Forecast the Exacerbation in Patients of Chronic Obstructive Pulmonary Disease with Clinical Indicators Using Machine Learning Techniques
Abstract
1. Introduction
2. Related Work
3. Methods and Materials
3.1. Study Design and Subjects
3.2. Data Collection and Experimental Procedure
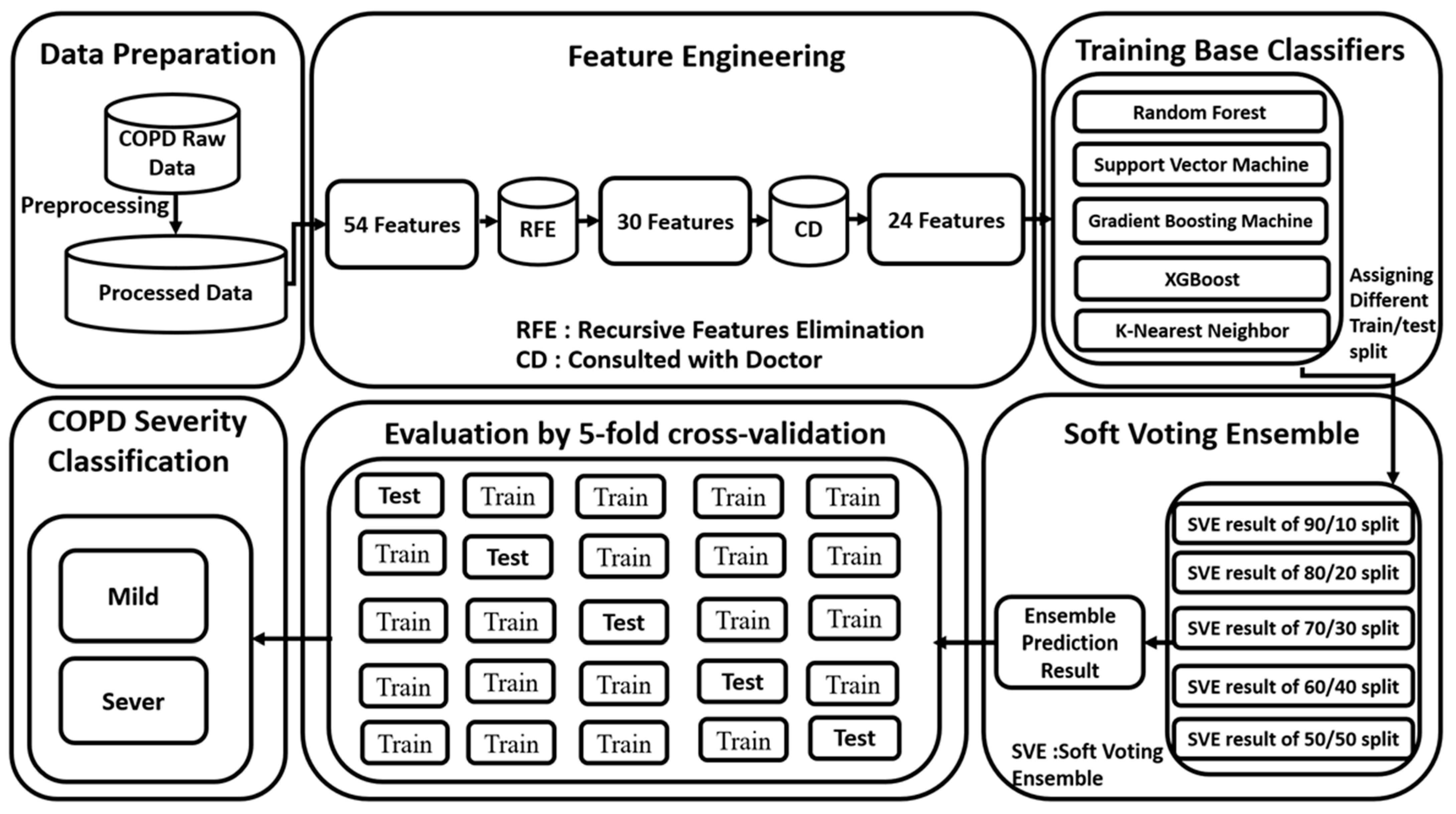
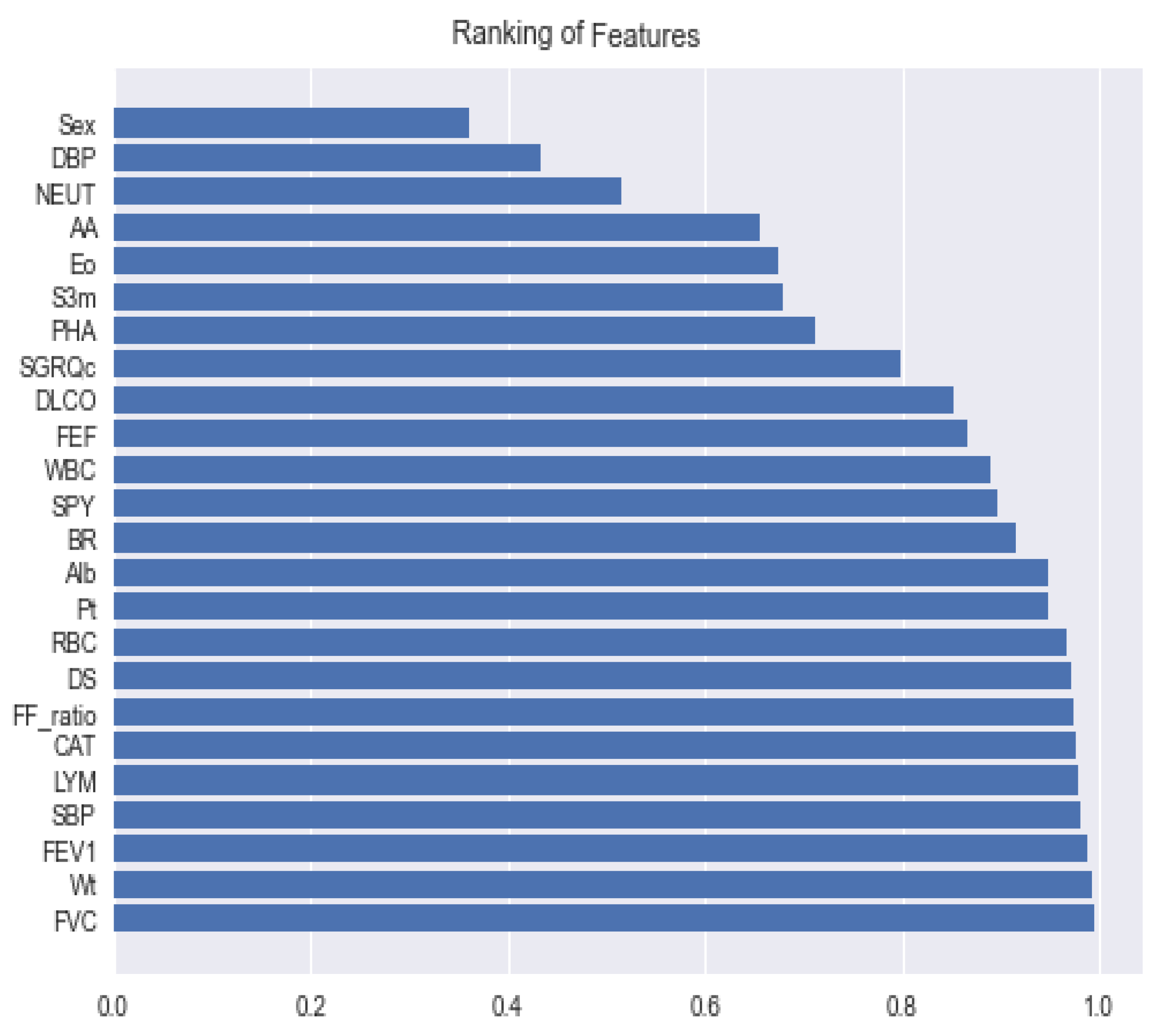
3.3. Feature Engineering
3.4. Machine-Learning Algorithm and Evaluation Metrics
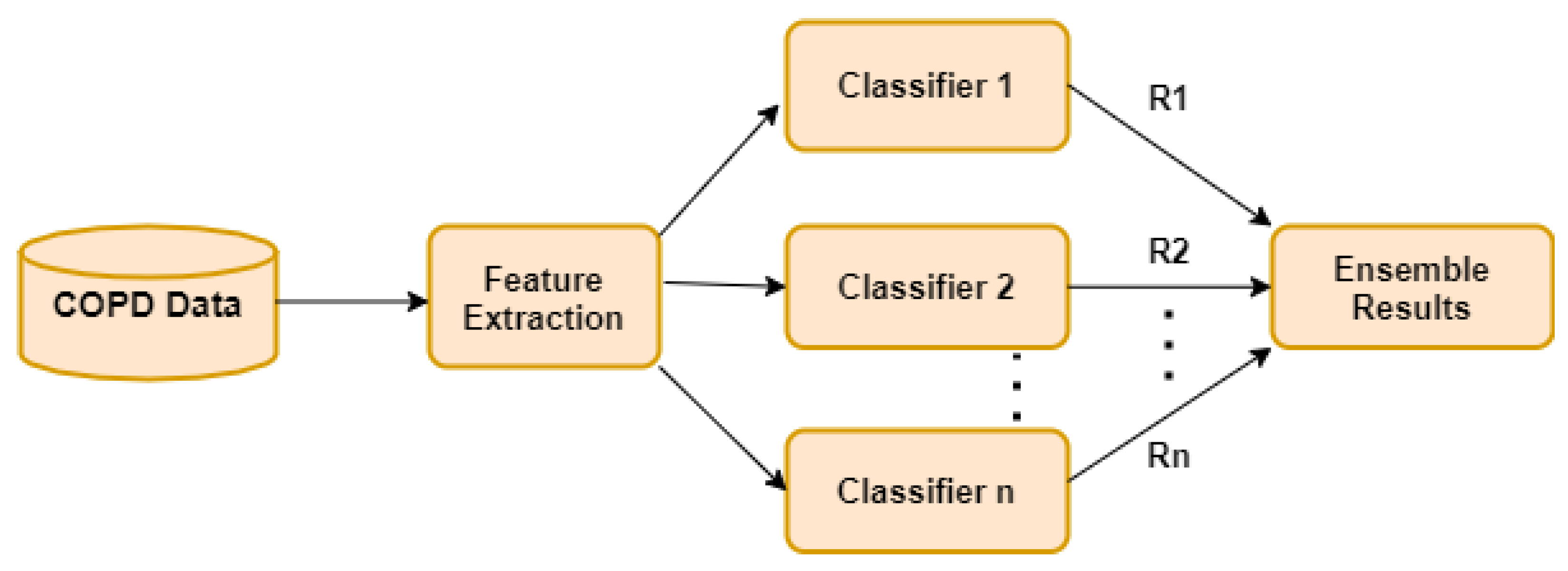
3.5. The Complete Framework of the Proposed Study
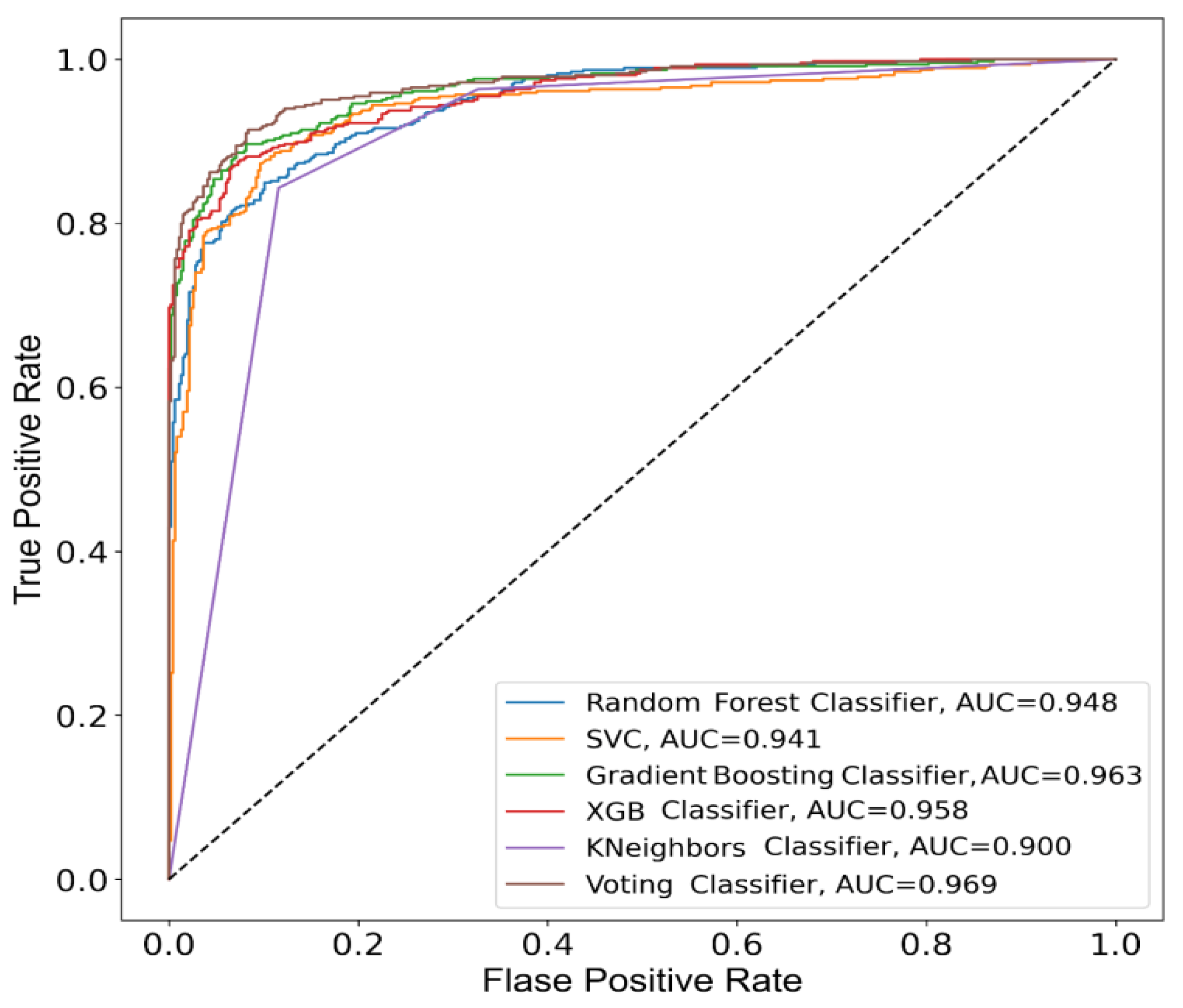
4. Results
5. Discussion
6. Conclusions and Future Work
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| COPD | Chronic obstructive pulmonary disease |
| SVE | Soft voting ensemble |
| ML | Machine learning |
| AI | Artificial intelligence |
| AUC | Area under curve |
| ROC | Receiver operating characteristic |
References
- Hamet, P.; Tremblay, J. Artificial intelligence in medicine. Metabolism 2017, 69, S36–S40. [Google Scholar] [CrossRef] [PubMed]
- Johnson, K.W.; Soto, J.T.; Glicksberg, B.S.; Shameer, K.; Miotto, R.; Ali, M.; Ashley, E.; Dudley, J.T. Artificial intelligence in cardiology. J. Am. Coll. Cardiol. 2018, 71, 2668–2679. [Google Scholar] [PubMed]
- Bini, S.A. Artificial intelligence, machine learning, deep learning, and cognitive computing: What do these terms mean and how will they impact health care? J. Arthroplast. 2018, 33, 2358–2361. [Google Scholar] [CrossRef]
- Saleh, L.; Mcheick, H.; Ajami, H.; Mili, H.; Dargham, J. Comparison of Machine Learning Algorithms to Increase Prediction Accuracy of COPD Domain. In Proceedings of the International Conference on Smart Homes and Health Telematics, Paris, France, 29–31 August 2017; Springer: Cham, Switzerland, 2017; pp. 247–254. [Google Scholar]
- Connors, A.F., Jr.; Dawson, N.V.; Thomas, C.; Harrell, F.E., Jr.; Desbiens, N.; Fulkerson, W.J.; Kussin, P.; Bellamy, P.; Goldman, L.; Knaus, W.A. Outcomes following acute exacerbation of severe chronic obstructive lung disease. The SUPPORT investigators (Study to Understand Prognoses and Preferences for Outcomes and Risks of Treatments). Am. J. Respir. Crit. Care Med. 1996, 154, 959–967. [Google Scholar] [CrossRef] [PubMed]
- Lareau, S.; Moseson, E.; Slatore, C.G. Patient information series: Exacerbation of COPD. Am. J. Respir. Crit. Care Med. 2014, 189, P11–P12. [Google Scholar] [PubMed]
- Chronic Respiratory Diseases. Burden of COPD. Available online: https://www.who.int/respiratory/copd/burden/en/ (accessed on 25 December 2020).
- Bhogal, S.K.; McGillivray, D.; Bourbeau, J.; Benedetti, A.; Bartlett, S.; Ducharme, F.M. Early administration of systemic corticosteroids reduces hospital admission rates for children with moderate and severe asthma exacerbation. Ann. Emerg. Med. 2012, 60, 84–91. [Google Scholar]
- Shope, T.R.; Cabana, M.D.; Zorc, J.J. Early predictors of admission or prolonged emergency department treatment for children with acute asthma. Ambul. Child Health 2001, 7, 11–21. [Google Scholar] [CrossRef]
- Chronic Obstructive Pulmonary Disease (COPD). Available online: https://www.who.int/news-room/fact-sheets/detail/chronic-obstructive-pulmonary-disease-(copd) (accessed on 25 December 2020).
- Anzueto, A. Impact of exacerbations on COPD. Eur. Respir. Rev. Off. J. Eur. Respir. Soc. 2010, 19, 113–118. [Google Scholar] [CrossRef]
- U.S. Department of Health and Human Services, National Institutes of Health, National Heart, Lung, and Blood Institute. COPD External Icon. Available online: https://www.nhlbi.nih.gov/science/lung-diseases (accessed on 27 December 2020).
- US Department of Health and Human Services. The Health Consequences of Smoking—50 Years of Progress: A Report of the Surgeon General; Centers for Disease Control and Prevention (US): Atlanta, GA, USA, 2014. [Google Scholar]
- Rao, K.M. Diagnosis and management of chronic cough due to extrapulmonary etiologies. Indian J. Clin. Pract. 2014, 25, 437–442. [Google Scholar]
- Kerkhof, M.; Freeman, D.; Jones, R.; Chisholm, A.; Price, D.B. Predicting frequent COPD exacerbations using primary care data [Corrigendum]. Int. J. Chronic Obstr. Pulm. Dis. 2018, 13, 2471–2472. [Google Scholar] [CrossRef]
- Chen, C.Z.; Ou, C.Y.; Hsu, C.H.; Hsiue, T.R. Validation of the GOLD 2013 classification in predicting exacerbations and mortality in Taiwanese patients with chronic obstructive pulmonary disease. J. Formos. Med. Assoc. 2015, 114, 1258–1266. [Google Scholar] [CrossRef][Green Version]
- Husebø, G.R.; Bakke, P.S.; Aanerud, M.; Hardie, J.A.; Ueland, T.; Grønseth, R.; Persson, L.J.; Aukrust, P.; Eagan, T.M. Predictors of exacerbations in chronic obstructive pulmonary disease-results from the Bergen COPD cohort study. PLoS ONE 2014, 9, e109721. [Google Scholar] [CrossRef] [PubMed][Green Version]
- To, T.; Zhu, J.; Larsen, K.; Simatovic, J.; Feldman, L.; Ryckman, K.; Gershon, A.; Lougheed, M.D.; Licskai, C.; Chen, H.; et al. Progression from asthma to chronic obstructive pulmonary disease. Is air pollution a risk factor? Am. J. Respir. Crit. Care Med. 2016, 194, 429–438. [Google Scholar]
- Kurai, D.; Saraya, T.; Ishii, H.; Takizawa, H. Virus-induced exacerbations in asthma and COPD. Front. Microbiol. 2013, 4, 293. [Google Scholar]
- Wedzicha, J.A. Role of viruses in exacerbations of chronic obstructive pulmonary disease. Proc. Am. Thorac. Soc. 2004, 1, 115–120. [Google Scholar]
- Buch, V.H.; Ahmed, I.; Maruthappu, M. Artificial intelligence in medicine: Current trends and future possibilities. Br. J. Gen. Pr. 2018, 68, 143–144. [Google Scholar] [CrossRef] [PubMed]
- Deo, R.C. Machine learning in medicine. Circulation 2015, 132, 1920–1930. [Google Scholar] [CrossRef]
- Peek, N.; Combi, C.; Marin, R.; Bellazzi, R. Thirty years of artificial intelligence in medicine (AIME) conferences: A review of research themes. Artif. Intell. Med. 2015, 65, 61–73. [Google Scholar]
- Battineni, G.; Sagaro, G.G.; Nalini, C.; Amenta, F.; Tayebati, S.K. Comparative Machine-Learning Approach: A Follow-Up Study on Type 2 Diabetes Predictions by Cross-Validation Methods. Machines 2019, 7, 74. [Google Scholar]
- Aich, S.; Pradhan, P.M.; Park, J.; Sethi, N.; Vathsa, V.S.S.; Kim, H.C. A validation study of freezing of gait (FoG) detection and machine-learning-based FoG prediction using estimated gait characteristics with a wearable accelerometer. Sensors 2018, 18, 3287. [Google Scholar] [CrossRef]
- Chang, W.; Liu, Y.; Xiao, Y.; Yuan, X.; Xu, X.; Zhang, S.; Zhou, S. A machine-learning-based prediction method for hypertension outcomes based on medical data. Diagnostics 2019, 9, 178. [Google Scholar] [CrossRef] [PubMed]
- Abedi, V.; Avula, V.; Chaudhary, D.; Shahjouei, S.; Khan, A.; Griessenauer, C.J.; Li, J.; Zand, R. Prediction of Long-Term Stroke Recurrence Using Machine Learning Models. J. Clin. Med. 2021, 10, 1286. [Google Scholar] [CrossRef] [PubMed]
- Aich, S.; Pradhan, P.M.; Park, J.; Kim, H.C. A machine learning approach to distinguish Parkinson’s disease (PD) patient’s with shuffling gait from older adults based on gait signals using 3D motion analysis. Int. J. Eng. Technol. 2018, 7, 153–156. [Google Scholar] [CrossRef]
- Aich, S.; Pradhan, P.M.; Chakraborty, S.; Kim, H.C.; Kim, H.T.; Lee, H.G.; Kim, I.H.; Joo, M.; Seong, S.J.; Park, J. Design of a Machine Learning-Assisted Wearable Accelerometer-Based Automated System for Studying the Effect of Dopaminergic Medicine on Gait Characteristics of Parkinson’s Patients. J. Healthc. Eng. 2020, 2020, 1823268. [Google Scholar] [CrossRef] [PubMed]
- Saha, S.; Ekbal, A. Combining multiple classifiers using vote based classifier ensemble technique for named entity recognition. Data Knowl. Eng. 2013, 85, 15–39. [Google Scholar] [CrossRef]
- Tan, P.N.; Steinbach, M.; Kumar, V. Methods for constructing an ensemble classifier. In Introduction to Data Mining; Pearson Education: New York, NY, USA, 2006; Volume 5. [Google Scholar]
- Saqlain, M.; Jargalsaikhan, B.; Lee, J.Y. A voting ensemble classifier for wafer map defect patterns identification in semiconductor manufacturing. IEEE Trans. Semicond. Manuf. 2019, 32, 171–182. [Google Scholar]
- Zhang, C.; Ma, Y. (Eds.) Ensemble Machine Learning: Methods and Applications; Springer: New York, NY, USA, 2012. [Google Scholar]
- Chawla, N.V.; Bowyer, K.W.; Hall, L.O.; Kegelmeyer, W.P. SMOTE: Synthetic minority over-sampling technique. J. Artif. Intell. Res. 2002, 16, 321–357. [Google Scholar] [CrossRef]
- Peng, J.; Chen, C.; Zhou, M.; Xie, X.; Zhou, Y.; Luo, C.H. A machine-learning approach to forecast aggravation risk in patients with acute exacerbation of chronic obstructive pulmonary disease with clinical indicators. Sci. Rep. 2020, 10, 1–9. [Google Scholar]
- Nunavath, V.; Goodwin, M.; Fidje, J.T.; Moe, C.E. Deep Neural Networks for Prediction of Exacerbations of Patients with Chronic Obstructive Pulmonary Disease. In Proceedings of the International Conference on Engineering Applications of Neural Networks, Bristol, UK, 3–5 September 2018; Springer: Cham, Switzerland, 2018; pp. 217–228. [Google Scholar]
- Vora, S.; Shah, C. COPD Classification Using Machine Learning Algorithms. Int. Res. J. Eng. Technol. 2019, 6, 608–611. [Google Scholar]
- Fernandez-Granero, M.A.; Sanchez-Morillo, D.; Leon-Jimenez, A. An artificial intelligence approach to early predict symptom-based exacerbations of COPD. Biotechnol. Biotechnol. Equip. 2018, 32, 778–784. [Google Scholar] [CrossRef]
- Amaral, J.L.; Faria, A.C.; Lopes, A.J.; Jansen, J.M.; Melo, P.L. Automatic Identification of Chronic Obstructive Pulmonary Disease Based on Forced Oscillation Measurements and Artificial Neural Networks. In Proceedings of the 2010 Annual International Conference of the IEEE Engineering in Medicine and Biology, Buenos Aires, Argentina, 31 August–4 September 2010; pp. 1394–1397. [Google Scholar]
- Kanwade, A.; Bairagi, V.K. Classification of COPD and normal lung airways using feature extraction of electromyographic signals. J. King Saud Univ. Comput. Inf. Sci. 2019, 31, 506–513. [Google Scholar] [CrossRef]
- Fang, Y.; Wang, H.; Wang, L.; Di, R.; Song, Y. Diagnosis of copd based on a knowledge graph and integrated model. IEEE Access 2019, 7, 46004–46013. [Google Scholar] [CrossRef]
- Hakim, M.A.; Garden, F.L.; Jennings, M.D.; Dobler, C.C. Performance of the LACE index to predict 30-day hospital readmissions in patients with chronic obstructive pulmonary disease. Clin. Epidemiol. 2018, 10, 51. [Google Scholar] [CrossRef]
- Amalakuhan, B.; Kiljanek, L.; Parvathaneni, A.; Hester, M.; Cheriyath, P.; Fischman, D. A prediction model for COPD readmissions: Catching up, catching our breath, and improving a national problem. J. Community Hosp. Intern. Med. Perspect. 2012, 2, 9915. [Google Scholar]
- Badnjevic, A.; Cifrek, M.; Koruga, D. Classification of Chronic Obstructive Pulmonary Disease (COPD) Using Integrated Software Suite. In Proceedings of the XIII Mediterranean Conference on Medical and Biological Engineering and Computing, Seville, Spain, 25–28 September 2013; Springer: Cham, Switzerland, 2014; pp. 911–914. [Google Scholar]
- Barúa, M.; Nazeran, H.; Nava, P.; Granda, V.; Diong, B. Classification of Pulmonary Diseases Based on Impulse Oscillometric Measurements of Lung Function Using Neural Networks. In Proceedings of the 26th Annual International Conference of the IEEE Engineering in Medicine and Biology Society, San Francisco, CA, USA, 1–5 September 2004; Volume 2, pp. 3848–3851. [Google Scholar]
- Er, O.; Temurtas, F. A study on chronic obstructive pulmonary disease diagnosis using multilayer neural networks. J. Med Syst. 2008, 32, 429–432. [Google Scholar] [CrossRef] [PubMed]
- Fernandez-Granero, M.A.; Sanchez-Morillo, D.; Leon-Jimenez, A. Computerised analysis of telemonitored respiratory sounds for predicting acute exacerbations of COPD. Sensors 2015, 15, 26978–26996. [Google Scholar] [CrossRef] [PubMed]
- Işık, Ü.; Güven, A.; Büyükoğlan, H. Chronic Obstructive Pulmonary Disease Classification with Artificial Neural Networks. In Proceedings of the IEEE 2015 Medical Technologies National Conference (TIPTEKNO), Bodrum, Turkey, 15–18 October 2015; pp. 1–4. [Google Scholar]
- Swaminathan, S.; Qirko, K.; Smith, T.; Corcoran, E.; Wysham, N.G.; Bazaz, G.; Kappel, G.; Gerber, A.N. A machine learning approach to triaging patients with chronic obstructive pulmonary disease. PLoS ONE 2017, 12, e0188532. [Google Scholar]
- Yang, C.; Delcher, C.; Shenkman, E.; Ranka, S. Predicting 30-day all-cause readmissions from hospital inpatient discharge data. In Proceedings of the 2016 IEEE 18th International Conference on e-Health Networking, Applications and Services (Healthcom), Munich, Germany, 14–16 September 2016; pp. 1–6. [Google Scholar]
- Raghavan, N.; Lam, Y.M.; Webb, K.A.; Guenette, J.A.; Amornputtisathaporn, N.; Raghavan, R.; Tan, W.C.; Bourbeau, J.; O’Donnell, D.E. Components of the COPD Assessment Test (CAT) associated with a diagnosis of COPD in a random population sample. COPD J. Chronic Obstr. Pulm. Dis. 2012, 9, 175–183. [Google Scholar] [CrossRef]
- Xia, J.; Sun, L.; Xu, S.; Xiang, Q.; Zhao, J.; Xiong, W.; Xu, Y.; Chu, S. A Model Using Support Vector Machines Recursive Feature Elimination (SVM-RFE) Algorithm to Classify Whether COPD Patients Have Been Continuously Managed According to GOLD Guidelines. Int. J. Chronic Obstr. Pulm. Dis. 2020, 15, 2779. [Google Scholar] [CrossRef] [PubMed]
- Spathis, D.; Vlamos, P. Diagnosing asthma and chronic obstructive pulmonary disease with machine learning. Health Inform. J. 2019, 25, 811–827. [Google Scholar]
- Tang, J.; Alelyani, S.; Liu, H. Feature selection for classification: A review. Data Classif. Algorithms Appl. 2014. [Google Scholar] [CrossRef]
- Kumar, V.; Minz, S. Feature selection: A literature review. SmartCR 2014, 4, 211–229. [Google Scholar] [CrossRef]
- Bolón-Canedo, V.; Sánchez-Marono, N.; Alonso-Betanzos, A.; Benítez, J.M.; Herrera, F. A review of microarray datasets and applied feature selection methods. Inf. Sci. 2014, 282, 111–135. [Google Scholar]
- Ang, J.C.; Mirzal, A.; Haron, H.; Hamed, H.N.A. Supervised, unsupervised, and semi-supervised feature selection: A review on gene selection. IEEE/ACM Trans. Comput. Biol. Bioinform. 2015, 13, 971–989. [Google Scholar] [CrossRef]
- Jeon, H.; Oh, S. Hybrid-Recursive Feature Elimination for Efficient Feature Selection. Appl. Sci. 2020, 10, 3211. [Google Scholar] [CrossRef]
- Aich, S.; Youn, J.; Chakraborty, S.; Pradhan, P.M.; Park, J.H.; Park, S.; Park, J. A Supervised Machine Learning Approach to Detect the On/Off State in Parkinson’s Disease Using Wearable Based Gait Signals. Diagnostics 2020, 10, 421. [Google Scholar] [CrossRef]
- Mcheick, H.; Saleh, L.; Ajami, H.; Mili, H. Context relevant prediction model for COPD domain using bayesian belief network. Sensors 2017, 17, 1486. [Google Scholar] [CrossRef] [PubMed]
- Do Sun Kwon, Y.J.C.; Kim, T.H.; Byun, M.K.; Cho, J.H.; Kim, H.J.; Park, H.J. FEF25-75% Values in Patients with Normal Lung Function Can Predict the Development of Chronic Obstructive Pulmonary Disease. Int. J. Chronic Obstr. Pulm. Dis. 2020, 15, 2913. [Google Scholar] [CrossRef] [PubMed]
- Ryynänen, O.P.; Soini, E.J.; Lindqvist, A.; Kilpeläinen, M.; Laitinen, T. Bayesian predictors of very poor health related quality of life and mortality in patients with COPD. BMC Med Inform. Decis. Mak. 2013, 13, 1–10. [Google Scholar]
- Zinellu, E.; Fois, A.G.; Sotgiu, E.; Mellino, S.; Mangoni, A.A.; Carru, C.; Zinellu, A.; Pirina, P. Serum Albumin Concentrations in Stable Chronic Obstructive Pulmonary Disease: A Systematic Review and Meta-Analysis. J. Clin. Med. 2021, 10, 269. [Google Scholar] [CrossRef]
- Lee, J.H.; You, H.S.; Kwon, Y.J.; Lee, J.W. Relationship between White Blood Cell Count and Pulmonary Function Test Results in Korean Adults: The 2007-2009 Korea National Health and Nutrition Examination Survey. Korean J. Fam. Pract. 2018, 8, 52–58. [Google Scholar]
- Verberne, L.D.; Leemrijse, C.J.; Swinkels, I.C.; van Dijk, C.E.; de Bakker, D.H.; Nielen, M.M. Overweight in patients with chronic obstructive pulmonary disease needs more attention: A cross-sectional study in general practice. NPJ Prim. Care Respir. Med. 2017, 27, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Global Initiative for Chronic Obstructive Lung Disease. Global Strategy for the Diagnosis, Management, and Prevention of Chronic Obstructive Pulmonary Disease. 2017 Report. Available online: http://www.goldcopd.org (accessed on 26 February 2021).
- Maclay, J.D.; McAllister, D.A.; Johnston, S.; Raftis, J.; McGuinnes, C.; Deans, A.; Newby, D.E.; Mills, N.L.; MacNee, W. Increased platelet activation in patients with stable and acute exacerbation of COPD. Thorax 2011, 66, 769–774. [Google Scholar] [CrossRef] [PubMed]
- Sin, D.D. The Devastating Power of Platelets in COPD Exacerbations: Can Aspirin Save Lives in COPD? Thorax 2014, 69, 603–604. [Google Scholar] [CrossRef] [PubMed]
- Arslan, S.; Yildiz, G.; Özdemir, L.; Kaysoydu, E.; Özdemir, B. Association between blood pressure, inflammation and spirometry parameters in chronic obstructive pulmonary disease. Korean J. Intern. Med. 2019, 34, 108. [Google Scholar]
- Ozgul, G.; Seyhan, E.C.; Ozgul, M.A.; Gunluoglu, M.Z. Red blood cell distribution width in patients with chronic obstructive pulmonary disease and healthy subjects. Arch. Bronconeumol. 2017, 53, 107–113. [Google Scholar] [CrossRef]
- Sears, M.R. Smoking, Asthma, Chronic Airflow Obstruction and COPD. Eur. Respir. J. 2015, 45, 586–588. [Google Scholar] [CrossRef] [PubMed]
| Reference | Features | Classifiers | Outcomes | Performance Indices |
|---|---|---|---|---|
| [36] | Clinical | LSTM, ANN, SVM | 92.86% | Accuracy |
| [38] | Clinical | DTF | 75.8% | Accuracy |
| [39] | Clinical | ANN | More than 90% | Sensitivity, Specificity, AUC |
| [40] | Clinical | Naïve Bayes, SVM | 87.8% | Accuracy |
| [43] | Clinical | RF | 75% | Sensitivity, Specificity |
| [44] | Clinical | ANN | 92% | Accuracy |
| [46] | Clinical | MLNN | 94.46% | Accuracy |
| [49] | Clinical | GBDT, LR | 89.1% | Accuracy |
| [51] | Clinical | LR | 77.6% | Sensitivity, Specificity |
| No. | Attributes | Value | Description |
|---|---|---|---|
| 1 | Sex | M/F | Male/Female |
| 2 | DBP | Numerical | Diastolic blood pressure (DBP) |
| 3 | NEUT | Numerical | Neutrophil (NEUT) |
| 4 | AA | Yes, no | Availability of asthma (AA) |
| 5 | EO | Numerical | Eosinophils (EO) |
| 6 | Sputum3m | Yes, no | You have had phlegm almost every day for more than three months a year. Is it? |
| 7 | PHA | Yes, no | History of asthma (PHA) |
| 8 | SGRQc | Numerical | The St. George’s Respiratory Questionnaire (SGRQc). Over the past year, you’ve had several respiratory symptoms. Have you experienced it? |
| 9 | DLCO | Numerical | Diffusing capacity for carbon monoxide (DLCO) |
| 10 | FEF | Numerical | The forced mid-expiratory flow (FEF) |
| 11 | WBC | Numerical | White blood cell (WBC) |
| 12 | SPY | Numerical | Smoke per year (SPY) |
| 13 | BR | Numerical | Breath Result (BR) |
| 14 | Alb | Numerical | Albumin (Alb) |
| 15 | Pt | Numerical | Platelets (Pt) |
| 16 | RBC | Numerical | Red blood cells (RBC) |
| 17 | DS | Numerical | Duration of Smoke (DS) |
| 18 | FF_ratio | Numerical | The ratio FEV1/FVC |
| 19 | CAT | Numerical | COPD Assessment Test (CAT) |
| 20 | LYM | Numerical | Lymphocytes (LYM) |
| 21 | SBP | Numerical | Systolic blood pressure (SBP) |
| 22 | FEV1 | Numerical | Forced expiratory volume in one second (FEV1) |
| 23 | Wt | Numerical | Weight (Wt) |
| 24 | FVC | Numerical | FVC (forced vital capacity): maximum volume of air that can be exhaled during a forced maneuver |
| Classifier | Specification |
|---|---|
| Random Forest | n_estimators = 500, random_state = 0, criterion = ‘gini’, max_depth = 15, min_samples_split = 5, min_samples_leaf = 5 |
| Support Vector Machine | kernel = ‘rbf’, degree = 4, gamma = 7.9, C = 20, decision_function_shape = ‘ovr’, probability = True, random_state = 0 |
| Gradient Boosting Machine | learning_rate = 0.1, n_estimators = 500, max_depth = 15, min_samples_split = 5, min_samples_leaf = 5, subsample = 1, max_features = ‘sqrt’, random_state = 10 |
| XGBoost | random_state = 0, silent = False, scale_pos_weight = 2, learning_rate = 0.1, colsample_bytree = 0.4, subsample = 0.9, objective = ‘binary:logistic’, n_estimators = 500, reg_alpha = 0.01, max_depth = 15, gamma = 7 |
| K-nearest neighbor | n_neighbors = 2, weights = ‘uniform’, algorithm = ‘auto’, leaf_size = 40, p = 2, metric = ‘manhattan’ |
| Classifier | 5-Fold Cross Validation (%) | |||||
|---|---|---|---|---|---|---|
| 1st Fold | 2nd Fold | 3rd Fold | 4th Fold | 5th Fold | Average | |
| Random Forest | 84.0268 | 83.8926 | 86.3087 | 88.9784 | 87.2311 | 86.0875 |
| Support Vector Machine | 87.1140 | 87.5167 | 89.1275 | 91.5322 | 88.1720 | 88.6925 |
| Gradient Boosting Machine | 88.8590 | 88.4563 | 90.7382 | 91.5322 | 91.1290 | 90.1429 |
| XGBoost | 84.9664 | 84.0268 | 87.5167 | 90.0537 | 86.4247 | 86.5976 |
| K-nearest neighbor | 84.4295 | 85.3691 | 87.3825 | 86.2903 | 86.6935 | 86.0329 |
| Soft voting ensemble (SVE) | 90.2013 | 88.1879 | 92.2147 | 93.6827 | 91.1290 | 91.0831 |
| Classifier | Disease Severity | Precision | Recall | F-Measure |
|---|---|---|---|---|
| RF | Mild | 85.3360 | 89.9141 | 87.5652 |
| Severe | 89.3181 | 84.5161 | 86.8507 | |
| SVM | Mild | 89.2070 | 86.9098 | 88.0434 |
| Severe | 87.2117 | 89.4623 | 88.3227 | |
| GBM | Mild | 87.7263 | 93.5622 | 90.5503 |
| Severe | 93.0875 | 86.8817 | 89.8776 | |
| XGB | Mild | 89.3569 | 86.4806 | 87.8951 |
| Severe | 86.8750 | 89.6774 | 88.2539 | |
| KNN | Mild | 84.9484 | 88.4120 | 86.6456 |
| Severe | 87.8923 | 84.3010 | 86.0591 | |
| SVE | Mild | 91.3606 | 90.7725 | 91.0656 |
| Severe | 90.8119 | 91.3978 | 91.1039 |
| Classifier | Accuracy | Precision | Recall | F-Measure | AUC |
|---|---|---|---|---|---|
| RF | 87.2180 | 89.9141 | 85.3360 | 87.5652 | 94.7875 |
| SVM | 88.1847 | 86.9098 | 89.2070 | 88.0434 | 94.0616 |
| GBM | 90.2255 | 93.5622 | 87.7263 | 90.5503 | 96.3192 |
| XGB | 88.0773 | 86.4806 | 89.3569 | 87.8952 | 95.8452 |
| KNN | 86.3587 | 88.4120 | 84.9484 | 86.6456 | 90.0259 |
| SVE | 91.0849 | 90.7725 | 91.3607 | 91.0656 | 96.8656 |
| Classifier | Different Division of Training Set (%) and Testing Set (%) | |||||
|---|---|---|---|---|---|---|
| 90/10 | 80/20 | 70/30 | 60/40 | 50/50 | Mean ± STD | |
| Random Forest | 87.9167 | 85.3360 | 84.5222 | 84.3592 | 84.0260 | 85.2320 ± 1.5762 |
| Support Vector Machine | 92.3076 | 89.2070 | 87.2675 | 86.1490 | 84.3485 | 87.8559 ± 3.0497 |
| Gradient Boosting Machine | 90.0000 | 87.7263 | 87.6177 | 87.4747 | 86.5853 | 87.7822 ± 1.1592 |
| XGBoost | 90.9090 | 89.3569 | 89.5434 | 89.3289 | 88.4892 | 89.1192 ± 0.4288 |
| K-nearest neighbor | 91.071 | 84.9484 | 84.0599 | 80.8593 | 79.3822 | 84.0641 ± 4.5296 |
| Soft voting ensemble (SVE) | 94.5945 | 91.3607 | 90.7172 | 89.5288 | 89.0691 | 91.0540 ± 2.1799 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hussain, A.; Choi, H.-E.; Kim, H.-J.; Aich, S.; Saqlain, M.; Kim, H.-C. Forecast the Exacerbation in Patients of Chronic Obstructive Pulmonary Disease with Clinical Indicators Using Machine Learning Techniques. Diagnostics 2021, 11, 829. https://doi.org/10.3390/diagnostics11050829
Hussain A, Choi H-E, Kim H-J, Aich S, Saqlain M, Kim H-C. Forecast the Exacerbation in Patients of Chronic Obstructive Pulmonary Disease with Clinical Indicators Using Machine Learning Techniques. Diagnostics. 2021; 11(5):829. https://doi.org/10.3390/diagnostics11050829
Chicago/Turabian StyleHussain, Ali, Hee-Eun Choi, Hyo-Jung Kim, Satyabrata Aich, Muhammad Saqlain, and Hee-Cheol Kim. 2021. "Forecast the Exacerbation in Patients of Chronic Obstructive Pulmonary Disease with Clinical Indicators Using Machine Learning Techniques" Diagnostics 11, no. 5: 829. https://doi.org/10.3390/diagnostics11050829
APA StyleHussain, A., Choi, H.-E., Kim, H.-J., Aich, S., Saqlain, M., & Kim, H.-C. (2021). Forecast the Exacerbation in Patients of Chronic Obstructive Pulmonary Disease with Clinical Indicators Using Machine Learning Techniques. Diagnostics, 11(5), 829. https://doi.org/10.3390/diagnostics11050829









