawa, A.; Kataoka, M.; Isoda, H.; Le Bihan, D.; Nakamoto, Y. Investigation of breast cancer microstructure and microvasculature from time-dependent DWI and CEST in correlation with histological biomarkers. Sci. Rep. 2022, 12, 6523. [Google Scholar] [CrossRef]
- Meng, N.; Wang, X.; Sun, J.; Han, D.; Bai, Y.; Wei, W.; Wang, Z.; Jia, F.; Wang, K.; Wang, M. A comparative study of the value of amide proton transfer-weighted imaging and diffusion kurtosis imaging in the diagnosis and evaluation of breast cancer. Eur. Radiol. 2021, 31, 1707–1717. [Google Scholar] [CrossRef]
- Jensen, J.H.; Helpern, J.A.; Ramani, A.; Lu, H.; Kaczynski, K. Diffusional kurtosis imaging: The quantification of non-gaussian water diffusion by means of magnetic resonance imaging. Magn. Reson. Med. Off. J. Int. Soc. Magn. Reson. Med. 2005, 53, 1432–1440. [Google Scholar] [CrossRef]
- Jensen, J.H.; Helpern, J.A. MRI quantification of non-Gaussian water diffusion by kurtosis analysis. NMR Biomed. 2010, 23, 698–710. [Google Scholar] [CrossRef]
- Wang, W.; Zhang, X.; Zhu, L.; Chen, Y.; Dou, W.; Zhao, F.; Zhou, Z.; Sun, Z. Prediction of Prognostic Factors and Genotypes in Patients with Breast Cancer Using Multiple Mathematical Models of MR Diffusion Imaging. Front. Oncol. 2022, 12, 825264. [Google Scholar] [CrossRef] [PubMed]
- Fardanesh, R.; Marino, M.A.; Avendano, D.; Leithner, D.; Pinker, K.; Thakur, S.B. Proton MR spectroscopy in the breast: Technical innovations and clinical applications. J. Magn. Reson. Imaging 2019, 50, 1033–1046. [Google Scholar] [CrossRef] [PubMed]
- Baltzer, P.A.; Dietzel, M. Breast lesions: Diagnosis by using proton MR spectroscopy at 1.5 and 3.0 T—systematic review and meta-analysis. Radiology 2013, 267, 735–746. [Google Scholar] [CrossRef] [PubMed]
- Sharma, U.; Jagannathan, N.R. In vivo MR spectroscopy for breast cancer diagnosis. BJR Open 2019, 1, 20180040. [Google Scholar] [CrossRef]
- Galati, F.; Luciani, M.L.; Caramanico, C.; Moffa, G.; Catalano, C.; Pediconi, F. Breast magnetic resonance spectroscopy at 3 T in biopsy-proven breast cancers: Does Choline peak correlate with prognostic factors? Investig. Radiol. 2019, 54, 767–773. [Google Scholar] [CrossRef]
- Thakur, S.B.; Horvat, J.V.; Hancu, I.; Sutton, O.M.; Bernard-Davila, B.; Weber, M.; Oh, J.H.; Marino, M.A.; Avendano, D.; Leithner, D. Quantitative in vivo proton MR spectroscopic assessment of lipid metabolism: Value for breast cancer diagnosis and prognosis. J. Magn. Reson. Imaging 2019, 50, 239–249. [Google Scholar] [CrossRef]
- Prvulovic Bunovic, N.; Sveljo, O.; Kozic, D.; Boban, J. Is Elevated Choline on Magnetic Resonance Spectroscopy a Reliable Marker of Breast Lesion Malignancy? Front. Oncol. 2021, 11, 610354. [Google Scholar] [CrossRef]
- Chen, L.; Liu, M.; Bao, J.; Xia, Y.; Zhang, J.; Zhang, L.; Huang, X.; Wang, J. The correlation between apparent diffusion coefficient and tumor cellularity in patients: A meta-analysis. PLoS ONE 2013, 8, e79008. [Google Scholar] [CrossRef]
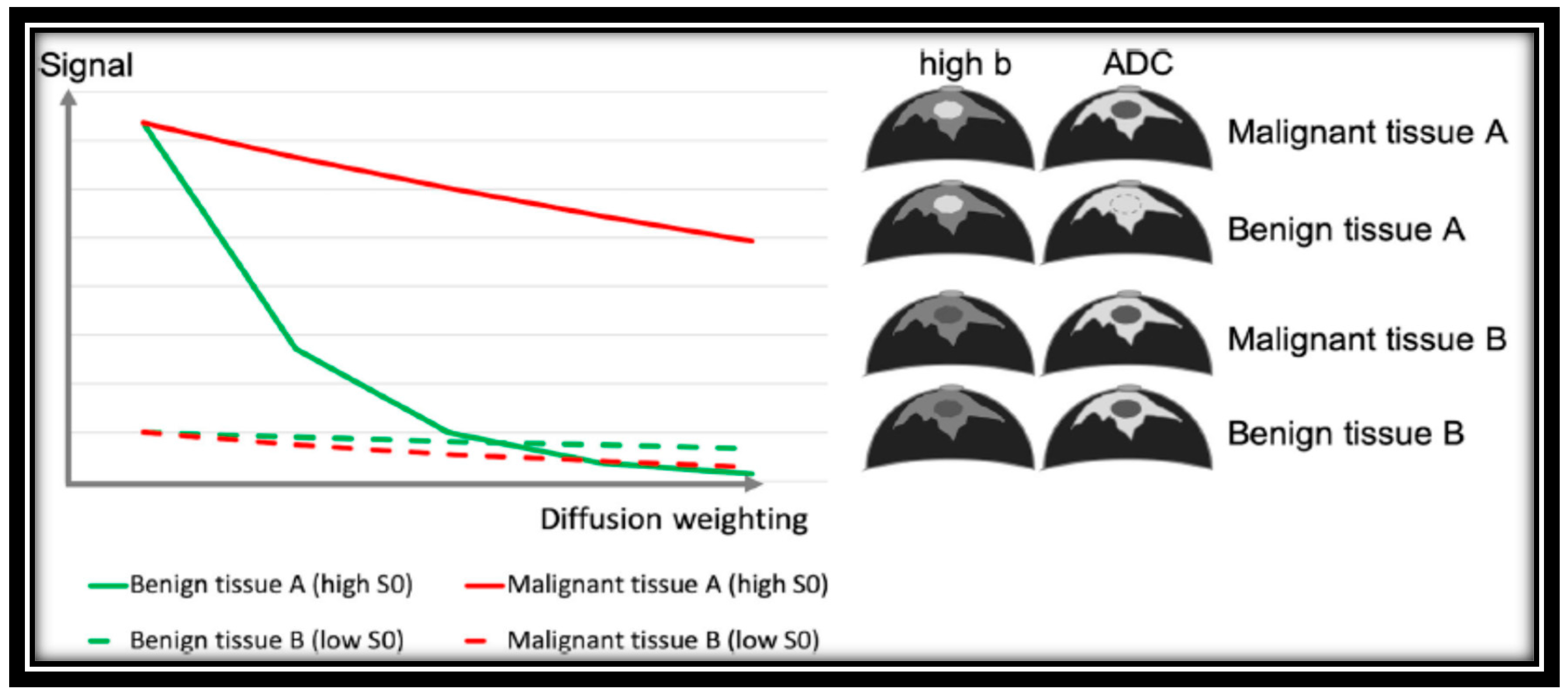
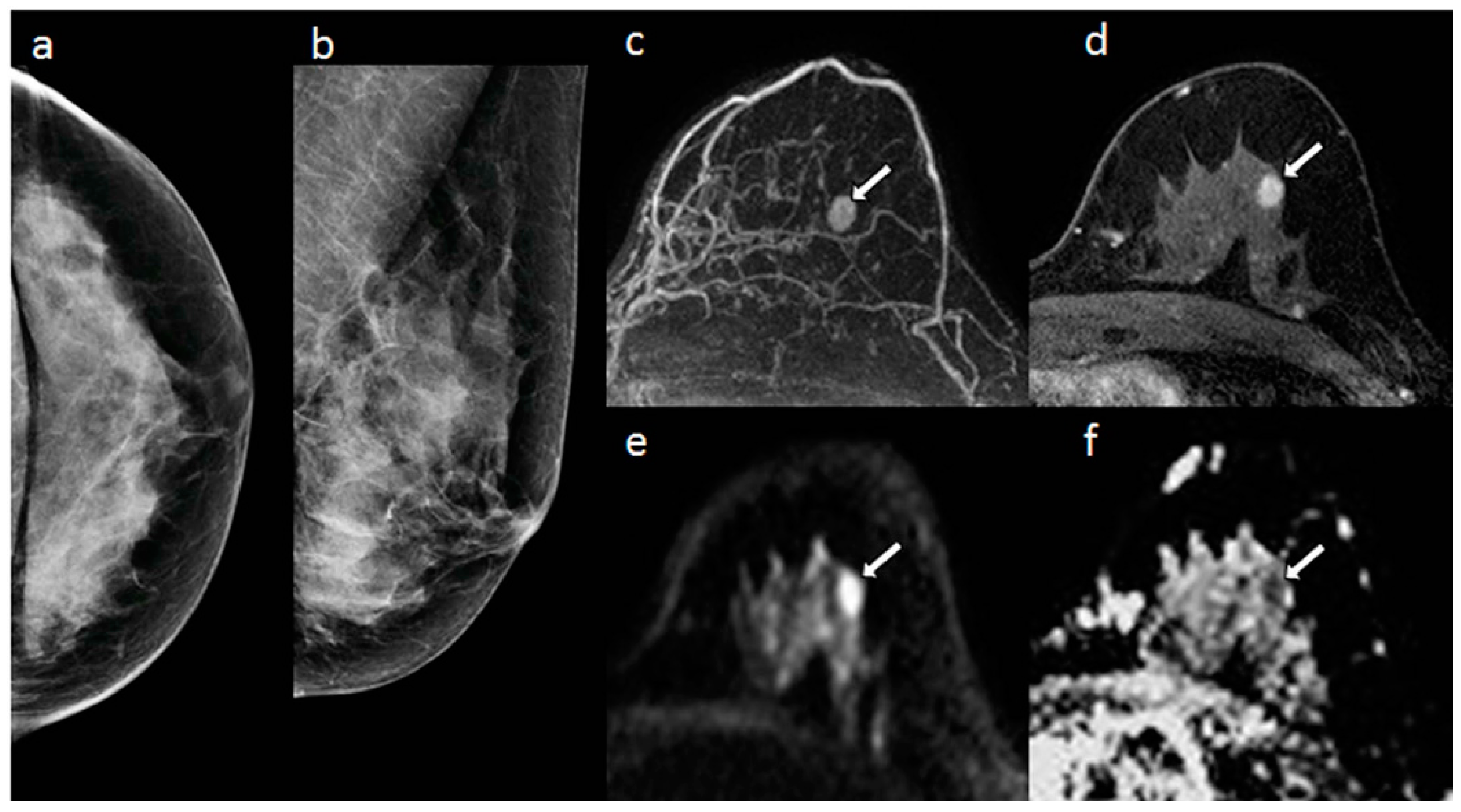

| Ref. | λ1 | λ2 | λ3 | MD | FA | λ1–λ3 |
|---|---|---|---|---|---|---|
| Noam Nissan et al. [76] in pregnancy-associated breast cancer | 1.17 ± 0.11 | 0.95 ± 0.11 | 0.74 ± 0.11 | 0.95 ± 0.11 | 0.25 ± 0.05± | 0.43 ± 0.07 |
| Haran et al. [79] (median % change responders) | 55.7 (43.6–77) | 55.4 (42.3–74.2) | 61.5 (41.3–81.0) | 55.6 (42.4–71.8) | 1.3 (214.3–20.8) | 55.4 (42.4–100.1) |
| Onaygil et al. [78] | 1.91 ± 0.30 * 1.27 ± 0.19 ** | 1.68 ± 0.28 * 1.01 ± 0.20 ** | 1.46 ± 0.27 * 0.81 ± 0.24 ** | 1.68 ± 0.27 * 1.03 ± 0.19 ** | 0.14 ± 0.05 * 0.24 ± 0.14 ** | 0.45 ± 0.17 * 0.48 ± 0.25 ** |
| Ref. | (ADC: ×103 mm2/s) Malignant | (ADC: ×103 mm2/s) Benign |
|---|---|---|
| Egnell et al. [52] b-values (0, 200, 600, 1200, 1800, 2400, 3000) s/mm2 | =1.04 (0.96–1.20) | =1.75 (1.51–1.86) |
| Pereira et al. [3] b-values (0, 250, 500, 750, and 1000) | 0.907 | 1.45 |
| Sinha et al. [29] | 1.36 ± 0.36 | 2.01 ± 0.46 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shahbazi-Gahrouei, D.; Aminolroayaei, F.; Nematollahi, H.; Ghaderian, M.; Gahrouei, S.S. Advanced Magnetic Resonance Imaging Modalities for Breast Cancer Diagnosis: An Overview of Recent Findings and Perspectives. Diagnostics 2022, 12, 2741. https://doi.org/10.3390/diagnostics12112741
Shahbazi-Gahrouei D, Aminolroayaei F, Nematollahi H, Ghaderian M, Gahrouei SS. Advanced Magnetic Resonance Imaging Modalities for Breast Cancer Diagnosis: An Overview of Recent Findings and Perspectives. Diagnostics. 2022; 12(11):2741. https://doi.org/10.3390/diagnostics12112741
Chicago/Turabian StyleShahbazi-Gahrouei, Daryoush, Fahimeh Aminolroayaei, Hamide Nematollahi, Mohammad Ghaderian, and Sogand Shahbazi Gahrouei. 2022. "Advanced Magnetic Resonance Imaging Modalities for Breast Cancer Diagnosis: An Overview of Recent Findings and Perspectives" Diagnostics 12, no. 11: 2741. https://doi.org/10.3390/diagnostics12112741
APA StyleShahbazi-Gahrouei, D., Aminolroayaei, F., Nematollahi, H., Ghaderian, M., & Gahrouei, S. S. (2022). Advanced Magnetic Resonance Imaging Modalities for Breast Cancer Diagnosis: An Overview of Recent Findings and Perspectives. Diagnostics, 12(11), 2741. https://doi.org/10.3390/diagnostics12112741







