FDG-PET/CT for Response Monitoring in Metastatic Breast Cancer: The Feasibility and Benefits of Applying PERCIST
Abstract
1. Introduction
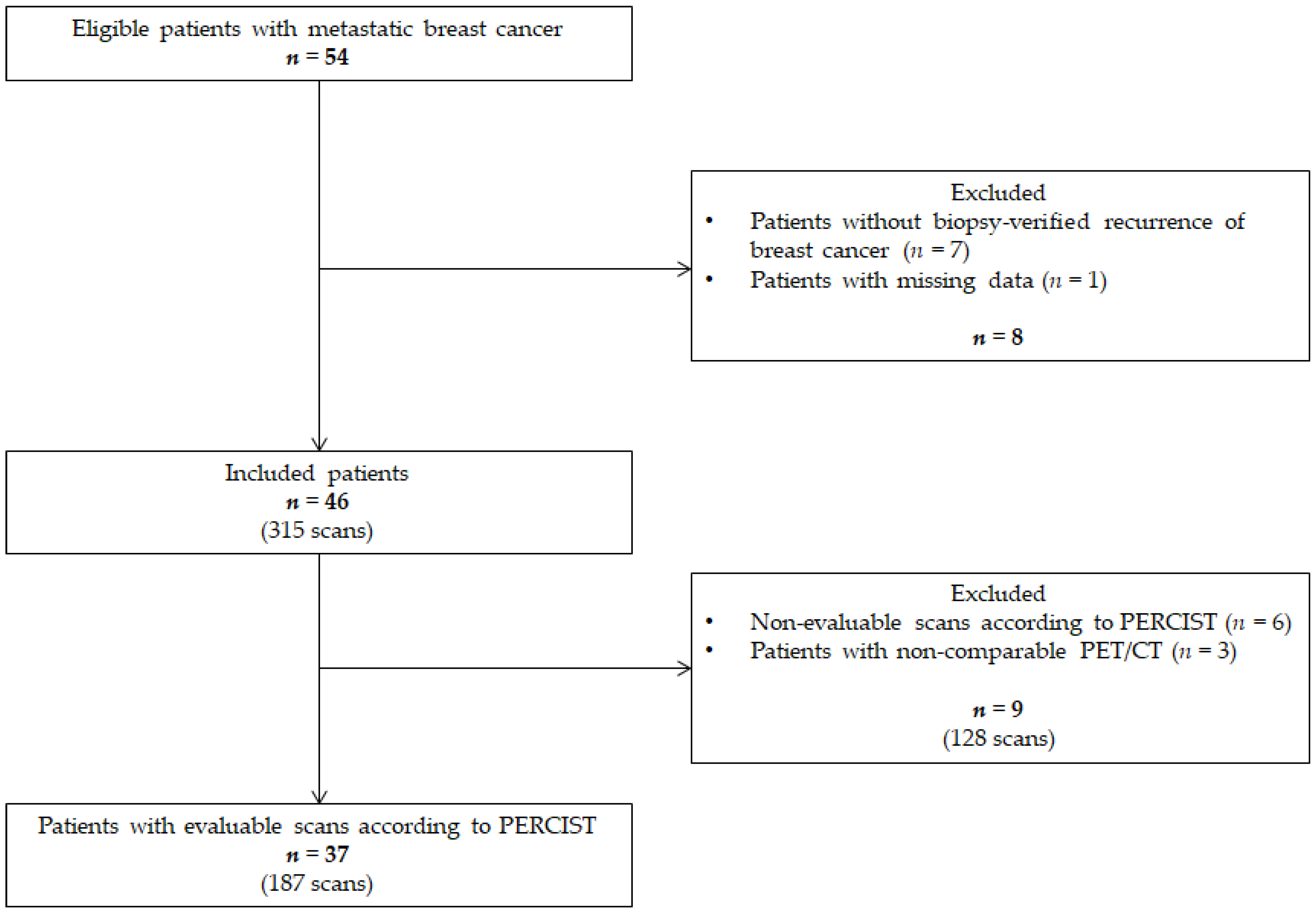
2. Materials and Methods
2.1. Setting and Participants
2.2. Imaging Techniques and Standardization Protocol
2.3. Visual Assessment of Scans
2.4. Assessment of Comparability by PERCIST
2.5. Assessment of Measurability by PERCIST
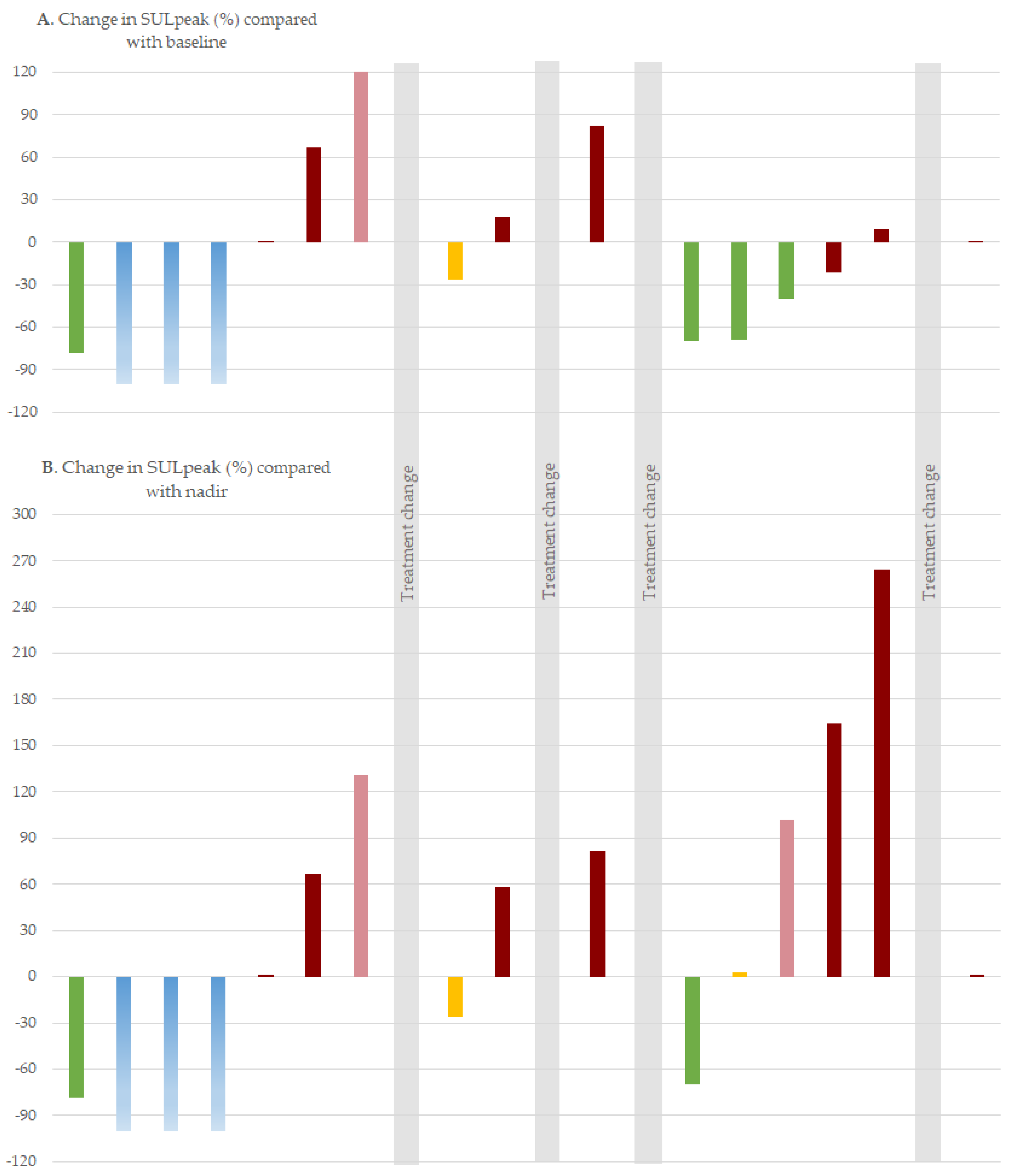
2.6. Definition of PERCISTnadir
2.7. Response Categorization by PERCIST
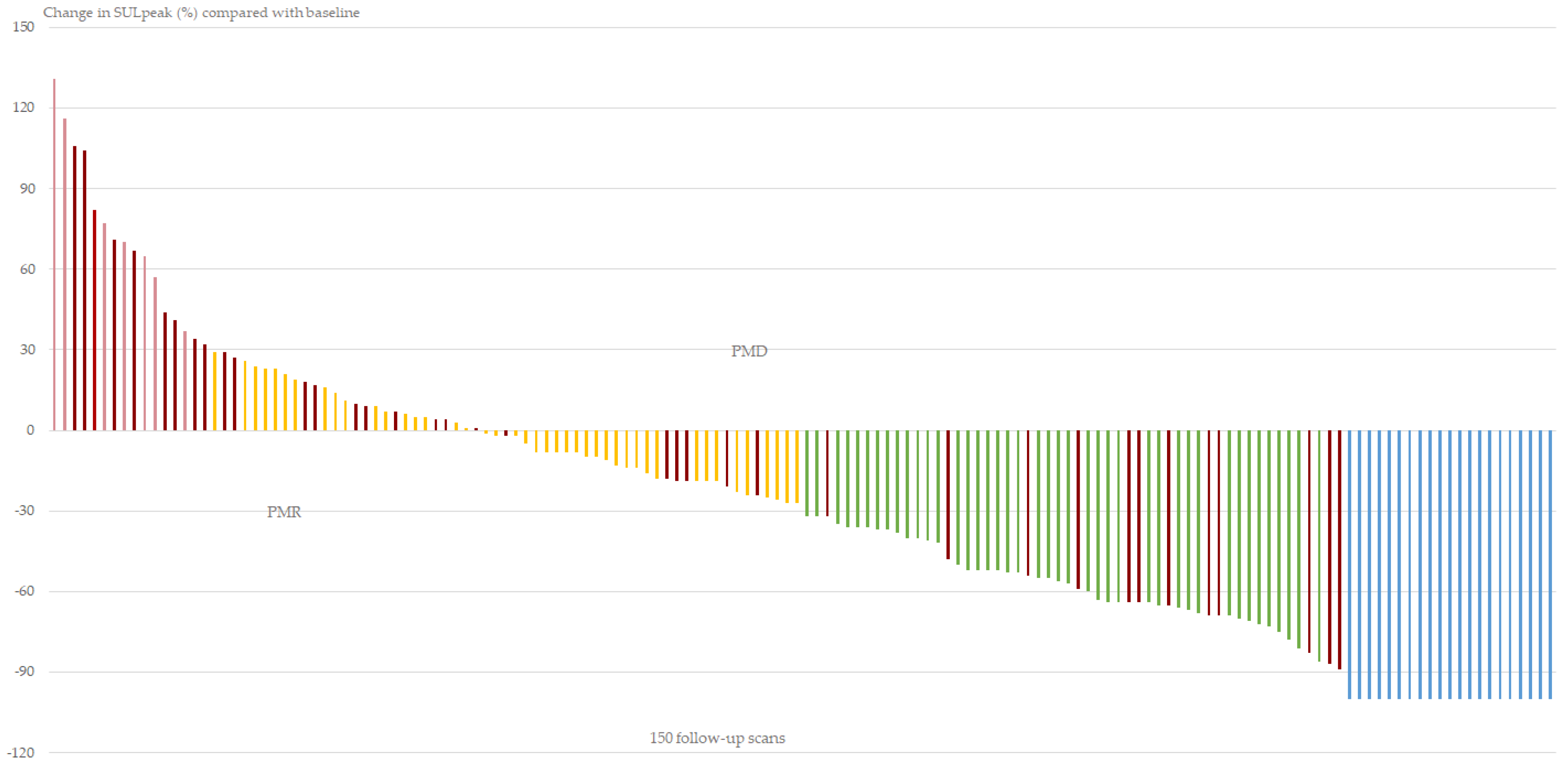
3. Results
3.1. Patients Characteristics
3.2. Applicability of PERCIST
3.3. Agreement between PERCIST and Visual Assessment for Response Categorization
3.4. Response Categories
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ferlay, J.; Steliarova-Foucher, E.; Lortet-Tieulent, J.; Rosso, S.; Coebergh, J.W.; Comber, H.; Forman, D.; Bray, F. Cancer incidence and mortality patterns in Europe: Estimates for 40 countries in 2012. Eur. J. Cancer 2013, 49, 1374–1403. [Google Scholar] [CrossRef] [PubMed]
- Harbeck, N.; Penault-Llorca, F.; Cortes, J.; Gnant, M.; Houssami, N.; Poortmans, P.; Ruddy, K.; Tsang, J.; Cardoso, F. Breast cancer. Nat. Rev. Dis. Prim. 2019, 5, 66. [Google Scholar] [CrossRef]
- Eisenhauer, E.A.; Therasse, P.; Bogaerts, J.; Schwartz, L.H.; Sargent, D.; Ford, R.; Dancey, J.; Arbuck, S.; Gwyther, S.; Mooney, M.; et al. New response evaluation criteria in solid tumours: Revised RECIST guideline (version 1.1). Eur. J. Cancer 2009, 45, 228–247. [Google Scholar] [CrossRef]
- Semiglazov, V. RECIST for response (clinical and imaging) in neoadjuvant clinical trials in operable breast cancer. J. Natl. Cancer Inst. Monogr. 2015, 2015, 21–23. [Google Scholar] [CrossRef]
- Schwartz, L.H.; Litiere, S.; de Vries, E.; Ford, R.; Gwyther, S.; Mandrekar, S.; Shankar, L.; Bogaerts, J.; Chen, A.; Dancey, J.; et al. RECIST 1.1-Update and clarification: From the RECIST committee. Eur. J. Cancer 2016, 62, 132–137. [Google Scholar] [CrossRef]
- Cardoso, F.; Paluch-Shimon, S.; Senkus, E.; Curigliano, G.; Aapro, M.S.; André, F.; Barrios, C.H.; Bergh, J.; Bhattacharyya, G.S.; Biganzoli, L.; et al. 5th ESO-ESMO international consensus guidelines for advanced breast cancer (ABC 5)(†). Ann. Oncol. Off. J. Eur. Soc. Med Oncol. 2020. [CrossRef] [PubMed]
- Yang, H.-L.; Liu, T.; Wang, X.-M.; Xu, Y.; Deng, S.-M. Diagnosis of bone metastases: A meta-analysis comparing 18 FDG PET, CT, MRI and bone scintigraphy. Eur. Radiol. 2011, 21, 2604–2617. [Google Scholar] [CrossRef] [PubMed]
- Li, J.-N.; Zhong, R.; Zhou, X.-H. Prediction of Bone Metastasis in Breast Cancer Based on Minimal Driver Gene Set in Gene Dependency Network. Genes 2019, 10, 466. [Google Scholar] [CrossRef] [PubMed]
- Hildebrandt, M.G.; Gerke, O.; Baun, C.; Falch, K.; Hansen, J.A.; Farahani, Z.A.; Petersen, H.; Larsen, L.B.; Duvnjak, S.; Buskevica, I.; et al. [18F] Fluorodeoxyglucose (FDG)-Positron Emission Tomography (PET)/Computed Tomography (CT) in Suspected Recurrent Breast Cancer: A Prospective Comparative Study of Dual-Time-Point FDG-PET/CT, Contrast-Enhanced CT, and Bone Scintigraphy. J. Clin. Oncol. 2016, 34, 1889–1897. [Google Scholar] [CrossRef]
- Ulaner, G.A. PET/CT for Patients with Breast Cancer: Where Is the Clinical Impact? AJR Am. J. Roentgenol. 2019, 213, 254–265. [Google Scholar] [CrossRef]
- O, J.H.; Lodge, M.A.; Wahl, R.L. Practical PERCIST: A Simplified Guide to PET Response Criteria in Solid Tumors 1.0. Radiology 2016, 280, 576–584. [Google Scholar] [CrossRef]
- Wahl, R.L.; Jacene, H.; Kasamon, Y.; Lodge, M.A. From RECIST to PERCIST: Evolving Considerations for PET response criteria in solid tumors. J. Nucl. Med. Off. Publ. Soc. Nucl. Med. 2009, 50 (Suppl. 1), 122s–150s. [Google Scholar] [CrossRef]
- Wahl, R.L. PERCIST in Perspective. Nucl. Med. Mol. Imaging 2018, 52, 1–4. [Google Scholar]
- Helland, F.; Hallin Henriksen, M.; Gerke, O.; Vogsen, M.; Hoilund-Carlsen, P.F.; Hildebrandt, M.G. FDG-PET/CT Versus Contrast-Enhanced CT for Response Evaluation in Metastatic Breast Cancer: A Systematic Review. Diagnostics 2019, 9, 106. [Google Scholar] [CrossRef]
- Riedl, C.C.; Pinker, K.; Ulaner, G.A.; Ong, L.T.; Baltzer, P.; Jochelson, M.S.; McArthur, H.L.; Gonen, M.; Dickler, M.; Weber, W.A. Comparison of FDG-PET/CT and contrast-enhanced CT for monitoring therapy response in patients with metastatic breast cancer. Eur. J. Nucl. Med. Mol. Imaging 2017, 44, 1428–1437. [Google Scholar] [CrossRef] [PubMed]
- Pinker, K.; Riedl, C.C.; Ong, L.; Jochelson, M.; Ulaner, G.A.; McArthur, H.; Dickler, M.; Gönen, M.; Weber, W.A. The impact that number of analyzed metastatic breast cancer lesions has on response assessment by 18F-FDG PET/CT using PERCIST. J. Nucl. Med. Off. Publ. Soc. Nucl. Med. 2016, 57, 1102. [Google Scholar] [CrossRef]
- Hildebrandt, M.G.; Lauridsen, J.F.; Vogsen, M.; Holm, J.; Vilstrup, M.H.; Braad, P.E.; Gerke, O.; Thomassen, M.; Ewertz, M.; Hoilund-Carlsen, P.F.; et al. FDG-PET/CT for Response Monitoring in Metastatic Breast Cancer: Today, Tomorrow, and Beyond. Cancers 2019, 11, 1190. [Google Scholar] [CrossRef] [PubMed]
- Groheux, D. Role of Fludeoxyglucose in Breast Cancer: Treatment Response. PET Clin. 2018, 13, 395–414. [Google Scholar] [CrossRef]
- Boellaard, R.; Delgado-Bolton, R.; Oyen, W.J.; Giammarile, F.; Tatsch, K.; Eschner, W.; Verzijlbergen, F.J.; Barrington, S.F.; Pike, L.C.; Weber, W.A.; et al. FDG PET/CT: EANM procedure guidelines for tumour imaging: Version 2.0. Eur. J. Nucl. Med. Mol. Imaging 2015, 42, 328–354. [Google Scholar] [CrossRef]
- Sørensen, J.S.; Vilstrup, M.H.; Holm, J.; Vogsen, M.; Bülow, J.L.; Ljungstrøm, L.; Braad, P.E.; Gerke, O.; Hildebrandt, M.G. Interrater Agreement and Reliability of PERCIST and Visual Assessment When Using 18F-FDG-PET/CT for Response Monitoring of Metastatic Breast Cancer. Diagnostics 2020, 10, 1001. [Google Scholar] [CrossRef]
- Depardon, E.; Kanoun, S.; Humbert, O.; Bertaut, A.; Riedinger, J.-M.; Tal, I.; Vrigneaud, J.-M.; Lasserre, M.; Toubeau, M.; Berriolo-Riedinger, A. FDG PET/CT for prognostic stratification of patients with metastatic breast cancer treated with first line systemic therapy: Comparison of EORTC criteria and PERCIST. PLoS ONE 2018, 13, e0199529. [Google Scholar] [CrossRef] [PubMed]
- Willemsen, A.E.; Vlenterie, M.; van Herpen, C.M.; van Erp, N.P.; van der Graaf, W.T.; de Geus-Oei, L.F.; Oyen, W.J. Positron emission tomography response criteria in solid tumours criteria for quantitative analysis of [18F]-fluorodeoxyglucose positron emission tomography with integrated computed tomography for treatment response assessment in metastasised solid tumours: All that glitters is not gold. Eur. J. Cancer 2016, 56, 54–58. [Google Scholar] [CrossRef] [PubMed]
| n (%) | ||
|---|---|---|
| Type of Surgery | Breast conserving | 11 (29.7) |
| Mastectomy | 21 (56.8) | |
| No surgery | 4 (10.8) | |
| Other | 1 (2.70) | |
| Histology | Invasive ductal carcinoma | 26 (70.3) |
| Invasive lobular carcinoma | 3 (8.11) | |
| Invasive carcinoma unspecified | 5 (13.5) | |
| Unknown | 3 (8.11) | |
| Size | ≤10 mm | 2 (5.41) |
| 11–20 mm | 11 (29.7) | |
| 21–50 mm | 11 (29.7) | |
| ≥50 mm | 6 (16.2) | |
| ≤10 mm | 2 (5.41) | |
| Lymph node involvement | 0 or micro-metastases | 9 (24.3) |
| 1–3 | 10 (27.0) | |
| 4–9 | 7 (18.9) | |
| ≥10 | 3 (8.11) | |
| Unknown or no surgery | 8 (21.6) | |
| Grade | I | 5 (13.5) |
| II | 11 (29.7) | |
| III | 9 (24.3) | |
| Unknown | 12 (32.4) | |
| ER-status | Positive | 29 (78.4) |
| Negative | 4 (10.8) | |
| Unknown | 4 (10.8) | |
| HER2-status | Positive | 9 (24.3) |
| Normal | 18 (48.6) | |
| Unknown | 10 (27.0) | |
| Medical treatment | Neo-adjuvant +/− adjuvant | 9 (24.3) |
| Adjuvant only | 22 (59.5) | |
| No medical treatment | 6 (16.2) | |
| 37 (100) |
| n (%) | ||
|---|---|---|
| ER-status | Positive | 30 (81.1) |
| Negative | 6 (16.2) | |
| Unknown | 1 (2.70) | |
| HER2-status | Positive | 9 (24.3) |
| Normal | 23 (62.2) | |
| Unknown | 5 (13.5) | |
| Location of biopsy * | Bone | 11 (29.7) |
| Lymph node | 9 (24.3) | |
| Liver | 6 (16.2) | |
| Other | 4 (10.8) | |
| Breast | 2 (5.40) | |
| Lung | 2 (5.40) | |
| Skin | 2 (5.40) | |
| Brain | 1 (2.70) | |
| 37 (100) |
| Visual Assessment | PERCIST | |||||
|---|---|---|---|---|---|---|
| CMR | PMR | SMD | PMD | Sum | ||
| CMR | 15 | 0 | 0 | 0 | 15 | |
| PMR | 5 | 37 | 22 | 11 | 75 | |
| SMD | 0 | 4 | 13 | 2 | 19 | |
| PMD | 0 | 0 | 7 | 28 | 35 | |
| Mixed Response | 0 | 0 | 1 | 1 | 2 | |
| Equivocal Answer | 1 | 1 | 0 | 2 | 4 | |
| Sum | 21 | 42 | 43 | 44 | 150 | |
| Agreement | Expected agreement | Kappa | Std. Error | Z | Prob > Z | |
| 62% | 25.9% | 0.49 | 0.04 | 11.0 | <0.0001 | |
| Overall agreement: 0.62 [95% CI 0.54–0.70] | Standard Error: 0.04 | |||||
| PERCISTnadir | ||||||
|---|---|---|---|---|---|---|
| CMR | PMR | SMD | PMD | Sum | ||
| PERCISTbaseline | CMR | 21 | 0 | 0 | 0 | 21 |
| PMR | 0 | 29 | 10 | 3 | 42 | |
| SMD | 0 | 2 | 34 | 7 | 43 | |
| PMD | 0 | 0 | 0 | 44 | 44 | |
| Sum | 21 | 31 | 44 | 54 | 150 | |
| Agreement | Expected agreement | Kappa | Std. Error | Z | Prob > Z | |
| 85.3% | 26.7% | 0.79 | 0.048 | 16.6 | <0.0001 | |
| Overall agreement 0.85 [95% CI 0.79–0.91] | Standard Error: 0.03 | |||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vogsen, M.; Bülow, J.L.; Ljungstrøm, L.; Oltmann, H.R.; Alamdari, T.A.; Naghavi-Behzad, M.; Braad, P.-E.; Gerke, O.; Hildebrandt, M.G. FDG-PET/CT for Response Monitoring in Metastatic Breast Cancer: The Feasibility and Benefits of Applying PERCIST. Diagnostics 2021, 11, 723. https://doi.org/10.3390/diagnostics11040723
Vogsen M, Bülow JL, Ljungstrøm L, Oltmann HR, Alamdari TA, Naghavi-Behzad M, Braad P-E, Gerke O, Hildebrandt MG. FDG-PET/CT for Response Monitoring in Metastatic Breast Cancer: The Feasibility and Benefits of Applying PERCIST. Diagnostics. 2021; 11(4):723. https://doi.org/10.3390/diagnostics11040723
Chicago/Turabian StyleVogsen, Marianne, Jakob Lykke Bülow, Lasse Ljungstrøm, Hjalte Rasmus Oltmann, Tural Asgharzadeh Alamdari, Mohammad Naghavi-Behzad, Poul-Erik Braad, Oke Gerke, and Malene Grubbe Hildebrandt. 2021. "FDG-PET/CT for Response Monitoring in Metastatic Breast Cancer: The Feasibility and Benefits of Applying PERCIST" Diagnostics 11, no. 4: 723. https://doi.org/10.3390/diagnostics11040723
APA StyleVogsen, M., Bülow, J. L., Ljungstrøm, L., Oltmann, H. R., Alamdari, T. A., Naghavi-Behzad, M., Braad, P.-E., Gerke, O., & Hildebrandt, M. G. (2021). FDG-PET/CT for Response Monitoring in Metastatic Breast Cancer: The Feasibility and Benefits of Applying PERCIST. Diagnostics, 11(4), 723. https://doi.org/10.3390/diagnostics11040723








