Prognostic Value of C-Reactive Protein-to-Albumin Ratio in Head and Neck Cancer: A Meta-Analysis
Abstract
1. Introduction
2. Materials and Methods
2.1. Data Sources and Search Strategy
2.2. Inclusion and Exclusion Criteria
2.3. Data Extraction and Quality Assessment
2.4. Quality Assessment
2.5. Statistical Analysis
3. Results
3.1. Study Characteristics
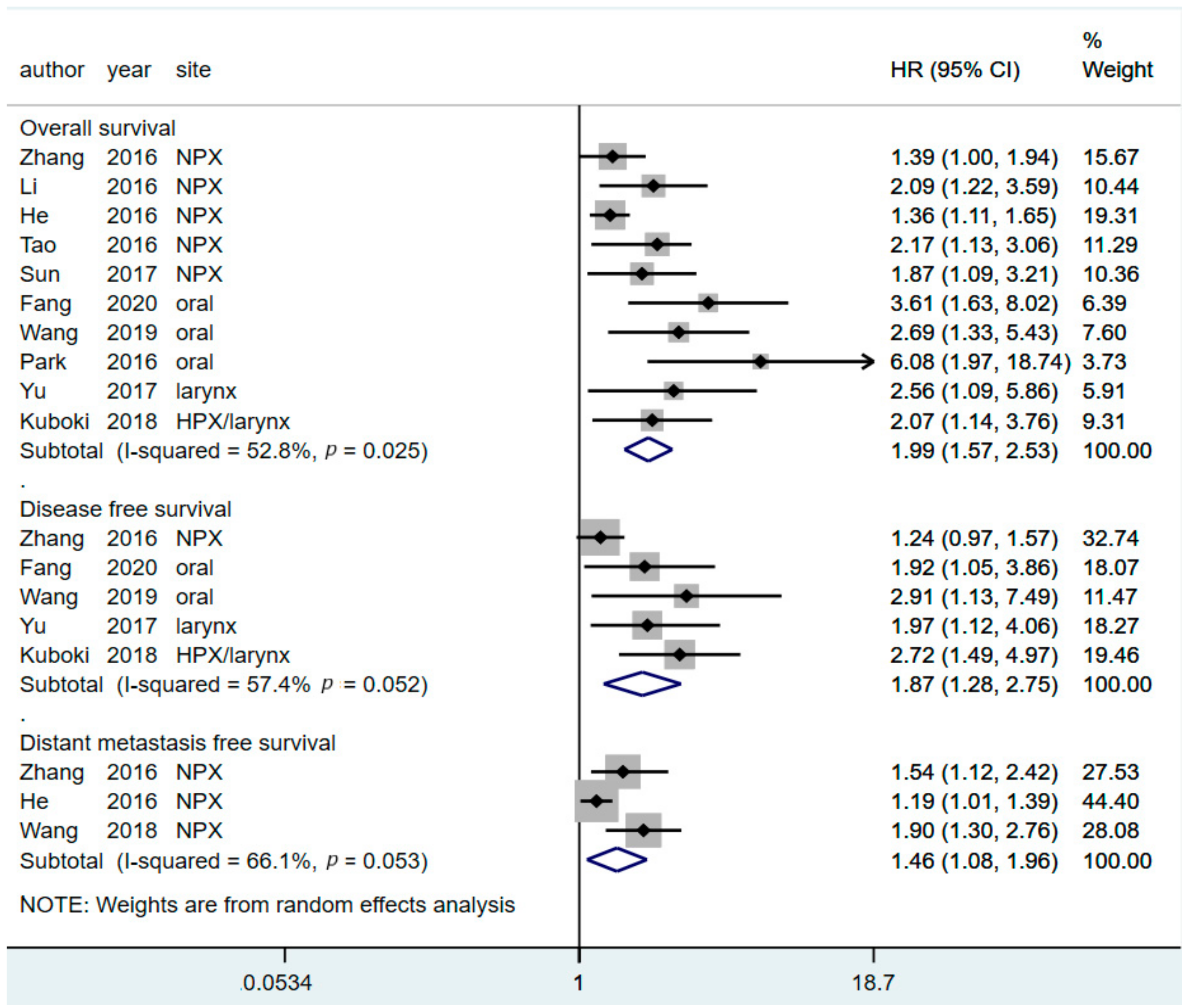
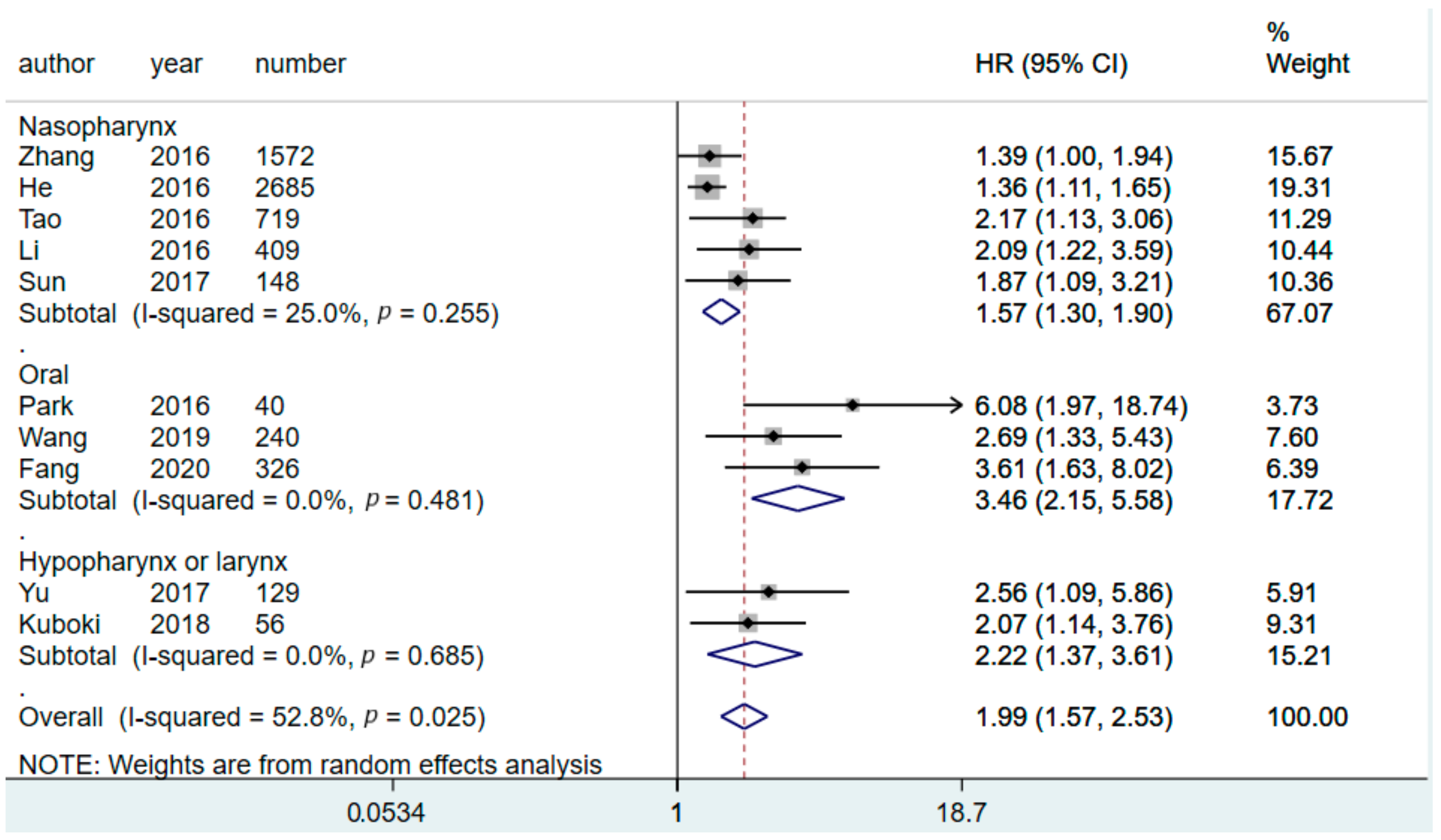
3.2. Prognostic Value of CAR for Predicting OS
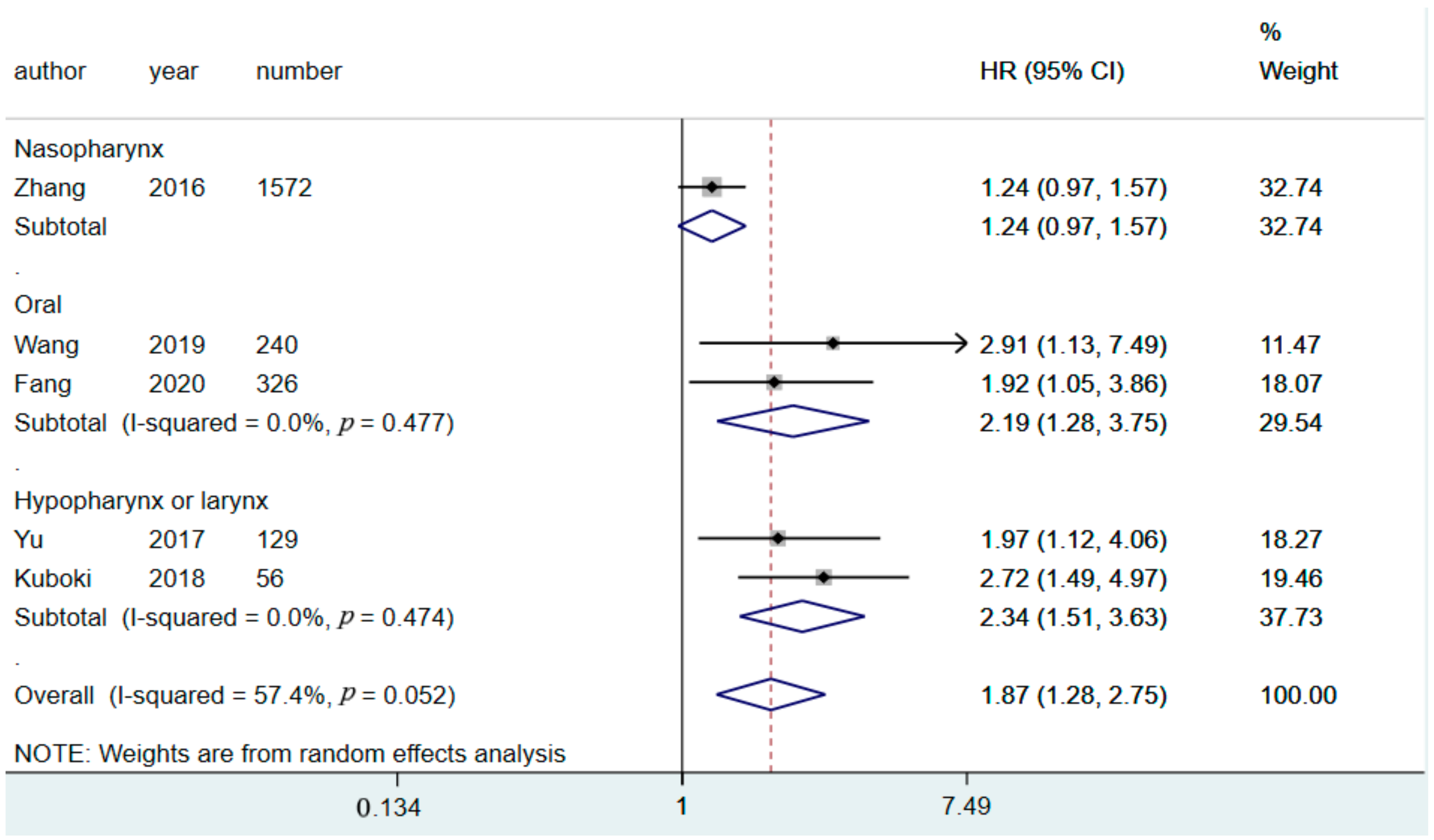
3.3. Prognostic Value of CAR for Predicting DFS
3.4. Prognostic Value of CAR in Predicting DMFS
3.5. Metaregression
3.6. Subgroup Analysis
3.7. Publication Bias
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Bray, F.; Ferlay, J.; Soerjomataram, I.; Siegel, R.L.; Torre, L.A.; Jemal, A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2018, 68, 394–424. [Google Scholar] [CrossRef]
- O-charoenrat, P.; Pillai, G.; Patel, S.; Fisher, C.; Archer, D.; Eccles, S.; Rhys-Evans, P. Tumour thickness predicts cervical nodal metastases and survival in early oral tongue cancer. Oral Oncol. 2003, 39, 386–390. [Google Scholar] [CrossRef]
- Colevas, A.D.; Yom, S.S.; Pfister, D.G.; Spencer, S.; Adelstein, D.; Adkins, D.; Brizel, D.M.; Burtness, B.; Busse, P.M.; Caudell, J.J.; et al. NCCN Guidelines Insights: Head and Neck Cancers, Version 1.2018. J. Natl. Compr. Cancer Netw. JNCCN 2018, 16, 479–490. [Google Scholar] [CrossRef] [PubMed]
- Pfister, D.G.; Spencer, S.; Brizel, D.M.; Adelstein, D.; Adkins, D.; Anzai, Y.; Bruce, J.Y.; Busse, P.M.; Caudell, J.J.; Cmelak, A.J.; et al. Head and neck cancers, Version 2.2014. Clinical practice guidelines in oncology. J. Natl. Compr. Cancer Netw. JNCCN 2014, 12, 1454–1487. [Google Scholar] [CrossRef] [PubMed]
- Diakos, C.I.; Charles, K.A.; McMillan, D.C.; Clarke, S.J. Cancer-related inflammation and treatment effectiveness. Lancet Oncol. 2014, 15, e493–e503. [Google Scholar] [CrossRef]
- Grivennikov, S.I.; Greten, F.R.; Karin, M. Immunity, inflammation, and cancer. Cell 2010, 140, 883–899. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.J.; Ma, Y.; Deng, F.; Ju, W.B.; Sun, X.Y.; Wang, H. The prognostic value of C-reactive protein/albumin ratio in human malignancies: An updated meta-analysis. OncoTargets Ther. 2017, 10, 3059–3070. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Yang, X.; Feng, J.F. A novel inflammation-based prognostic score for patients with esophageal squamous cell carcinoma: The c-reactive protein/prognostic nutritional index ratio. Oncotarget 2016, 7, 62123–62132. [Google Scholar] [CrossRef]
- Chen, Z.; Shao, Y.; Fan, M.; Zhuang, Q.; Wang, K.; Cao, W.; Xu, X.; He, X. Prognostic significance of preoperative C-reactive protein: Albumin ratio in patients with clear cell renal cell carcinoma. Int. J. Clin. Exp. Pathol. 2015, 8, 14893–14900. [Google Scholar]
- Haruki, K.; Shiba, H.; Shirai, Y.; Horiuchi, T.; Iwase, R.; Fujiwara, Y.; Furukawa, K.; Misawa, T.; Yanaga, K. The C-reactive Protein to Albumin Ratio Predicts Long-Term Outcomes in Patients with Pancreatic Cancer After Pancreatic Resection. World J. Surg. 2016, 40, 2254–2260. [Google Scholar] [CrossRef]
- Kinoshita, A.; Onoda, H.; Imai, N.; Iwaku, A.; Oishi, M.; Tanaka, K.; Fushiya, N.; Koike, K.; Nishino, H.; Matsushima, M. The C-reactive protein/albumin ratio, a novel inflammation-based prognostic score, predicts outcomes in patients with hepatocellular carcinoma. Ann. Surg. Oncol. 2015, 22, 803–810. [Google Scholar] [CrossRef]
- Ni, X.F.; Wu, J.; Ji, M.; Shao, Y.-J.; Xu, B.; Jiang, J.-T.; Wu, C.-P. Effect of C-reactive protein/albumin ratio on prognosis in advanced non-small-cell lung cancer. Asia-Pac. J. Clin. Oncol. 2018, 14, 402–409. [Google Scholar] [CrossRef] [PubMed]
- Shibutani, M.; Maeda, K.; Nagahara, H.; Iseki, Y.; Ikeya, T.; Hirakawa, K. Prognostic Significance of the Preoperative Ratio of C-Reactive Protein to Albumin in Patients with Colorectal Cancer. Anticancer. Res. 2016, 36, 995–1001. [Google Scholar]
- Gao, N.; Yang, R.N.; Meng, Z.; Wang, W.H. The prognostic value of C-reactive protein/albumin ratio in nasopharyngeal carcinoma: A meta-analysis. Biosci. Rep. 2018, 38. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Liu, H.; He, M.; Liu, M.; Zhou, G.; Gong, P.; Ma, J.; Wang, Q.; Xiong, W.; Ren, Z.; et al. Prognostic value of pretreatment C-reactive protein/albumin ratio in nasopharyngeal carcinoma: A meta-analysis of published literature. Medicine 2018, 97, e11574. [Google Scholar] [CrossRef]
- McInnes, M.D.F.; Moher, D.; Thombs, B.D.; McGrath, T.A.; Bossuyt, P.M.; Clifford, T.; Cohen, J.F.; Deeks, J.J.; Gatsonis, C.; Hooft, L.; et al. Preferred Reporting Items for a Systematic Review and Meta-analysis of Diagnostic Test Accuracy Studies: The PRISMA-DTA Statement. JAMA 2018, 319, 388–396. [Google Scholar] [CrossRef] [PubMed]
- Wells, G.A.; Tugwell, P.; O’Connell, D.; Peterson, J.; Welch, V.; Losos, M.; Tugwell, P. The Newcastle-Ottawa Scale (NOS) for assessing the quality of nonrandomized studies in meta-analyses. 2015. Available online: http://www.ohri.ca/programs/clinical_epidemiology/oxford.asp (accessed on 26 February 2020).
- Wang, Y.; Yang, L.; Xia, L.; Chen, Y. High C-reactive protein/albumin ratio predicts unfavorable distant metastasis-free survival in nasopharyngeal carcinoma: A propensity score-matched analysis. Cancer Manag. Res. 2018, 10, 371–381. [Google Scholar] [CrossRef] [PubMed]
- Park, H.C.; Kim, M.Y.; Kim, C.H. C-reactive protein/albumin ratio as prognostic score in oral squamous cell carcinoma. J. Korean Assoc. Oral Maxillofac. Surg. 2016, 42, 243–250. [Google Scholar] [CrossRef]
- He, S.; Wang, Y.; Chen, H.; Yang, L.; Liang, S.; Lu, L.; Chen, Y. C-Reactive Protein/Albumin Ratio (CAR) as a Prognostic Factor in Patients with Non-Metastatic Nasopharyngeal Carcinoma. J. Cancer 2016, 7, 2360–2366. [Google Scholar] [CrossRef][Green Version]
- Kuboki, A.; Kanaya, H.; Nakayama, T.; Konno, W.; Goto, K.; Nakajima, I.; Kashiwagi, T.; Hirabayashi, H.; Haruna, S.I. Prognostic value of C-reactive protein/albumin ratio for patients with hypopharyngeal and laryngeal cancer undergoing invasive surgery involving laryngectomy. Head Neck 2019, 41, 1342–1350. [Google Scholar] [CrossRef] [PubMed]
- Li, J.P.; Chen, S.L.; Liu, X.M.; He, X.; Xing, S.; Liu, Y.-J.; Lin, Y.-H.; Liu, W.-L. A Novel Inflammation-Based Stage (I Stage) Predicts Overall Survival of Patients with Nasopharyngeal Carcinoma. Int. J. Mol. Sci. 2016, 17, 1900. [Google Scholar] [CrossRef]
- Sun, P.; Chen, C.; Xia, Y.; Bi, X.; Liu, P.; Zhang, F.; Yang, H.; An, X.; Jiang, W.; Wang, F. The Ratio of C-Reactive Protein/Albumin is a Novel Inflammatory Predictor of Overall Survival in Cisplatin-Based Treated Patients with Metastatic Nasopharyngeal Carcinoma. Dis. Markers 2017, 2017, 6570808. [Google Scholar] [CrossRef]
- Tao, C.J.; Chen, Y.Y.; Jiang, F.; Feng, X.-L.; Jin, Q.-F.; Jin, T.; Piao, Y.-F.; Chen, X.-Z. The C-reactive Protein/Albumin Ratio Is an independent Prognostic Factor for Overall Survival in Patients with Nasopharyngeal Carcinoma Receiving Intensity-Modulated Radiotherapy. J. Cancer 2016, 7, 2005–2011. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Wang, Q.; Song, X.; Zhao, Y.; He, Q.; Shi, M.; Xu, P.; Ni, S.; Chen, Y.; Lin, J.; Zhang, L. Preoperative high c-reactive protein/albumin ratio is a poor prognostic factor of oral squamous cell carcinoma. Future Oncol. 2019, 15, 2277–2286. [Google Scholar] [CrossRef] [PubMed]
- Yu, S.T.; Zhou, Z.; Cai, Q.; Liang, F.; Han, P.; Chen, R.; Huang, X.-M. Prognostic value of the C-reactive protein/albumin ratio in patients with laryngeal squamous cell carcinoma. OncoTargets Ther. 2017, 10, 879–884. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhou, G.Q.; Liu, X.; Chen, L.; Li, W.-F.; Tang, L.-L.; Liu, Q.; Sun, Y.; Ma, J. Exploration and Validation of C-Reactive Protein/Albumin Ratio as a Novel Inflammation-Based Prognostic Marker in Nasopharyngeal Carcinoma. J. Cancer 2016, 7, 1406–1412. [Google Scholar] [CrossRef]
- Fang, K.H.; Lai, C.H.; Hsu, C.M.; Huang, E.; Tsai, M.-S.; Chang, G.-H.; Lee, Y.-C.; Tsai, Y.-T. A retrospective study on the prognostic value of preoperative C-reactive protein to albumin ratio in patients with oral cavity squamous cell carcinoma. PeerJ 2020, 8, e9361. [Google Scholar] [CrossRef]
- Fernandes, J.V.; Cobucci, R.N.; Jatoba, C.A.; Fernandes, T.A.; de Azevedo, J.W.; de Araujo, J.M. The role of the mediators of inflammation in cancer development. Pathol. Oncol. Res. POR 2015, 21, 527–534. [Google Scholar] [CrossRef] [PubMed]
- Hanahan, D.; Weinberg, R.A. Hallmarks of cancer: The next generation. Cell 2011, 144, 646–674. [Google Scholar] [CrossRef]
- Singh, R.; Mishra, M.K.; Aggarwal, H. Inflammation, Immunity, and Cancer. Mediat. Inflamm. 2017, 2017, 6027305. [Google Scholar] [CrossRef] [PubMed]
- Pepys, M.B.; Hirschfield, G.M. C-reactive protein: A critical update. J. Clin. Investig. 2003, 111, 1805–1812. [Google Scholar] [CrossRef] [PubMed]
- Danan, D.; Shonka, D.C., Jr.; Selman, Y.; Chow, Z.; Smolkin, M.E.; Jameson, M.J. Prognostic value of albumin in patients with head and neck cancer. Laryngoscope 2016, 126, 1567–1571. [Google Scholar] [CrossRef]
- Bano, G.; Trevisan, C.; Carraro, S.; Solmi, M.; Luchini, C.; Stubbs, B.; Manzato, E.; Sergi, G.; Veronese, N. Inflammation and sarcopenia: A systematic review and meta-analysis. Maturitas 2017, 96, 10–15. [Google Scholar] [CrossRef] [PubMed]
- Ishizuka, M.; Nagata, H.; Takagi, K.; Iwasaki, Y.; Shibuya, N.; Kubota, K. Clinical Significance of the C-Reactive Protein to Albumin Ratio for Survival After Surgery for Colorectal Cancer. Ann. Surg. Oncol. 2016, 23, 900–907. [Google Scholar] [CrossRef] [PubMed]
- Bussu, F.; Miccichè, F.; Rigante, M.; DiNapoli, N.; Parrilla, C.; Bonomo, P.; Cadoni, G.; Mantini, G.; Galli, J.; Rufini, V.; et al. Oncologic outcomes in advanced laryngeal squamous cell carcinomas treated with different modalities in a single institution: A retrospective analysis of 65 cases. Head Neck 2012, 34, 573–579. [Google Scholar] [CrossRef] [PubMed]
- Lala, M.; Chirovsky, D.; Cheng, J.D.; Mayawala, K. Clinical outcomes with therapies for previously treated recurrent/metastatic head-and-neck squamous cell carcinoma (R/M HNSCC): A systematic literature review. Oral Oncol. 2018, 84, 108–120. [Google Scholar] [CrossRef] [PubMed]

| First Author | Year | Country | Sample Size | Tumor Site | Cancer Stage | Curative/ Palliative | Treatment | Cut-Off Value Resource | CAR Cut-Off (mg/g) | Outcome | Median Follow-Up (Months) | NOS |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Li et al. [22] | 2016 | China | 409 | NPX | I–IV | Curative | R/C+R | Unknown | 0.03 | OS | 53.7 | 7 |
| He et al. [20] | 2016 | China | 2685 | NPX | I–III | Curative | R/C+R | ROC curve | 0.064 | OS, DMFS | 46.3 | 7 |
| Zhang et al. [27] | 2016 | China | 1572 | NPX | I–IV | Curative | R/C+R | ROC curve | 0.05 | OS | 50 | 8 |
| Wang et al. [18] | 2018 | China | 756 | NPX | I–IV | Curative | R/C+R | Cutoff Finder | 0.081 | DMFS | 68.8 | 8 |
| Tao et al. [24] | 2016 | China | 719 | NPX | I–IV | Curative | R/C+R | ROC curve | 0.141 | OS | 47 | 7 |
| Sun et al. [23] | 2017 | China | 148 | NPX | IVb | Palliative | C | Cutoff Finder | 0.189 | OS | 21.8 | 8 |
| Kuboki et al. [21] | 2018 | Japan | 56 | HPX/Larynx | I–IV | Curative | OP | ROC curve | 0.32 | OS, DFS | 38 | 8 |
| Yu et al. [26] | 2017 | China | 129 | Larynx | I–IV | Curative | OP/OP+C+R | ROC curve | 0.047 | OS | 77 | 7 |
| Wang et al. [25] | 2019 | China | 240 | Oral | I–IV | Curative | OP/OP+C+R | Cutoff Finder | 0.525 | OS, DFS | 72.39 | 8 |
| Park et al. [19] | 2016 | Korea | 40 | Oral | I–IV | Curative | OP/OP+C+R | ROC curve | 0.085 | OS, DFS | 35.38 | 7 |
| Fang et al. [28] | 2020 | Taiwan | 326 | Oral | I-IV | Curative | OP/OP+C+R | ROC curve | 0.195 | OS, DFS | 48 | 7 |
| Subgroup | Number of Studies | Number of Patients | Pooled HR with 95% CI | Subgroup p Value | Heterogeneity | |
|---|---|---|---|---|---|---|
| I2 % | p Value | |||||
| Overall Survival | ||||||
| Tumor location | ||||||
| Nasopharynx | 5 | 5533 | 1.57 (1.30–1.90) | 0.03 | 25 | 0.26 |
| Hypopharynx/larynx | 2 | 185 | 2.22 (1.37–3.61) | 0 | 0.68 | |
| Oral cavity | 3 | 606 | 3.46 (2.15–5.58) | 0 | 0.48 | |
| Sample size | ||||||
| ≤500 | 7 | 5302 | 2.39 (1.86–3.09) | 0.001 | 0 | 0.54 |
| >500 | 3 | 1022 | 1.44 (1.22–1.69) | 33 | 0.23 | |
| Country | ||||||
| China | 7 | 5902 | 1.57 (1.36–1.81) | 0.01 | 35 | 0.16 |
| Others | 3 | 422 | 2.89 (1.86–4.48) | 37 | 0.20 | |
| Cut-off value for CAR | ||||||
| ≤0.1 | 5 | 4706 | 1.77 (1.27–2.47) | 0.26 | 60 | 0.04 |
| >0.1 | 5 | 1618 | 2.27 (1.73–2.97) | 0 | 0.71 | |
| Follow up period | ||||||
| ≥50 months | 4 | 2350 | 1.88 (1.34–2.64) | 0.62 | 33 | 0.21 |
| <50 months | 6 | 3974 | 2.14 (1.48–3.09) | 65 | 0.01 | |
| Disease Free Survival | ||||||
| Tumor location | ||||||
| Nasopharynx | 1 | 1572 | 1.24 (0.97–1.57) | 0.02 | 0 | NA |
| Hypopharynx/larynx | 2 | 185 | 2.34 (1.51–3.63) | 0 | 0.47 | |
| Oral cavity | 2 | 566 | 2.19 (1.28–3.75) | 0 | 0.48 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Luan, C.-W.; Yang, H.-Y.; Tsai, Y.-T.; Hsieh, M.-C.; Chou, H.-H.; Chen, K.-S. Prognostic Value of C-Reactive Protein-to-Albumin Ratio in Head and Neck Cancer: A Meta-Analysis. Diagnostics 2021, 11, 403. https://doi.org/10.3390/diagnostics11030403
Luan C-W, Yang H-Y, Tsai Y-T, Hsieh M-C, Chou H-H, Chen K-S. Prognostic Value of C-Reactive Protein-to-Albumin Ratio in Head and Neck Cancer: A Meta-Analysis. Diagnostics. 2021; 11(3):403. https://doi.org/10.3390/diagnostics11030403
Chicago/Turabian StyleLuan, Chih-Wei, Hsin-Yi Yang, Yao-Te Tsai, Meng-Chiao Hsieh, Hsin-Hsu Chou, and Kuo-Su Chen. 2021. "Prognostic Value of C-Reactive Protein-to-Albumin Ratio in Head and Neck Cancer: A Meta-Analysis" Diagnostics 11, no. 3: 403. https://doi.org/10.3390/diagnostics11030403
APA StyleLuan, C.-W., Yang, H.-Y., Tsai, Y.-T., Hsieh, M.-C., Chou, H.-H., & Chen, K.-S. (2021). Prognostic Value of C-Reactive Protein-to-Albumin Ratio in Head and Neck Cancer: A Meta-Analysis. Diagnostics, 11(3), 403. https://doi.org/10.3390/diagnostics11030403






