Volumetric Imaging of Lung Tissue at Micrometer Resolution: Clinical Applications of Micro-CT for the Diagnosis of Pulmonary Diseases
Abstract
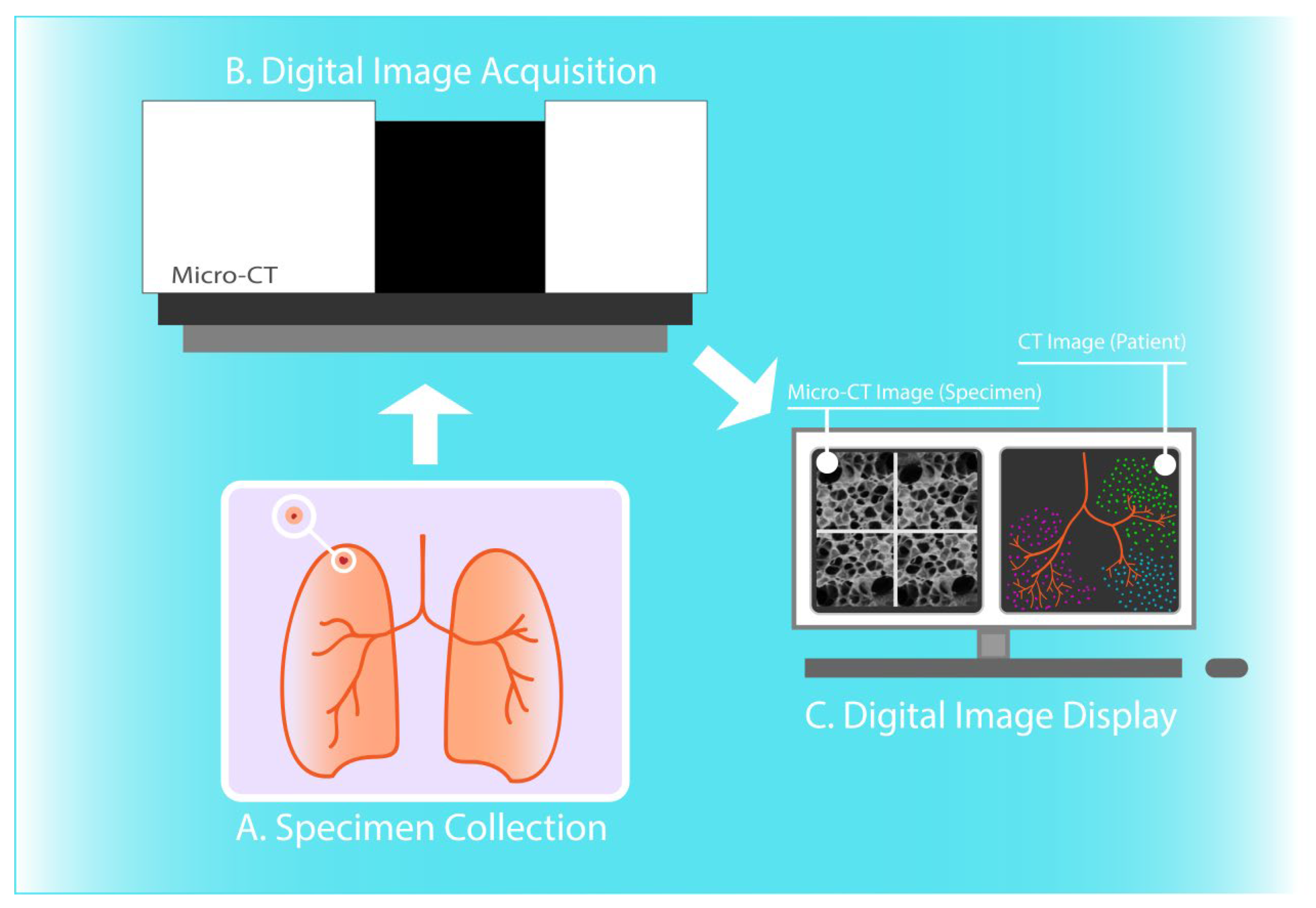
:1. Introduction
2. Materials and Methods
2.1. Search Strategy
2.2. Eligibility Criteria
2.3. Selection of Studies
2.4. Data Extraction and Tabulation
2.5. Quality Assessment
3. Results
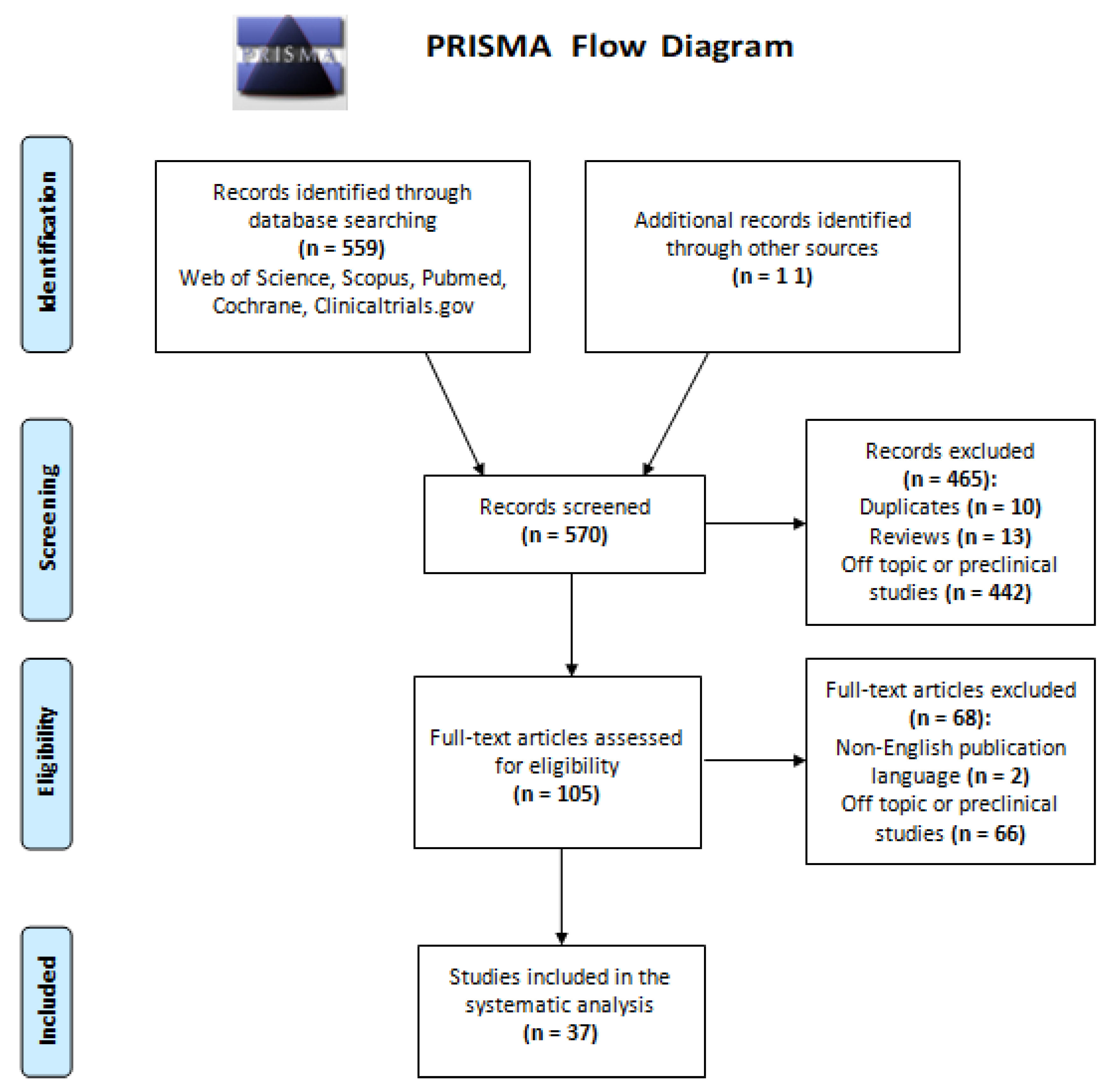
3.1. Selection of Eligible Studies
3.2. Study Quality
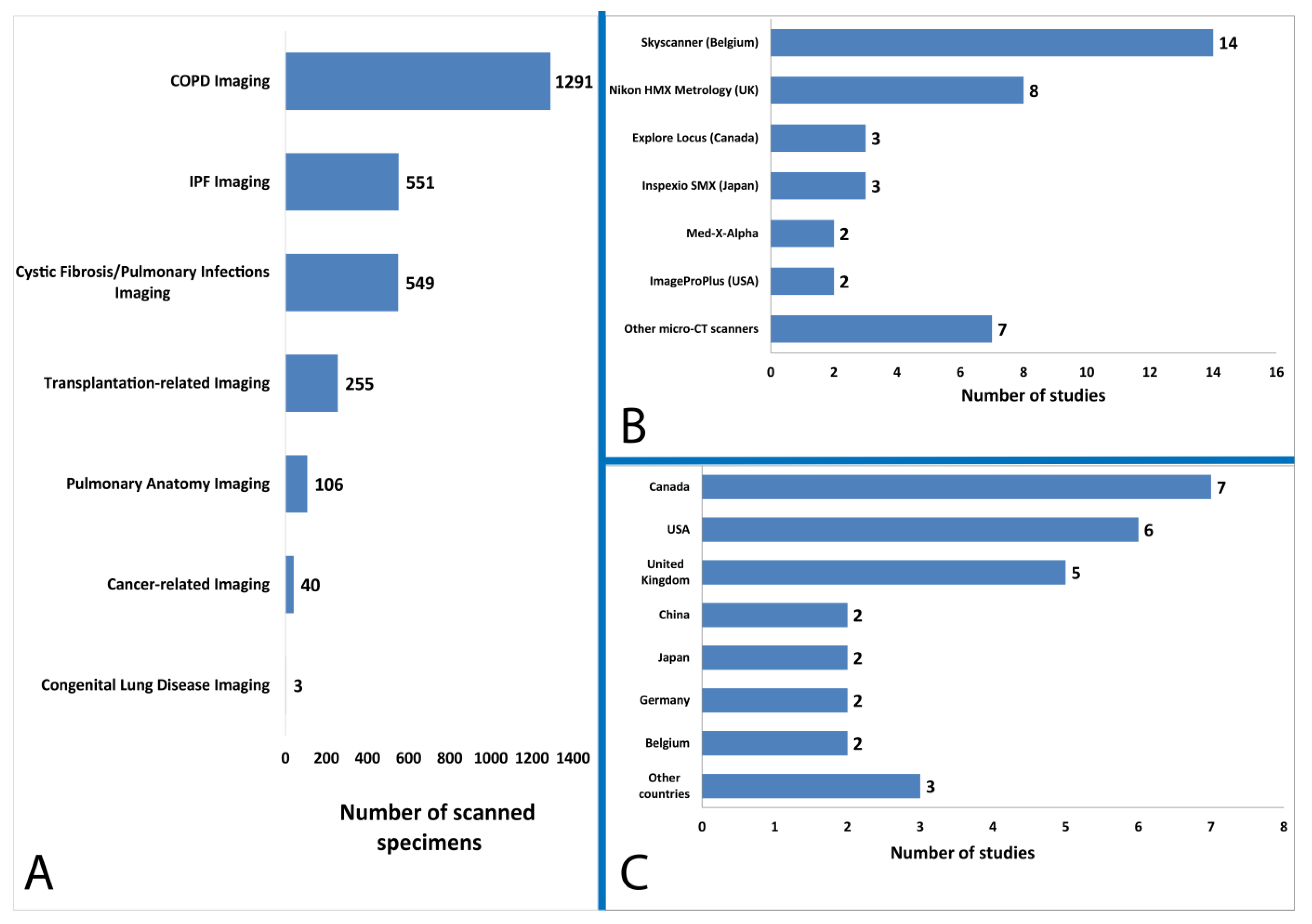
3.3. Characteristics of Included Studies
4. Discussion
4.1. Visualization and Quantitative Analysis of the Lung Microanatomy in Chronic Obstructive and Restrictive Pulmonary Disease
4.2. Non-Destructive Imaging of Surgical Specimens for Lung Cancer Diagnosis
4.3. Evaluation of Lung Allograft Specimens upon Transplant Rejection or Prior to Transplantation
4.4. Assessing COVID-19-Related Alterations of Lung Tissue
4.5. Example of a Potential Future Micro-CT Application in Lung Cancer Management
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Hutchinson, J.C.; Shelmerdine, S.C.; Simcock, I.C.; Sebire, N.J.; Arthurs, O.J. Early clinical applications for imaging at microscopic detail: Microfocus computed tomography (micro-CT). Br. J. Radiol. 2017, 90, 20170113. [Google Scholar] [CrossRef]
- Ritman, E.L. Current status of developments and applications of Micro-CT. Annu. Rev. Biomed. Eng. 2011, 13, 531–552. [Google Scholar] [CrossRef] [PubMed]
- Papazoglou, A.S.; Karagiannidis, E.; Moysidis, D.V.; Sofidis, G.; Bompoti, A.; Stalikas, N.; Panteris, E.; Arvanitidis, C.; Herrman, M.D.; Michaelson, J.S.; et al. Current clinical applications and potential perspective of micro-computed tomography in cardiovascular imaging: A systematic scoping review. Hell. J. Cardiol. 2021. [Google Scholar] [CrossRef] [PubMed]
- Du Plessis, A.; Broeckhoven, C.; Guelpa, A.; le Roux, S. Laboratory x-ray micro-computed tomography: A user guideline for biological samples. GigaScience 2017, 6, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Cecchini, M.J.; Tarmey, T.; Ferreira, A.; Mangaonkar, A.A.; Ferrer, A.; Patnaik, M.M.; Wylam, M.E.; Jenkins, S.M.; Spears, G.M.; Yi, E.S.; et al. Pathology, radiology, and genetics of interstitial lung disease in patients with shortened telomeres. Am. J. Surg. Pathol. 2021, 47, 871–884. [Google Scholar] [CrossRef]
- Ritman, E.L. Micro-Computed tomography of the lungs and pulmonary-vascular system. Proc. Am. Thorac. Soc. 2005, 2, 477–480. [Google Scholar] [CrossRef]
- Wells, W.A.; Thrall, M.; Sorokina, A.; Fine, J.; Krishnamurthy, S.; Haroon, A.; Rao, B.; Shevchuk, M.M.; Wolfsen, H.C.; Tearney, G.J.; et al. In vivo and ex vivo microscopy: Moving toward the integration of optical imaging technologies into pathology practice. Arch. Pathol. Lab. Med. 2018, 143, 288–298. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, J.T.C.; Glaser, A.K.; Bera, K.; True, L.D.; Reder, N.P.; Eliceiri, K.W.; Madabhushi, A. Harnessing non-destructive 3D pathology. Nat. Biomed. Eng. 2021, 5, 203–218. [Google Scholar] [CrossRef]
- DiCorpo, D.; Tiwari, A.; Tang, R.; Griffin, M.; Aftreth, O.; Bautista, P.; Hughes, K.; Gershenfeld, N.; Michaelson, J. The role of Micro-CT in imaging breast cancer specimens. Breast Cancer Res. Treat. 2020, 180, 343–357. [Google Scholar] [CrossRef]
- Mori, K. From macro-scale to micro-scale computational anatomy: A perspective on the next 20 years. Med. Image Anal. 2016, 33, 159–164. [Google Scholar] [CrossRef]
- Hochhegger, B.; Langer, F.W.; Irion, K.; Souza, A.; Moreira, J.; Baldisserotto, M.; Pallaoro, Y.; Muller, E.; Medeiros, T.M.; Altmayer, S.; et al. Pulmonary acinus: Understanding the computed tomography findings from an acinar perspective. Lung 2019, 197, 259–265. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Chen, H.; Ambalavanan, N.; Liu, G.; Antony, V.B.; Ding, Q.; Nath, H.; Eary, J.F.; Thannickal, V.J. Noninvasive imaging of experimental lung fibrosis. Am. J. Respir. Cell Mol. Biol. 2015, 53, 8–13. [Google Scholar] [CrossRef] [PubMed]
- Tricco, A.C.; Lillie, E.; Zarin, W.; O’Brien, K.K.; Colquhoun, H.; Levac, D.; Moher, D.; Peters, M.D.J.; Horsley, T.; Weeks, L.; et al. PRISMA Extension for Scoping Reviews (PRISMA-ScR): Checklist and Explanation. Ann. Intern. Med. 2018, 169, 467–473. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ouzzani, M.; Hammady, H.; Fedorowicz, Z.; Elmagarmid, A. Rayyan—A web and mobile app for systematic reviews. Syst. Rev. 2016, 5, 210. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lo, C.K.-L.; Mertz, D.; Loeb, M. Newcastle-Ottawa scale: Comparing reviewers’ to authors’ assessments. BMC Med. Res. Methodol. 2014, 14, 45. [Google Scholar] [CrossRef] [Green Version]
- Stang, A. Critical evaluation of the Newcastle-Ottawa scale for the assessment of the quality of nonrandomized studies in meta-analyses. Eur. J. Epidemiol. 2010, 25, 603–605. [Google Scholar] [CrossRef] [Green Version]
- Kayı Cangır, A.; Dizbay Sak, S.; Güneş, G.; Orhan, K. Differentiation of benign and malignant regions in paraffin embedded tissue blocks of pulmonary adenocarcinoma using micro CT scanning of paraffin tissue blocks: A pilot study for method validation. Surg. Today 2021, 51, 1594–1601. [Google Scholar] [CrossRef]
- Kirby, M.; Tanabe, N.; Vasilescu, D.M.; Cooper, J.D.; McDonough, J.; Verleden, S.; Vanaudenaerde, B.; Sin, D.D.; Tan, W.C.; Coxson, H.O.; et al. Computed tomography total airway count is associated with the number of micro–computed tomography terminal bronchioles. Am. J. Respir. Crit. Care Med. 2020, 201, 613–615. [Google Scholar] [CrossRef]
- Tanabe, N.; McDonough, J.E.; Vasilescu, D.M.; Ikezoe, K.; Verleden, S.; Xu, F.; Wuyts, W.A.; Vanaudenaerde, B.; Colby, T.V.; Hogg, J.C. Pathology of idiopathic pulmonary fibrosis assessed by a combination of microcomputed tomography, histology, and immunohistochemistry. Am. J. Pathol. 2020, 190, 2427–2435. [Google Scholar] [CrossRef]
- Verleden, S.; Tanabe, N.; McDonough, J.; Vasilescu, D.M.; Xu, F.; Wuyts, W.A.; Piloni, D.; De Sadeleer, L.; Willems, S.; Mai, C.; et al. Small airways pathology in idiopathic pulmonary fibrosis: A retrospective cohort study. Lancet Respir. Med. 2020, 8, 573–584. [Google Scholar] [CrossRef]
- Nakamura, S.; Mori, K.; Iwano, S.; Kawaguchi, K.; Fukui, T.; Hakiri, S.; Ozeki, N.; Oda, M.; Yokoi, K. Micro-computed tomography images of lung adenocarcinoma: Detection of lepidic growth patterns. Nagoya J. Med. Sci. 2020, 82, 25–31. [Google Scholar] [PubMed]
- Verleden, S.E.; Kirby, M.; Everaerts, S.; Vanstapel, A.; McDonough, J.E.; Verbeken, E.K.; Braubach, P.; Boone, M.N.; Aslam, D.; Verschakelen, J.; et al. Small airway loss in the physiologically ageing lung: A cross-sectional study in unused donor lungs. Lancet Respir. Med. 2021, 9, 167–174. [Google Scholar] [CrossRef]
- Norvik, C.; Westöö, C.K.; Peruzzi, N.; Lovric, G.; Van Der Have, O.; Mokso, R.; Jeremiasen, I.; Brunnström, H.; Galambos, C.; Bech, M.; et al. Synchrotron-based phase-contrast micro-CT as a tool for understanding pulmonary vascular pathobiology and the 3-D microanatomy of alveolar capillary dysplasia. Am. J. Physiol. Cell. Mol. Physiol. 2020, 318, L65–L75. [Google Scholar] [CrossRef]
- Umetani, K.; Okamoto, T.; Saito, K.; Kawata, Y.; Niki, N. 36M-pixel synchrotron radiation micro-CT for whole secondary pulmonary lobule visualization from a large human lung specimen. Eur. J. Radiol. Open 2020, 7, 100262. [Google Scholar] [CrossRef] [PubMed]
- Vasilescu, D.M.; Phillion, A.B.; Kinose, D.; Verleden, S.; Vanaudenaerde, B.; Verleden, G.M.; Van Raemdonck, D.; Stevenson, C.S.; Hague, C.J.; Han, M.K.; et al. Comprehensive stereological assessment of the human lung using multiresolution computed tomography. J. Appl. Physiol. 2020, 128, 1604–1616. [Google Scholar] [CrossRef]
- Everaerts, S.; McDonough, J.; Verleden, S.; Josipovic, I.; Boone, M.; Dubbeldam, A.; Mathyssen, C.; Serré, J.; Dupont, L.J.; Gayan-Ramirez, G.; et al. Airway morphometry in COPD with bronchiectasis: A view on all airway generations. Eur. Respir. J. 2019, 54, 1802166. [Google Scholar] [CrossRef]
- Katsamenis, O.L.; Olding, M.; Warner, J.A.; Chatelet, D.S.; Jones, M.G.; Sgalla, G.; Smit, B.; Larkin, O.J.; Haig, I.; Richeldi, L.; et al. X-ray Micro-Computed tomography for nondestructive three-dimensional (3D) X-ray histology. Am. J. Pathol. 2019, 189, 1608–1620. [Google Scholar] [CrossRef] [Green Version]
- Troschel, F.M.; Gottumukkala, R.V.; Dicorpo, D.; Mario, J.; Ott, H.C.; Wright, C.D.; Muniappan, A.; Lanuti, M.; Yang, K.; Shepard, J.O.; et al. Feasibility of perioperative micro–computed tomography of human lung cancer specimens: A pilot study. Arch. Pathol. Lab. Med. 2018, 143, 319–325. [Google Scholar] [CrossRef]
- Shelmerdine, S.C.; Ashworth, M.T.; Calder, A.D.; Muthialu, N.; Arthurs, O. Micro-CT of tracheal stenosis in trisomy 21. Thorax 2019, 74, 419–420. [Google Scholar] [CrossRef] [Green Version]
- Vasilescu, D.M.; Martinez, F.J.; Marchetti, N.; Galbán, C.J.; Hatt, C.; Meldrum, C.A.; Dass, C.; Tanabe, N.; Reddy, R.M.; Lagstein, A.; et al. Noninvasive imaging biomarker identifies small airway damage in severe chronic obstructive pulmonary disease. Am. J. Respir. Crit. Care Med. 2019, 200, 575–581. [Google Scholar] [CrossRef]
- Robinson, S.K.; Ramsden, J.J.; Warner, J.; Lackie, P.M.; Roose, T. Correlative 3D imaging and microfluidic modelling of human pulmonary lymphatics using immunohistochemistry and high-resolution μCT. Sci. Rep. 2019, 9, 6415. [Google Scholar] [CrossRef]
- McDonough, J.E.; Ahangari, F.; Li, Q.; Jain, S.; Verleden, S.E.; Herazo-Maya, J.; Vukmirovic, M.; Deiuliis, G.; Tzouvelekis, A.; Tanabe, N.; et al. Transcriptional regulatory model of fibrosis progression in the human lung. JCI Insight 2019, 4, e131597. [Google Scholar] [CrossRef] [PubMed]
- Verleden, S.E.; Gheysens, O.; Goffin, K.E.; Vanaudenaerde, B.M.; Verbeken, E.K.; Weynand, B.; Van Raemdonck, D.E.; Verleden, G.M.; Vos, R. Role of 18F-FDG PET/CT in restrictive allograft syndrome after lung transplantation. Transplantation 2019, 103, 823–831. [Google Scholar] [CrossRef]
- Tanabe, N.; Vasilescu, D.M.; Kirby, M.; Coxson, H.O.; Verleden, S.; Vanaudenaerde, B.; Kinose, D.; Nakano, Y.; Paré, P.D.; Hogg, J.C. Analysis of airway pathology in COPD using a combination of computed tomography, micro-computed tomography and histology. Eur. Respir. J. 2018, 51, 1701245. [Google Scholar] [CrossRef] [PubMed]
- Everaerts, S.; Lammertyn, E.J.; Martens, D.S.; De Sadeleer, L.J.; Maes, K.; Van Batenburg, A.A.; Goldschmeding, R.; Van Moorsel, C.H.M.; Dupont, L.J.; Wuyts, W.A.; et al. The aging lung: Tissue telomere shortening in health and disease. Respir. Res. 2018, 19, 95. [Google Scholar] [CrossRef] [PubMed]
- Verleden, S.E.; Martens, A.; Ordies, S.; Heigl, T.; Bellon, H.; Vandermeulen, E.; Van Herck, A.; Sacreas, A.; Verschakelen, J.; Coudyzer, W.; et al. Radiological analysis of unused donor lungs: A tool to improve donor acceptance for transplantation? Arab. Archaeol. Epigr. 2017, 17, 1912–1921. [Google Scholar] [CrossRef]
- Suzuki, M.; Sze, M.A.; Campbell, J.D.; Ii, J.F.B.; Lenburg, M.E.; McDonough, J.; Elliott, W.M.; Cooper, J.D.; Spira, A.; Hogg, J.C. The cellular and molecular determinants of emphysematous destruction in COPD. Sci. Rep. 2017, 7, 9562. [Google Scholar] [CrossRef] [Green Version]
- Tanabe, N.; Vasilescu, D.M.; McDonough, J.; Kinose, D.; Suzuki, M.; Cooper, J.D.; Paré, P.D.; Hogg, J.C. Micro–Computed tomography comparison of preterminal bronchioles in centrilobular and panlobular emphysema. Am. J. Respir. Crit. Care Med. 2017, 195, 630–638. [Google Scholar] [CrossRef]
- Mai, C.; Verleden, S.; McDonough, J.; Willems, S.; De Wever, W.; Coolen, J.; Dubbeldam, A.; Van Raemdonck, D.; Verbeken, E.K.; Verleden, G.M.; et al. Thin-Section CT features of idiopathic pulmonary fibrosis correlated with Micro-CT and histologic analysis. Radiology 2017, 283, 252–263. [Google Scholar] [CrossRef]
- Vasilescu, D.M.; Phillion, A.B.; Tanabe, N.; Kinose, D.; Paige, D.F.; Kantrowitz, J.J.; Liu, G.; Liu, H.; Fishbane, N.; Verleden, S.; et al. Nondestructive cryomicro-CT imaging enables structural and molecular analysis of human lung tissue. J. Appl. Physiol. 2017, 122, 161–169. [Google Scholar] [CrossRef] [Green Version]
- Guan, C.-S.; Ma, D.-Q.; Cui, D.; Chen, J.-H.; Chen, B.-D.; Zhang, Y.-S.; Liu, W.-H. Short linear shadows connecting pulmonary segmental arteries to oblique fissures in volumetric thin-section CT images: Comparing CT, micro-CT and histopathology. Eur. Radiol. 2015, 26, 2740–2748. [Google Scholar] [CrossRef] [PubMed]
- Jones, M.G.; Fabre, A.; Schneider, P.; Cinetto, F.; Sgalla, G.; Mavrogordato, M.; Jogai, S.; Alzetani, A.; Marshall, B.; O’Reilly, K.M.; et al. Three-dimensional characterization of fibroblast foci in idiopathic pulmonary fibrosis. JCI Insight 2016, 1, e86375. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Boon, M.; Verleden, S.; Bosch, B.; Lammertyn, M.E.J.; McDonough, J.E.; Mai, C.; Verschakelen, J.; De Corput, M.K.-V.; Tiddens, H.A.W.; Proesmans, M.; et al. Morphometric analysis of explant lungs in cystic fibrosis. Am. J. Respir. Crit. Care Med. 2016, 193, 516–526. [Google Scholar] [CrossRef] [Green Version]
- Scott, A.E.; Vasilescu, D.M.; Seal, K.A.D.; Keyes, S.D.; Mavrogordato, M.N.; Hogg, J.C.; Sinclair, I.; Warner, J.A.; Hackett, T.-L.; Lackie, P.M. Three dimensional imaging of paraffin embedded human lung tissue samples by Micro-Computed tomography. PLoS ONE 2015, 10, e0126230. [Google Scholar] [CrossRef] [Green Version]
- Verleden, S.E.; Vasilescu, A.M.; McDonough, J.E.; Ruttens, D.; Vos, R.; Vandermeulen, E.; Bellon, H.; Geenens, R.; Verbeken, E.K.; Verschakelen, J.; et al. Linking clinical phenotypes of chronic lung allograft dysfunction to changes in lung structure. Eur. Respir. J. 2015, 46, 1430–1439. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zuo, Y.-Z.; Liu, C.; Liu, S.-W. Pulmonary intersegmental planes: Imaging appearance and possible reasons leading to their visualization. Radiology 2013, 267, 267–275. [Google Scholar] [CrossRef] [PubMed]
- Kampschulte, M.; Schneider, C.R.; Litzlbauer, H.D.; Tscholl, D.; Schneider, C.; Zeiner, C.; Krombach, G.A.; Ritman, E.L.; Bohle, R.M.; Langheinirch, A.C. Quantitative 3D micro-CT imaging of human lung tissue. In RöFo-Fortschritte auf dem Gebiet der Röntgenstrahlen und der Bildgebenden Verfahren; Georg Thieme Verlag KG: New York, NY, USA, 2013; Volume 185, pp. 869–876. [Google Scholar]
- Okubo, Y.; Ishiwatari, T.; Izumi, H.; Sato, F.; Aki, K.; Sasai, D.; Ando, T.; Shinozaki, M.; Natori, K.; Tochigi, N.; et al. Pathophysiological implication of reversed CT halo sign in invasive pulmonary mucormycosis: A rare case report. Diagn. Pathol. 2013, 8, 82. [Google Scholar] [CrossRef] [Green Version]
- Campbell, J.D.; McDonough, J.E.; Zeskind, J.E.; Hackett, T.L.; Pechkovsky, D.V.; Brandsma, C.-A.; Suzuki, M.; Gosselink, J.V.; Liu, G.; Alekseyev, Y.O.; et al. A gene expression signature of emphysema-related lung destruction and its reversal by the tripeptide GHK. Genome Med. 2012, 4, 67. [Google Scholar] [CrossRef] [Green Version]
- Litzlbauer, H.D.; Korbel, K.; Kline, T.L.; Jorgensen, S.M.; Eaker, D.R.; Bohle, R.M.; Ritman, E.L.; Langheinrich, A.C. Synchrotron-based Micro-CT imaging of the human lung acinus. Anat. Rec. Adv. Integr. Anat. Evol. Biol. 2010, 293, 1607–1614. [Google Scholar] [CrossRef] [Green Version]
- Hogg, J.C.; McDonough, J.E.; Sanchez, P.G.; Cooper, J.D.; Coxson, H.O.; Elliott, W.M.; Naiman, D.; Pochettino, M.; Horng, D.; Gefter, W.B.; et al. Micro-Computed Tomography measurements of peripheral lung pathology in chronic obstructive pulmonary disease. Proc. Am. Thorac. Soc. 2009, 6, 546–549. [Google Scholar] [CrossRef] [Green Version]
- Hogg, J.C.; McDonough, J.; Gosselink, J.V.; Hayashi, S. What Drives the peripheral lung-remodeling process in chronic obstructive pulmonary disease? Proc. Am. Thorac. Soc. 2009, 6, 668–672. [Google Scholar] [CrossRef]
- Watz, H.; Breithecker, A.; Rau, W.S.; Kriete, A. Micro-CT of the human lung: Imaging of alveoli and virtual endoscopy of an alveolar duct in a normal lung and in a lung with centrilobular Emphysema—Initial observations. Radiology 2005, 236, 1053–1058. [Google Scholar] [CrossRef] [PubMed]
- Dame Carroll, J.R.; Chandra, A.; Jones, A.S.; Berend, N.; Magnussen, J.S.; King, G.G. Airway dimensions measured from micro-computed tomography and high-resolution computed tomography. Eur. Respir. J. 2006, 28, 712–720. [Google Scholar] [CrossRef] [Green Version]
- Hariri, L.P.; Mino-Kenudson, M.; Mark, E.J.; Suter, M.J. In vivo optical coherence tomography: The role of the pathologist. Arch. Pathol. Lab. Med. 2012, 136, 1492–1501. [Google Scholar] [CrossRef] [PubMed]
- Adams, D.C.; Hariri, L.P.; Miller, A.J.; Wang, Y.; Cho, J.L.; Villiger, M.; Holz, J.A.; Szabari, M.V.; Hamilos, D.L.; Harris, R.S.; et al. Birefringence microscopy platform for assessing airway smooth muscle structure and function in vivo. Sci. Transl. Med. 2016, 8, 359ra131. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hariri, L.P.; Roden, A.C.; Chung, J.H.; Danoff, S.K.; Manjarres, D.C.G.; Hartwig, M.; Kheir, F.; King, C.; Kreider, M.; Lynch, D.A.; et al. The role of surgical lung biopsy in the diagnosis of fibrotic interstitial lung disease: Perspective from the pulmonary fibrosis foundation. Ann. Am. Thorac. Soc. 2021, 18, 1601–1609. [Google Scholar] [CrossRef] [PubMed]
- Taghavi-Farahabadi, M.; Mahmoudi, M.; Soudi, S.; Hashemi, S.M. Hypothesis for the management and treatment of the COVID-19-induced acute respiratory distress syndrome and lung injury using mesenchymal stem cell-derived exosomes. Med. Hypotheses 2020, 144, 109865. [Google Scholar] [CrossRef] [PubMed]
- Boudewijns, R.; Thibaut, H.J.; Kaptein, S.J.F.; Li, R.; Vergote, V.; Seldeslachts, L.; Van Weyenbergh, J.; De Keyzer, C.; Bervoets, L.; Sharma, S.; et al. STAT2 signaling restricts viral dissemination but drives severe pneumonia in SARS-CoV-2 infected hamsters. Nat. Commun. 2020, 11, 5838. [Google Scholar] [CrossRef] [PubMed]
- Kawakami, T.; Nabeshima, K.; Hamasaki, M.; Iwasaki, A.; Shirakusa, T.; Iwasaki, H. Small cluster invasion: A possible link between micropapillary pattern and lymph node metastasis in pT1 lung adenocarcinomas. Virchows Arch. 2008, 454, 61–70. [Google Scholar] [CrossRef] [PubMed]
- Travis, W.D.; Brambilla, E.; Nicholson, A.G.; Yatabe, Y.; Austin, J.H.M.; Beasley, M.B.; Chirieac, L.R.; Dacic, S.; Duhig, E.; Flieder, D.B.; et al. The 2015 World Health Organization classification of lung tumors: Impact of genetic, clinical and radiologic advances since the 2004 classification. J. Thorac. Oncol. 2015, 10, 1243–1260. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yokoyama, S.; Murakami, T.; Tao, H.; Onoda, H.; Hara, A.; Miyazaki, R.; Furukawa, M.; Hayashi, M.; Inokawa, H.; Okabe, K.; et al. Tumor spread through air spaces identifies a distinct subgroup with poor prognosis in surgically resected lung pleomorphic carcinoma. Chest 2018, 154, 838–847. [Google Scholar] [CrossRef] [PubMed]
- Kadota, K.; Kushida, Y.; Katsuki, N.; Ishikawa, R.; Ibuki, E.; Motoyama, M.; Nii, K.; Yokomise, H.; Bandoh, S.; Haba, R. Tumor spread through air spaces is an independent predictor of recurrence-free survival in patients with resected lung squamous cell carcinoma. Am. J. Surg. Pathol. 2017, 41, 1077–1086. [Google Scholar] [CrossRef] [PubMed]
- Yin, Q.; Wang, H.; Cui, H.; Wang, W.; Yang, G.; Qie, P.; Xun, X.; Han, S.; Liu, H. Meta-analysis of association between CT-based features and tumor spread through air spaces in lung adenocarcinoma. J. Cardiothorac. Surg. 2020, 15, 243. [Google Scholar] [CrossRef] [PubMed]
- Ledda, R.E.; Milanese, G.; Gnetti, L.; Borghesi, A.; Sverzellati, N.; Silva, M. Spread through air spaces in lung adenocarcinoma: Is radiology reliable yet? J. Thorac. Dis. 2019, 11, S256–S261. [Google Scholar] [CrossRef] [PubMed]
- Onozato, Y.; Nakajima, T.; Yokota, H.; Morimoto, J.; Nishiyama, A.; Toyoda, T.; Inage, T.; Tanaka, K.; Sakairi, Y.; Suzuki, H.; et al. Radiomics is feasible for prediction of spread through air spaces in patients with nonsmall cell lung cancer. Sci. Rep. 2021, 11, 13526. [Google Scholar] [CrossRef] [PubMed]
| Studies | Selection (****) | Comparability (**) | Outcomes (***) | Total Score | |||||
|---|---|---|---|---|---|---|---|---|---|
| Representativeness of the Sample (*) | Sample Size (*) | Non-Respondents (*) | Ascertainment of the Exposure-Risk Factor (*) | Based on the Design (*) | Based on the Analysis (*) | Assessment of the Outcome (**) | Statistical-Test (*) | ||
| Kayı Cangır, A. [17], 2021 | * | * | * | * | * | 5 | |||
| Kirby, M. [18], 2020 | * | * | * | * | * | 5 | |||
| Tanabe, N. [19], 2020 | * | * | * | * | ** | 6 | |||
| Verleden, S. [20], 2020 | * | * | * | * | * | * | * | 7 | |
| Nakamura, S. [21], 2020 | * | * | * | * | * | * | 6 | ||
| Verleden, S. [22], 2020 | * | * | * | ** | 5 | ||||
| Norvik, C. [23], 2020 | * | * | * | * | 5 | ||||
| Umetani, K. [24], 2020 | * | * | ** | 4 | |||||
| Vasilescu, D. [25], 2020 | * | * | ** | 4 | |||||
| Everaerts, S. [26], 2019 | * | * | * | * | ** | * | 7 | ||
| Katsamenis, O. [27], 2019 | * | * | ** | 4 | |||||
| Troschel, F. [28], 2019 | * | * | * | ** | 5 | ||||
| Shelmerdine, S.C. [29], 2019 | * | * | * | * | 4 | ||||
| Vasilescu, D. [30], 2019 | * | * | * | ** | 5 | ||||
| Robinson, S.K. [31], 2019 | * | * | ** | * | 5 | ||||
| McDonough, J. [32], 2019 | * | * | * | *** | 6 | ||||
| Verleden, S. [33], 2019 | * | * | * | * | * | *** | 8 | ||
| Tanabe, N. [34], 2018 | * | * | * | ** | 5 | ||||
| Everaerts, S. [35], 2018 | * | * | * | * | ** | * | 7 | ||
| Verleden, S. [36], 2017 | * | * | * | * | ** | 6 | |||
| Suzuki, M. [37], 2017 | * | * | * | * | * | 5 | |||
| Tanabe, N. [38], 2017 | * | * | * | * | ** | 6 | |||
| Mai, C. [39], 2017 | * | * | * | ** | * | 7 | |||
| Vasilescu, D. [40], 2017 | * | * | * | ** | 5 | ||||
| Guan, C.S. [41], 2016 | * | * | * | * | 4 | ||||
| Jones, M. [42], 2016 | * | * | * | * | 4 | ||||
| Boon, M. [43], 2016 | * | * | * | * | ** | 6 | |||
| Scott, A. [44], 2015 | * | * | * | * | * | 5 | |||
| Verleden, S. [45], 2015 | * | * | * | * | ** | * | 7 | ||
| Zuo, Y.Z. [46], 2013 | * | * | * | ** | 5 | ||||
| Kampschulte, M. [47], 2013 | * | * | * | * | * | 5 | |||
| Okubo, Y. [48], 2013 | * | * | * | * | * | 5 | |||
| Campell, J. [49], 2012 | * | * | * | * | * | 5 | |||
| Litzlbauer, H. [50], 2010 | * | * | * | ** | 5 | ||||
| Hogg, J. [51], 2009 | * | * | * | ** | 5 | ||||
| Hogg, J. [52], 2009 | * | * | * | * | * | ** | * | 8 | |
| Watz, H. [53], 2005 | * | * | * | * | * | 5 | |||
| Main Author, Year, Country | Study Design | Main Outcome Assessed via Micro-CT | Number of Participants/Specimens (n: 74/105) | Cryo-MicroCT | Micro-CT Scanner |
|---|---|---|---|---|---|
| Verleden, S. [22], 2020, Belgium | Cs | Micro-CT was used to assess the number, length, and diameter of terminal bronchioles | 32/32 | No/not reported | Skyscan 1172 (Bruker, Kontich, Belgium) |
| Umetani, K. [24], 2020, USA | Cs | Micro-CT was used for whole secondary pulmonary lobule visualization | 1/1 | No/not reported | BL20B2, SPring-8 (Englewood, CO, USA) |
| Vasilescu, D. [25], 2020, Canada | Cs | Micro-CT was used as a part of a multi-resolution CT imaging in order to extract specific volumetric findings | 13/13 | Yes | XT H 225 (Nikon Metrology Inc, Brighton, MI, USA) |
| Katsamenis, O. [27], 2019, UK | Cs | Micro-CT was used to enable nondestructive 3D-X-ray histology, and to examine its use and benefits in the exemplar of human lung biopsy specimens | 2/2 | No/not reported | Med-X (Nikon X-Tek Systems Ltd.& Southampton, UK) |
| Vasilescu, D. [40], 2017, Canada | Cs | Micro-CT was used to image unfixed frozen human lung samples under conditions allowing the tissue to be afterwards used for immunohistochemistry | 1/1 | No/not reported | Nikon HMX-225 micro-CT scanner (Nikon Metrology, Tring, UK) |
| Guan, C.S. [41], 2016, China | Cs | Micro-CT was used to retrospectively evaluate short linear shadows connecting pulmonary segmental arteries to oblique fissures in thin-section CT images and determine their anatomical basis | 11/11 | No/not reported | Siemens micro-CT scanner (Siemens Medical Solutions, Knoxville, TN, USA) |
| Scott, A. [44], 2015, UK | Cs | Micro-CT was used to visualize, assess and analyze the 3D lung morphology | 4/4 | No/not reported | Nikon HMX-225 micro-CT scanner (Nikon Metrology, Tring, UK) |
| Zuo, Y.Z. [46], 2013, China | Cs | Micro-CT was used to describe the normal imaging appearance of pulmonary intersegmental planes compared to thoracic CT scans | 10/30 | No/not reported | SkyScan 1176 (Bruker, Aartselaar, Belgium) |
| Litzibauer, H. [50], 2010, Germany | Cs | High-resolution synchrotron-based micro-CT was used to generate a complete dataset of the intact three-dimensional architecture of the human acinus | 1/12 | No/not reported | X2B beamline, National Synchrotron Light Source (Brookhaven National Laboratories, Germany) |
| Main Author, Year, Country | Study Design | Main Outcome Assessed via Micro-CT | Number of Participants/Specimens (n: 120/903) | Cryo-MicroCT | Micro-CT Scanner |
|---|---|---|---|---|---|
| Kirby, M. [18], 2019, Canada | Cs | Micro-CT was used to estimate the number of terminal bronchioles and their association with total airway count (assessed by multidetector CT) | 22/133 | Yes | XT H 225 (Nikon Metrology Inc, Brighton, MI, USA) |
| Everaerts, S. [26], 2019, Belgium | Cs | Micro-CT was used to investigate and compare the airway generations between COPD lungs with and without bronchiectasis and unused donor lungs | 21/66 | Yes (in 60 specimens) | Skyscan 1172 (Bruker, Kontich, Belgium) |
| Vasilescu, D. [30], 2019, Canada | Cs | Micro-CT was used to assess terminal bronchioles in emphysema | 55/55 | Yes | XT H 225 (Nikon Metrology Inc, Brighton, MI, USA) |
| Tanabe, N. [34], 2018, Canada | Cs | Micro-CT was used to measure the mean linear intercept and the numbers of terminal bronchioles/mL lung in each tissue core | 15/15 | Yes | XT H 225 (Nikon Metrology Inc, Brighton, MI, USA) |
| Everaerts, S. [35], 2018, Belgium | Cs | Micro-CT was used to measure surface density and determine the extent of normal tissue within each sample of normal lungs and end-stage COPD lungs. | 24/280 | Yes | Skyscan 1172 (Bruker, Kontich, Belgium) |
| Suzuki, M. [37], 2017, USA | Cs | Micro-CT was used to measure the mean linear intercept and the numbers of terminal bronchioles/mL lung in each tissue core | 8/61 | Yes | eXplore Locus SP MicroCT scanner (GE Healthcare) |
| Tanabe, N. [38], 2017, Canada | Cs | Micro-CT was used to study small airways pathology in centrilobular and panlobular emphysema and show that these airway alterations are more visible with micro-CT scanners, rather than with thoracic multidetector computed tomography | 20/95 | No/not reported | Locus SP MicroCT (GE Healthcare, Chicago, IL, USA), Scanco MicroCT35 (Scanco Medical, Brüttisellen, Switzerland), MicroXCT-400 (Zeiss, Oberkochen, Germany), HMX 225ST (Nikon Metrology, Leuven, Belgium) |
| Kampschulte, M. [47], 2013, Germany | Cs | Micro-CT was used to obtain quantitative volumetric and morphologic information of changes in soft tissue, respiratory tracts and vascularization in fibrotic, emphysematous and non-diseased human lung specimens. | 32/32 | No/not reported | Not reported |
| Hogg, J. [52], 2009, Canada | Cs | Micro-CT was used to measure the number and lumen area of terminal bronchioles in COPD lungs | 52/530 | No/not reported | Micro-CT scanner (Biomedical Imaging Resource, Mayo Clinic, Rochester, MN, USA) |
| Hogg, J. [51], 2009, Canada | Cs | Micro-CT was used to examine bronchiolar remodelling and alveolar destruction in COPD | 8/8 | No/not reported | Not reported |
| Watz, H. [53], 2005, Germany | Cs | Micro-CT was used to investigate the appearance of human lung parenchyma at the structural level of alveoli in a patient with centrilobular emphysema | 1/12 | No/not reported | CT 20; Scanco Medical, Bassersdorf, Switzerland |
| Main Author, Year, Country | Study Design | Main Outcome Assessed via Micro-CT | Number of Participants/Specimens (n: 60/529) | Cryo-MicroCT | Micro-CT Scanner |
|---|---|---|---|---|---|
| Tanabe, N. [19], 2020, USA | Cs | Micro-CT was used to examine associations between histopathologic features of usual interstitial pneumonia and IPF in explanted lungs and to measure alveolar surface density, total lung volume taken up by tissue (%), and terminal bronchiolar number | 16/96 | No/not reported | Skyscan 1172 (Bruker, Kontich, Belgium) |
| Verleden, S. [20], 2020, Belgium | Cohort | Micro-CT was used to anatomically identify terminal bronchioles and count them per mL of lung tissue | 21/240 | Yes | Skyscan 1172 (Bruker, Kontich, Belgium) |
| McDonough, J. [32], 2019, USA | Cs | Micro-CT was used for the assessment of the extent of fibrosis in each sample via measuring alveolar surface density | 10/95 | Yes | Skyscan 1172 (Bruker, Kontich, Belgium) |
| Mai, C. [39], 2017, Belgium | Cs | Micro-CT was used to study underlying lung changes responsible for the CT features of IPF and to gain insight into the way IPF proceeds through the lungs and progresses over time | 9/94 | No/not reported | Skyscan 1172 (Bruker, Kontich, Belgium) |
| Jones, M. [42] 2016, UK | Cs | Micro-CT was used to characterize fibroblast foci morphology in lung specimens | 4/4 | No/Not reported | Nikon HMX-225 micro-CT scanner (Nikon Metrology, Tring, UK) |
| Kampschulte, M. [47], 2013, Germany | Cs | Micro-CT was used to obtain quantitative volumetric and morphologic information of changes in soft tissue, respiratory tracts and vascularization in fibrotic, emphysematous and non-diseased human lung specimens. | 22/22 | No/not reported | Not reported |
| Main Author, Year, Country | Study Design | Main Outcome Assessed via Micro-CT | Number of Participants/Specimens (n: 155/763) | Cryo-Micro-CT | Micro-CT Scanner |
|---|---|---|---|---|---|
| Kayı Cangır, A. [17], 2021, Turkey | Cs | Micro-CT was used to evaluate pulmonary adenocarcinoma specimens by comparing tumoral and non-tumoral areas and correlating micro-CT findings with hematoxylin and eosin sections. | 3/3 | No/not reported | Skyscan 1275 (Bruker, Kontich, Belgium) |
| Nakamura, S. [21], 2020, Japan | Cs | Micro-CT was used to distinguish areas of normal lung tissue and lung adenocarcinoma | 10/10 | No/not reported | InspeXio SMX-100CT (Shimadzu, Kyoto, Japan) |
| Norvik, C. [23], 2020, Sweden | Cs | Micro-CT was used to evaluate the micro-anatomy of normal lung tissue and microvascular anomalies of ACD/MPV (alveolar capillary dysplasia with misalignment of pulmonary veins) | 2/2 | No/not reported | X02DA TOMCAT beamline, Swiss Light Source (Villigen, Switzerland) |
| Shelmerdine, S.C. [29], 2019, UK | Cs | Micro-CT was used for post-mortem investigation of an excised stenotic infant trachea | 1/1 | No/not reported | Med-X Alpha (Nikon Metrology, Tring, UK) |
| Troschel, F. [28], 2019, USA | Cs | Micro-CT was used to peri-operatively evaluate fresh surgical lung resection specimens from patients with a presumptive diagnosis of lung cancer | 21/22 | No/not reported | Skyscan 1275 (Bruker, Kontich, Belgium)/XT H 225 (Nikon Metrology Inc, Brighton, MI, USA) |
| Robinson, S.K. [31], 2019, UK | Cs | Micro-CT was used to identify via mathematic modelling and Immunohistochemistry lymphatic heterogeneity within and between lung tissue | 2/4 | No/not reported | Nikon Metrology micro-CT scanner (Nikon Metrology, Tring Herts, UK) |
| Verleden, S. [33], 2019, Belgium | Cohort | Micro-CT findings were compared to 18F-FDG PET/CT scan findings in a patient with restrictive allograft syndrome undergoing redo transplantation | 1/1 | Yes | Skyscan 1172 (Bruker, Kontich, Belgium) |
| Everaerts, S. [35], 2018, Belgium | Cs | Micro-CT was used to measure surface density and determine the extent of normal tissue within each sample of normal lungs and lungs with end-stage cystic fibrosis, chronic hypersensitivity pneumonitis, bronchiolitis obliterans and restrictive allograft syndromes. | 46/280 | Yes | Skyscan 1172 (Bruker, Kontich, Belgium) |
| Verleden, S. [36], 2017, Belgium | Cs | Micro-CT was used to assess mass and density of donor lungs, aiding in decision-making to accept or decline lung allografts | 28/28 | Yes | Skyscan 1172 (Bruker, Kontich, Belgium) |
| Boon, M. [43], 2016, Netherlands | Cs | Micro-CT was used to quantify the involvement of small and large airways in end-stage cystic fibrosis | 18/167 | No/not reported | Not reported |
| Verleden, S. [45], 2015, Belgium | Cs | Micro-CT was used to evaluate lungs from patients with chronic lung allograft dysfunction | 24/246 | Yes | Skyscan 1172 (Bruker, Kontich, Belgium) |
| Okubo, Y. [48], 2013, Japan | Cs | Micro-CT was used to evaluate the pathophysiological implications of the reversed CT halo sign in a patient with invasive pulmonary mucormycosis and a patient with invasive pulmonary aspergillosis | 2/2 | No/not reported | InspeXio SMX-100CT (Shimadzu, Kyoto, Japan) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bompoti, A.; Papazoglou, A.S.; Moysidis, D.V.; Otountzidis, N.; Karagiannidis, E.; Stalikas, N.; Panteris, E.; Ganesh, V.; Sanctuary, T.; Arvanitidis, C.; et al. Volumetric Imaging of Lung Tissue at Micrometer Resolution: Clinical Applications of Micro-CT for the Diagnosis of Pulmonary Diseases. Diagnostics 2021, 11, 2075. https://doi.org/10.3390/diagnostics11112075
Bompoti A, Papazoglou AS, Moysidis DV, Otountzidis N, Karagiannidis E, Stalikas N, Panteris E, Ganesh V, Sanctuary T, Arvanitidis C, et al. Volumetric Imaging of Lung Tissue at Micrometer Resolution: Clinical Applications of Micro-CT for the Diagnosis of Pulmonary Diseases. Diagnostics. 2021; 11(11):2075. https://doi.org/10.3390/diagnostics11112075
Chicago/Turabian StyleBompoti, Andreana, Andreas S. Papazoglou, Dimitrios V. Moysidis, Nikolaos Otountzidis, Efstratios Karagiannidis, Nikolaos Stalikas, Eleftherios Panteris, Vijayakumar Ganesh, Thomas Sanctuary, Christos Arvanitidis, and et al. 2021. "Volumetric Imaging of Lung Tissue at Micrometer Resolution: Clinical Applications of Micro-CT for the Diagnosis of Pulmonary Diseases" Diagnostics 11, no. 11: 2075. https://doi.org/10.3390/diagnostics11112075
APA StyleBompoti, A., Papazoglou, A. S., Moysidis, D. V., Otountzidis, N., Karagiannidis, E., Stalikas, N., Panteris, E., Ganesh, V., Sanctuary, T., Arvanitidis, C., Sianos, G., Michaelson, J. S., & Herrmann, M. D. (2021). Volumetric Imaging of Lung Tissue at Micrometer Resolution: Clinical Applications of Micro-CT for the Diagnosis of Pulmonary Diseases. Diagnostics, 11(11), 2075. https://doi.org/10.3390/diagnostics11112075








