Demystifying Supervised Learning in Healthcare 4.0: A New Reality of Transforming Diagnostic Medicine
Abstract
1. Introduction
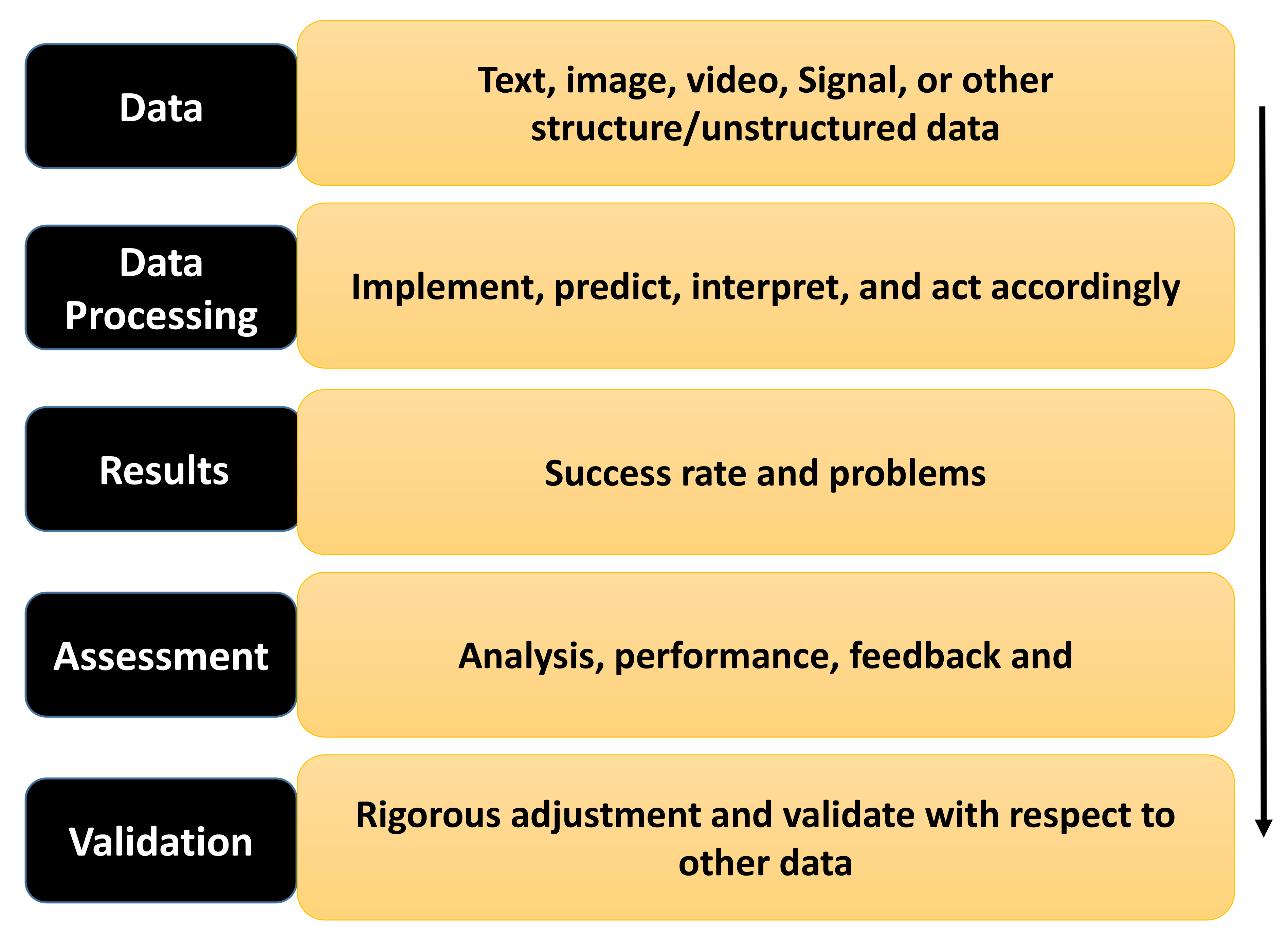
1.1. The Major Data Types Analyzed
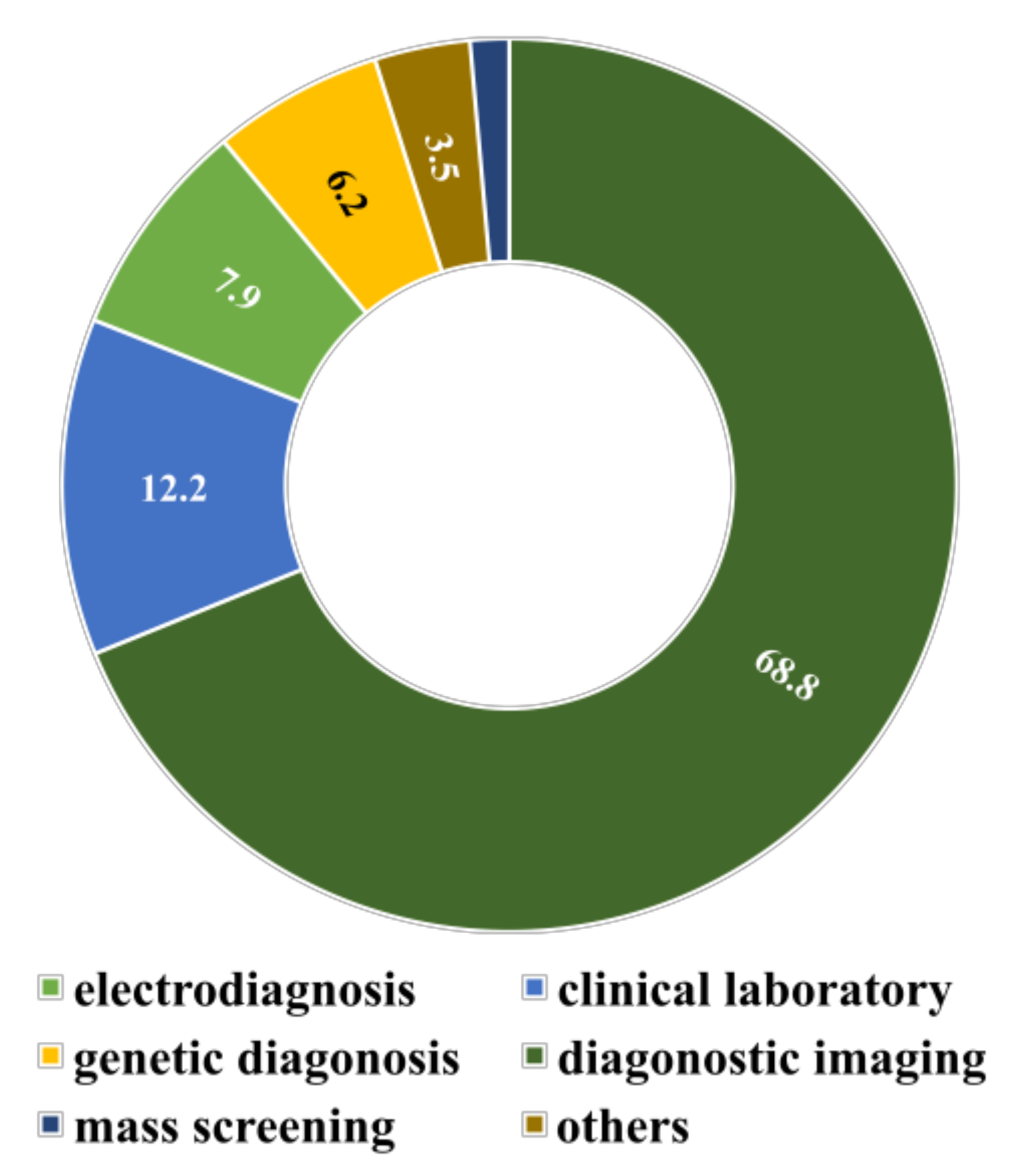
1.2. Major Disease Types Currently Tackled by SML
- Neurology: Movement of the cortical controller and its restoration in a human using SML was implemented with limited accuracy in patients with quadriplegia [11]. Later, a human and machine interface was created based on release time after re-innervation of the target muscle spinal motor to regulate the prostheses' upper limb [12].
- Cardiology: Heart disease diagnosis from cardiac images is a great application in SML. Arterys received authorization from Food and Drug Administration (FDA)-USA to market their cardio deep learning (DL) method that provides computerized, editable segmentations on cardiac MRI images [13].
1.3. Long-Term Prospect
2. Growing Research Areas
2.1. Connected/Augmented Care
2.1.1. AI-Based Chatbots
2.1.2. Ambient and Intelligent Care
2.1.3. Digital Consultation
2.1.4. Internet of Things for Patient Monitoring
2.2. Precision Diagnostics
2.2.1. Analytic Imaging
2.2.2. Diabetic Retinopathy
2.2.3. Faster Results with More Precision and Accuracy
2.2.4. Precision Therapeutics
2.2.5. Immunomics and Synthetic Biology
2.2.6. Cancer Diagnosis
2.3. Drug Discovery
2.4. Surgical Robotics
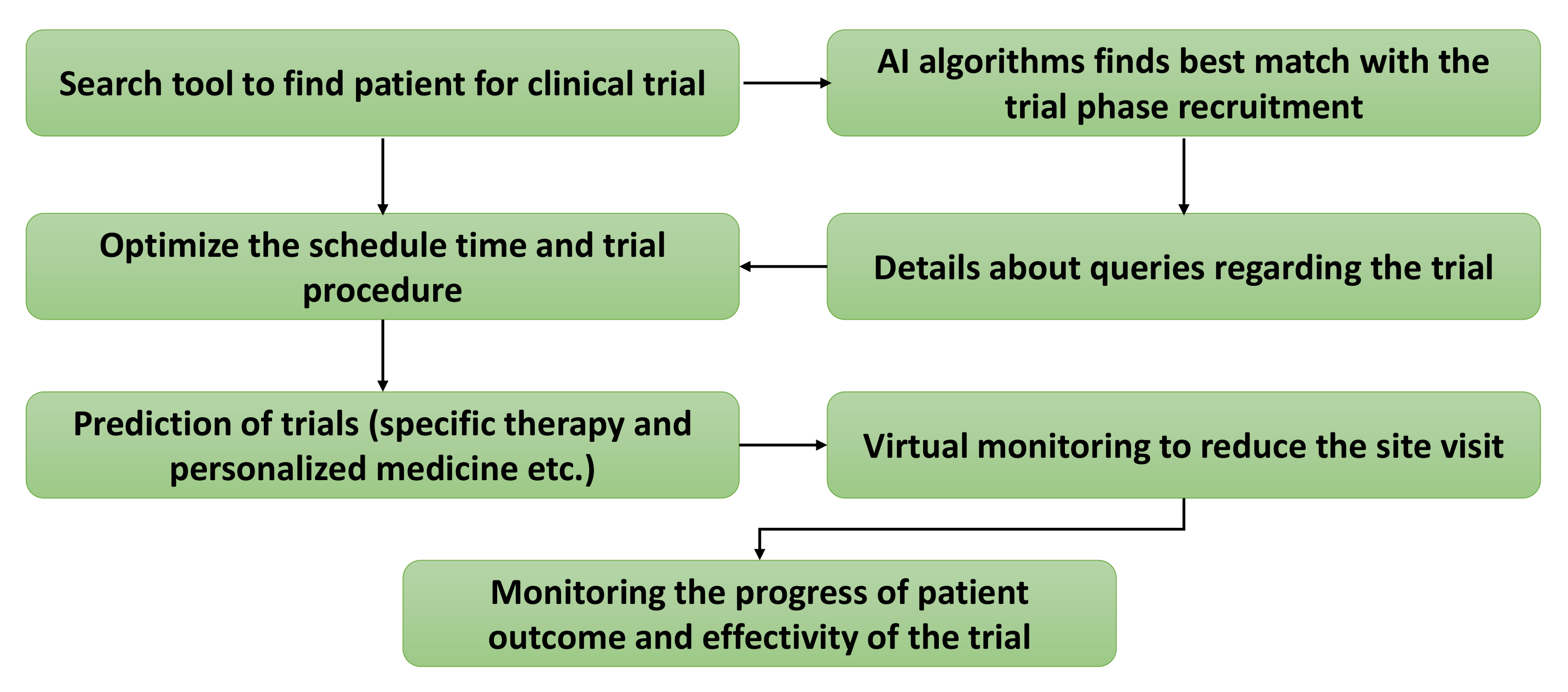
2.5. Clinical Trials
2.6. Nanotechnology Research
2.7. Prediction of Pandemic Outbreak
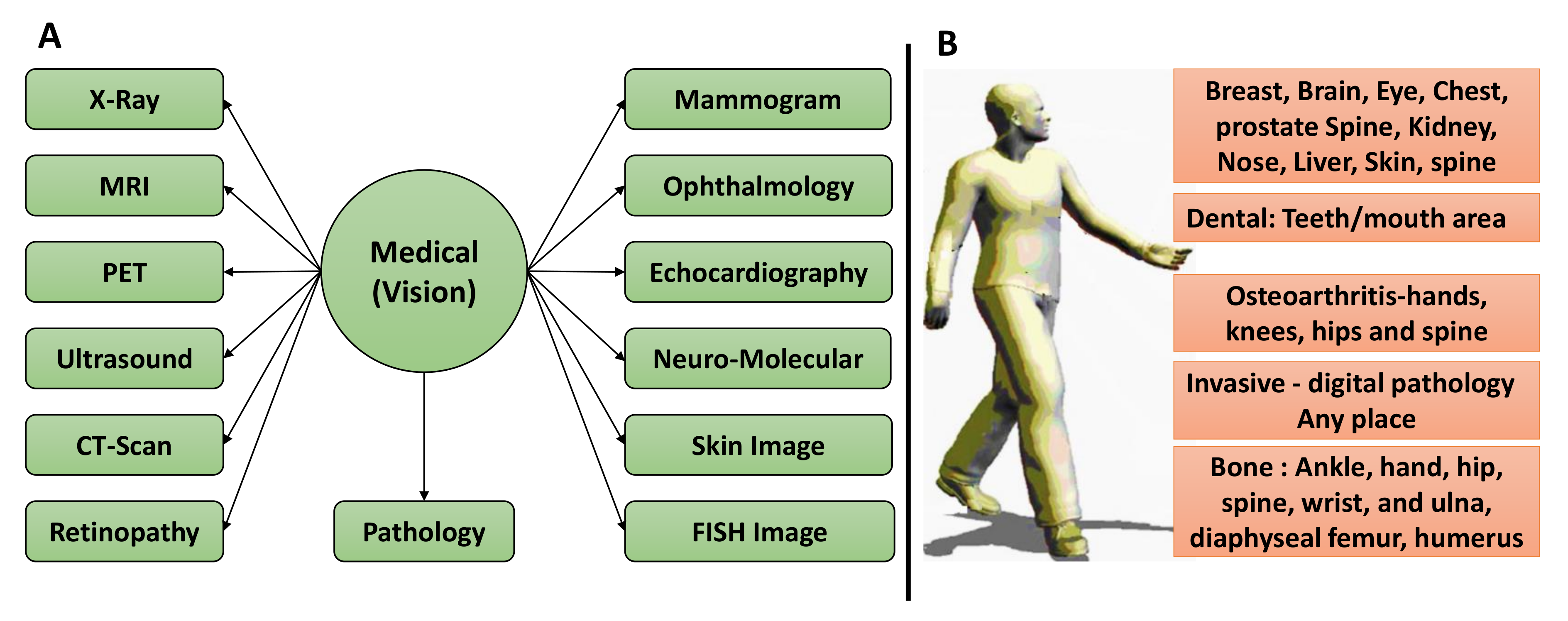
2.8. Computer Vision in Precision Medicine and Diagnostics from Medical Imaging
2.9. Advances in SML-Based Clinical Imaging
2.10. Current Stages (in Industry)
3. A Use Case of New Non-Invasive Diagnostics Development Approaches
4. Comparative Analysis
5. Challenges from the SML Implementation Side
5.1. Data Issues
5.2. Data-Snake Oil
5.3. Interdisciplinary Team Building
5.4. Reproducibility
5.5. Personalized Medicine
5.6. Moving into Clinical Practice
5.7. Use of Hospital Data
- Electronic health records: Such data is obtained at the point of care at a medical care facility or hospital. These data include administrative and demographic information, diagnosis, treatment, etc. Sites such as NIH and the Stanford center for clinical informatics provide access to such databases as mediated or collaborative access.
- Administrative data: These data are often associated with electronic health records and are reported to a government agency such as the Agency for Healthcare Research and Quality (AHRQ).
- Claims data: These data describe the billable interactions between insured patients and healthcare delivery systems. The sources of claims data can be obtained from the government, such as Medicare, and commercial health firms, such as United Healthcare.
- Patient/disease registries: These data include clinical information systems that track a narrow range of data for specific chronic conditions such as cancer, heart disease, and asthma.
- Health surveys: These consist of data which represents an accurate evaluation of population health and surveys conducted for common chronic conditions.
- Clinical trials, registries, and databases: ClinicalTrials.gov, Cochrane Library, European Union Clinical Trials Database clinical research datasets, The National Heart, Lung, and Blood Institute (NHLBI), Biomedical Translational Research Information System (BTRIS), and the National Institute of Mental Health (NIMH).
5.8. Data and Algorithms
5.9. Causal AI
5.10. Product Level Development
5.11. Effectiveness and Trust in AI-Augmented Healthcare
5.12. Precision Medicine
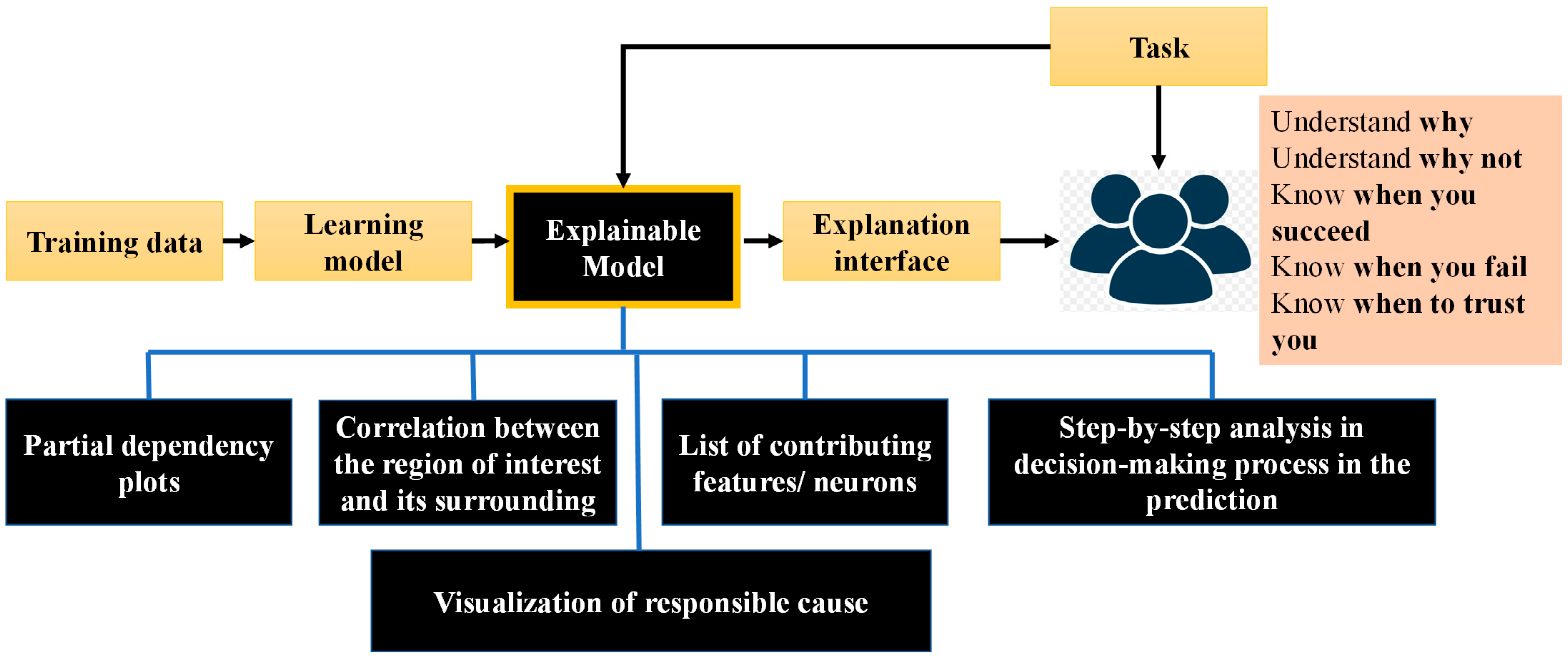
6. Explainability in Healthcare
7. Ethical Implications
8. Radiomics
9. The Future of SML in Biomedical and Healthcare
10. Conclusions and Discussion
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kumar, A.; Gadag, S.; Nayak, U.Y. The Beginning of a New Era: Artificial Intelligence in Healthcare. Adv. Pharm. Bull. 2021, 11, 414–425. [Google Scholar] [CrossRef] [PubMed]
- Zhavoronkov, A. Artificial intelligence for drug discovery, biomarker development, and generation of novel chemistry. Mol. Pharm. 2018, 15, 4311–4313. [Google Scholar] [CrossRef] [PubMed]
- Reddy, S.; Fox, J.; Purohit, M.P. Artificial intelligence-enabled healthcare delivery. J. R. Soc. Med. 2019, 112, 22–28. [Google Scholar] [CrossRef] [PubMed]
- Jiang, F.; Jiang, Y.; Zhi, H.; Dong, Y.; Li, H.; Ma, S.; Wang, Y.; Dong, Q.; Shen, H.; Wang, Y. Artificial intelligence in healthcare: Past, present and future. Stroke Vasc. Neurol. 2017, 2, 230–243. [Google Scholar] [CrossRef]
- Aggarwal, M.; Madhukar, M. IBM’s Watson analytics for health care: A miracle made true. In Cloud Computing Systems and Applications in Healthcare; IGI Global: Hershey, PA, USA, 2016. [Google Scholar]
- Velik, R. AI reloaded: Objectives, potentials, and challenges of the novel field of brain-like artificial intelligence. Brain 2012, 3, 25–54. [Google Scholar]
- Gbedemah, L. Introducing the Global AI Index. Available online: https://www.tortoisemedia.com/wp-content/uploads/sites/2/2020/02/191203_AI_Part1_v3.pdf (accessed on 18 June 2021).
- The Aims of AI—ST112 A2018. Available online: https://web.colby.edu/st112a2018/2018/04/14/the-aims-of-ai/ (accessed on 18 June 2021).
- Somashekhar, S.P.; Kumarc, R.; Rauthan, A.; Arun, K.R.; Patil, P.; Ramya, Y.E. Double blinded validation study to assess performance of IBM artificial intelligence platform. " Watson for Oncology in comparison with Manipal multidisciplinary tumour board—first study of 638. Cancer Res. 2017, 77, S6-07. [Google Scholar]
- Esteva, A.; Kuprel, B.; Novoa, R.A.; Ko, J.; Swetter, S.M.; Blau, H.M.; Thrun, S. Dermatologist-level classification of skin Cancer with deep neural networks. Nature 2017, 542, 115–118. [Google Scholar] [CrossRef]
- Bouton, C.E.; Shaikhouni, A.; Annetta, N.V.; Esteva, A.; Kuprel, B.; Novoa, R.A.; Ko, J.; Swetter, S.M.; Blau, H.M.; Thrun, S. Restoring cortical control of functional movement in a human with quadriplegia. Nature 2016, 533, 247–250. [Google Scholar] [CrossRef]
- Farina, D.; Vujaklija, I.; Sartori, M.; Kapelner, T.; Negro, F.; Jiang, N.; Bergmeister, K.; Andalib, A.; Principe, J.; Aszmann, O.C. Man/machine interface based on the discharge timings of spinal motor neurons after targeted muscle reinnervation. Nat. Biomed. Eng. 2017, 1, 0025. [Google Scholar] [CrossRef]
- Marr, B. First FDA approval for clinical Cloud-Based Deep Learning in Healthcare. 2017. Available online: https://www.forbes.com/sites/bernardmarr/2017/01/20/first-fda-approval-for-clinical-cloud-based-deep-learning-in-healthcare/#7a0ed8dc161c (accessed on 1 June 2017).
- Bajwa, J.; Munir, U.; Nori, A.; Williams, B. Artificial intelligence in healthcare: Transforming the practice of medicine. Future Healthc. J. 2021, 8, e188–e194. [Google Scholar] [CrossRef]
- Wang, A.; Nguyen, D.; Sridhar, A.R.; Gollakota, S. Using smart speakers to contactlessly monitor heart rhythms. Commun. Biol. 2021, 4, 319. [Google Scholar] [CrossRef] [PubMed]
- Fan, X.; Chao, D.; Zhang, Z.; Wang, D.; Li, X.; Tian, F. Utilization of Self-Diagnosis Health Chatbots in Real-World Settings: Case Study. J. Med. Internet Res. 2021, 23, e19928. [Google Scholar] [CrossRef] [PubMed]
- Schachner, T.; Keller, R.V.; Wangenheim, F. Artificial Intelligence-Based Conversational Agents for Chronic Conditions: Systematic Literature Review. J. Med. Internet Res. 2020, 22, e20701. [Google Scholar] [CrossRef] [PubMed]
- Kataria, S.; Ravindran, V. Musculoskeletal care—at the confluence of data science, sensors, engineering, and computation. BMC Musculoskelet. Disord. 2022, 23, 169. [Google Scholar] [CrossRef]
- Nascimento, L.M.S.d.; Bonfati, L.V.; Freitas, M.L.B.; Mendes Junior, J.J.A.; Siqueira, H.V.; Stevan, S.L., Jr. Sensors and Systems for Physical Rehabilitation and Health Monitoring—A Review. Sensors 2020, 20, 4063. [Google Scholar] [CrossRef]
- Mittal, A.; Kumar, D.; Mittal, M.; Saba, T.; Abunadi, I.; Rehman, A.; Roy, S. Detecting Pneumonia Using Convolutions and Dynamic Capsule Routing for Chest X-ray Images. Sensors 2020, 20, 1068. [Google Scholar] [CrossRef]
- Patel, B.; Dhayal, K.; Roy, S.; Shah, R. Computerized skin cancer lesion identification using the combination of clustering and entropy. In Proceedings of the 2017 International Conference on Big Data Analytics and Computational Intelligence (ICBDAC), Chirala, India, 23–25 March 2017; pp. 46–51. [Google Scholar] [CrossRef]
- Zhou, L.Q.; Wu, X.L.; Huang, S.Y.; Wu, G.G.; Ye, H.R.; Wei, Q.; Bao, L.Y.; Deng, Y.B.; Li, X.R.; Cui, X.W. Lymph Node Metastasis Prediction from Primary Breast Cancer US Images Using Deep Learning. Radiology 2020, 294, 19–28. [Google Scholar] [CrossRef]
- Chorba, J.S.; Shapiro, A.M.; Le, L.; Maidens, J.; Prince, J.; Pham, S.; Kanzawa, M.M.; Barbosa, D.N.; Currie, C.; Brooks, C.; et al. Deep Learning Algorithm for Automated Cardiac Murmur Detection via a Digital Stethoscope Platform. J. Am. Heart Assoc. 2021, 10, e019905. [Google Scholar] [CrossRef]
- Anirban, M.; Roy, S.; Purkait, R.; Konar, S.; Majumder, A.; Chatterjee, M.; Roy, S.; Kr Setua, S. Glaucoma Detection from Retinal Fundus Images Using RNFL Texture Analysis. In Advancement of Machine Intelligence in Interactive Medical Image Analysis, Algorithms for Intelligent Systems; Verma, O., Roy, S., Pandey, S.M., Eds.; Springer: Singapore, 2020. [Google Scholar] [CrossRef]
- Roy, S.; Mitra, A.; Roy, S.; Setua, S.K. Blood vessel segmentation of retinal image using Clifford matched filter and Clifford convolution. Multimed. Tools Appl. 2019, 78, 34839–34865. [Google Scholar] [CrossRef]
- Romeo, V.; Clauser, P.; Rasul, S.; Kapetas, P.; Gibbs, P.; Baltzer, P.A.T.; Hacker, M.; Woitek, R.; Helbich, T.H.; Pinker, K. AI-enhanced simultaneous multiparametric 18F-FDG PET/MRI for accurate breast cancer diagnosis. Eur. J. Nucl. Med. Mol. Imaging 2022, 49, 596–608. [Google Scholar] [CrossRef]
- Roy, S.; Bhattacharyya, D.; Bandyopadhyay, S.K.; Kim, T.H. An improved brain MR image binarization method as a preprocessing for abnormality detection and features extraction. Front. Comput. Sci. 2017, 11, 717–727. [Google Scholar] [CrossRef]
- Oktay, O.; Nanavati, J.; Schwaighofer, A.; Carter, D.; Bristow, M.; Tanno, R.; Jena, R.; Barnett, G.; Noble, D.; Rimmer, Y.; et al. Evaluation of Deep Learning to Augment Image-Guided Radiotherapy for Head and Neck and Prostate Cancers. JAMA Netw. Open 2020, 3, e2027426, Erratum in: JAMA Netw Open. 2020, 3, e2032624. [Google Scholar] [CrossRef] [PubMed]
- Biswas, N.; Chakrabarti, S. Artificial Intelligence (AI)-Based Systems Biology Approaches in Multi-Omics Data Analysis of Cancer. Front. Oncol. 2020, 10, 588221. [Google Scholar] [CrossRef]
- Bartel, M.J.; Brahmbhatt, B.S.; Wallace, M.B. Management of colorectal T1 carcinoma treated by endoscopic resection from the Western perspective. Dig. Endosc. 2016, 28, 330–341. [Google Scholar] [CrossRef]
- Kudo, S.E.; Ichimasa, K.; Villard, B.; Mori, Y.; Misawa, M.; Saito, S.; Hotta, K.; Saito, Y.; Matsuda, T.; Yamada, K.; et al. Artificial Intelligence System to Determine Risk of T1 Colorectal Cancer Metastasis to Lymph Node. Gastroenterology 2021, 160, 1075–1084.e2. [Google Scholar] [CrossRef] [PubMed]
- Georg, S.; Kriegsmann, M.; Zgorzelski, C.; Brobei, A.; Goeppert, B.; Dietrich, S.; Mechtersheimer, G.; Kriegsmann, K. Deep Learning for the Classification of Non-Hodgkin Lymphoma on Histopathological Images. Cancers 2021, 13, 2419. [Google Scholar] [CrossRef]
- Paul, D.; Sanap, G.; Shenoy, S.; Kalyane, D.; Kalia, K.; Tekade, R.K. Artificial intelligence in drug discovery and development. Drug Discov. Today 2021, 26, 80–93. [Google Scholar] [CrossRef] [PubMed]
- Bajorath, J.; Kearnes, S.; Walters, W.P.; Meanwell, N.A.; Georg, G.I.; Wang, S. Artificial Intelligence in Drug Discovery: Into the Great Wide Open. J. Med. Chem. 2020, 63, 8651–8652. [Google Scholar] [CrossRef]
- Kabiraj, A.; Pal, D.; Ganguly, D.; Chatterjee, K.; Roy, S. Number plate recognition from enhanced super-resolution using generative adversarial network. Multimed Tools Appl. 2022. [Google Scholar] [CrossRef]
- Himanshu, K.; Patel, M.; Roy, S. Prediction of arteriovenous nicking for hypertensive retinopathy using deep learning. In Computational Intelligence in Data Mining; Springer: Singapore, 2020; pp. 141–149. [Google Scholar]
- Flier, J.S. Academia and industry: Allocating credit for discovery and development of new therapies. J. Clin. Investig. 2019, 129, 2172–2174. [Google Scholar] [CrossRef]
- Rehman, A.; Harouni, M.; Karchegani NH, S.; Saba, T.; Bahaj, S.A.; Roy, S. Identity verification using palm print microscopic images based on median robust extended local binary pattern features and k-nearest neighbor classifier. Microsc. Res. Tech. 2022, 85, 1224–1237. [Google Scholar] [CrossRef] [PubMed]
- Martin, L.; Hutchens, M.; Hawkins, C. Trial watch: Clinical trial cycle times continue to increase despite industry efforts. Nat. Rev. Drug Discov. 2017, 16, 157. [Google Scholar] [CrossRef] [PubMed]
- Roy, S.; Whitehead, T.D.; Li, S.; Ademuyiwa, F.O.; Wahl, R.L.; Dehdashti, F.; Shoghi, K.I. Co-clinical FDG-PET radiomic signature in predicting response to neoadjuvant chemotherapy in triple-negative breast cancer. Eur. J. Nucl. Med. Mol. Imaging 2021, 49, 550–562, Erratum in: Eur. J. Nucl. Med. Mol. Imaging 2021. [Google Scholar] [CrossRef] [PubMed]
- Fisher, C.K.; Smith, A.M.; Walsh, J.R. Machine learning for comprehensive forecasting of Alzheimer’s Disease progression. Sci. Rep. 2019, 9, 13622. [Google Scholar] [CrossRef]
- Peng, Z.; Guo, Z.; Ullah, S.; Melagraki, G.; Afantitis, A.; Lynch, I. Nanotechnology and artificial intelligence to enable sustainable and precision agriculture. Nat. Plants 2021, 7, 864–876. [Google Scholar] [CrossRef]
- Lewis, T.G.; Al Mannai, W.I. Predicting the Size and Duration of the COVID-19 Pandemic. Front. Appl. Math. Stat. 2021, 6, 611854. [Google Scholar] [CrossRef]
- Shah, S.; Mulahuwaish, A.; Ghafoor, K.Z.; Maghdid, H.S. Prediction of global spread of COVID-19 pandemic: A review and research challenges. Artif. Intell. Rev. 2021, 55, 1607–1628. [Google Scholar] [CrossRef]
- Noorbakhsh-Sabet, N.; Zand, R.; Zhang, Y.; Abedi, V.R. Artificial Intelligence Transforms the Future of Health Care. Am. J. Med. 2019, 132, 795–801. [Google Scholar] [CrossRef]
- McCall, B. COVID-19 and artificial intelligence: Protecting health-care workers and curbing the spread. Lancet Digit. Health 2020, 2, e166–e167. [Google Scholar] [CrossRef]
- Choy, G.; Khalilzadeh, O.; Michalski, M.; Do, S.; Samir, A.E.; Pianykh, O.S.; Geis, J.R.; Pandharipande, P.V.; Brink, J.A.; Dreyer, K.J. Current Applications and Future Impact of Machine Learning in Radiology. Radiology 2018, 288, 318–328. [Google Scholar] [CrossRef]
- Roy, S.; Ghosh, P.; Bandyopadhyay, S.K. Contour Extraction and Segmentation of Cerebral Hemorrhage from MRI of Brain by Gamma Transformation Approach. In Advances in Intelligent Systems and Computing, Proceedings of the 3rd International Conference on Frontiers of Intelligent Computing: Theory and Applications (FICTA) 2014; Karnataka, I., Satapathy, S., Biswal, B., Udgata, S., Mandal, J., Eds.; Springer: Cham, Switzerland, 2015; Volume 328, p. 328. [Google Scholar] [CrossRef]
- Weston, A.D.; Korfiatis, P.; Kline, T.L.; Philbrick, K.A.; Kostandy, P.; Sakinis, T.; Sugimoto, M.; Takahashi, N.; Erickson, B.J. Automated Abdominal Segmentation of CT Scans for Body Composition Analysis Using Deep Learning. Radiology 2019, 290, 669–679. [Google Scholar] [CrossRef] [PubMed]
- Yash, D.; Choksi, S.; Roy, S. Prediction of two year survival among patients of non-small cell lung cancer. In Computer Aided Intervention and Diagnostics in Clinical and Medical Images; Springer: Cham, Switzerland, 2019; pp. 169–177. [Google Scholar]
- Suganyadevi, S.; Seethalakshmi, V.; Balasamy, K. A review on deep learning in medical image analysis. Int. J. Multimed. Info. Retr. 2022, 11, 19–38. [Google Scholar] [CrossRef] [PubMed]
- American College of Radiology Data Science Institute. FDA Cleared AI Algorithms. Available online: https://www.acrdsi.org/DSI-Services/FDA-Cleared-AI-Algorithms (accessed on 7 May 2020).
- Gruber, N.; Antholzer, S.; Jaschke, W.; Kremser, C.; Haltmeier, M. A Joint Deep Learning Approach for Automated Liver and Tumor Segmentation. 2019. Available online: https://arxiv.org/abs/1902.07971 (accessed on 18 December 2020).
- Rammah, Y.; Gupta, G.; Yousef, N.; Khari, M. A holistic overview of deep learning approach in medical imaging. Multimed. Syst. 2022, 28, 881–914. [Google Scholar] [CrossRef]
- Gulshan, V.; Peng, L.; Coram, M.; Stumpe, M.C.; Wu, D.; Narayanaswamy, A.; Venugopalan, S.; Widner, K.; Madams, T.; Cuadros, J. Development and Validation of a Deep Learning Algorithm for Detection of Diabetic Retinopathy in Retinal Fundus Photographs. JAMA 2016, 316, 2402–2410. [Google Scholar] [CrossRef]
- De Fauw, J.; Ledsam, J.R.; Romera-Paredes, B.; Nikolov, S.; Tomasev, N.; Blackwell, S.; Askham, H.; Glorot, X.; O’Donoghue, B.; Visentin, D. Clinically applicable deep learning for diagnosis and referral in retinal disease. Nat. Med. 2018, 24, 1342–1350. [Google Scholar] [CrossRef] [PubMed]
- Emmanuel, M.; Cerny, M.; Cadrin-Chênevert, A.; Hamilton, V.; Derennes, T.; Ilinca, A.; Vandenbroucke-Menu, F.; Turcotte, S.; Kadoury, S.; Tang, A. Deep learning workflow in radiology: A primer. Insights Imaging 2020, 11, 22. [Google Scholar] [CrossRef]
- Diaz, O.; Kushibar, K.; Osuala, R.; Linardos, A.; Garrucho, L.; Igual, L.; Radeva, P.; Prior, F.; Gkontra, P.; Lekadir, K. Data preparation for artificial intelligence in medical imaging: A comprehensive guide to open-access platforms and tools. Phys. Med. 2021, 83, 25–37. [Google Scholar] [CrossRef]
- Razzak, M.I.; Naz, S.; Zaib, A. Deep Learning for Medical Image Processing: Overview, Challenges and the Future. In Classification in BioApps. Lecture Notes in Computational Vision and Biomechanics; Dey, N., Ashour, A., Borra, S., Eds.; Springer: Cham, Switzerland, 2018; Volume 26. [Google Scholar] [CrossRef]
- Debojyoti, P.; Reddy, P.B.; Roy, S. Attention UW-Net: A fully connected model for automatic segmentation and annotation of chest X-ray. Comput. Biol. Med. 2022, 150, 106083. [Google Scholar]
- Ayhan, M.S.; Kümmerle, L.B.; Kühlewein, L.; Inhoffen, W.; Aliyeva, G.; Ziemssen, F.; Berens, P. Clinical validation of saliency maps for understanding deep neural networks in ophthalmology. Med. Image Anal. 2022, 77, 102364. [Google Scholar] [CrossRef]
- Santos, E.S.D.; Veras, R.d.M.S.; Aires, K.R.T.; Portela, H.M.B.F.; Junior, G.B.; Santos, J.D.; Tavares, J.M.R.S. Semi-automatic segmentation of skin lesions based on superpixels and hybrid texture information. Med. Image Anal. 2022, 77, 102363. [Google Scholar] [CrossRef]
- Amirata, G.; Ouyang, D.; Abid, A.; He, B.; Chen, J.H.; Harrington, R.A.; Liang, D.H.; Ashley, E.A.; Zou, J.Y. Deep learning interpretation of echocardiograms. NPJ Digit. Med. 2020, 3, 10. [Google Scholar] [CrossRef]
- Bulten, W.; Kartasalo, K.; Chen, P.H.C.; Ström, P.; Pinckaers, H.; Nagpal, K.; Cai, Y.; Steiner, D.F.; van Boven, H.; Vink, R. Artificial intelligence for diagnosis and Gleason grading of prostate cancer: The PANDA challenge. Nat. Med. 2022, 28, 154–163. [Google Scholar] [CrossRef] [PubMed]
- Kumar, P.R.; Lin, Y.; Agarwal, A.; Ho, M.; Yeh, J.A. A pilot study for the prediction of liver function related scores using breath biomarkers and machine learning. Sci. Rep. 2022, 12, 2032. [Google Scholar] [CrossRef]
- Dutta, K.; Roy, S.; Whitehead, T.D.; Luo, J.; Jha, A.K.; Li, S.; Quirk, J.D.; Shoghi, K.I. Deep Learning Segmentation of Triple-Negative Breast Cancer (TNBC) Patient Derived Tumor Xenograft (PDX) and Sensitivity of Radiomic Pipeline to Tumor Probability Boundary. Cancers 2021, 13, 3795. [Google Scholar] [CrossRef] [PubMed]
- Anand, I.; Negi, H.; Kumar, D.; Mittal, M.; Kim, T.H.; Roy, S. Residual u-network for breast tumor segmentation from magnetic resonance images. Comput. Mater. Contin. 2021, 67, 3107–3127. [Google Scholar] [CrossRef]
- Roy, S.; Whitehead, T.D.; Quirk, J.D.; Salter, A.; Ademuyiwa, F.O.; Li, S.; An, H.; Shoghi, K.I. Optimal co-clinical radiomics: Sensitivity of radiomic features to tumour volume, image noise and resolution in co-clinical T1-weighted and T2-weighted magnetic resonance imaging. EBioMedicine 2020, 59, 102963. [Google Scholar] [CrossRef]
- Varadarajan, A.V.; Bavishi, P.; Ruamviboonsuk, P.; Chotcomwongse, P.; Venugopalan, S.; Narayanaswamy, A.; Cuadros, J.; Kanai, K.; Bresnick, G.; Tadarati, M. Predicting optical coherence tomography-derived diabetic macular edema grades from fundus photographs using deep learning. Nat. Commun. 2020, 11, 130. [Google Scholar] [CrossRef]
- Chetty, G.; Yamin, M.; White, M. A low resource 3D U-Net based deep learning model for medical image analysis. Int. J. Inf. Tecnol. 2022, 14, 95–103. [Google Scholar] [CrossRef]
- Savaikar, M.A.; Whitehead, T.; Roy, S.; Strong, L.; Fettig, N.; Prmeau, T.; Luo, J.; Li, S.; Wahl, R.L.; Shoghi, K.I. Preclinical PERCIST and 25% of SUVmax Threshold: Precision Imaging of Response to Therapy in Co-clinical 18F-FDG PET Imaging of Triple-Negative Breast Cancer Patient-Derived Tumor Xenografts. J. Nucl. Med. 2020, 61, 842–849. [Google Scholar] [CrossRef]
- Wang, C.W.; Huang, S.C.; Lee, Y.C.; Shen, Y.J.; Meng, S.I.; Gaol, J.L. Deep learning for bone marrow cell detection and classification on whole-slide images. Med. Image Anal. 2022, 75, 102270. [Google Scholar] [CrossRef]
- Min, L.; Wang, S.; Chen, H.; Liu, Y. A pilot study of a deep learning approach to detect marginal bone loss around implants. BMC Oral Health 2022, 22, 11. [Google Scholar] [CrossRef]
- Wu-in, L.W.; Hong, S.; Nam, H.; Kang, W.; Yang, Z.; Noh, E. External validation of deep learning-based bone-age software: A preliminary study with real world data. Sci. Rep. 2022, 12, 1232. [Google Scholar] [CrossRef]
- Roy, S.; Bandyopadhyay, S.K. Abnormal regions detection and quantification with accuracy estimation from MRI of brain. In Proceedings of the 2013 2nd International Symposium on Instrumentation and Measurement, Sensor Network and Automation (IMSNA), Toronto, ON, Canada, 23–24 December 2013; IEEE: Piscataway, NJ, USA; pp. 611–615. [Google Scholar]
- Mall, P.K.; Singh, P.K. BoostNet: A method to enhance the performance of deep learning model on musculoskeletal radiographs X-ray images. Int. J. Syst. Assur. Eng. Manag. 2022, 13, 658–672. [Google Scholar] [CrossRef]
- Roy, S.; Bhattacharyya, D.; Bandyopadhyay, S.K.; Kim, T.H. An iterative implementation of level set for precise segmentation of brain tissues and abnormality detection from MR images. IETE J. Res. 2017, 63, 769–783. [Google Scholar] [CrossRef]
- Roy, S.; Bhattacharyya, D.; Bandyopadhyay, S.K.; Kim, T.H. An effective method for computerized prediction and segmentation of multiple sclerosis lesions in brain MRI. Comput. Methods Programs Biomed. 2017, 140, 307–320. [Google Scholar] [CrossRef]
- Guan, B.; Liu, F.; Mizaian, A.H.; Demehri, S.; Samsonov, A.; Guermazi, A.; Kijowski, R. Deep learning approach to predict pain progression in knee osteoarthritis. Skelet. Radiol. 2022, 51, 363–373. [Google Scholar] [CrossRef] [PubMed]
- Osamu, I.; Kanavati, F.; Kato, K.; Rambeau, M.; Arihiro, K.; Tsuneki, M. Deep Learning Models for Histopathological Classification of Gastric and Colonic Epithelial Tumours. Sci. Rep. 2020, 10, 1504. [Google Scholar] [CrossRef] [PubMed]
- Abbas, A.; Abdelsamea, M.M.; Gaber, M.M. Classification of COVID-19 in chest X-ray images using DeTraC deep convolutional neural network. Appl. Intell. 2021, 51, 854–864. [Google Scholar] [CrossRef]
- Wulczyn, E.; Steiner, D.F.; Xu, Z.; Sadhwani, A.; Wang, H.; Flament-Auvigne, I.; Mermel, C.H.; Chen, P.H.C.; Liu, Y.; Stumpe, M.C.; et al. Deep learning-based survival prediction for multiple cancer types using histopathology images. PLoS ONE 2020, 15, e0233678. [Google Scholar] [CrossRef]
- Sabanayagam, C.; Xu, D.; Ting, D.S.; Nusinovici, S.; Banu, R.; Hamzah, H.; Lim, C.; Tham, Y.C.; Cheung, C.Y.; Tai, E.S.; et al. A deep learning algorithm to detect chronic kidney disease from retinal photographs in community-based populations. Lancet Digit. Health 2020, 2, e295–e302. [Google Scholar] [CrossRef]
- Zhang, Y.Z.; Wang, Z.H.; Zhang, J.; Wang, C.C.; Wang, Y.S.; Chen, H.; Shan, L.H.; Huo, J.N.; Gu, J.H.; Ma, X. Deep learning model for classifying endometrial lesions. J. Transl. Med. 2021, 19, 10. [Google Scholar] [CrossRef] [PubMed]
- Pinochet, P.; Eude, F.; Becker, S.; Shah, V.; Sibille, L.; Toledano, M.N.; Modzelewski, R.; Vera, P.; Decazes, P. Evaluation of an Automatic Classification Algorithm Using Convolutional Neural Networks in Oncological Positron Emission Tomography. Front. Med. 2021, 8, 628179. [Google Scholar] [CrossRef] [PubMed]
- Sadik, M.; López-Urdaneta, J.; Ulén, J.; Enqvist, O.; Krupic, A.; Kumar, R.; Andersson, P.O.; Trägårdh, E. Artificial intelligence could alert for focal skeleton/bone marrow uptake in Hodgkin’s lymphoma patients staged with FDG-PET/CT. Sci. Rep. 2021, 11, 10382. [Google Scholar] [CrossRef] [PubMed]
- Venugopalan, J.; Tong, L.; Hassanzadeh, H.R.; Wang, M.D. Multimodal deep learning models for early detection of Alzheimer’s disease stage. Sci. Rep. 2021, 11, 3254. [Google Scholar] [CrossRef]
- Mohamed, E.A.; Rashed, E.A.; Gaber, T.; Karam, O. Deep learning model for fully automated breast cancer detection system from thermograms. PLoS ONE 2022, 17, e0262349. [Google Scholar] [CrossRef]
- TariqSadiq, M.; Aziz, M.Z.; Almogren, A.; Yousaf, A.; Siuly, S.; UrRehman, A. Exploiting pretrained CNN models for the development of an EEG-based robust BCI framework. Comput. Biol. Med. 2022, 143, 105242. [Google Scholar]
- Pereira, M.R.; Bertolini, D.; Teixeira, O.L.; Silla, N.C.; Costa, M.G.Y. COVID-19 identification in chest X-ray images on flat and hierarchical classification scenarios. Comput. Methods Programs Biomed. 2020, 194, 105532. [Google Scholar] [CrossRef]
- Bailly, A.; Blanc, C.; Francis, É.; Guillotin, T.; Jamal, F.; Wakim, B.; Roy, P. Effects of dataset size and interactions on the prediction performance of logistic regression and deep learning models. Comput. Methods Programs Biomed. 2022, 213, 106504. [Google Scholar] [CrossRef]
- Sánchez-Cauce, R.; Pérez-Martín, J.; Luque, M. Multi-input convolutional neural network for breast cancer detection using thermal images and clinical data. Comput. Methods Programs Biomed. 2021, 204, 106045. [Google Scholar] [CrossRef]
- Wallis, D.; Soussan, M.; Lacroix, M.; Akl, P.; Duboucher, C.; Buvat, I. An [18F]FDG-PET/CT deep learning method for fully automated detection of pathological mediastinal lymph nodes in lung cancer patients. Eur. J. Nucl. Med. Mol. Imaging 2022, 49, 881–888. [Google Scholar] [CrossRef]
- Kerstin, J.; Brynolfsson, J.; Sahlstedt, H.; Nickols, N.G.; Rettig, M.; Probst, S.; Morris, M.J.; Bjartell, A.; Eiber, M.; Anand, A. Analytical performance of a PROMISE: Automated anatomic contextualization, detection, and quantification of [18F]DCFPyL (PSMA) imaging for standardized reporting. Eur. J. Nucl. Med. Mol. Imaging 2022, 49, 1041–1051. [Google Scholar]
- Capobianco, N.; Sibille, L.; Chantadisai, M.; Gafita, A.; Langbein, T.; Platsch, G.; Solari, E.L.; Shah, V.; Spottiswoode, B.; Eiber, M.; et al. Whole-body uptake classification and prostate cancer staging in 68Ga-PSMA-11 PET/CT using dual-tracer learning. Eur. J. Nucl. Med. Mol. Imaging 2022, 49, 517–526. [Google Scholar] [CrossRef] [PubMed]
- Etminani, K.; Soliman, A.; Davidsson, A.; Chang, J.R.; Martínez-Sanchis, B.; Byttner, S.; Camacho, V.; Bauckneht, M.; Stegeran, R.; Ressner, M. A 3D deep learning model to predict the diagnosis of dementia with Lewy bodies, Alzheimer’s disease, and mild cognitive impairment using brain 18F-FDG PET. Eur. J. Nucl. Med. Mol. Imaging 2022, 49, 563–584. [Google Scholar] [CrossRef] [PubMed]
- Mehranian, A.; Wollenweber, S.D.; Walker, M.D.; Bradley, K.M.; Fielding, P.A.; Su, K.H.; Johnsen, R.; Kotasidis, F.; Jansen, F.P.; McGowan, D.R. Image enhancement of whole-body oncology [18F]-FDG PET scans using deep neural networks to reduce noise. Eur. J. Nucl. Med. Mol. Imaging 2022, 49, 539–549. [Google Scholar] [CrossRef]
- Xue, S.; Guo, R.; Bohn, K.P.; Matzke, J.; Viscione, M.; Alberts, I.; Meng, H.; Sun, C.; Zhang, M.; Zhang, M. A cross-scanner and cross-tracer deep learning method for the recovery of standard-dose imaging quality from low-dose PET. Eur. J. Nucl. Med. Mol. Imaging 2021, 49, 1843–1856. [Google Scholar] [CrossRef]
- Wang, L.; Ding, W.; Mo, Y.; Shi, D.; Zhang, S.; Zhong, L.; Wang, K.; Wang, J.; Huang, C.; Zhang, S. Distinguishing nontuberculous mycobacteria from Mycobacterium tuberculosis lung disease from CT images using a deep learning framework. Eur. J. Nucl. Med. Mol. Imaging 2021, 48, 4293–4306. [Google Scholar] [CrossRef]
- Merali, Z.; Wang, J.Z.; Badhiwala, J.H.; Witiw, C.D.; Wilson, J.R.; Fehlings, M.G. A deep learning model for detection of cervical spinal cord compression in MRI scans. Sci. Rep. 2021, 11, 10473. [Google Scholar] [CrossRef]
- Awan, M.J.; Mohd Rahim, M.S.; Salim, N.; Mohammed, M.A.; Garcia-Zapirain, B.; Abdulkareem, K.H. Efficient Detection of Knee Anterior Cruciate Ligament from Magnetic Resonance Imaging Using Deep Learning Approach. Diagnostics 2021, 11, 105. [Google Scholar] [CrossRef]
- Awan, M.J.; Rahim, M.S.M.; Salim, N.; Rehman, A.; Nobanee, H. Machine Learning-Based Performance Comparison to Diagnose Anterior Cruciate Ligament Tears. J. Healthc. Eng. 2022, 2022, 2550120. [Google Scholar] [CrossRef]
- Awan, M.J.; Rahim, M.S.M.; Salim, N.; Rehman, A.; Garcia-Zapirain, B. Automated Knee MR Images Segmentation of Anterior Cruciate Ligament Tears. Sensors 2022, 22, 1552. [Google Scholar] [CrossRef]
- Saeedizadeh, N.; Minaee, S.; Kafieh, R.; Yazdani, S.; Sonka, M. COVID TV-Unet: Segmenting COVID-19 chest CT images using connectivity imposed Unet. Comput. Methods Programs Biomed. Update 2021, 1, 100007. [Google Scholar] [CrossRef] [PubMed]
- Stefano, A.; Comelli, A. Customized Efficient Neural Network for COVID-19 Infected Region Identification in CT Images. J. Imaging 2021, 7, 131. [Google Scholar] [CrossRef] [PubMed]
- Barragán-Montero, A.; Javaid, U.; Valdés, G.; Nguyen, D.; Desbordes, P.; Macq, B.; Willems, S.; Vandewinckele, L.; Holmström, M.; Löfman, F. Artificial intelligence and machine learning for medical imaging: A technology review. Phys. Med. 2021, 83, 242–256. [Google Scholar] [CrossRef] [PubMed]
- Madai, V.I.; Higgins, D.V. Artificial Intelligence in Healthcare: Lost in Translation? arXiv 2021, arXiv:2107.13454 [cs.AI]. [Google Scholar] [CrossRef]
- Ji, M.; Genchev, G.Z.; Huang, H.; Xu, T.; Lu, H.; Yu, G. Evaluation Framework for Successful Artificial Intelligence-Enabled Clinical Decision Support Systems: Mixed Methods Study. J. Med. Internet Res. 2021, 23, e25929. [Google Scholar] [CrossRef]
- Cancer Information and Resources. Available online: https://www.cancer.org/research/cancer-facts-statistics.html (accessed on 1 January 2022).
- The Mayo Clinic Cancer Centre. Available online: https://www.mayoclinic.org/departments-centers/mayo-clinic-cancer-center (accessed on 1 January 2022).
- Commonly Abused Prescription and OTC Drugs. Available online: https://www.webmd.com/mental-health/addiction/ss/slideshow-commonly-abused-drugs (accessed on 1 January 2022).
- Hutson, M. Artificial intelligence faces reproducibility crisis. Science 2018, 359, 725–726, Available online (accessed on 18 June 2021). [Google Scholar] [CrossRef]
- Miotto, R.; Wang, F.; Wang, S.; Jiang, X.; Dudley, J.T. Deep learning for healthcare: Review, opportunities and challenges. Brief Bioinform. 2018, 19, 1236–1246. [Google Scholar] [CrossRef]
- Kimmelman, J.; Tannock, I. The paradox of precision medicine. Nat. Rev. Clin. Oncol. 2018, 15, 341–342. [Google Scholar] [CrossRef]
- Sanchis-Gomar, F.; Perez-Quilis, C.; Leischik, R.; Lucia, A. Epidemiology of coronary heart disease and acute coronary syndrome. Ann. Transl. Med. 2016, 4, 256. [Google Scholar] [CrossRef]
- Abouelmehdi, K.; Beni-Hessane, A.; Khaloufi, H. Big healthcare data: Preserving security and privacy. J. Big Data 2018, 5, 1. [Google Scholar] [CrossRef]
- Krois, J.; Garcia Cantu, A.; Chaurasia, A.; Patil, R.; Chaudhari, P.K.; Gaudin, R.; Gehrung, S.; Schwendicke, F. Generalizability of deep learning models for dental image analysis. Sci. Rep. 2021, 11, 6102. [Google Scholar] [CrossRef] [PubMed]
- Higgins, D.; Madai, V.I. From Bit to Bedside: A Practical Framework for Artificial Intelligence Product Development in Healthcare. Adv. Intell. Syst. 2020, 2, 2000052. [Google Scholar] [CrossRef]
- Johner-Institut/Ai-Guideline. Available online: https://github.com/johner-institut/ai-guideline (accessed on 31 July 2021).
- Panch, T.; Mattie, H.; Celi, L.A. The "inconvenient truth" about AI in healthcare. NPJ Digit. Med. 2019, 2, 77. [Google Scholar] [CrossRef]
- Davahli, M.R.; Karwowski, W.; Fiok, K.; Wan, T.; Parsaei, H.R. Controlling Safety of Artificial Intelligence-Based Systems in Healthcare. Symmetry 2021, 13, 102. [Google Scholar] [CrossRef]
- England, J.R.; Cheng, P.M. Artificial intelligence for medical image analysis: Aguide for authors and reviewers. Am. J. Roentgenol. 2019, 212, 513–519. [Google Scholar] [CrossRef] [PubMed]
- Srivastava, V.; Kumar, D.; Roy, S. A median based quadrilateral local quantized ternary pattern technique for the classification of dermatoscopic images of skin cancer. Comput. Electr. Eng. 2022, 102, 108259. [Google Scholar] [CrossRef]
- Asan, O.; Bayrak, A.E.; Choudhury, A. An Artificial Intelligence and Human Trust in Healthcare: Focus on Clinicians. J. Med. Internet Res. 2020, 22, e15154. [Google Scholar] [CrossRef]
- Datta, B.; Dutta, K.; Roy, S. Data hiding in virtual bit-plane using efficient Lucas number sequences. Multimed Tools Appl. 2020, 79, 22673–22703. [Google Scholar] [CrossRef]
- Simonyan, K.; Vedaldi, A.; Zisserman, A. Deep inside convolutional networks: Visualising image classification models and saliency maps. arXiv 2013, arXiv:1312.6034. [Google Scholar]
- Selvaraju, R.R.; Cogswell, M.; Das, A.; Vedantam, R.; Parikh, D.; Batra, D. Grad-cam: Visual explanations from deep networks via gradient-based localization. In Proceedings of the IEEE International Conference on Computer Vision, Cambridge, MA, USA, 20–23 June 1995; pp. 618–626. [Google Scholar]
- Zhang, Z.; Xie, Y.; Xing, F.; McGough, M.; Yang, L. Mdnet: A semantically and visually interpretable medical image diagnosis network. In Proceedings of the IEEE Conference on Computer Vision and Pattern Recognition, San Juan, PR, USA, 17–19 June 1997; pp. 6428–6436. [Google Scholar]
- Kabiraj, A.; Meena, T.; Reddy, P.B.; Roy, S. Detection and Classification of Lung Disease Using Deep Learning Architecture from X-ray images. In International Symposium on Visual Computing; Springer LNCS: Berlin/Heidelberg, Germany, 2022; pp. 1–12. [Google Scholar]
- Yang, G.; Ye, Q.; Xia, J. Unbox the black-box for the medical explainable AI via multi-modal and multi-centre data fusion: A mini-review, two showcases and beyond. Inf. Fusion 2022, 77, 29–52. [Google Scholar] [CrossRef]
- Meena, T.; Roy, S. Bone Fracture Detection Using Deep Supervised Learning from Radiological Images: A Paradigm Shift. Diagnostics 2022, 12, 2420. [Google Scholar] [CrossRef]
- Pasini, G.; Bini, F.; Russo, G.; Comelli, A.; Marinozzi, F.; Stefano, A. matRadiomics: A Novel and Complete Radiomics Framework, from Image Visualization to Predictive Model. J. Imaging 2022, 8, 221. [Google Scholar] [CrossRef] [PubMed]
- Van Griethuysen, J.J.; Fedorov, A.; Parmar, C.; Hosny, A.; Aucoin, N.; Narayan, V.; Beets-Tan, R.G.; Fillion-Robin, J.C.; Pieper, S.; Aerts, H.J. Computational Radiomics System to Decode the Radiographic Phenotype. Cancer Res. 2017, 77, e104–e107. [Google Scholar] [CrossRef] [PubMed]
- Nioche, C.; Orlhac, F.; Boughdad, S.; Reuzé, S.; Goya-Outi, J.; Robert, C.; Pellot-Barakat, C.; Soussan, M.; Frouin, F.; Buvat, I. LIFEx: A Freeware for Radiomic Feature Calculation in Multimodality Imaging to Accelerate Advances in the Characterization of Tumor Heterogeneity. Cancer Res. 2018, 78, 4786–4789. [Google Scholar] [CrossRef] [PubMed]
- Gunjan, V.K.; Singh, N.; Shaik, F.; Roy, S. Detection of lung cancer in CT scans using grey wolf optimization algorithm and recurrent neural network. Health Technol. 2022. [Google Scholar] [CrossRef]

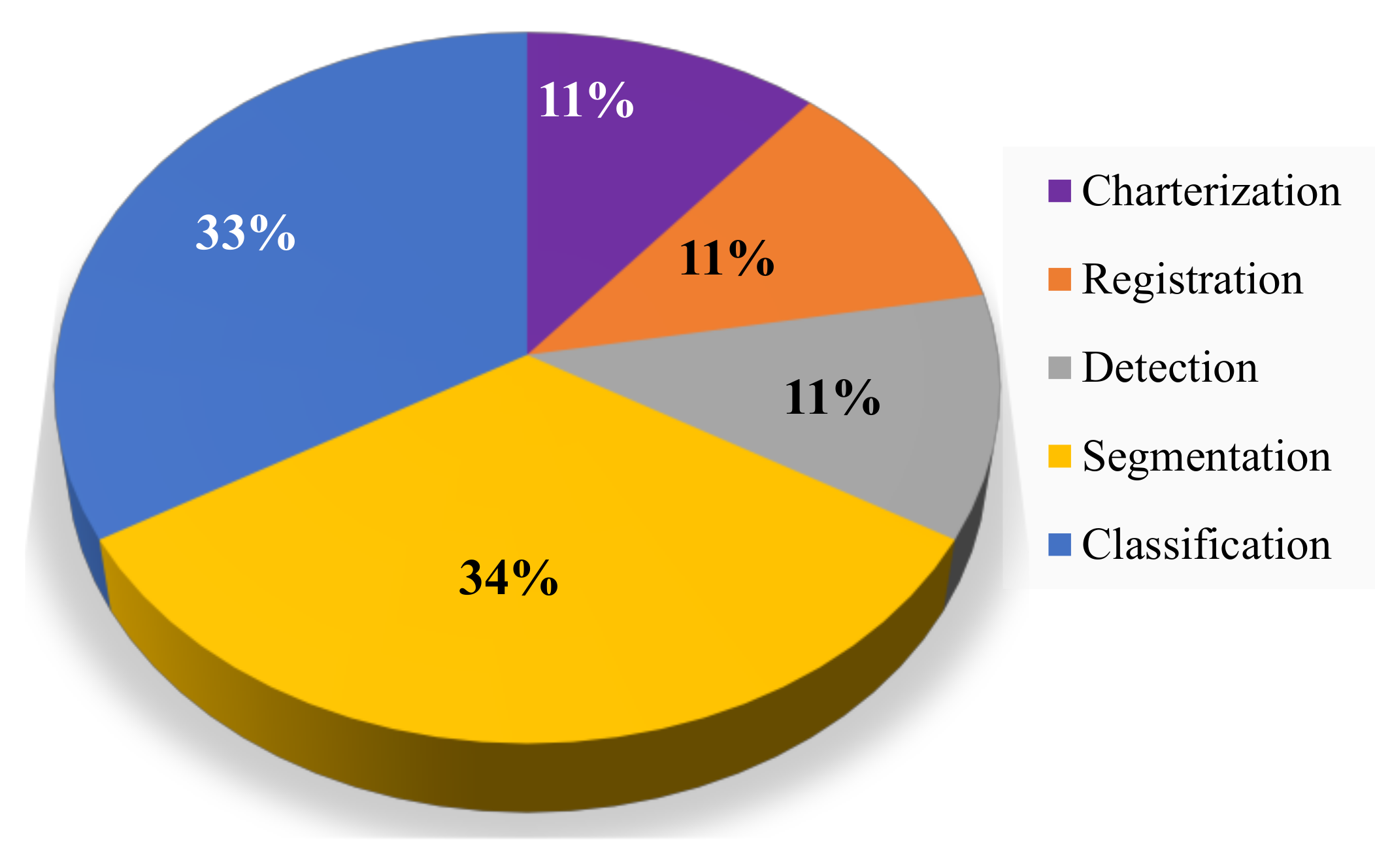
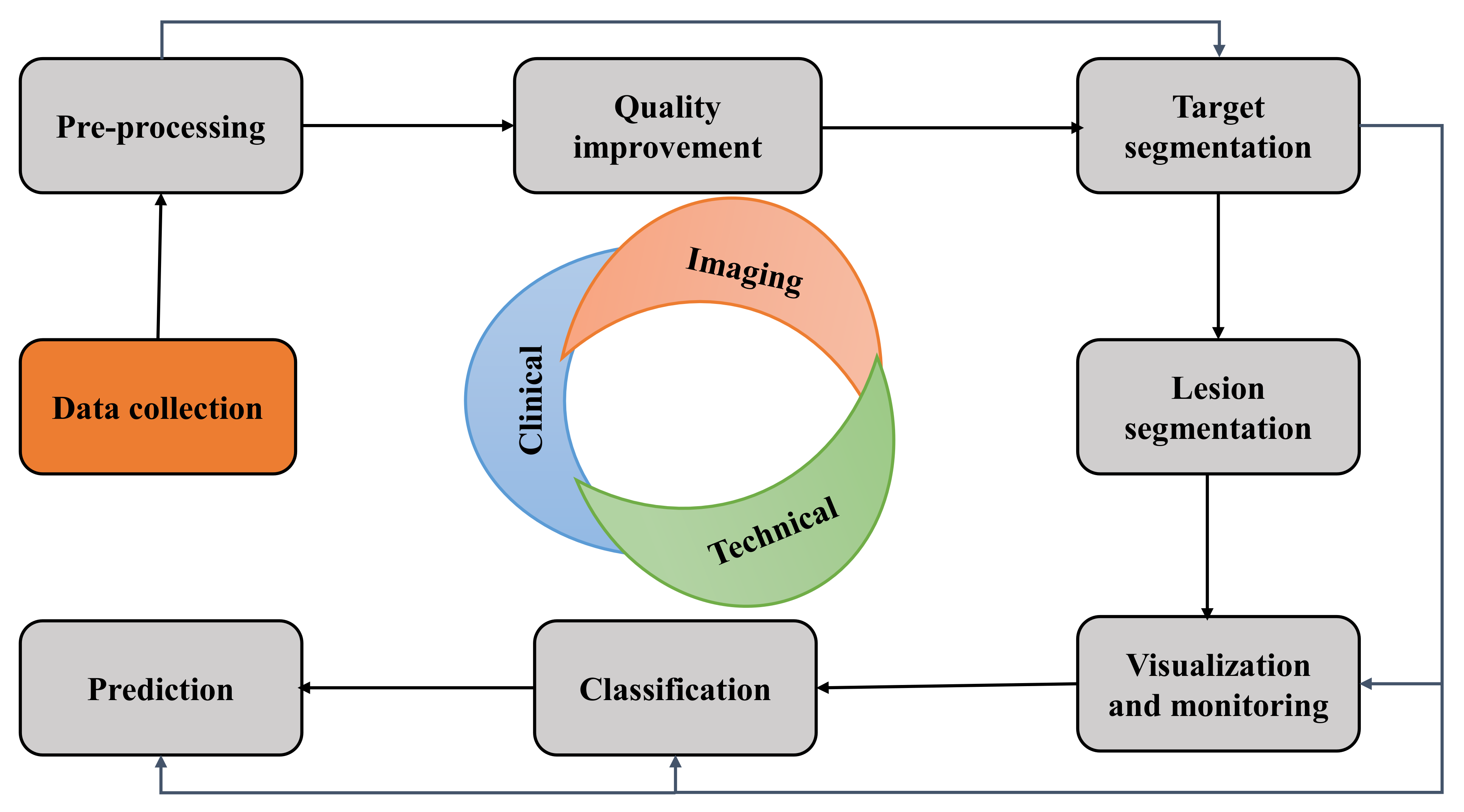
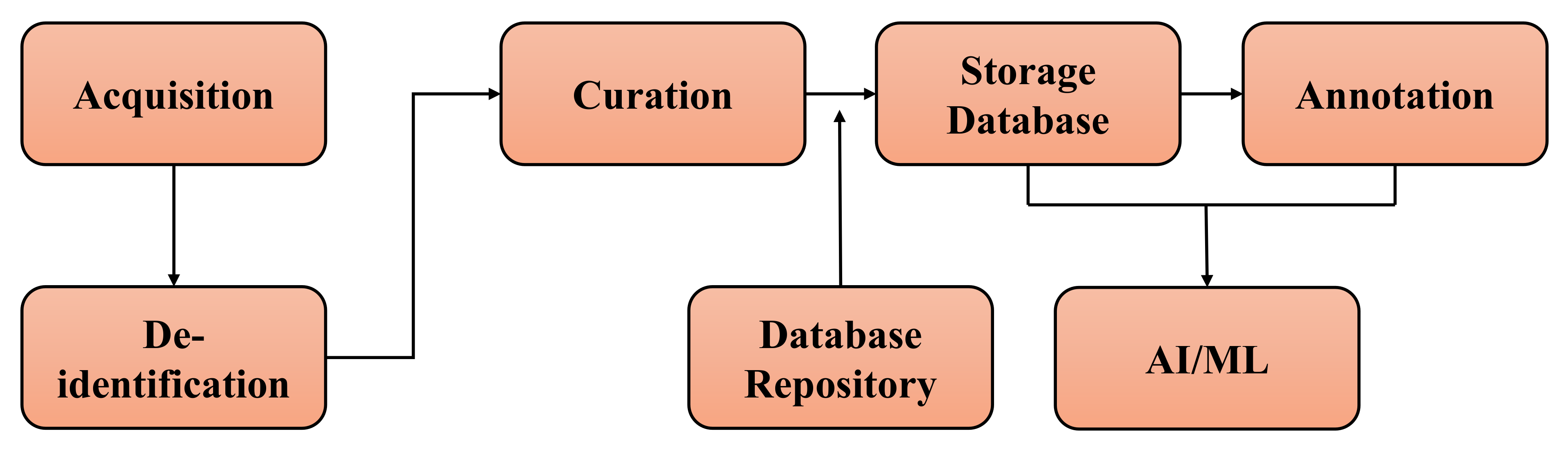
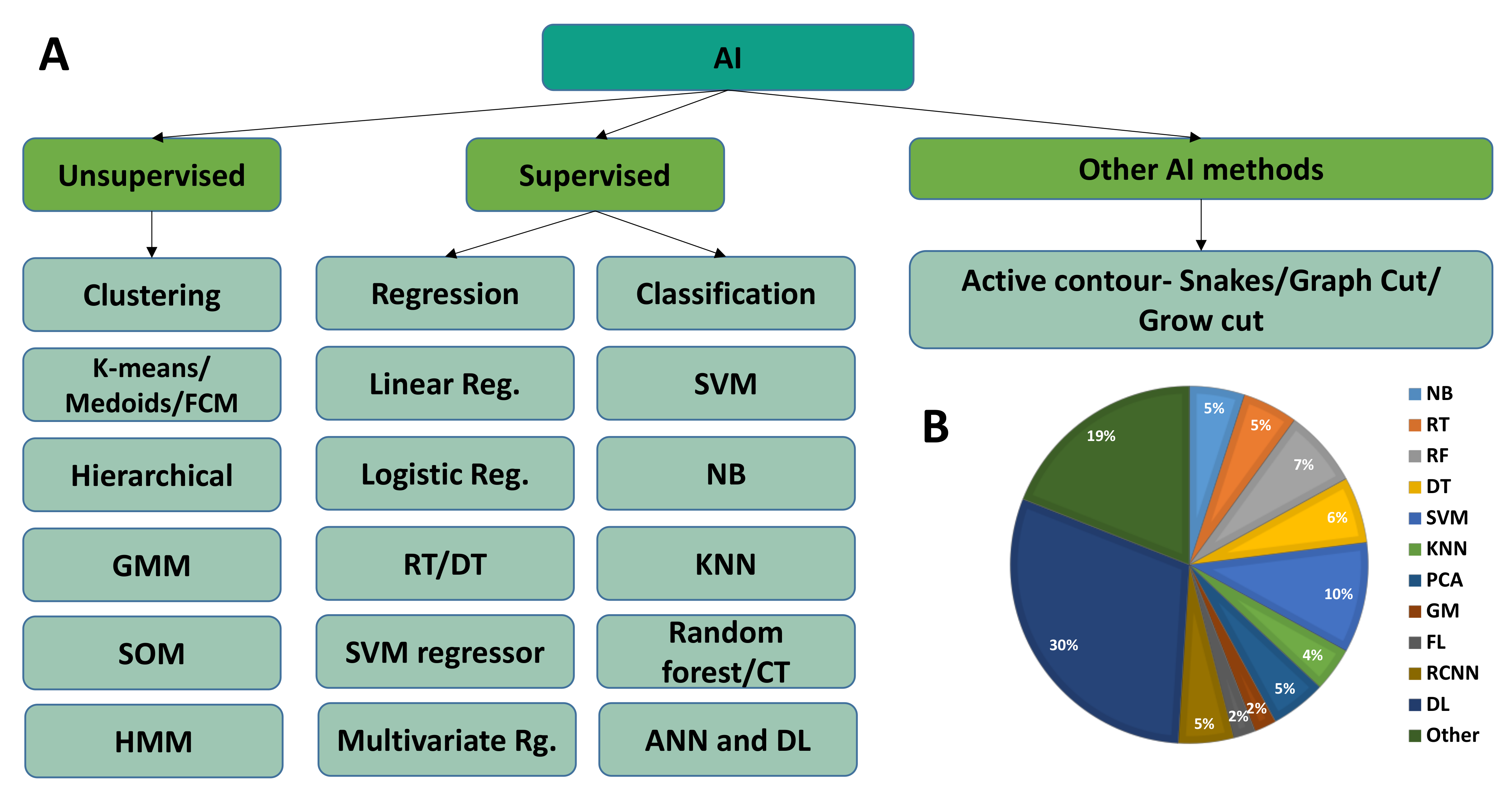
| Industry/Company Name | Task Performed |
|---|---|
| Google Brain | DR screening solution and also working on AI to improve breast cancer detection |
| DeepMind | Diagnosis and referral in retinal disease |
| Microsoft | Optimization of cancer treatment and radiotherapy-based planning |
| IBM | AI in healthcare and life sciences (e.g., predict the specificity of T cell receptors) |
| Qure | AI-assisted chest X-ray solution |
| In med prognostic | Volumetric analysis of the brain using AI |
| Tamil nadu e govt agency | AI-based cataract detection system |
| Niramai Health analytix | Breast cancer screening solution |
| Remidio | DR screening tool |
| Arterys | Proper blood flow visualization and quantification from MRI of whole-body images |
| Gauss Surgical Inc | Real-time tracking and monitoring of blood loss after surgery |
| Zebra Medical Vision | AI chest X-ray triage |
| Freenome | Cancer detection from blood cells imaging |
| Viz | Early signs of stroke prediction and detection |
| DiA Imaging | AI-powered ultrasound image analysis solution |
| RetinAi | “Discovery Platform” Glaucoma, DR, and macular degeneration (MR) data collection and analysis |
| BrainMiner | AI-based brain MRI scan analyzing systems |
| Lunit | AI-based automated chest X-ray interpretation |
| Technique | Advantage | Disadvantage |
|---|---|---|
| Deep Neural Network (DNN): Minimum two layers, many complex nonlinear associations among the layers. Mainly used for regression and classification purpose. | High accuracy | The very slow learning process. Nontrivial training procedure due to back propagation to the aforementioned layer. |
| Convolutional Neural Network (CNN): Transformation of 2D to 3D convolutional filters, and also works well for 2D data. | High accuracy on 2D data and fast learning model. | Need lots of labeled data for perdition. |
| Recurrent Neural Network (RNN): Weight sharing across all neurons and steps and sequential learning. | Provides high accuracy for visualization aspect of medical image analytics problems. | Many issues due to gradient vanishing and need a large amount of data. |
| Deep Boltzmann machine (DBM): It consists of unidirectional linking between all hidden layers. | More robust inference as top-down feedback incorporates. | Parameter optimization is not promising for large data. |
| Deep Belief Network (DBN): The greedy approach used in each layer. Unidirectional association between two layers at the top of layers. | Work well for similar data. | Training is expansive due to initialization problems. |
| Deep Autoencoder (DAE): The no. of I/P is equal to the number of O/P., reduced dimensionality. | Labeled data is not needed. | Training may suffer due to convergence and vanishing problems. |
| Detection | Segmentation | Classification | Prediction | |
|---|---|---|---|---|
| Features | Using various shapes such as circles, rectangles, and squares and labels using different binary masks, overlays, etc. | Texture, shape, position, and intensity Higher order features | Texture patches 3D Radiomics features Morphological features Imaging metrics | Texture and shape Survival time Dynamic modeling Imaging metrics |
| Model architectures | CNN | UNet/Unet++ | Dense net | Efficient net/CNN |
| Performance metrics |
| Mainly IoU and F measure, precision, recall, relative area error, and accuracy | Area under the curve (AUC) or receiver operating characteristic (ROC) Confusion matrix | AUC/ROC Confusion matrix Coefficient of determination |
| SL. NO. | Authors | Application | Dataset | Method Used | Reported Success |
|---|---|---|---|---|---|
| 1 | Murat SeçkinAyhan et al. [61] | Ophthalmology | Kaggle, Asia Pacific Tele-Ophthalmology Society, Messidor and Indian Diabetic, Retinopathy Image Dataset (IDRiD) | Deep neural networks (DNNs) | Accuracy ≥ 90% |
| 2 | Elineide S. dos Santos. et al. [62] | Skin cancerdermoscopic images | PH and DermIS datasets | Linear iterative clustering (SLIC0) algorithm | Accuracy = 96.78% |
| 3 | Ghorbani, A et al. [63] | Interpretation of echocardiograms from heart | The Stanford Echocardiography Database | CNN- EchoNet | Area under the curve = 0.89 |
| 4 | Bulten, W. et al. [64] | Grading and diagnosis of prostate cancer | Data from Radboud University Medical Center and Karolinska Institutet for PANDA challenge and competition | Multiple AI and machine learning methods | Error or miss: 1% for cancer and 1.8% for the pathologist |
| 5 | Rakesh Kumar Patnaik et al. [65] | Prediction of liver function from quick breath monitoring | A pilot study at a local university | Different regression and support vector machines (SVM) and various forms of decision trees | Correlation p < 0.01 between unhealthy and healthy samples |
| 6 | S Roy et al. [40] | Treatment response prediction of triple negative breast cancer—Co clinical trial (PET) | WashU medical school-imaging facilities | Radiomics, support vector machine, and naïve Bayes | Accuracy ≥ 72% |
| 7 | K Dutta et al. [66] | Preclinical breast cancer segmentation (MRI) | WashU medical school-imaging facilities | Dense UNet deep network | F-measure ≥ 94.8% |
| 8 | I. Anand et al. [67] | Breast tumor segmentation from magnetic resonance images (MRI) | RIDER breast MRI dataset | ResU-Net Architect | Accuracy = 73.22% and dice coefficient = 85.32% |
| 9 | S Roy et al. [68] | Preclinical breast cancer optimal radiomics (MRI) | WashU medical school-imaging facilities | Statistics and radiomics | SNR between 28 to 33/NA |
| 10 | Varadarajan et al. [69] | Edema grades of diabetic macular from fundus image | Rajavithi Hospital clinical data from 2010 to 2018 | Deep neural network | Area under the curve = 0.89 |
| 11 | Chetty, G et al. [70] | Tumor lesion segmentation from brain MRI | BraTS open challenge data set, 2018 | Modified UNET architecture | Dice score = 94% |
| 12 | M A. Savaikar et al. [71] | Prediction of response to carboplatin therapy for triple negative breast cancer (TNBC) from mice PET images | WashU medical school-imaging facilities | Change in imaging metrics a standard uptake value | F score of SUVmax = 73% SUV25 = 72% SUV(SS) = 74% |
| 13 | Ching-WeiWang et al. [72] | Segmentation and classification of bone marrow from Hematopathology image | Own institute dataset | Hierarchical deep learning framework | recall and accuracy of 0.905 ± 0.078 and 0.989 ± 0.006 |
| 14 | Liu, M. et al. [73] | Detect marginal bone loss around implants | Peking University School and Hospital of Stomatology | RCNN or region-based CNN architecture | Sensitivity 81% |
| 15 | Lea, W.Wi et al. [74] | Bone-age software on real-world data | Home hospital-pediatric clinic | Deep learning (DL)–based software | Correlation = 98% and p ≤ 0.025 |
| 16 | A Mitra et al. [75] | Glaucoma detection and analysis from retinal fundus image | ESSIDOR and Kaggle datasets | Deep Convolution Neural Network (CNN) | Accuracy was 99% on both two datasets |
| 17 | Mall, P.K et al. [76] | Musculoskeletal radiographs, X-ray images | Musculoskeletal radiograph (MURA) | Combination of ChampNet with CLAHE and other types | Highest accuracy was 96% |
| 18 | S Roy et al. [77] | Brain abnormality segmentation from MRI of brain images | Brainweb database | Iterative Level Set | Accuracy ≥ 75% |
| 19 | S Roy et al. [78] | Multiple small target lesion detection and segmentation from MRI of the brain | Harvard brain atlas network | Hybrid level set and thresholding | correct detection ration = 92.6% |
| 20 | Guan B et al. [79] | Predict pain progression in knee osteoarthritis | University of Wisconsin | deep learning (DL) | Area under the curve = 0.77 |
| 21 | Iizuka, O. et al. [80] | Detection and classification of gastric and colonic epithelial tumors from histopathological slides | The Cancer Genome Atlas open dataset | CNNs and RCNN (recurrent neural net.) | Area under the curves (AUC) was 0.97 and 0.99 |
| 22 | Abbas, A. et al. [81] | COVID-19 detection classification from chest X-ray | Images were collected from several hospitals around the world | Decompose, Transfer, and Compose based on deep CNN | Accuracy of 93.1% (with a sensitivity of 100%) |
| 23 | Wulczyn E et al. [82] | Multiple cancer types from histopathology images | The Cancer Genome Atlas open dataset | Multiple deep CNN modules | Disease specific survival prediction was significant p < 0.0001 |
| 24 | Sabanayagam C et al. [83] | Chronic kidney disease prediction from retinal images | Institute data-Singapore Eye hospital | Deep learning algorithm (CNN) | AUC was 0·88 and 0·71 in internal and external validation |
| 25 | Zhang, Y. et al. [84] | Classifying endometrial lesions | Hengjing Hospital of China Medical University | Tuned VGGNet-16 model | Accuracy = 91% |
| 26 | Pierre Pinochet et al. [85] | Suspected cancer location detection from PET and computed tomography (CT) images | Clinical data from the Henri Becquerel Cancer Center | CNNs | Dice score was 65% |
| 27 | May Sadik et al. [86] | Bone marrow uptake estimation and detection in Hodgkin’s lymphoma patients from PET images | Sahlgrenska University Hospital | CNN | Sensitivity = 65% and Specificity = 98%. |
| 28 | Janani Venugopalan et al. [87] | Early prediction of Alzheimer’s disease stage from MRI images | ADNI-Alzheimer’s Disease Neuroimaging Initiative open database | Novel deep learning and multi-modality data | accuracy of 0.75 ± 0.11 |
| 29 | Esraa A. Mohamed et al. [88] | Breast cancer detection system from thermograms | DMR-IR real data | U-Net network and world-class deep learning model | Accuracy = 99%, |
| 30 | Muhammad TariqSadiq et al. [89] | EEG-based robust brain–computer interface framework | -- | Pre-trained CNN models | average classification accuracy of 99.52% |
| 31 | Rodolfo M. Pereira et al. [90] | Analysis of chest X-Ray and identification of COVID-19 from X-ray | RYDLS-20 | Pre-trained CNN model | F1-Score of 0.89 |
| 32 | Alexandre Bailly et al. [91] | Performance prediction of various machine learning methods on simulated data | Simulated data | Deep neural network and logistic regression | AUC = 0.80 and 0.85 for neural net and penalized regression. |
| 33 | Raquel Sánchez-Cauce et al. [92] | Cancer localization from breast thermal image | DMR database | Deep CNN architecture | 97% accuracy |
| 34 | Wallis, D. et al. [93] | Identification and localization of lymph nodes from pathological mediastinal | Clinical data obtained by the authors | U-Net and 3D CNN | Sensitivity = 87% |
| 35 | Johnsson, K. et al. [94] | Standard report generation and automated annotation from PET imaging data | Progenics Pharmaceuticals, Inc., USA | Deep learning | Dice scores ≥ 0.79. |
| 36 | Capobianco, N. et al. [95] | Whole body PET uptake and prostate cancer grading classification | Institute data-Technical University of Munich | Deep learning methods | 80.4% average precision |
| 37 | Etminani, K. et al. [96] | Alzheimer's and dementia prediction and diagnosis of cognitive impairment | European DLB (EDLB) Consortium | 3D deep learning model | AUC = 0.96 |
| 38 | Mehranian, A. et al. [97] | Whole-body PET oncology scans quality enhancement and noise reduction | Hospitals-Oxford University | Deep learning | Scan time reduction by 50% |
| 39 | Xue, S. et al. [98] | Low to standard dose in PET imaging quality conversion | Harvard Medical School and Massachusetts General Hospital | Cross-scanner and tracer-based deep neural network | Conversion significantly p < 0.05 and normalized dose acquisition p < 0.05 |
| 40 | Wang, L. et al. [99] | Differentiating between nontuberculous mycobacteria and Mycobacterium tuberculosis from a chest CT image | Data was collected from 2014 to 2020 from Tianjin Haihe Hospital | deep learning framework (3D-ResNet) | AUCs = 0.86 |
| 41 | Merali, Z et al. [100] | Spinal cord compression identification and detection from cervical MRI scans | University of Toronto-Home institute data | CNN model | AUC = 0.94 |
| 42 | Awan, M.J. et al. [101] | Detection of anterior cruciate ligament injury from MRI scans using deep learning | Clinical Hospital Centre Rijeka | ResNet-14 | AUC = 0.99 |
| 43 | Awan, M.J. et al. [102] | Comparative analysis of machine learning models to identify the condition of three anterior cruciate ligament tears | Clinical Hospital Centre Rijeka | Random forest, categorical boosting, light gradient boosting machines, and highly randomized classifier | AUC = 0.99 |
| 44 | Awan, M.J. et al. [103] | Automatic segmentation of the anterior cruciate ligament tears from MRI | Clinical Hospital Center in Rijeka, Croatia | U-Net | Accuracy = 98.4 |
| 45 | Saeedizadeh N. et al. [104] | A segmentation framework to detect chest regions in CT images which are infected by COVID-19 | COVID-19 CT segmentation dataset | U-Net (COVID-19 TV-U-Net) | Dice score = 86% |
| 46 | Stefano, A. et al. [105] | Automated identification and segmentation of COVID-19-infected regions using CT | COVID-19 Lung CT Lesion Segmentation Challenge—2020 (COVID-19-20) | Deep learning framework (C-ENET) | Dice score = 75% |
| Learning Type | Methods | Applications |
|---|---|---|
| Basic learning frameworks | ||
| Supervised learning | Different types of classification and regression trees (CART), RF, NB, SVM, ANNs, and RNNs |
|
| Unsupervised learning | Dimensionality reduction (e.g., PCA, LDA), different clustering—unsupervised (e.g., K-medoids, K-means), and auto encoders |
|
| Reinforcement learning | Markov model and reinforcement Q-learning |
|
| Hybrid learning frameworks | ||
| Semi-supervised learning | Generative adversarial networks |
|
| Self-supervised learning | Augmentation, texture features, patch extraction, and rotation |
|
| Complex learning strategies | ||
| Transfer learning | Causative, perspiration, and unsupervised | - Toxicity estimate of radiotherapy dose - Clinical practices adaptation - Model simplification |
| Ensemble learning | Bagging bootstrap aggregating and boosting (e.g., AdaBoost, gradient boosting) | - Prediction of radiotherapy dose - Uncertainty Estimation - Stratification of patients |
| Task/Type | Management |
|---|---|
| Team |
|
| Cohorting |
|
| Data |
|
| Model |
|
| Hardware |
|
| Regulatory |
|
| Clinical adoption |
|
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Roy, S.; Meena, T.; Lim, S.-J. Demystifying Supervised Learning in Healthcare 4.0: A New Reality of Transforming Diagnostic Medicine. Diagnostics 2022, 12, 2549. https://doi.org/10.3390/diagnostics12102549
Roy S, Meena T, Lim S-J. Demystifying Supervised Learning in Healthcare 4.0: A New Reality of Transforming Diagnostic Medicine. Diagnostics. 2022; 12(10):2549. https://doi.org/10.3390/diagnostics12102549
Chicago/Turabian StyleRoy, Sudipta, Tanushree Meena, and Se-Jung Lim. 2022. "Demystifying Supervised Learning in Healthcare 4.0: A New Reality of Transforming Diagnostic Medicine" Diagnostics 12, no. 10: 2549. https://doi.org/10.3390/diagnostics12102549
APA StyleRoy, S., Meena, T., & Lim, S.-J. (2022). Demystifying Supervised Learning in Healthcare 4.0: A New Reality of Transforming Diagnostic Medicine. Diagnostics, 12(10), 2549. https://doi.org/10.3390/diagnostics12102549







