Does the Keele STarT Back Screening Tool Contribute to Effectiveness in Treatment and Cost and Loss of Follow-Up of the Mechanical Diagnosis and Therapy for Patients with Low Back Pain?
Abstract
1. Introduction
2. Materials and Methods
2.1. Design
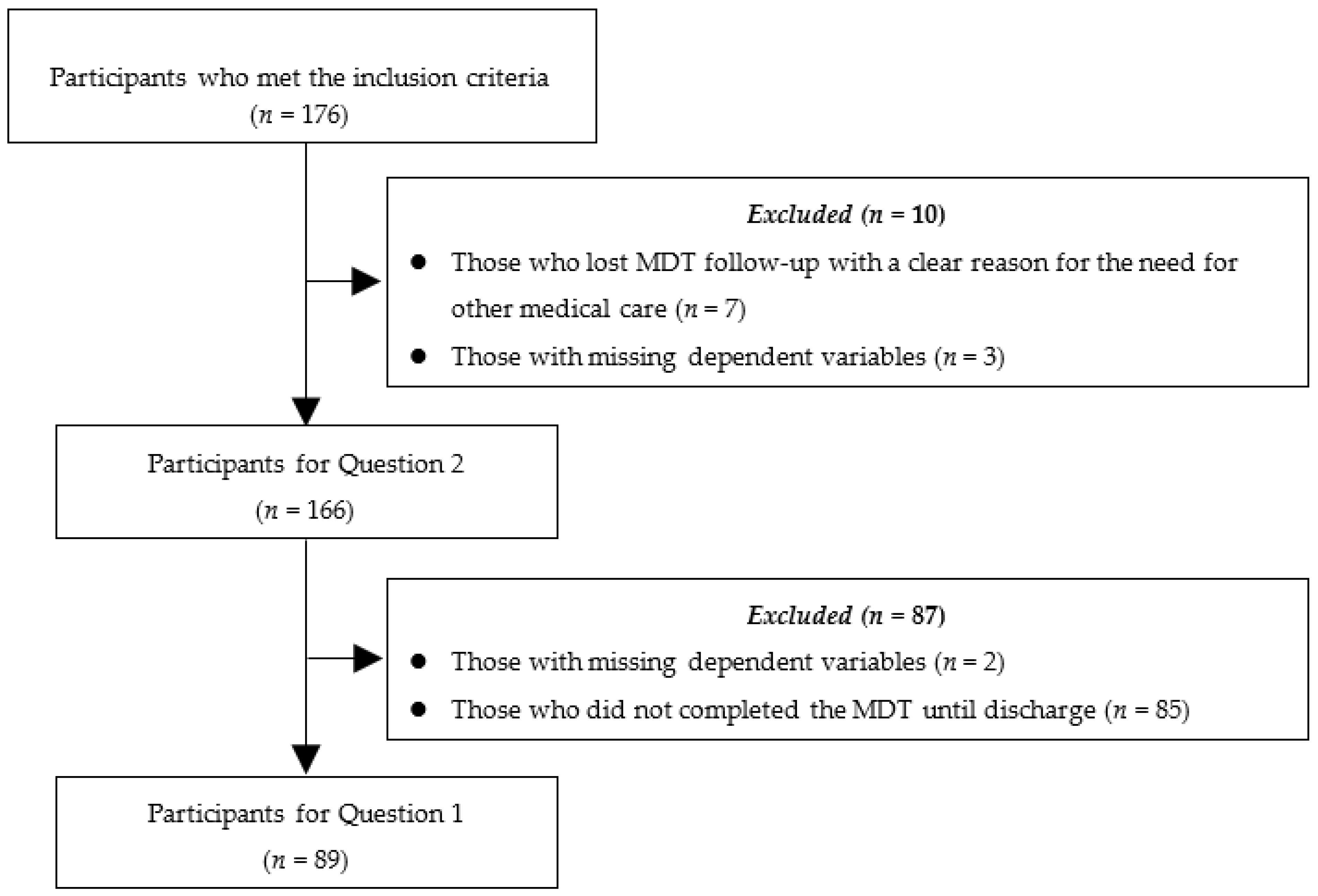
2.2. Participants
2.3. Exposure
2.4. Outcomes
2.4.1. Outcomes for Question 1
2.4.2. Outcomes for Question 2
2.5. Statistics
3. Results
3.1. Question 1
3.2. Question 2
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Foster, N.E.; Hill, J.C.; O’Sullivan, P.; Hancock, M. Stratified models of care. Best Pract. Res. Clin. Rheumatol. 2013, 27, 649–661. [Google Scholar] [CrossRef] [PubMed]
- Costa Lda, C.; Koes, B.W.; Pransky, G.; Borkan, J.; Maher, C.G.; Smeets, R.J. Primary care research priorities in low back pain: An update. Spine 2013, 38, 148–156. [Google Scholar] [CrossRef] [PubMed]
- Borkan, J.M.; Cherkin, D.C. An agenda for primary care research on low back pain. Spine 1996, 21, 2880–2884. [Google Scholar] [CrossRef] [PubMed]
- Halliday, M.H.; Garcia, A.N.; Amorim, A.B.; Machado, G.C.; Hayden, J.A.; Pappas, E.; Ferreira, P.H.; Hancock, M.J. Treatment effect sizes of Mechanical Diagnosis and Therapy for pain and disability in patients with low back pain: A systematic review. J. Orthop. Sports Phys. Ther. 2019, 49, 219–229. [Google Scholar] [CrossRef]
- Manca, A.; Dumville, J.C.; Torgerson, D.J.; Klaber Moffett, J.A.; Mooney, M.P.; Jackson, D.A.; Eaton, S. Randomized trial of two physiotherapy interventions for primary care back and neck pain patients: Cost effectiveness analysis. Rheumatology 2007, 46, 1495–1501. [Google Scholar] [CrossRef]
- Deutscher, D.; Werneke, M.W.; Gottlieb, D.; Fritz, J.M.; Resnik, L. Physical therapists’ level of McKenzie education, functional outcomes, and utilization in patients with low back pain. J. Orthop. Sports Phys. Ther. 2014, 44, 925–936. [Google Scholar] [CrossRef]
- Donelson, R.; Spratt, K.; McClellan, W.S.; Gray, R.; Miller, J.M.; Gatmaitan, E. The cost impact of a quality-assured mechanical assessment in primary low back pain care. J. Man. Manip. Ther. 2019, 27, 277–286. [Google Scholar] [CrossRef]
- Foster, N.E.; Thompson, K.A.; Baxter, G.D.; Allen, J.M. Management of nonspecific low back pain by physiotherapists in Britain and Ireland. A descriptive questionnaire of current clinical practice. Spine 1999, 24, 1332–1342. [Google Scholar] [CrossRef]
- Ladeira, C.E.; Samuel Cheng, M.; Hill, C.J. Physical therapists’ treatment choices for non-specific low back pain in Florida: An electronic survey. J. Man. Manip. Ther. 2015, 23, 109–118. [Google Scholar] [CrossRef]
- Battie, M.C.; Cherkin, D.C.; Dunn, R.; Ciol, M.A.; Wheeler, K.J. Managing low back pain: Attitudes and treatment preferences of physical therapists. Phys. Ther. 1994, 74, 219–226. [Google Scholar] [CrossRef]
- Davies, C.; Nitz, A.J.; Mattacola, C.G.; Kitzman, P.; Howell, D.; Viele, K.; Baxter, D.; Brockopp, D. Practice patterns when treating patients with low back pain: A survey of physical therapists. Physiother. Theory Pract. 2014, 30, 399–408. [Google Scholar] [CrossRef] [PubMed]
- Takasaki, H. Mechanical Diagnosis and Therapy enhances attitude towards self-management in people with musculoskeletal disorders: A preliminary evidence with a before-after design. SAGE Open Med. 2017, 5, 2050312117740986. [Google Scholar] [CrossRef] [PubMed]
- Takasaki, H.; Saiki, T.; Iwasada, Y. McKenzie therapists adhere more to evidence-based guidelines and have a more biopsychosocial perspective on the management of patients with low back pain than general physical therapists in Japan. Open J. Ther. Rehabil. 2014, 2, 173–181. [Google Scholar] [CrossRef]
- May, S.; Donelson, R. McKenzie Method. In Evidence-Based Management of Low Back Pain; Dagenais, S., Haldeman, S., Eds.; Mosby, Inc.: St. Louis, MO, USA, 2012; pp. 122–133. [Google Scholar]
- Edmond, S.L.; Werneke, M.W.; Hart, D.L. Association between centralization, depression, somatization, and disability among patients with nonspecific low back pain. J. Orthop. Sports Phys. Ther. 2010, 40, 801–810. [Google Scholar] [CrossRef]
- Lam, O.T.; Strenger, D.M.; Chan-Fee, M.; Pham, P.T.; Preuss, R.A.; Robbins, S.M. Effectiveness of the McKenzie Method of Mechanical Diagnosis and Therapy for treating low back pain: Literature review with meta-analysis. J. Orthop. Sports Phys. Ther. 2018, 48, 476–490. [Google Scholar] [CrossRef]
- Cruz, E.B.; Canhão, H.; Fernandes, R.; Caeiro, C.; Branco, J.C.; Rodrigues, A.M.; Pimentel-Santos, F.; Gomes, L.A.; Paiva, S.; Pinto, I.; et al. Prognostic indicators for poor outcomes in low back pain patients consulted in primary care. PLoS ONE 2020, 15, e0229265. [Google Scholar] [CrossRef]
- Ramond, A.; Bouton, C.; Richard, I.; Roquelaure, Y.; Baufreton, C.; Legrand, E.; Huez, J.F. Psychosocial risk factors for chronic low back pain in primary care—A systematic review. Fam. Pract. 2011, 28, 12–21. [Google Scholar] [CrossRef]
- Garcia, A.N.; Costa, L.; de Souza, F.S.; de Almeida, M.O.; Araujo, A.C.; Hancock, M.; Costa, L.O.P. Reliability of the Mechanical Diagnosis and Therapy system in patients with spinal pain: A systematic review. J. Orthop. Sports Phys. Ther. 2018, 48, 923–933. [Google Scholar] [CrossRef]
- Hefford, C. McKenzie classification of mechanical spinal pain: Profile of syndromes and directions of preference. Man. Ther. 2008, 13, 75–81. [Google Scholar] [CrossRef]
- Yarznbowicz, R.; Tao, M.; Owens, A.; Wlodarski, M.; Dolutan, J. Pain pattern classification and directional preference are associated with clinical outcomes for patients with low back pain. J. Man. Manip. Ther. 2018, 26, 18–24. [Google Scholar] [CrossRef]
- Yarznbowicz, R.; Tao, M. Directional preference constructs for patients’ low back pain in the absence of centralization. J. Man. Manip. Ther. 2018, 26, 281–291. [Google Scholar] [CrossRef] [PubMed]
- May, S. Classification by McKenzie Mechanical Syndromes: A survey of McKenzie-trained faculty. J. Manip. Physiol. Ther. 2006, 29, 637–642. [Google Scholar] [CrossRef] [PubMed]
- Werneke, M.W.; Edmond, S.; Deutscher, D.; Ward, J.; Grigsby, D.; Young, M.; McGill, T.; McClenahan, B.; Weinberg, J.; Davidow, A.L. Effect of adding McKenzie syndrome, centralization, directional preference, and psychosocial classification variables to a risk-adjusted model predicting functional status outcomes for patients with lumbar impairments. J. Orthop. Sports Phys. Ther. 2016, 46, 726–741. [Google Scholar] [CrossRef] [PubMed]
- Delitto, A.; George, S.Z.; Van Dillen, L.R.; Whitman, J.M.; Sowa, G.; Shekelle, P.; Denninger, T.R.; Godges, J.J. Orthopaedic Section of the American Physical Therapy, A. Low back pain. J. Orthop. Sports Phys. Ther. 2012, 42, A1-57. [Google Scholar] [CrossRef] [PubMed]
- Hill, J.C.; Dunn, K.M.; Lewis, M.; Mullis, R.; Main, C.J.; Foster, N.E.; Hay, E.M. A primary care back pain screening tool: Identifying patient subgroups for initial treatment. Arthritis Rheum. 2008, 59, 632–641. [Google Scholar] [CrossRef]
- Medeiros, F.C.; Costa, L.O.P.; Oliveira, I.S.; Costa, L. A responsiveness analysis of the Subgroups for Targeted Treatment (STarT) Back Screening Tool in patients with nonspecific low back pain. J. Orthop. Sports Phys. Ther. 2019, 49, 725–735. [Google Scholar] [CrossRef]
- Hill, J.C.; Whitehurst, D.G.; Lewis, M.; Bryan, S.; Dunn, K.M.; Foster, N.E.; Konstantinou, K.; Main, C.J.; Mason, E.; Somerville, S.; et al. Comparison of stratified primary care management for low back pain with current best practice (STarT Back): A randomised controlled trial. Lancet 2011, 378, 1560–1571. [Google Scholar] [CrossRef]
- Takasaki, H.; Gabel, C.P. Cross-cultural adaptation of the 12-item Örebro musculoskeletal screening questionnaire to Japanese (ÖMSQ-12-J), reliability and clinicians’ impressions for practicality. J. Phys. Ther. Sci. 2017, 29, 1409–1415. [Google Scholar] [CrossRef][Green Version]
- Miki, T.; Kondo, Y.; Takebayashi, T.; Takasaki, H. Difference between physical therapist estimation and psychological patient-reported outcome measures in patients with low back pain. PLoS ONE 2020, 15, e0227999. [Google Scholar] [CrossRef]
- McKenzie, R.; May, S. The Lumbar Spine: Mechanical Diagnosis and Therapy, 2nd ed.; Spinal Publications New Zealand Ltd.: Waikenae, New Zealand, 2003. [Google Scholar]
- Hashimoto, S.; Hirokado, M.; Takasaki, H. The most common classification in the mechanical diagnosis and therapy for patients with a primary complaint of non-acute knee pain was Spinal Derangement: A retrospective chart review. J. Man. Manip. Ther. 2019, 27, 33–42. [Google Scholar] [CrossRef]
- Shand, L.; Higgins, R.; Murphy, B.; Jackson, A. Development and validation of the healthcare providers patient-activation scale. Patient Educ. Couns. 2019, 102, 1550–1557. [Google Scholar] [CrossRef]
- Matsudaira, K.; Oka, H.; Kikuchi, N.; Haga, Y.; Sawada, T.; Tanaka, S. Psychometric properties of the Japanese version of the STarT Back Tool in patients with low back pain. PLoS ONE 2016, 11, e0152019. [Google Scholar] [CrossRef]
- Matsudaira, K.; Kikuchi, N.; Kawaguchi, M.; Inuzuka, K.; Arisaka, M.; Hara, N.; Isomura, T. Development of a Japanese version of the STarT (Subgrouping for Targeted Treatment) Back screening tool: Translation and linguistic validation. J. Musculoskelet. Pain Res. 2013, 5, 11–19. [Google Scholar]
- Takasaki, H.; Aoki, S.; May, S. No increase in 6-week treatment effect of Mechanical Diagnosis and Therapy with the use of the LUMOback in people with non-acute non-specific low back pain and a directional preference of extension: A pilot randomized controlled trial. Physiotherapy 2018, 104, 347–353. [Google Scholar] [CrossRef]
- Werneke, M.W.; Hart, D.L.; Resnik, L.; Stratford, P.W.; Reyes, A. Centralization: Prevalence and effect on treatment outcomes using a standardized operational definition and measurement method. J. Orthop. Sports Phys. Ther. 2008, 38, 116–125. [Google Scholar] [CrossRef]
- Stanton, T.R.; Latimer, J.; Maher, C.G.; Hancock, M. Definitions of recurrence of an episode of low back pain: A systematic review. Spine 2009, 34, E316–E322. [Google Scholar] [CrossRef]
- Nakamura, M.; Miyamoto, K.; Shimizu, K. Validation of the Japanese version of the Roland-Morris Disability Questionnaire for Japanese patients with lumbar spinal diseases. Spine 2003, 28, 2414–2418. [Google Scholar] [CrossRef]
- Moore, D.S.; Notz, W.; Fligner, M.A. The Basic Practice of Statistics; W.H. Freeman and Company: New York, NY, USA, 2013. [Google Scholar]
- Common Miscomceptions of the McKenzie Method® of Mechanical Diagnosis and Therapy®—MDT. Available online: https://www.mckenzieinstituteaustralia.org/clinicians/common-misconceptions/ (accessed on 30 May 2020).
- Takasaki, H.; Miki, T.; Hall, T. Development of the Working Alliance Inventory-Short Form Japanese version through factor analysis and test–retest reliability. Physiother. Theory Pract. 2020, 36, 444–449. [Google Scholar] [CrossRef]
- Takasaki, H.; Hall, T. A Japanese version of the Patient Attitudes and Beliefs Scale for patients with musculoskeletal disorders. Physiother. Theory Pract. 2019, 1–9. [Google Scholar] [CrossRef]
- Kawazoe, S.; Takasaki, H. Development of a preliminary draft for the Exercise Adherence Rating Scale in Japanese. In Proceedings of the 24th Conference of the Society of Japanese Manual Physical Therapy, Fukuoka, Japan, 17 March 2019. [Google Scholar]
- May, S.; Runge, N.; Aina, A. Centralization and directional preference: An updated systematic review with synthesis of previous evidence. Musculoskelet. Sci. Pract. 2018, 38, 53–62. [Google Scholar] [CrossRef]
- Takasaki, H.; May, S. Mechanical diagnosis and therapy has similar effects on pain and disability as ‘wait and see’ and other approaches in people with neck pain: A systematic review. J. Physiother. 2014, 60, 78–84. [Google Scholar] [CrossRef]
| Variables | Question 1 (n = 89) | Question 2 (n = 166) |
|---|---|---|
| Age, years | 61.5 (13.8) | 61.6 (14.7) |
| Gender (n of male:n of female) | 37:52 | 65:101 |
| The shortest distance from the patient address to the hospital, km | 7.6 (6.6) | 7.5 (6.6) |
| Symptom area, 0–11 | 3.8 (1.8) | 3.7 (1.8) |
| Duration of the current episode of LBP, months | 7.7 (17.1) | 12.0 (30.7) |
| The number of medical diagnoses | 2.4 (1.7) | 2.7 (1.9) |
| RMDQ, 0–24 | 8.7 (5.6) | 8.8 (5.3) |
| NRS-LBP, 0–10 | 4.8 (2.6) | 4.9 (2.6) |
| Model | Unstandardized Coefficients (B) | Standardized Coefficients (β) | p-Value (95% Confidence Intervals) |
|---|---|---|---|
| (Constant) | −15.38 | 0.60 (−73.90–43.14) | |
| MDT subgroup | 106.41 | 0.43 | <0.001 (59.24–153.58) |
| Model | Unstandardized Coefficients (B) | Standardized Coefficients (β) | p-Value (95% Confidence Intervals) |
|---|---|---|---|
| (Constant) | −0.41 | 0.83 (−4.13–3.31) | |
| MDT subgroup | 3.85 | 0.35 | 0.001 (1.70–6.00) |
| SBST | 1.89 | 0.22 | 0.026 (0.23–3.55) |
| Model | Partial Regression Coefficient (B) | p-Value | Odd Ratio (95% Confidence Intervals) |
|---|---|---|---|
| SBST | 1.53 | <0.001 | 4.61 (2.13–10.00) |
| Distance | −1.59 | 0.03 | 0.20 (0.05–0.88) |
| (Constant) | −4.13 | <0.001 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tsuge, T.; Takasaki, H.; Toda, M. Does the Keele STarT Back Screening Tool Contribute to Effectiveness in Treatment and Cost and Loss of Follow-Up of the Mechanical Diagnosis and Therapy for Patients with Low Back Pain? Diagnostics 2020, 10, 536. https://doi.org/10.3390/diagnostics10080536
Tsuge T, Takasaki H, Toda M. Does the Keele STarT Back Screening Tool Contribute to Effectiveness in Treatment and Cost and Loss of Follow-Up of the Mechanical Diagnosis and Therapy for Patients with Low Back Pain? Diagnostics. 2020; 10(8):536. https://doi.org/10.3390/diagnostics10080536
Chicago/Turabian StyleTsuge, Takahiro, Hiroshi Takasaki, and Michio Toda. 2020. "Does the Keele STarT Back Screening Tool Contribute to Effectiveness in Treatment and Cost and Loss of Follow-Up of the Mechanical Diagnosis and Therapy for Patients with Low Back Pain?" Diagnostics 10, no. 8: 536. https://doi.org/10.3390/diagnostics10080536
APA StyleTsuge, T., Takasaki, H., & Toda, M. (2020). Does the Keele STarT Back Screening Tool Contribute to Effectiveness in Treatment and Cost and Loss of Follow-Up of the Mechanical Diagnosis and Therapy for Patients with Low Back Pain? Diagnostics, 10(8), 536. https://doi.org/10.3390/diagnostics10080536





