Myocardial Pathology in COVID-19-Associated Cardiac Injury: A Systematic Review
Abstract
:1. Introduction
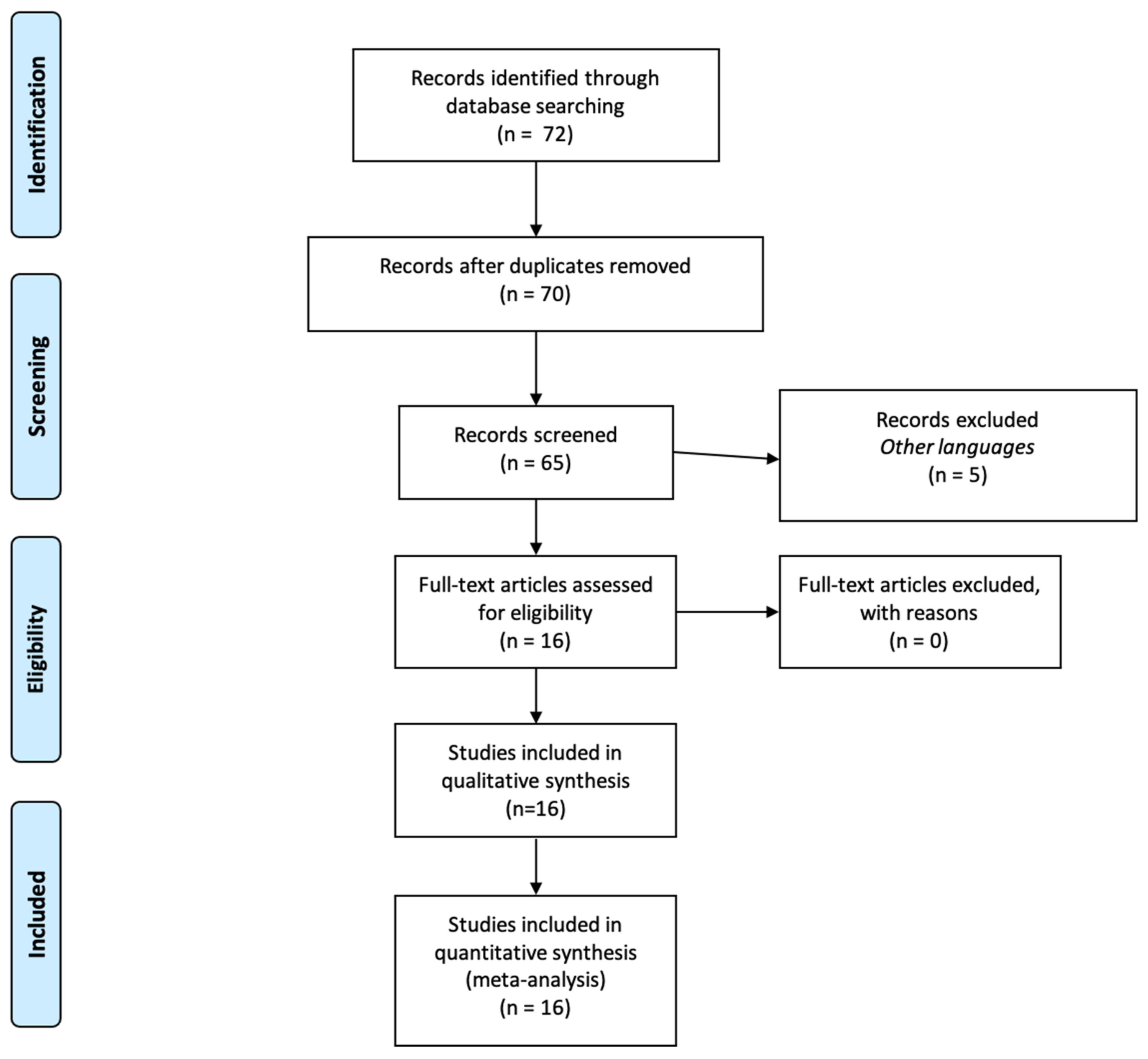
2. Materials and Methods
3. Results
3.1. Autopsy Data
| Authors | Number of Cases | Age | M:F | Cardiac Death | Cardiac Infection | Macroscopic | Microscopic | IHC | ||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Weight | Ventricular Dilatation/ Myocyte Hypertrophy | Necrosis | Lymphocytic Infiltration Parenchyma | Microthrombi/ Cardiac Vessels Involvement | ||||||||
| Lindner et al. 2020 [15] | 39 | 85 (78–89) | 16:23 | 1 Prior HF, 38 ARDS | 15 (+) interstitial cells within the cardiac tissue | Not reported | None | None | 0 | - | Yes | |
| Fox et al. 2020 [16] | 22 | 68.5 (44–79) | 10:12 | 0 | 16 | 340 to 1010 g | 9 RVD Se R:L >1:1 Right Hypertrophy | Individual cell dropout/necrosis/apoptosis | 0 | - | Yes | |
| Jacobs et al., 2020 [17] | 1 | 48 | 1 | 1 Fulminant Myocarditis due to COVID-19 | 1 | 605 | 1 | piecemeal necrosis | 1 | - | Yes | |
| Pellegrini et al. 2020 [19] | 40 | 74 | 29:11 | 3 HF, 37 RF | 8 | Not reported | Not reported | 14 (3 infarction, 11 focal necrosis) | 9 | 9 | No | |
| Gauchotte et al. 2021 [20] | 1 | 69 | 1 | 1 Fulminant myocarditis due to COVID-19 | 1 | 403 | none | None | 1 | - | Yes | |
| Bulfamante et al., 2020 [21] | 6 | 59.5 (54–69) | 1:5 | 6 RF | 6 | 410–750 | 1 LVH (prior), 6 RVD (lung overload) | 6 | 6 | - | Yes | |
| Del Nonno et al., 2020 [22] | 9 | 70 (35–92) | 7:2 | 9 (6 shock, 3 sudden death) | 0 | 450 | No supplementary available | 9 | 9 | 9 | No | |
| Basso et al. 2020 [23] | 21 | 69 (44–86) | 15:6 | 1 HF, 1 Sudden death, 19 RF | Not reported | Not reported | Not reported | 4 | 3 criteria /11 infiltration | 3 | Yes | |
| Hanley et al. 2020 [24] | 10 | 73 (52–79) | 6:3 | 9 RF | 2 | 450 (312–513) | 4 LVH | 1 | 0 | 6 | No | |
| Rapkiewicz et al., 2020 [25] | 7 | 57 (44–65) | 3:4 | 7 RF | Not reported | Not Reported | Not reported | 3 | 1 | 7 | No | |
| Cîrstea et al., 2020 [26] | 1 | 30 | 0:1 | 1 HF | Not reported | Not reported | Not reported | 0 | 1 | 1 | No | |
| Bradley et al., 2020 [27] | 14 | 73.5 (42–84) | 6:8 | 1 VF, 1 LVF, prior 12 RF | 0 | Not reported | 13 CH | 1 | 1 | 0 | No | |
| Duarte-Neto et al., 2020 [30] | 10 | 69 (33–83) | 5:5 | 10 RF | Not reported | Not reported | 9 CH | 0 | 2 | 2 | No | |
| Schaller et al., 2020 [32] | 10 | 69 (64–90) | 7:4 | Not reported | Not reported | Not reported | Not reported | Not reported | 4 | 0 | No | |
| Buja et al., 2020 [33] | 3 | 48 (34–62) | 3:0 | 2RF, 1 SD | Not reported | 720 (420–1070) | 2CH | 0 | 2 | 0 | No | |
| Bois et al., 2021 [34] | 15 | 78 (71–86) | 12:3 | Not reported | 5 | 443.1 (286.3–545.0) | Not reported | 9 | 5 | 12 | Yes | |
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- World Health Organization. WHO Characterizes COVID-19 as a Pandemic. 2020. Available online: https://www.who.int/director-general/speeches/detail/who-director-general-s-opening-remarks-at-the-media-briefing-on-covid-19---11-march-2020 (accessed on 14 August 2021).
- Wang, H.; Li, X.; Li, T.; Zhang, S.; Wang, L.; Wu, X.; Liu, J. The genetic sequence, origin, and diagnosis of SARS-CoV-2. Eur. J. Clin. Microbiol. Infect. Dis. 2020, 39, 1629–1635. [Google Scholar] [CrossRef]
- Zhu, Z.; Zhang, Z.; Chen, W.; Cai, Z.; Ge, X.; Zhu, H.; Jiang, T.; Tan, W.; Peng, Y. Predicting the receptor-binding domain usage of the coronavirus based on kmer frequency on spike protein. Infect. Genet. Evol. 2018, 61, 183–184. [Google Scholar] [CrossRef]
- He, Y.; Zhou, Y.; Liu, S.; Kou, Z.; Li, W.; Farzan, M.; Jiang, S. Receptor-binding domain of SARS-CoV spike protein induces highly potent neutralizing antibodies: Implication for developing subunit vaccine. Biochem. Biophys. Res. Commun. 2004, 324, 773–781. [Google Scholar] [CrossRef] [PubMed]
- Van Doremalen, N.; Bushmaker, T.; Morris, D.H.; Holbrook, M.G.; Gamble, A.; Williamson, B.N.; Tamin, A.; Harcourt, J.L.; Thornburg, N.J.; Gerber, S.I.; et al. Aerosol and Surface Stability of SARS-CoV-2 as Compared with SARS-CoV-1. N. Engl. J. Med. 2020, 382, 1564–1567. [Google Scholar] [CrossRef]
- Gandhi, R.T.; Lynch, J.B.; Del Rio, C. Mild or Moderate Covid-19. N. Engl. J. Med. 2020, 383, 1757–1766. [Google Scholar] [CrossRef] [PubMed]
- Xu, Z.; Shi, L.; Wang, Y.; Zhang, J.; Huang, L.; Zhang, C.; Liu, S.; Zhao, P.; Liu, H.; Zhu, L.; et al. Pathological findings of COVID-19 associated with acute respiratory distress syndrome. Lancet Respir. Med. 2020, 8, 420–422. [Google Scholar] [CrossRef]
- Maiese, A.; Passaro, G.; Matteis, A.; Fazio, V.; Raffaele, R.; Paolo, M.D. Thromboinflammatory response in SARS-CoV-2 sepsis. Med.-Leg. J. 2020, 88, 78–80. [Google Scholar] [CrossRef]
- Maccio, U.; Zinkernagel, A.S.; Shambat, S.M.; Zeng, X.; Cathomas, G.; Ruschitzka, F.; Schuepbach, R.A.; Moch, H.; Varga, Z. SARS-CoV-2 leads to a small vessel endotheliitis in the heart. EBioMedicine 2021, 63, 103182. [Google Scholar] [CrossRef] [PubMed]
- Shi, S.; Qin, M.; Shen, B.; Cai, Y.; Liu, T.; Yang, F.; Gong, W.; Liu, X.; Liang, J.; Zhao, Q.; et al. Association of Cardiac Injury with Mortality in Hospitalized Patients With COVID-19 in Wuhan, China. JAMA Cardiol. 2020, 5, 802–810. [Google Scholar] [CrossRef] [Green Version]
- Guo, T.; Fan, Y.; Chen, M.; Wu, X.; Zhang, L.; He, T.; Wang, H.; Wan, J.; Wang, X.; Lu, Z. Cardiovascular Implications of Fatal Outcomes of Patients with Coronavirus Disease 2019 (COVID-19). JAMA Cardiol. 2020, 5, 811–818. [Google Scholar] [CrossRef] [Green Version]
- Wendel Garcia, P.D.; Fumeaux, T.; Guerci, P.; Heuberger, D.M.; Montomoli, J.; Roche-Campo, F.; Schuepbach, R.A.; Hilty, M.P.; RISC-19-ICU Investigators. Prognostic factors associated with mortality risk and disease progression in 639 critically ill patients with COVID-19 in Europe: Initial report of the international RISC-19-ICU prospective observational cohort. EClinicalMedicine 2020, 25, 100449. [Google Scholar] [CrossRef]
- Maiese, A.; Manetti, A.C.; La Russa, R.; Di Paolo, M.; Turillazzi, E.; Frati, P.; Fineschi, V. Autopsy findings in COVID-19-related deaths: A literature review. Forensic. Sci. Med. Pathol. 2020, 7, 279–296. [Google Scholar] [CrossRef]
- Liberati, A.; Altman, D.G.; Tetzlaff, J.; Mulrow, C.; GÃtzsche, P.C.; Ioannidis, J.P.A.; Clarke, M.; Devereaux, P.J.; Kleijnen, J.; Moher, D. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate healthcare interventions: Explanation and elaboration. BMJ 2009, 339, b2700. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lindner, D.; Fitzek, A.; Bräuninger, H.; Aleshcheva, G.; Edler, C.; Meissner, K.; Scherschel, K.; Kirchhof, P.; Escher, F.; Schultheiss, H.P.; et al. Association of Cardiac Infection With SARS-CoV-2 in Confirmed COVID-19 Autopsy Cases. JAMA Cardiol. 2020, 5, 1281–1285. [Google Scholar] [CrossRef] [PubMed]
- Fox, S.E.; Li, G.; Akmatbekov, A.; Harbert, J.L.; Lameira, F.S.; Brown, J.Q.; Vander Heide, R.S. Unexpected Features of Cardiac Pathology in COVID-19 Infection. Circulation 2020, 142, 1123–1125. [Google Scholar] [CrossRef]
- Jacobs, W.; Lammens, M.; Kerckhofs, A.; Voets, E.; Van San, E.; Van Coillie, S.; Peleman, C.; Mergeay, M.; Sirimsi, S.; Matheeussen, V.; et al. Fatal lymphocytic cardiac damage in coronavirus disease 2019 (COVID-19): Autopsy reveals a ferroptosis signature. ESC Heart Fail. 2020, 7, 3772–3781. [Google Scholar] [CrossRef]
- Aretz, H.T.; Billingham, M.E.; Edwards, W.D.; Factor, S.M.; Fallon, J.T.; Fenoglio, J.J.; Olsen, E.G., Jr.; Schoen, F.J. Myocarditis. A histopathologic definition and classification. Am. J. Cardiovasc. Pathol. 1987, 1, 3–14. [Google Scholar]
- Pellegrini, D.; Kawakami, R.; Guagliumi, G.; Sakamoto, A.; Kawai, K.; Gianatti, A.; Nasr, A.; Kutys, R.; Guo, L.; Cornelissen, A.; et al. Microthrombi as a major cause of cardiac injury in COVID-19: A pathologic study. Circulation 2021, 143, 1031–1042. [Google Scholar] [CrossRef]
- Gauchotte, G.; Venard, V.; Segondy, M.; Cadoz, C.; Esposito-Fava, A.; Barraud, D.; Louis, G. SARS-Cov-2 fulminant myocarditis: An autopsy and histopathological case study. Int. J. Legal. Med. 2021, 135, 577–581. [Google Scholar] [CrossRef]
- Bulfamante, G.P.; Perrucci, G.L.; Falleni, M.; Sommariva, E.; Tosi, D.; Martinelli, C.; Songia, P.; Poggio, P.; Carugo, S.; Pompilio, G. Evidence of SARS-CoV-2 transcriptional activity in cardiomyocytes of COVID-19 patients without clinical signs of cardiac involvement. medRxiv 2020, 8, 626. [Google Scholar] [CrossRef]
- Del Nonno, F.; Frustaci, A.; Verardo, R.; Chimenti, C.; Nicastri, E.; Antinori, A.; Petrosillo, N.; Lalle, E.; Agrati, C.; Ippolito, G.; et al. Virus-Negative Myopericarditis in Human Coronavirus Infection: Report from an Autopsy Series. Circ. Heart Fail. 2020, 13, e007636. [Google Scholar] [CrossRef]
- Basso, C.; Leone, O.; Rizzo, S.; De Gaspari, M.; van der Wal, A.C.; Aubry, M.C.; Bois, M.C.; Lin, P.T.; Maleszewski, J.J.; Stone, J.R. Pathological features of COVID-19-associated myocardial injury: A multicentre cardiovascular pathology study. Eur. Heart J. 2020, 41, 3827–3835. [Google Scholar] [CrossRef] [PubMed]
- Hanley, B.; Naresh, K.N.; Roufosse, C.; Nicholson, A.G.; Weir, J.; Cooke, G.S.; Thursz, M.; Manousou, P.; Corbett, R.; Goldin, R.; et al. Histopathological findings and viral tropism in UK patients with severe fatal COVID-19: A post-mortem study. Lancet Microbe 2020, 1, e245–e253. [Google Scholar] [CrossRef]
- Rapkiewicz, A.V.; Mai, X.; Carsons, S.E.; Pittaluga, S.; Kleiner, D.E.; Berger, J.S.; Thomas, S.; Adler, N.M.; Charytan, D.M.; Gasmi, B.; et al. Megakaryocytes and platelet-fibrin thrombi characterize multi-organ thrombosis at autopsy in COVID-19: A case series. EClinicalMedicine 2020, 24, 100434. [Google Scholar] [CrossRef] [PubMed]
- Cîrstea, A.E.; Buzulică, R.L.; Pirici, D.; Ceauşu, M.C.; Iman, R.V.; Gheorghe, O.M.; Neamţu, S.D.; Stanca, L.; Ene, R.; Kumar-Singh, S.; et al. Histopathological findings in the advanced natural evolution of the SARS-CoV-2 infection. Rom. J. Morphol. Embryol. 2020, 61, 209–218. [Google Scholar] [CrossRef]
- Bradley, B.T.; Maioli, H.; Johnston, R.; Chaudhry, I.; Fink, S.L.; Xu, H.; Najafian, B.; Deutsch, G.; Lacy, J.M.; Williams, T.; et al. Histopathology and ultrastructural findings of fatal COVID-19 infections in Washington State: A case series. Lancet 2020, 396, 320–332. [Google Scholar] [CrossRef]
- Zhou, F.; Yu, T.; Du, R.; Fan, G.; Liu, Y.; Liu, Z.; Xiang, J.; Wang, Y.; Song, B.; Gu, X.; et al. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: A retrospective cohort study. Lancet 2020, 395, 1054–1062. [Google Scholar] [CrossRef]
- Gautret, P.; Million, M.; Jarrot, P.A.; Camoin-Jau, L.; Colson, P.; Fenollar, F.; Leone, M.; La Scola, B.; Devaux, C.; Gaubert, J.Y.; et al. Natural history of COVID-19 and therapeutic options. Expert Rev. Clin. Immunol. 2020, 16, 1159–1184. [Google Scholar] [CrossRef]
- Duarte-Neto, A.N.; Monteiro, R.A.A.; da Silva, L.F.F.; Malheiros, D.M.A.C.; de Oliveira, E.P.; Theodoro-Filho, J.; Pinho, J.; Gomes-Gouvêa, M.S.; Salles, A.; de Oliveira, I.; et al. Pulmonary and systemic involvement in COVID-19 patients assessed with ultrasound-guided minimally invasive autopsy. Histopathology 2020, 77, 186–197. [Google Scholar] [CrossRef]
- D’Onofrio, V.; Donders, E.; Vanden Abeele, M.E.; Dubois, J.; Cartuyvels, R.; Achten, R.; Lammens, M.; Dendooven, A.; Driessen, A.; Augsburg, L.; et al. The clinical value of minimal invasive autopsy in COVID-19 patients. PLoS ONE 2020, 15, e0242300. [Google Scholar] [CrossRef]
- Schaller, T.; Hirschbühl, K.; Burkhardt, K.; Braun, G.; Trepel, M.; Märkl, B.; Claus, R. Postmortem Examination of Patients With COVID-19. JAMA 2020, 323, 2518–2520. [Google Scholar] [CrossRef]
- Buja, L.M.; Wolf, D.A.; Zhao, B.; Akkanti, B.; McDonald, M.; Lelenwa, L.; Reilly, N.; Ottaviani, G.; Elghetany, M.T.; Trujillo, D.O.; et al. The emerging spectrum of cardiopulmonary pathology of the coronavirus disease 2019 (COVID-19): Report of 3 autopsies from Houston, Texas, and review of autopsy findings from other United States cities. Cardiovasc. Pathol. 2020, 48, 107233. [Google Scholar] [CrossRef] [PubMed]
- Bois, M.C.; Boire, N.A.; Layman, A.J.; Aubry, M.C.; Alexander, M.P.; Roden, A.C.; Hagen, C.E.; Quinton, R.A.; Larsen, C.; Erben, Y.; et al. COVID-19-Associated Nonocclusive Fibrin Microthrombi in the Heart. Circulation 2021, 143, 230–243. [Google Scholar] [CrossRef] [PubMed]
- Gu, J.; Korteweg, C. Pathology and pathogenesis of severe acute respiratory syndrome. Am. J. Pathol. 2007, 170, 1136–1147. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Van den Brand, J.M.; Smits, S.L.; Haagmans, B.L. Pathogenesis of Middle East respiratory syndrome coronavirus. J. Pathol. 2015, 235, 175–184. [Google Scholar] [CrossRef] [Green Version]
- Ganji, A.; Farahani, I.; Khansarinejad, B.; Ghazavi, A.; Mosayebi, G. Increased expression of CD8 marker on T-cells in COVID-19 patients. Blood Cells Mol. Dis. 2020, 83, 102437. [Google Scholar] [CrossRef]
- McBride, J.A.; Striker, R. Imbalance in the game of T cells: What can the CD4/CD8 T-cell ratio tell us about HIV and health? PLoS Pathog. 2017, 13, e1006624. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Diao, B.; Wang, C.; Tan, Y.; Chen, X.; Liu, Y.; Ning, L.; Chen, L.; Li, M.; Liu, Y.; Wang, G.; et al. Reduction and Functional Exhaustion of T Cells in Patients with Coronavirus Disease 2019 (COVID-19). Front. Immunol. 2020, 11, 827. [Google Scholar] [CrossRef]
- Pallotto, C.; Suardi, L.R.; Esperti, S.; Tarquini, R.; Grifoni, E.; Meini, S.; Valoriani, A.; Di Martino, S.; Cei, F.; Sisti, E.; et al. Increased CD4/CD8 ratio as a risk factor for critical illness in coronavirus disease 2019 (COVID-19): A retrospective multicentre study. Infect. Dis. 2020, 52, 675–677. [Google Scholar] [CrossRef]
- Wang, F.; Nie, J.; Wang, H.; Zhao, Q.; Xiong, Y.; Deng, L.; Song, S.; Ma, Z.; Mo, P.; Zhang, Y. Characteristics of Peripheral Lymphocyte Subset Alteration in COVID-19 Pneumonia. J. Infect. Dis. 2020, 221, 1762–1769. [Google Scholar] [CrossRef] [Green Version]
- Wenzel, P.; Kopp, S.; Göbel, S.; Jansen, T.; Geyer, M.; Hahn, F.; Kreitner, K.F.; Escher, F.; Schultheiss, H.P.; Münzel, T. Evidence of SARS-CoV-2 mRNA in endomyocardial biopsies of patients with clinically suspected myocarditis tested negative for COVID-19 in nasopharyngeal swab. Cardiovasc. Res. 2020, 116, 1661–1663. [Google Scholar] [CrossRef] [PubMed]
- Halushka, M.K.; Vander Heide, R.S. Myocarditis is rare in COVID-19 autopsies: Cardiovascular findings across 277 postmortem examinations. Cardiovasc. Pathol. 2021, 50, 107300. [Google Scholar] [CrossRef]
- Zanza, C.; Racca, F.; Longhitano, Y.; Piccioni, A.; Franceschi, F.; Artico, M.; Abenavoli, L.; Maiese, A.; Passaro, G.; Volonnino, G.; et al. Risk Management and Treatment of Coagulation Disorders Related to COVID-19 Infection. Int. J. Environ. Res. Public Health 2021, 18, 1268. [Google Scholar] [CrossRef] [PubMed]
- Ng, A.C.T.; Delgado, V.; Bax, J.J. An international, multicentre survey of echocardiographic abnormalities in COVID-19 patients. Eur. Heart J. Cardiovasc. Imaging 2020, 21, 959–960. [Google Scholar] [CrossRef]
- Turner, A.J.; Hiscox, J.A.; Hooper, N.M. ACE2: From vasopeptidase to SARS virus receptor. Trends Pharmacol. Sci. 2004, 25, 294–297. [Google Scholar] [CrossRef]
- Chen, L.; Li, X.; Chen, M.; Feng, Y.; Xiong, C. The ACE2 expression in human heart indicates new potential mechanism of heart injury among patients infected with SARS-CoV-2. Cardiovasc. Res. 2020, 116, 1097–1100. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mahmoud-Elsayed, H.M.; Moody, W.E.; Bradlow, W.M.; Khan-Kheil, A.M.; Senior, J.; Hudsmith, L.E.; Steeds, R.P. Echocardiographic Findings in Patients With COVID-19 Pneumonia. Can. J. Cardiol. 2020, 36, 1203–1207. [Google Scholar] [CrossRef]
- Deng, Q.; Hu, B.; Zhang, Y.; Wang, H.; Zhou, X.; Hu, W.; Cheng, Y.; Yan, J.; Ping, H.; Zhou, Q. Suspected myocardial injury in patients with COVID-19: Evidence from front-line clinical observation in Wuhan, China. Int. J. Cardiol. 2020, 311, 116–121. [Google Scholar] [CrossRef]
- Lassen, M.C.H.; Skaarup, K.G.; Lind, J.N.; Alhakak, A.S.; Sengeløv, M.; Nielsen, A.B.; Espersen, C.; Ravnkilde, K.; Hauser, R.; Schöps, L.B.; et al. Echocardiographic abnormalities and predictors of mortality in hospitalized COVID-19 patients: The ECHOVID-19 study. ESC Heart Fail. 2020, 7, 4189–4197. [Google Scholar] [CrossRef]
- Cau, R.; Bassareo, P.; Saba, L. Cardiac Involvement in COVID-19-Assessment with Echocardiography and Cardiac Magnetic Resonance Imaging. SN Compr. Clin. Med. 2020, 2, 845–851. [Google Scholar] [CrossRef] [PubMed]
- Maiese, A.; Manetti, A.C.; Bosetti, C.; Del Duca, F.; La Russa, R.; Frati, P.; Di Paolo, M.; Turillazzi, E.; Fineschi, V. SARS-CoV-2 and the brain: A review of the current knowledge on neuropathology in COVID-19. Brain Pathol. 2021, 13, e13013. [Google Scholar] [CrossRef]
- Hofman, P.; Copin, M.C.; Tauziede-Espariat, A.; Adle-Biassette, H.; Fortarezza, F.; Passeron, T.; Salmon, I.; Calabrese, F. Les lésions histologiques associées à l’infection par le SARS-CoV-2 [Histopathological features due to the SARS-CoV-2]. Ann. Pathol. 2021, 41, 9–22. [Google Scholar] [CrossRef] [PubMed]
- Santurro, A.; Scopetti, M.; D’Errico, S.; Fineschi, V. A technical report from the Italian SARS-CoV-2 outbreak. Postmortem sampling and autopsy investigation in cases of suspected or probable COVID-19. Forensic Sci. Med. Pathol. 2020, 16, 471–476. [Google Scholar] [CrossRef] [PubMed]
- Li, A.; Garcia-Bengochea, Y.; Stechel, R.; Azari, B.M. Management of COVID-19 myopericarditis with reversal of cardiac dysfunction after blunting of cytokine storm: A case report. Eur. Heart J. Case Rep. 2020, 4, 1–6. [Google Scholar] [CrossRef]
- Frisoni, P.; Neri, M.; D’Errico, S.; Alfieri, L.; Bonuccelli, D.; Cingolani, M.; Di Paolo, M.; Gaudio, R.M.; Lestani, M.; Marti, M.; et al. Cytokine storm and histopathological findings in 60 cases of COVID-19-related death: From viral load research to immunohistochemical quantification of major players IL-1β, IL-6, IL-15 and TNF-α. Forensic Sci. Med. Pathol. 2021, 1–15. [Google Scholar] [CrossRef]

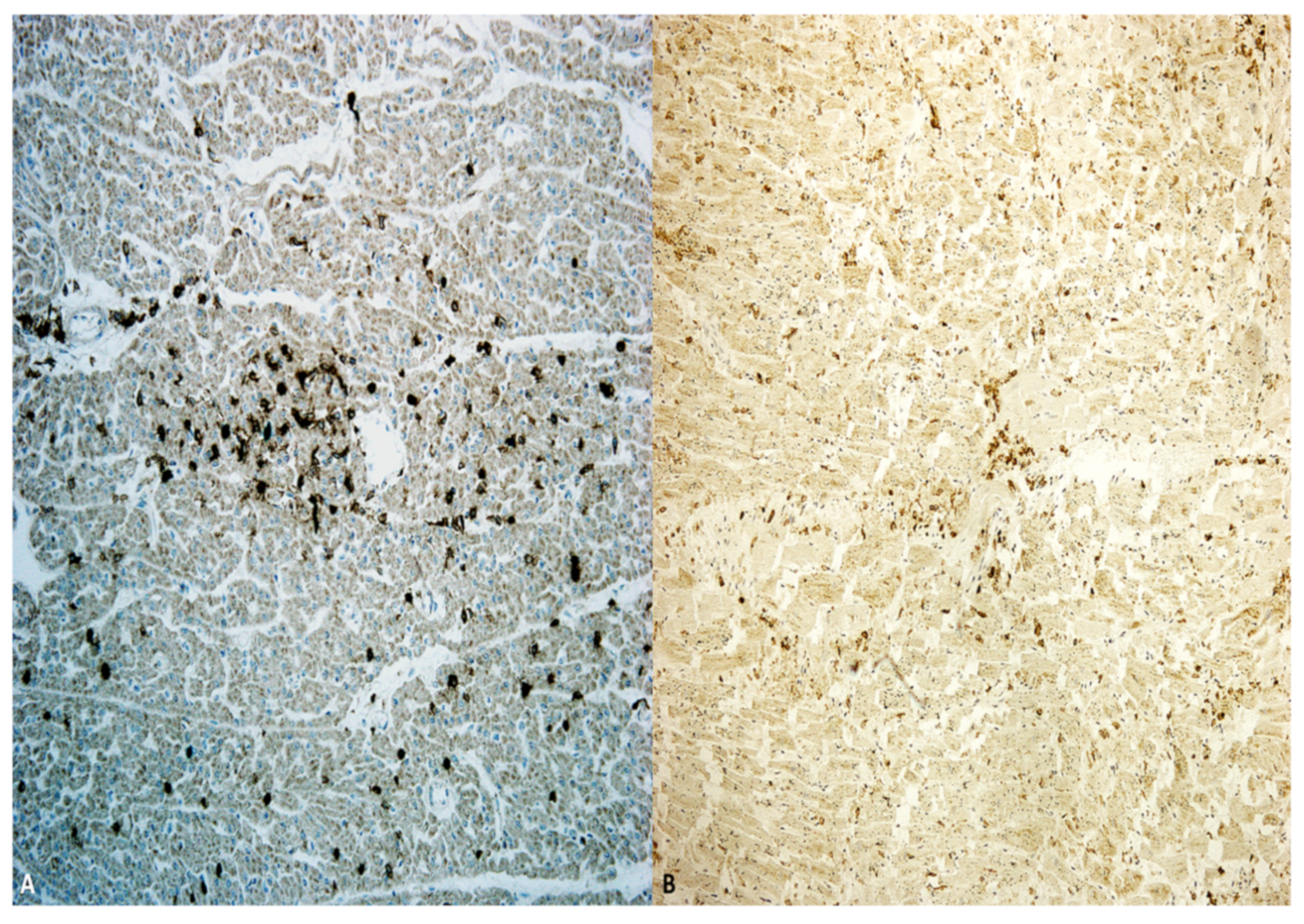
| Authors | Number of Cases | Tissue RNA Scope or TEM | CD3 | CD4 | CD8 | CD4/CD8 | CD68 |
|---|---|---|---|---|---|---|---|
| Lindner et al., 2020 [15] | 39 | 24 | 21 | - | - | 1 | 19 |
| Fox et al., 2020 [16] | 22 | 6 | 6 | 0 | 6 | <1 | - |
| Jacobs et al., 2020 [17] | 1 | 1 | 1 | 1 | 1 | 1:7 | 1 |
| Pellegrini et al., 2020 [19] | 40 | 40 | 8 | - | - | - | - |
| Gauchotte et al., 2021 [20] | 1 | 1 | 1 | 1 | 1 | 1:9 | 1 |
| Bulfamante et al., 2020 [21] | 6 | 6 | 6 | - | - | - | - |
| Del Nonno et al., 2020 [22] | 9 | - | - | - | - | - | - |
| Basso et al., 2020 [23] | 21 | 21 | 21 | 2 | 19 | <1 | 18 |
| Hanley et al., 2020 [24] | 10 | 9 | - | - | - | - | - |
| Rapkiewicz et al., 2020 [25] | 7 | 7 | 1 | 1 | 1 | <1 | 0 |
| Cîrstea et al., 2020 [26] | 1 | 1 | - | - | - | - | - |
| Bradley et al., 2020 [27] | 14 | - | - | - | - | - | - |
| Duarte-Neto et al., 2020 [30] | 10 | - | - | - | - | - | - |
| Schaller et al., 2020 [32] | 10 | 10 | - | - | - | - | - |
| Buja et al., 2020 [33] | 3 | 2 | 2 | 2 | 2 | 2:1 | 1 |
| Bois et al., 2021 [34] | 15 | 15 | - | - | - | - | - |
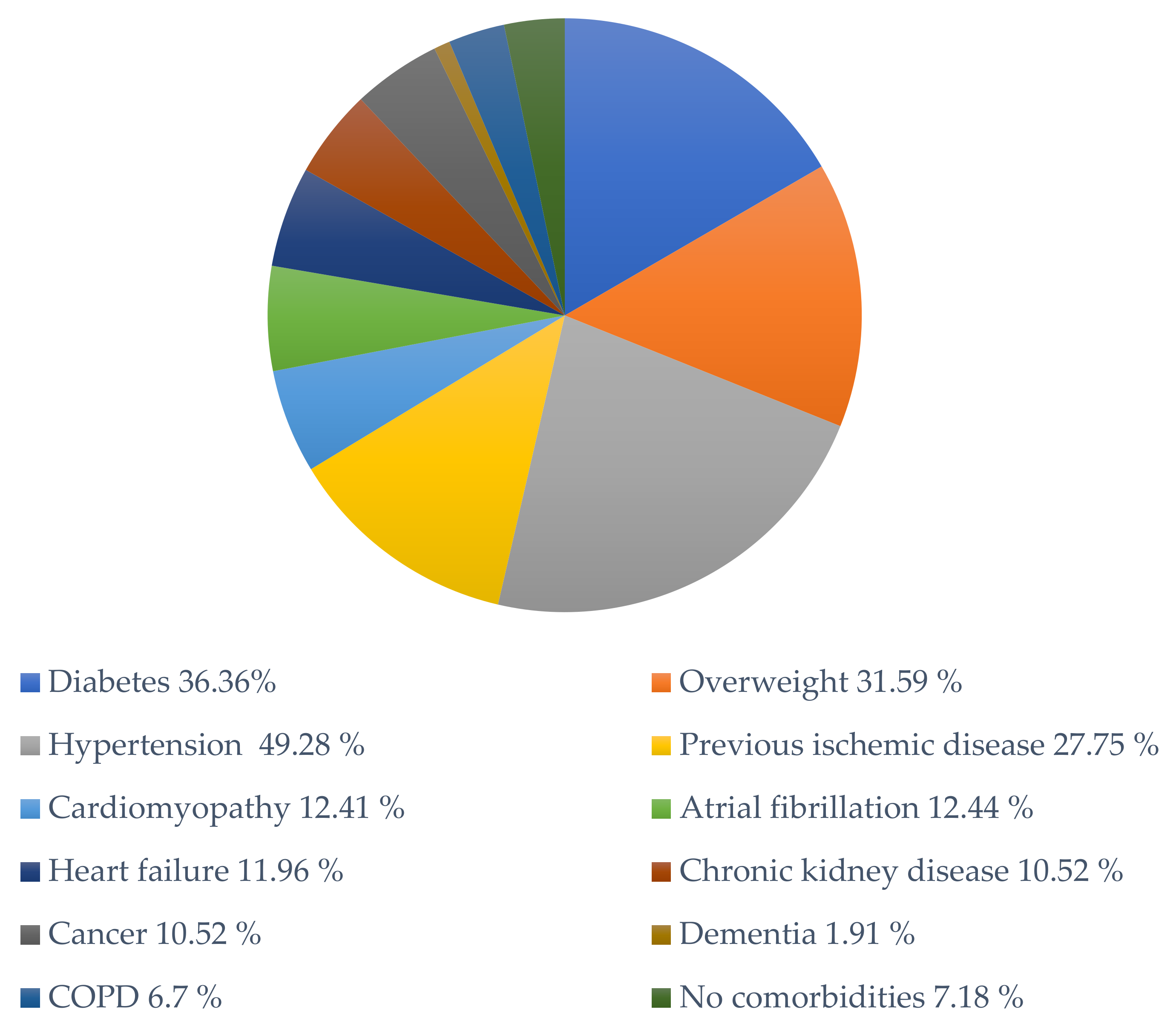
| Number of Cases | Diabetes | Overweight | Hypertension | Previous Ischemic Disease | Cardiomyopathy | Atrial Fibrillation | Heart Failure | Chronic Kidney Disease | Cancer | Dementia | COPD | No Comorbidities | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Lindner et al., 2020 [15] | 39 | 17 | - | 17 | 17 | - | - | 1 | - | - | - | 0 | - |
| Fox et al., 2020 [16] | 22 | 22 | 11 | 9 | 18 | 1 | 9 | 2 | 2 | 4 | 0 | 0 | 0 |
| Jacobs et al., 2020 [17] | 1 | - | 1 | 1 | - | 1 | - | 0 | 0 | 0 | 0 | 0 | 0 |
| Pellegrini et al., 2020 [19] | 40 | 11 | 40 | 29 | 8 | 0 | 3 | 0 | 7 | 5 | 0 | 0 | 0 |
| Gauchotte et al., 2021 [20] | 1 | 1 | 0 | 1 | 1 | 1 | 1 | 0 | 1 | 0 | 0 | 0 | 0 |
| Bulfamante et al., 2020 [21] | 6 | 0 | 0 | 2 | 1 | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 3 |
| Del Nonno et al., 2020 [22] | 9 | - | - | - | - | 2 | 0 | 6 | 0 | 1 | 1 | 0 | 3 |
| Basso et al., 2020 [23] | 21 | 7 | - | 16 | 3 | 7 | 4 | 7 | 2 | 4 | - | 1 | 0 |
| Hanley et al., 2020 [24] | 10 | 0 | 0 | 3 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 3 | 4 |
| Rapkiewicz et al., 2020 [25] | 7 | 5 | 5 | 6 | 1 | 1 | 0 | 0 | 1 | 2 | 0 | 1 | 1 |
| Cîrstea et al., 2020 [26] | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 1 |
| Bradley et al., 2020 [27] | 14 | 5 | 4 | 6 | 4 | 13 | 8 | 8 | 5 | 2 | 2 | 3 | 0 |
| Duarte-Neto et al., 2020 [30] | 10 | 5 | - | 5 | 4 | 5 | - | - | 1 | 1 | - | 3 | 0 |
| Schaller et al., 2020 [32] | 10 | 2 | 2 | 7 | 1 | 3 | - | - | 3 | 3 | 1 | 2 | 0 |
| Buja et al., 2020 [33] | 3 | 1 | 3 | 1 | 0 | 1 | 1 | 1 | 0 | 0 | 0 | 1 | 0 |
| Bois et al., 2021 [34] | 15 | - | - | - | - | - | - | - | - | - | - | - | 3 |
| Total | 209 | 76 | 66 | 103 | 58 | 36 | 26 | 25 | 22 | 22 | 4 | 14 | 15 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Maiese, A.; Frati, P.; Del Duca, F.; Santoro, P.; Manetti, A.C.; La Russa, R.; Di Paolo, M.; Turillazzi, E.; Fineschi, V. Myocardial Pathology in COVID-19-Associated Cardiac Injury: A Systematic Review. Diagnostics 2021, 11, 1647. https://doi.org/10.3390/diagnostics11091647
Maiese A, Frati P, Del Duca F, Santoro P, Manetti AC, La Russa R, Di Paolo M, Turillazzi E, Fineschi V. Myocardial Pathology in COVID-19-Associated Cardiac Injury: A Systematic Review. Diagnostics. 2021; 11(9):1647. https://doi.org/10.3390/diagnostics11091647
Chicago/Turabian StyleMaiese, Aniello, Paola Frati, Fabio Del Duca, Paola Santoro, Alice Chiara Manetti, Raffaele La Russa, Marco Di Paolo, Emanuela Turillazzi, and Vittorio Fineschi. 2021. "Myocardial Pathology in COVID-19-Associated Cardiac Injury: A Systematic Review" Diagnostics 11, no. 9: 1647. https://doi.org/10.3390/diagnostics11091647
APA StyleMaiese, A., Frati, P., Del Duca, F., Santoro, P., Manetti, A. C., La Russa, R., Di Paolo, M., Turillazzi, E., & Fineschi, V. (2021). Myocardial Pathology in COVID-19-Associated Cardiac Injury: A Systematic Review. Diagnostics, 11(9), 1647. https://doi.org/10.3390/diagnostics11091647








