Methods for Testing the Subjective Visual Vertical during the Chronic Phase of Menière’s Disease
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design
- Two or more episodes of vertigo or dizziness, each lasting for 20 min to 24 h;
- Fluctuating aural symptoms (hearing loss, tinnitus, or fullness) in the reported ear;
- Not better accounted for by another vestibular diagnosis;
- Patients were not examined during an acute phase of MD. Other exclusion criteria in both groups were pregnancy, lactation, and diseases of the cervical spine.
2.2. SVV Bucket Test
2.3. C-SVV® Goggles
2.4. Statistical Analysis
3. Results
3.1. Participants and Eligibility Criteria
3.2. Comparison of Patients with MD and of the Control Group Concerning the SVV Measurement Using the Bucket
3.3. Comparison of MD Patients and the Control Group Regarding SVV Measurement Using the C-SVV® Goggles
3.4. Comparison of Sensitivity and Specificity of Bucket Test and C-SVV®
3.5. Between-Group Comparison of Bucket Test Results and C-SVV® Goggles Results
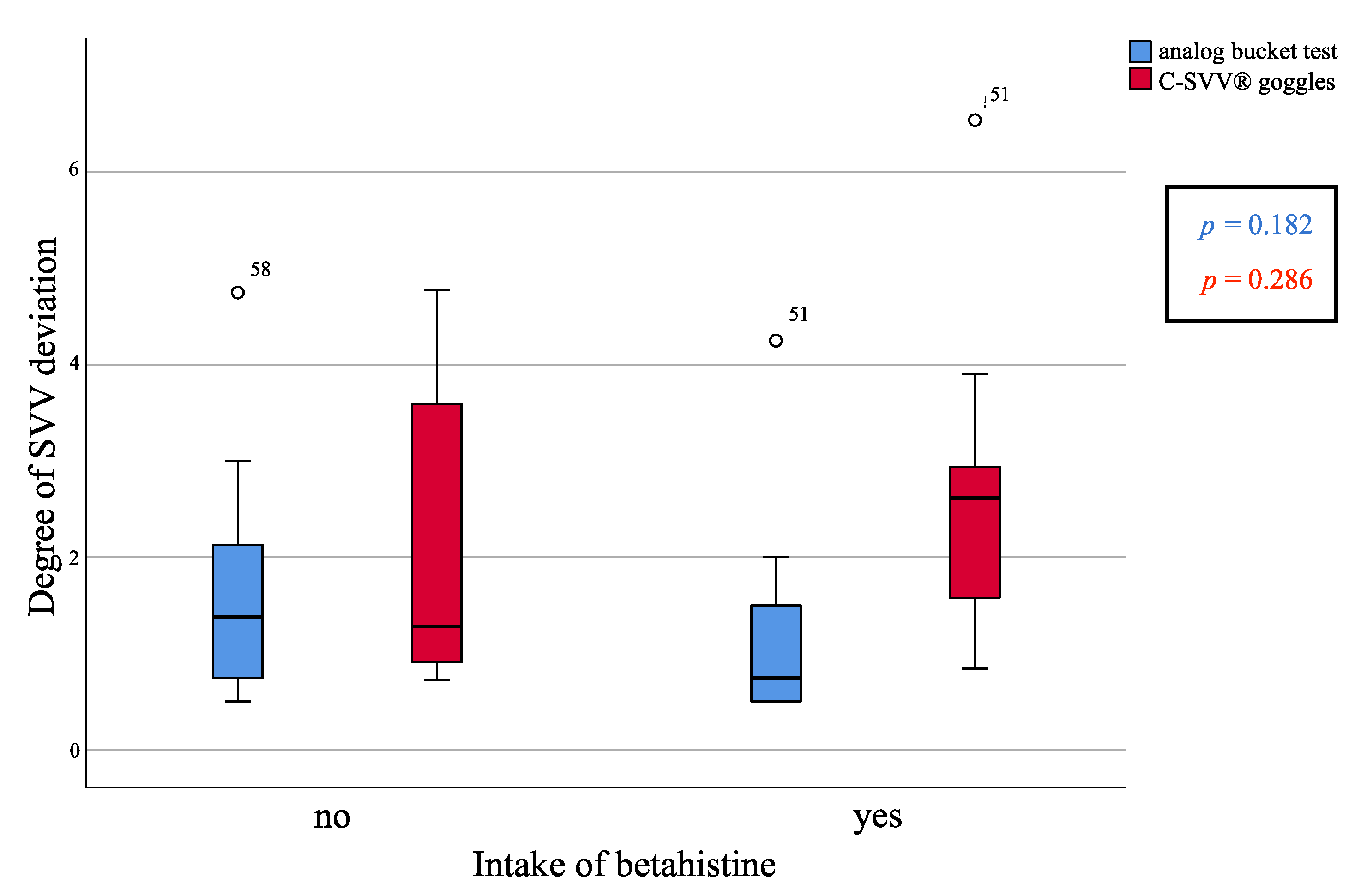
3.6. SVV Deviation and Betahistine Intake by MD Patients
4. Discussion
Study Limitations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Chetana, N.; Jayesh, R. Subjective Visual Vertical in Various Vestibular Disorders by Using a Simple Bucket Test. Indian J Otolaryngol. Head Neck Surg. 2015, 67, 180–184. [Google Scholar] [CrossRef]
- Clarke, A.H.; Schonfeld, U.; Hamann, C.; Scherer, H. Measuring unilateral otolith function via the otolith-ocular response and the subjective visual vertical. Acta Otolaryngol. Suppl. 2001, 545, 84–87. [Google Scholar] [PubMed]
- Van Nechel, C.; Toupet, M.; Bodson, I. The Subjective Visual Vertical. Cochlear Implant. Relat. Sci. Update 2000, 58, 77–87. [Google Scholar] [CrossRef]
- Glasauer, S.; Dieterich, M.; Brandt, T. Neuronal network-based mathematical modeling of perceived verticality in acute unilateral vestibular lesions: From nerve to thalamus and cortex. J. Neurol. 2018, 265, 101–112. [Google Scholar] [CrossRef] [PubMed]
- Dieterich, M.; Brandt, T. Perception of Verticality and Vestibular Disorders of Balance and Falls. Front. Neurol. 2019, 10, 172. [Google Scholar] [CrossRef] [PubMed]
- Wagner, J.H.; Basta, D.; Ernst, A. Diagnosis of otolith disorders in routine otolaryngology. HNO 2013, 61, 738–742. [Google Scholar] [CrossRef] [PubMed]
- Clarke, A.H.; Schönfeld, U.; Helling, K. Unilateral examination of utricle and saccule function. J. Vestib. Res. 2003, 13, 215–225. [Google Scholar]
- Dieterich, M.; Brandt, T. Ocular torsion and tilt of subjective visual vertical are sensitive brainstem signs. Ann. Neurol. 2004, 33, 292–299. [Google Scholar] [CrossRef] [PubMed]
- Zwergal, A.; Rettinger, N.; Frenzel, C.; Dieterich, M.; Brandt, T.; Strupp, M. A bucket of static vestibular function. Neurology 2009, 72, 1689–1692. [Google Scholar] [CrossRef] [PubMed]
- Bjasch, D.; Bockisch, C.J.; Straumann, D.; Tarnutzer, A.A. Differential Effects of Visual Feedback on Subjective Visual Vertical Accuracy and Precision. PLoS ONE 2012, 7, e49311. [Google Scholar] [CrossRef]
- Klatt, B.N.; Sparto, P.J.; Terhorst, L.; Winser, S.J.; Heyman, R.; Whitney, S.L. Relationship between subjective visual vertical and balance in individuals with multiple sclerosis. Physiother. Res. Int. 2019, 24, e1757. [Google Scholar] [CrossRef] [PubMed]
- Aoki, M. The impaired subjective perception of verticality independent of peripheral vestibular function in dizzy elderly with orthostatic hypotension. Aging Clin. Exp. Res. 2016, 29, 647–653. [Google Scholar] [CrossRef] [PubMed]
- Voß, L.J.; Zabaneh, S.I.; Hölzl, M.; Olze, H.; Stölzel, K. The subjective perception of the vertical-a valuable parameter for determination of peripheral vestibular disorder in Menière’s disease in the chronic phase? HNO 2019, 67, 282–292. [Google Scholar] [CrossRef] [PubMed]
- Lopez-Escamez, J.A.; Carey, J.; Chung, W.H.; Goebel, J.A.; Magnusson, M.; Mandala, M. Diagnostic criteria for Meniere’s disease. J Vestib. Res. 2015, 25, 1–7. [Google Scholar] [CrossRef]
- Gurkov, R.; Jerin, C.; Flatz, W.; Maxwell, R. Clinical manifestations of hydropic ear disease (Meniere’s). Eur. Arch. Otorhinolaryngol. 2019, 276, 27–40. [Google Scholar] [CrossRef]
- Kumagami, H.; Sainoo, Y.; Fujiyama, D.; Baba, A.; Oku, R.; Takasaki, K.; Shigeno, K.; Takahashi, H. Subjective visual vertical in acute attacks of Ménière’s disease. Otol Neurotol. 2009, 30, 206–209. [Google Scholar] [CrossRef] [PubMed]
- Magnan, J.; Özgirgin, O.N.; Trabalzini, F.; Lacour, M.; Lopez-Escamez, J.A.; Magnusson, M.; Güneri, E.A.; Guyot, J.P.; Nuti, D.; Mandala, M. European Position Statement on Diagnosis, and Treatment of Meniere’s Disease. J. Int. Adv. Otol. 2018, 14, 317–321. [Google Scholar] [CrossRef]
- Ihler, F.; Bertlich, M.; Sharaf, K.; Strieth, S.; Strupp, M.; Canis, M. Betahistine Exerts a Dose-Dependent Effect on Cochlear Stria Vascularis Blood Flow in Guinea Pigs In Vivo. PLoS ONE 2012, 7, e39086. [Google Scholar] [CrossRef] [PubMed]
- Adrion, C.; Fischer, C.S.; Wagner, J.; Gürkov, R.; Mansmann, U.; Strupp, M. Efficacy and safety of betahistine treatment in patients with Meniere’s disease: Primary results of a long term, multicentre, double blind, randomised, placebo controlled, dose defining trial (BEMED trial). BMJ 2016, 352, h6816. [Google Scholar] [CrossRef]
- Baier, B.; Thömke, F.; Wilting, J.; Heinze, C.; Geber, C.; Dieterich, M. A Pathway in the Brainstem for Roll-Tilt of the Subjective Visual Vertical: Evidence from a Lesion-Behavior Mapping Study. J. Neurosci. 2012, 32, 14854–14858. [Google Scholar] [CrossRef]
- Pinar, H.S.; Ardiç, F.N.; Topuz, B.; Kara, C.O. Subjective Visual Vertical and Subjective Visual Horizontal Measures in Patients with Chronic Dizziness. J. Otolaryngol. 2005, 34, 121–125. [Google Scholar] [CrossRef]
- Adams, M.E.; Heidenreich, K.D.; Kileny, P.R. Audiovestibular testing in patients with Meniere’s disease. Otolaryngol. Clin. N. Am. 2010, 43, 995–1009. [Google Scholar] [CrossRef]
- Basura, G.J.; Adams, M.E.; Monfared, A.; Schwartz, S.R.; Antonelli, P.J.; Burkard, R.; Bush, M.L.; Bykowski, J.; Colandrea, M.; Derebery, J.; et al. Clinical Practice Guideline: Meniere’s Disease. Otolaryngol. Head Neck Surg. 2020, 162, S1–S55. [Google Scholar] [CrossRef] [PubMed]
- Sugimoto, S.; Yoshida, T.; Teranishi, M.; Kobayashi, M.; Shimono, M.; Naganawa, S.; Sone, M. Significance of Endolymphatic Hydrops Herniation Into the Semicircular Canals Detected on MRI. Otol. Neurotol. 2018, 39, 1229–1234. [Google Scholar] [CrossRef] [PubMed]
- Attyé, A.; Eliezer, M.; Medici, M.; Tropres, I.; Dumas, G.; Krainik, A.; Schmerber, S. In vivo imaging of saccular hydrops in humans reflects sensorineural hearing loss rather than Meniere’s disease symptoms. Eur. Radiol. 2018, 28, 2916–2922. [Google Scholar] [CrossRef] [PubMed]
- Kahn, L.; Hautefort, C.; Guichard, J.P.; Toupet, M.; Jourdaine, C.; Vitaux, H.; Herman, P.; Kania, R.; Houdart, E.; Attyé, A.; et al. Relationship between video head impulse test, ocular and cervical vestibular evoked myogenic potentials, and compartmental magnetic resonance imaging classification in meniere’s disease. Laryngoscope 2020, 130, E444–E452. [Google Scholar] [CrossRef]
- Michelson, P.L.; McCaslin, D.L.; Jacobson, G.P.; Petrak, M.; English, L.; Hatton, K. Assessment of Subjective Visual Vertical (SVV) Using the “Bucket Test” and the Virtual SVV System. Am. J. Audiol. 2018, 27, 249–259. [Google Scholar] [CrossRef]
- Alexander, T.H.; Harris, J.P. Current epidemiology of Meniere’s syndrome. Otolaryngol. Clin. N. Am. 2010, 43, 965–970. [Google Scholar] [CrossRef] [PubMed]
- Sun, D.Q.; Zuniga, M.G.; Davalos-Bichara, M.; Carey, J.P.; Agrawal, Y. Evaluation of a bedside test of utricular function—The bucket test—In older individuals. Acta Oto-Laryngol. 2014, 134, 382–389. [Google Scholar] [CrossRef]

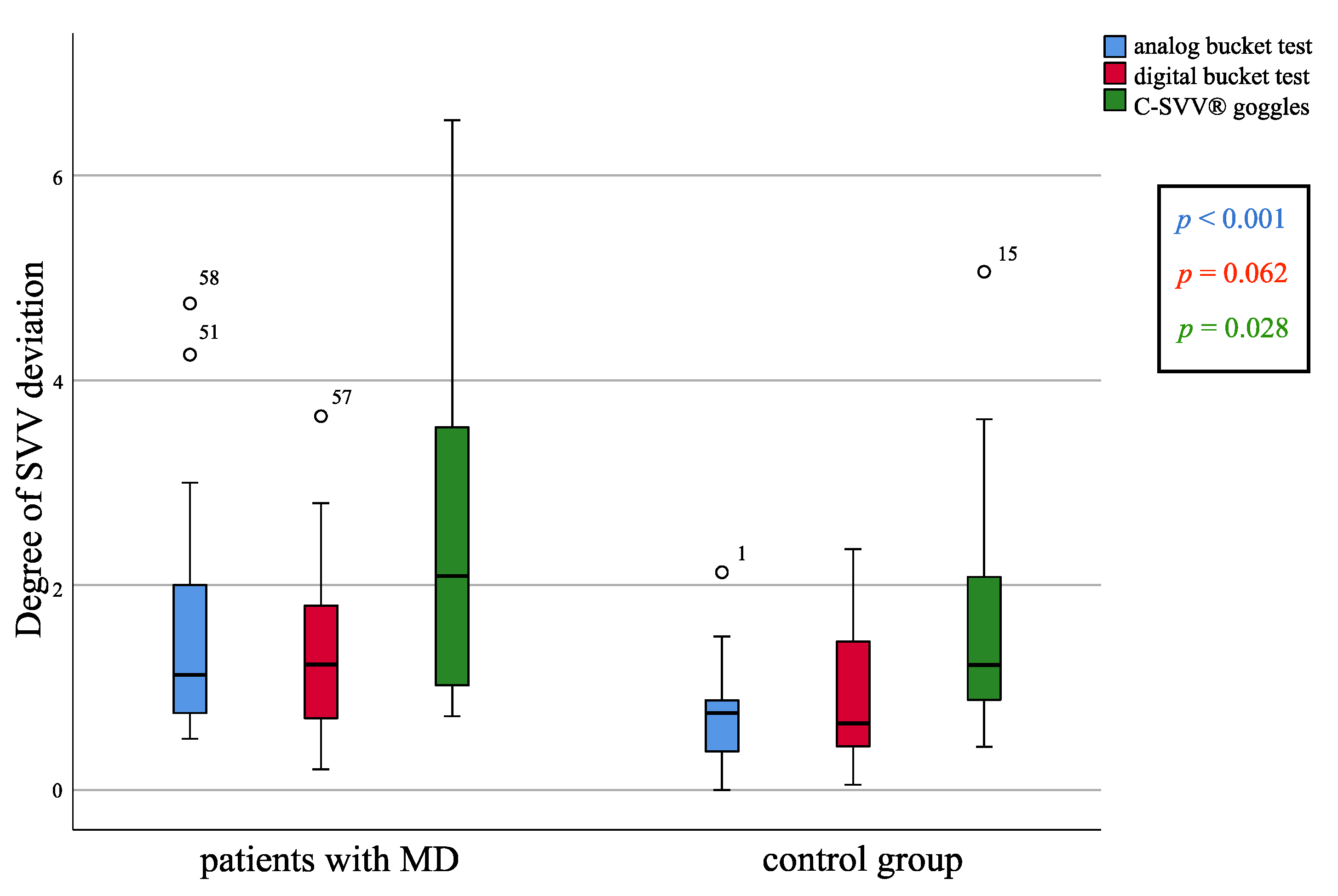

| Parameters Measured | Patients with MD (n = 26) | Control Group (n = 39) |
| Age, median (range), years | 54.50 (25–75) | 25.00 (20–59) |
| MD classification (Barany), definite/probable | 24/2 | - |
| Sex, female/male | 15/11 | 30/9 |
| Average DHI | 39.28 | - |
| Analog Bucket Test, Degrees | ||
| Average (±standard deviation) | 1.500 (±1.134) | 0.686 (±0.446) |
| Median | 1.125 | 0.750 |
| Difference in averages | –0.814 | |
| Digital Bucket Test (Visual Vertical App), Degrees | ||
| Average (±standard deviation) | 1.346 (±0.836) | 0.968 (0.701) |
| Median | 1.225 | 0.650 |
| Difference in averages | –0.378 | |
| C-SVV® Goggles, Degrees | ||
| Average (±standard deviation) | 2.391 (±1.567) | 1.550 (±0.986) |
| Median | 2.090 | 1.220 |
| Difference in averages | –0.841 | |
| AUC of ROC curves | ||
| Bucket test analog | 0.754 | |
| C-SVV®-goggles | 0.662 | |
| Influence of Intake of Betahistine (Analog Bucket Test), Degrees | ||
| No | ||
| Average (±standard deviation) | 1.656 (±1.125) | - |
| Median | 1.375 | - |
| Yes | ||
| Average (±standard deviation) | 1.250 (±1.161) | - |
| Median | 0.750 | - |
| Influence of Intake of Betahistine (C-SVV® Goggles), Degrees | ||
| No | ||
| Average (±standard deviation) | 2.191 (±1.559) | - |
| Median | 1.280 | - |
| Yes | ||
| Average (±standard deviation) | 2.710 (±1.607) | - |
| Median | 2.610 | - |
| (a) Confusion matrix for the analog bucket test with the new pathologic threshold of >1.125°. | |||
| Predicted Condition | Patients with Meniere’s Disease | Control Group | Accuracy |
| Negative | T.N.: 13 (50%) | T.N.: 35 (89.7%) | NPV = 72.9% |
| Positive | F.P.: 13 (50%) | F.P.: 4 (10.3%) | PPV = 76.5% |
| Specificity: 50% | Specificity: 89.7% | Accuracy = 73.84% | |
| (b) Confusion matrix for the C-SVV® goggles with the new pathologic threshold of >2.5°. | |||
| Predicted Condition | Patients with Meniere’s Disease | Control Group | Accuracy |
| Negative | F.N.: 14 (53.8%) | T.N.: 34 (87.2%) | NPV =70.2% |
| Positive | T.P.: 12 (46.2%) | F.P.: 5 (12.8%) | PPV = 70.6% |
| Sensitivity: 46.2% | Specificity: 87.2% | Accuracy = 69.23% | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zabaneh, S.I.; Voss, L.J.; Szczepek, A.J.; Olze, H.; Stölzel, K. Methods for Testing the Subjective Visual Vertical during the Chronic Phase of Menière’s Disease. Diagnostics 2021, 11, 249. https://doi.org/10.3390/diagnostics11020249
Zabaneh SI, Voss LJ, Szczepek AJ, Olze H, Stölzel K. Methods for Testing the Subjective Visual Vertical during the Chronic Phase of Menière’s Disease. Diagnostics. 2021; 11(2):249. https://doi.org/10.3390/diagnostics11020249
Chicago/Turabian StyleZabaneh, Samira Ira, Linda Josephine Voss, Agnieszka J. Szczepek, Heidi Olze, and Katharina Stölzel. 2021. "Methods for Testing the Subjective Visual Vertical during the Chronic Phase of Menière’s Disease" Diagnostics 11, no. 2: 249. https://doi.org/10.3390/diagnostics11020249
APA StyleZabaneh, S. I., Voss, L. J., Szczepek, A. J., Olze, H., & Stölzel, K. (2021). Methods for Testing the Subjective Visual Vertical during the Chronic Phase of Menière’s Disease. Diagnostics, 11(2), 249. https://doi.org/10.3390/diagnostics11020249








