Diagnostic Use of Post-therapy 131I-Meta-Iodobenzylguanidine Scintigraphy in Consolidation Therapy for Children with High-Risk Neuroblastoma
Abstract
1. Introduction
2. Materials and Methods
2.1. Ethical Considerations and Registration
2.2. Inclusion Criteria
2.3. Treatment
2.4. Imaging
2.5. Statistical Analysis
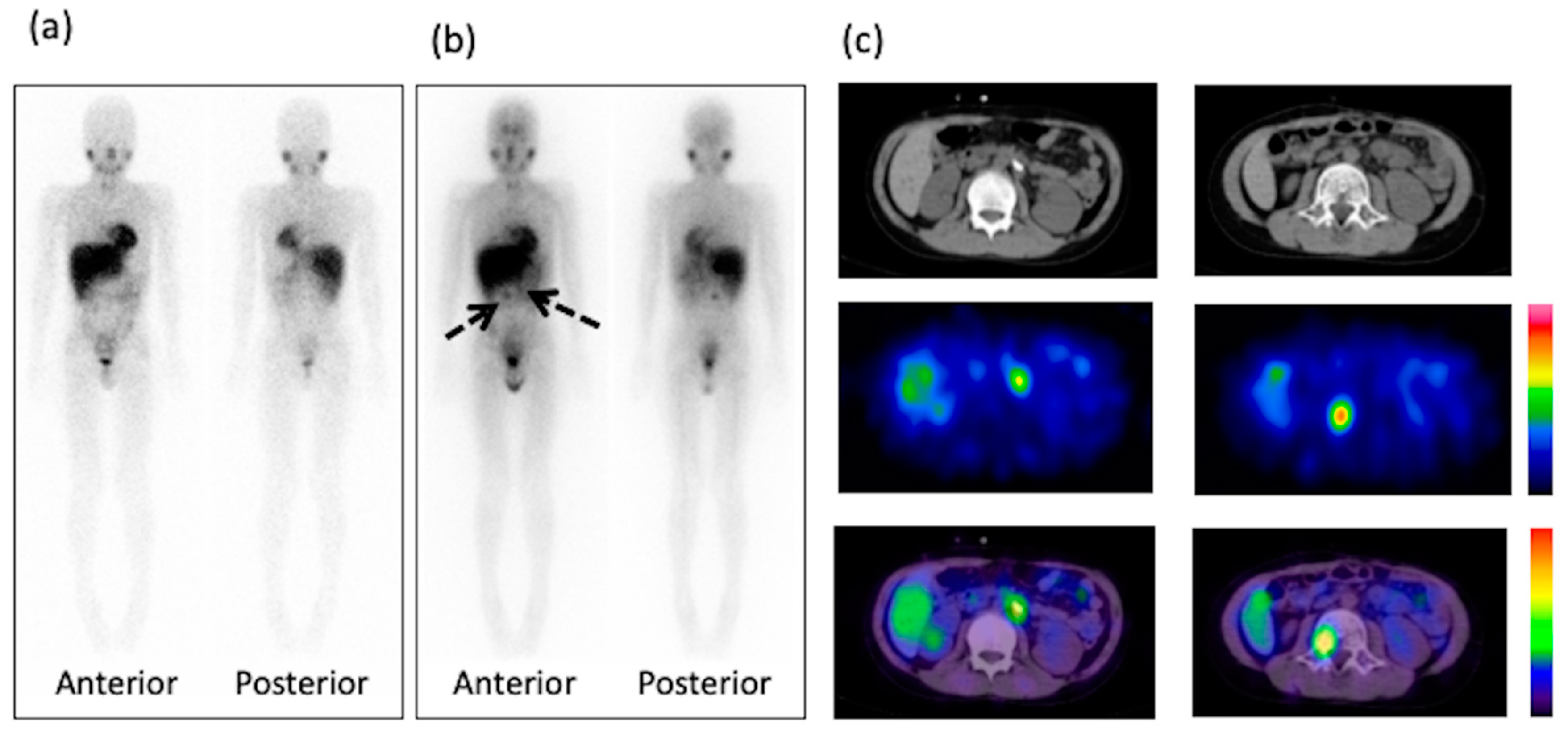
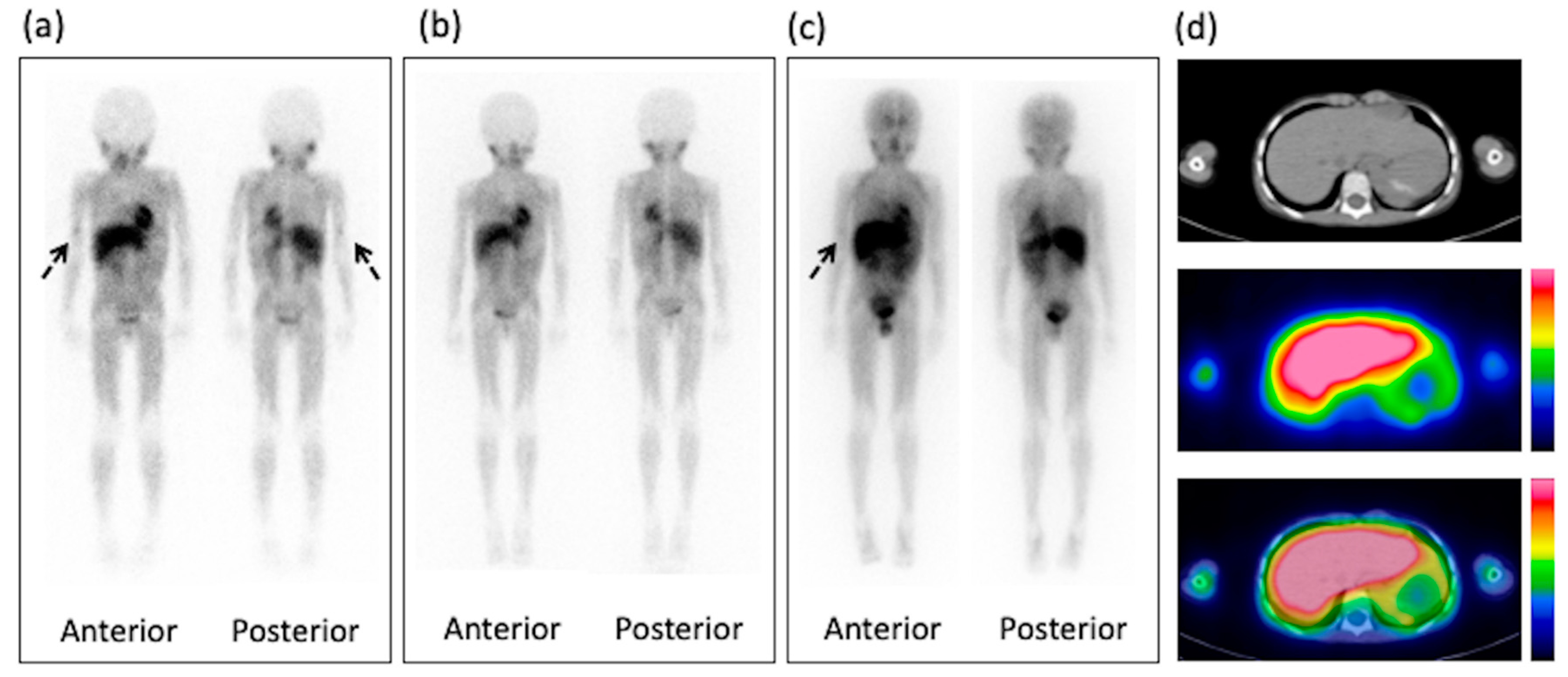
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Pfluger, T.; Piccardo, A. Neuroblastoma: MIBG Imaging and New Tracers. Semin. Nucl. Med. 2017, 47, 143–157. [Google Scholar] [CrossRef] [PubMed]
- Giammarile, F.; Chiti, A.; Lassmann, M.; Brans, B.; Flux, G.; EANM. EANM procedure guidelines for 131I-meta-iodobenzylguanidine (131I-mIBG) therapy. Eur. J. Nucl. Med. Mol. Imaging 2008, 35, 1039–1047. [Google Scholar] [CrossRef] [PubMed]
- Parisi, M.T.; Eslamy, H.; Park, J.R.; Shulkin, B.L.; Yanik, G.A. 131I-Metaiodobenzylguanidine Theranostics in Neuroblastoma: Historical Perspectives; Practical Applications. Semin. Nucl. Med. 2016, 46, 184–202. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.W.; Lee, S.; Cho, H.W.; Ma, Y.; Yoo, K.H.; Sung, K.W.; Koo, H.H.; Cho, E.J.; Lee, S.K.; Lim, D.H. Incorporation of high-dose 131I-metaiodobenzylguanidine treatment into tandem high-dose chemotherapy and autologous stem cell transplantation for high-risk neuroblastoma: Results of the SMC NB-2009 study. J. Hematol. Oncol. 2017, 10, 108. [Google Scholar] [CrossRef] [PubMed]
- Suh, J.K.; Koh, K.N.; Min, S.Y.; Kim, Y.S.; Kim, H.; Im, H.J.; Namgoong, J.M.; Kim, D.Y.; Ahn, S.D.; Lee, J.J.; et al. Feasibility and effectiveness of treatment strategy of tandem high-dose chemotherapy and autologous stem cell transplantation in combination with 131I-MIBG therapy for high-risk neuroblastoma. Pediatr. Transplant. 2020, 24, e13658. [Google Scholar] [CrossRef] [PubMed]
- Hishiki, T.; Matsumoto, K.; Ohira, M.; Kamijo, T.; Shichino, H.; Kuroda, T.; Yoneda, A.; Soejima, T.; Nakazawa, A.; Takimoto, T.; et al. Results of a phase II trial for high-risk neuroblastoma treatment protocol JN-H-07: A report from the Japan Childhood Cancer Group Neuroblastoma Committee (JNBSG). Int. J. Clin. Oncol. 2018, 23, 965–973. [Google Scholar] [CrossRef] [PubMed]
- Haas-Kogan, D.A.; Swift, P.S.; Selch, M.; Haase, G.M.; Seeger, R.C.; Gerbing, R.B.; Stram, D.O.; Matthay, K.K. Impact of radiotherapy for high-risk neuroblastoma: A Children’s Cancer Group study. Int. J. Radiat Oncol. Biol. Phys. 2003, 56, 28–39. [Google Scholar] [CrossRef]
- Panandiker, A.S.P.; McGregor, L.; Krasin, M.J.; Wu, S.J.; Xiong, X.P.; Merchant, T.E. Locoregional Tumor Progression after Radiation Therapy Influences Overall Survival in Pediatric Patients with Neuroblastoma. Int. J. Radiat. Oncol. 2010, 76, 1161–1165. [Google Scholar] [CrossRef] [PubMed]
- Giammarile, F.; Lumbroso, J.; Ricard, M.; Aubert, B.; Hartmann, O.; Schlumberger, M.; Parmentier, C. Radioiodinated metaiodobenzylguanidine in neuroblastoma: Influence of high dose on tumour site detection. Eur. J. Nucl. Med. 1995, 22, 1180–1183. [Google Scholar] [CrossRef] [PubMed]
- Kayano, D.; Taki, J.; Fukuoka, M.; Wakabayashi, H.; Inaki, A.; Nakamura, A.; Kinuya, S. Low-dose 123I-metaiodobenzylguanidine diagnostic scan is inferior to 131I-metaiodobenzylguanidine posttreatment scan in detection of malignant pheochromocytoma and paraganglioma. Nucl. Med. Commun. 2011, 32, 941–946. [Google Scholar] [CrossRef] [PubMed]
- Monsieurs, M.; Brans, B.; Bacher, K.; Dierckx, R.; Thierens, H. Patient dosimetry for 131I-MIBG therapy for neuroendocrine tumours based on 123I-MIBG scans. Eur. J. Nucl. Med. Mol. Imaging 2002, 29, 1581–1587. [Google Scholar] [CrossRef] [PubMed]
- Liu, B.; Zhuang, H.; Servaes, S. Comparison of [123I]MIBG and [131I]MIBG for imaging of neuroblastoma and other neural crest tumors. Q. J. Nucl. Med. Mol. Imaging 2013, 57, 21–28. [Google Scholar] [PubMed]
- Johnson, K.; McGlynn, B.; Saggio, J.; Baniewicz, D.; Zhuang, H.; Maris, J.M.; Mosse, Y.P. Safety and efficacy of tandem 131I-metaiodobenzylguanidine infusions in relapsed/refractory neuroblastoma. Pediatr. Blood Cancer 2011, 57, 1124–1129. [Google Scholar] [CrossRef] [PubMed]
- Fukuoka, M.; Taki, J.; Mochizuki, T.; Kinuya, S. Comparison of diagnostic value of I-123 MIBG and high-dose I-131 MIBG scintigraphy including incremental value of SPECT/CT over planar image in patients with malignant pheochromocytoma/paraganglioma and neuroblastoma. Clin. Nucl. Med. 2011, 36, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Kinuya, S.; Yoshinaga, K.; Higuchi, T.; Jinguji, M.; Kurihara, H.; Kawamoto, H. Draft guidelines regarding appropriate use of (131)I-MIBG radiotherapy for neuroendocrine tumors: Guideline Drafting Committee for Radiotherapy with (131)I-MIBG, Committee for Nuclear Oncology and Immunology, The Japanese Society of Nuclear Medicine. Ann. Nucl. Med. 2015, 29, 543–552. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Codreanu, I.; Servaes, S.; Zhuang, H. I-131 MIBG post-therapy scan is more sensitive than I-123 MIBG pretherapy scan in the evaluation of metastatic neuroblastoma. Nucl. Med. Commun. 2012, 33, 1134–1137. [Google Scholar] [CrossRef]
- Hickeson, M.P.; Charron, M.; Maris, J.M.; Brophy, P.; Kang, T.I.; Zhuang, H.; Khan, J.; Nevrotski, T. Biodistribution of post-therapeutic versus diagnostic (131)I-MIBG scans in children with neuroblastoma. Pediatr. Blood Cancer 2004, 42, 268–274. [Google Scholar] [CrossRef] [PubMed]
- Nakagawara, A.; Li, Y.; Izumi, H.; Muramori, K.; Inada, H.; Nishi, M. Neuroblastoma. Jpn. J. Clin. Oncol. 2018, 48, 214–241. [Google Scholar] [CrossRef] [PubMed]
- Mazloom, A.; Louis, C.U.; Nuchtern, J.; Kim, E.; Russell, H.; Allen-Rhoades, W.; Krance, R.; Paulino, A.C. Radiation therapy to the primary and postinduction chemotherapy MIBG-avid sites in high-risk neuroblastoma. Int. J. Radiat. Oncol. Biol. Phys. 2014, 90, 858–862. [Google Scholar] [CrossRef] [PubMed]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wakabayashi, H.; Kayano, D.; Inaki, A.; Araki, R.; Kuroda, R.; Akatani, N.; Yamase, T.; Watanabe, S.; Hiromasa, T.; Kunita, Y.; et al. Diagnostic Use of Post-therapy 131I-Meta-Iodobenzylguanidine Scintigraphy in Consolidation Therapy for Children with High-Risk Neuroblastoma. Diagnostics 2020, 10, 663. https://doi.org/10.3390/diagnostics10090663
Wakabayashi H, Kayano D, Inaki A, Araki R, Kuroda R, Akatani N, Yamase T, Watanabe S, Hiromasa T, Kunita Y, et al. Diagnostic Use of Post-therapy 131I-Meta-Iodobenzylguanidine Scintigraphy in Consolidation Therapy for Children with High-Risk Neuroblastoma. Diagnostics. 2020; 10(9):663. https://doi.org/10.3390/diagnostics10090663
Chicago/Turabian StyleWakabayashi, Hiroshi, Daiki Kayano, Anri Inaki, Raita Araki, Rie Kuroda, Norihito Akatani, Takafumi Yamase, Satoru Watanabe, Tomo Hiromasa, Yuji Kunita, and et al. 2020. "Diagnostic Use of Post-therapy 131I-Meta-Iodobenzylguanidine Scintigraphy in Consolidation Therapy for Children with High-Risk Neuroblastoma" Diagnostics 10, no. 9: 663. https://doi.org/10.3390/diagnostics10090663
APA StyleWakabayashi, H., Kayano, D., Inaki, A., Araki, R., Kuroda, R., Akatani, N., Yamase, T., Watanabe, S., Hiromasa, T., Kunita, Y., Mori, H., Saito, S., Ikawa, Y., Fujiki, T., & Kinuya, S. (2020). Diagnostic Use of Post-therapy 131I-Meta-Iodobenzylguanidine Scintigraphy in Consolidation Therapy for Children with High-Risk Neuroblastoma. Diagnostics, 10(9), 663. https://doi.org/10.3390/diagnostics10090663




