Changes in the 3D Corneal Structure and Morphogeometric Properties in Keratoconus after Corneal Collagen Crosslinking
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Population
2.2. Surgical Procedure
2.3. Examinations and Measurements
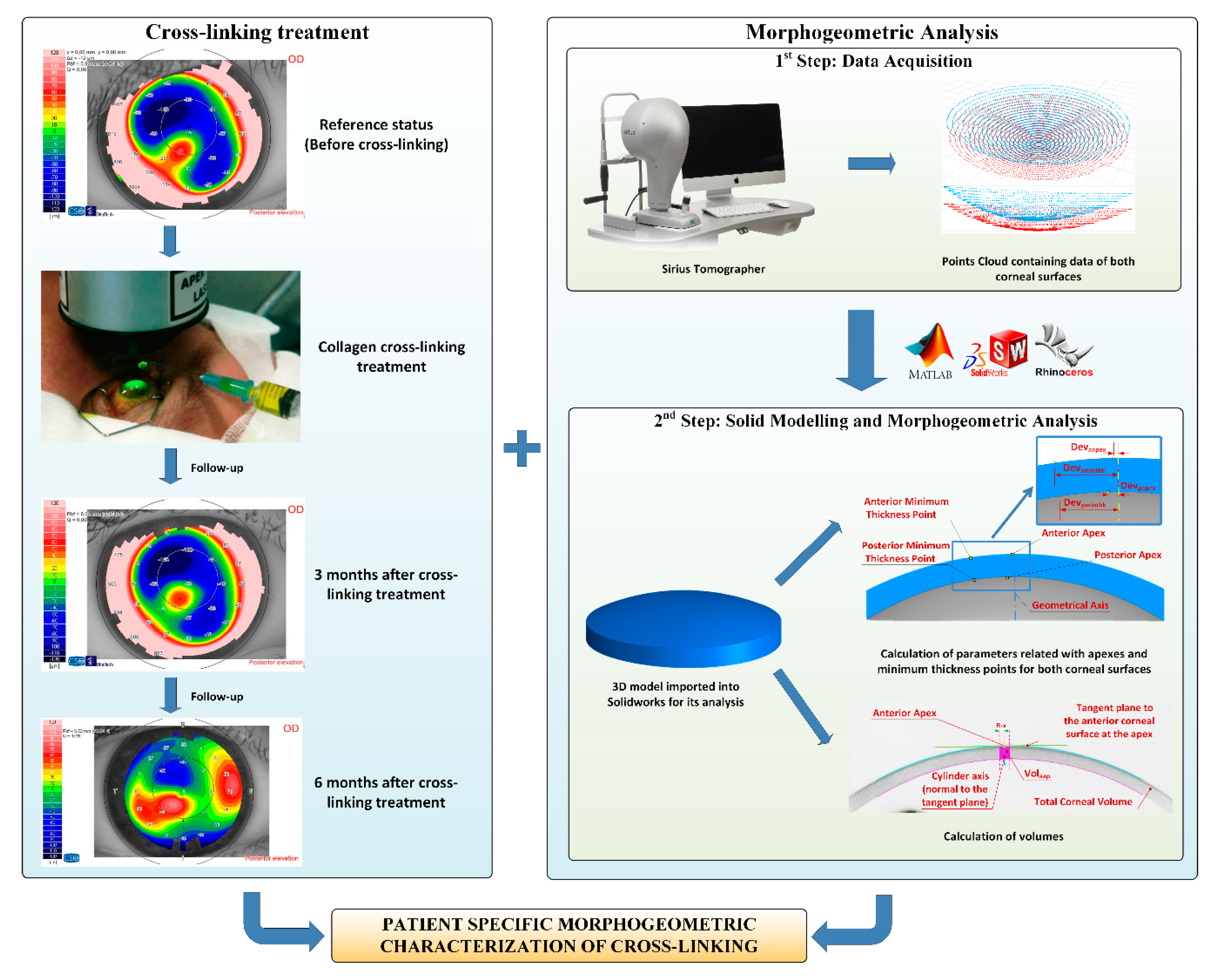
2.4. Morphogeometric Analysis
- Data Acquisition. A custom-made script programmed in the Matlab R2017b (Mathworks, Natick, USA) software was created to transform each point of the cloud provided by the tomographer in polar coordinates into the Cartesian format. This procedure has been meticulously described in several previous research works [8,9,10,11,12,13,14,15]. The program provides a. CSV file containing data of the points of clouds that describe both the anterior and posterior corneal surfaces for the area comprised between the geometric center of the cornea (r = 0 mm) and the mid-peripheral area (r = 4 mm). This zone usually contains most corneal anomalies for not only diseased, but also healthy eyes [8,9,10,11,12,13,14,15]. Afterward, both surfaces were imported to the Rhinoceros V 5.0 (MCNeel and Associates, Seattle, USA) software, and the “patch” function was used to find the surfaces that better fitted the clouds of points. The fixed configuration settings for the function were:
- •
- Sample point spacing: 256;
- •
- Surface span planes: 255 for both directions “u” and “v”;
- •
- Stiffness of the solution surface: 1.
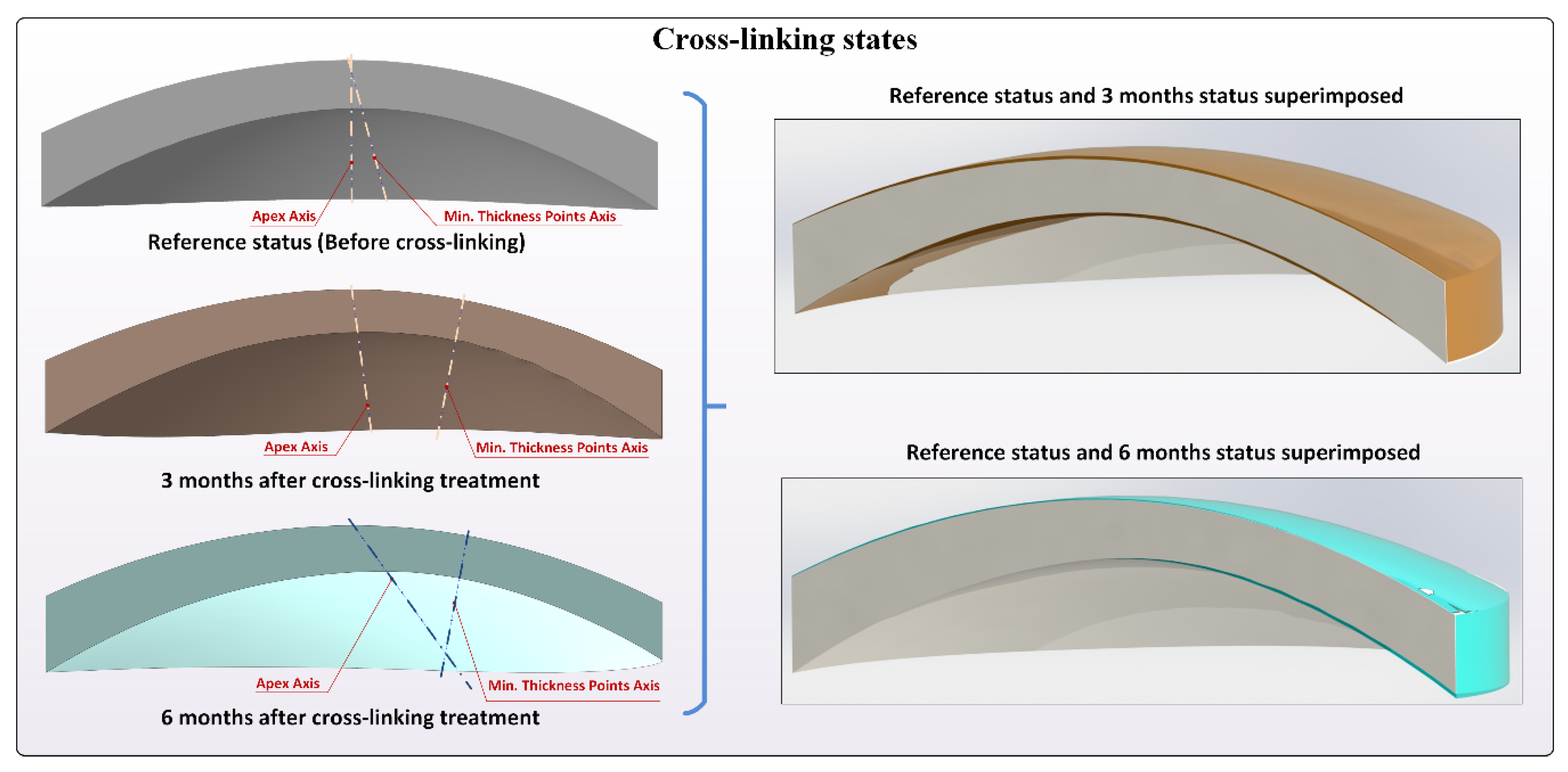
- Solid modeling and morphogeometric analysis. Finally, a customized three-dimensional model of the patient’s cornea was created from the surfaces previously generated in Rhinoceros by importing them to the SolidWorks V2018 (Dassault Systèmes, Vélizy-Villacoublay, France) software [8,9,10,11,12,13,14,15]. This model was then analyzed, and several morphogeometrical parameters were established. These parameters herein studied, as well as their particularities, have been previously described in several other articles [8,9,10,11,12,13,14,15]. This was also the case of several volumetric parameters related directly to the volumes around the anterior/posterior apices and the minimum thickness points [12].
- Aant: area of the anterior corneal surface [9].
- Apost: area of the posterior corneal surface [9].
- Atot: total area of the corneal surface [9].
- CV: total cornea volume [9].
- AAD: anterior apex deviation, which is the minimum mean distance between the point of maximum curvature (apex) of the anterior corneal surface and the z-axis [11].
- PAD: posterior apex deviation, which is the minimum mean distance between the point of maximum curvature of the posterior corneal surface and the z-axis [11].
2.5. Statistical Analysis
3. Results
4. Discussion
Author Contributions
Funding
Conflicts of Interest
References
- Arnalich-Montiel, F.; Alió Del Barrio, J.L.; Alió, J.L. Corneal surgery in keratoconus: Which type, which technique, which outcomes? Eye Vis. (Lond.) 2016, 3, 2. [Google Scholar] [CrossRef]
- Jhanji, V.; Sharma, N.; Vajpayee, R.B. Management of keratoconus: Current scenario. Br. J. Ophthalmol. 2010, 95, 1044–1050. [Google Scholar] [CrossRef] [PubMed]
- Coster, D.J.; Lowe, M.T.; Keane, M.C.; Williams, K.A. A Comparison of Lamellar and Penetrating Keratoplasty Outcomes. Ophthalmology 2014, 121, 979–987. [Google Scholar] [CrossRef]
- O’Brart, D.P.S.; Patel, P.; Lascaratos, G.; Wagh, V.K.; Tam, C.; Lee, J.; O’Brart, N.A. Corneal Cross-linking to Halt the Progression of Keratoconus and Corneal Ectasia: Seven-Year Follow-up. Am. J. Ophthalmol. 2015, 160, 1154–1163. [Google Scholar] [CrossRef] [PubMed]
- Seiler, T.; Spoerl, E.; Huhle, M.; Kamouna, A. Conservative therapy of keratoconus by enhancement of collagen cross-links. Investig. Ophthalmol. Vis. Sci. 1996, 37, 4671. [Google Scholar]
- O’Brart, D. Corneal Collagen Cross-Linking for Corneal Ectasias. In Keratoconus; Springer International Publishing: Cham, Switzerland, 2016; pp. 219–238. [Google Scholar] [CrossRef]
- Martínez-Abad, A.; Piñero, D.P. New perspectives on the detection and progression of keratoconus. J. Cataract Refract. Surg. 2017, 43, 1213–1227. [Google Scholar] [CrossRef]
- Cavas-Martínez, F.; Fernández-Pacheco, D.G.; De la Cruz-Sánchez, E.; Nieto Martínez, J.; Fernández Cañavate, F.J.; Vega-Estrada, A.; Plaza-Puche, A.B.; Alió, J.L. Geometrical custom modeling of human cornea in vivo and its use for the diagnosis of corneal ectasia. PLoS ONE 2014, 9, e110249. [Google Scholar] [CrossRef]
- Velázquez, J.S.; Cavas, F.; Piñero, D.P.; Cañavate, F.J.F.; Alio Del Barrio, J.; Alio, J.L. Morphogeometric analysis for characterization of keratoconus considering the spatial localization and projection of apex and minimum corneal thickness point. J. Adv. Res 2020, 24, 261–271. [Google Scholar] [CrossRef]
- Velázquez, J.S.; Cavas, F.; Alió Del Barrio, J.; Fernández-Pacheco, D.G.; Alió, J. Assessment of the Association between In Vivo Corneal Morphogeometrical Changes and Keratoconus Eyes with Severe Visual Limitation. J. Ophthalmol. 2019, 2019, 8731626. [Google Scholar] [CrossRef]
- Cavas-Martínez, F.; Fernández-Pacheco, D.G.; Parras, D.; Cañavate, F.J.F.; Bataille, L.; Alió, J. Study and characterization of morphogeometric parameters to assist diagnosis of keratoconus. Biomed. Eng. Online 2018, 17, 161. [Google Scholar] [CrossRef]
- Cavas-Martínez, F.; Bataille, L.; Fernández-Pacheco, D.G.; Cañavate, F.J.F.; Alio, J.L. Keratoconus Detection Based on a New Corneal Volumetric Analysis. Sci. Rep. 2017, 7, 15837. [Google Scholar] [CrossRef]
- Cavas-Martínez, F.; Bataille, L.; Fernández-Pacheco, D.G.; Cañavate, F.J.F.; Alió, J.L. A new approach to keratoconus detection based on corneal morphogeometric analysis. PLoS ONE 2017, 12, e0184569. [Google Scholar] [CrossRef] [PubMed]
- Velázquez, J.S.; Cavas, F.; Bolarín, J.M.; Alió, J.L. 3D Printed Personalized Corneal Models as a Tool for Improving Patient’s Knowledge of an Asymmetric Disease. Symmetry 2020, 12, 151. [Google Scholar] [CrossRef]
- Cavas-Martínez, F.; Piñero, D.; Fernández-Pacheco, D.; Mira, J.; Cañavate, F.; Alió, J. Assessment of Pattern and Shape Symmetry of Bilateral Normal Corneas by Scheimpflug Technology. Symmetry 2018, 10, 453. [Google Scholar] [CrossRef]
- Akhtar, S.; Almubrad, T.; Paladini, I.; Mencucci, R. Keratoconus corneal architecture after riboflavin/ultraviolet A cross-linking: Ultrastructural studies. Mol. Vis. 2013, 19, 1526. [Google Scholar] [PubMed]
- Choi, S.; Lee, S.-C.; Lee, H.-J.; Cheong, Y.; Jung, G.-B.; Jin, K.-H.; Park, H.-K. Structural response of human corneal and scleral tissues to collagen cross-linking treatment with riboflavin and ultraviolet A light. Lasers Med. Sci. 2012, 28, 1289–1296. [Google Scholar] [CrossRef] [PubMed]
- Jordan, C.; Patel, D.V.; Abeysekera, N.; McGhee, C.N.J. In Vivo Confocal Microscopy Analyses of Corneal Microstructural Changes in a Prospective Study of Collagen Cross-linking in Keratoconus. Ophthalmology 2014, 121, 469–474. [Google Scholar] [CrossRef] [PubMed]
- Uçakhan, Ö.Ö.; Bayraktutar, B. Morphology of the Corneal Limbus Following Standard and Accelerated Corneal Collagen Cross-Linking (9 mW/cm2) for Keratoconus. Cornea 2017, 36, 78–84. [Google Scholar] [CrossRef]
- Bradford, S.M.; Mikula, E.R.; Juhasz, T.; Brown, D.J.; Jester, J.V. Collagen fiber crimping following in vivo UVA-induced corneal crosslinking. Exp. Eye Res. 2018, 177, 173–180. [Google Scholar] [CrossRef]
- Müller, P.; Löffler, K.; Kohlhaas, M.; Holz, F.; Herwig-Carl, M. Morphologische Hornhautveränderungen nach Crosslinking bei Keratokonus. Klin. Mon. Für Augenheilkd. 2017, 235, 809–819. [Google Scholar] [CrossRef]
- Vinciguerra, P.; Albè, E.; Trazza, S.; Rosetta, P.; Vinciguerra, R.; Seiler, T.; Epstein, D. Refractive, Topographic, Tomographic, and Aberrometric Analysis of Keratoconic Eyes Undergoing Corneal Cross-Linking. Ophthalmology 2009, 116, 369–378. [Google Scholar] [CrossRef]
- Vinciguerra, P. Intraoperative and Postoperative Effects of Corneal Collagen Cross-linking on Progressive Keratoconus. Arch. Ophthalmol. 2009, 127, 1258. [Google Scholar] [CrossRef] [PubMed]
- Mazzotta, C.; Caporossi, T.; Denaro, R.; Bovone, C.; Sparano, C.; Paradiso, A.; Baiocchi, S.; Caporossi, A. Morphological and functional correlations in riboflavin UV A corneal collagen cross-linking for keratoconus. Acta Ophthalmol. 2010, 90, 259–265. [Google Scholar] [CrossRef] [PubMed]
- Toprak, I.; Yildirim, C. Scheimpflug Parameters after Corneal Collagen Crosslinking for Keratoconus. Eur. J. Ophthalmol. 2013, 23, 793–798. [Google Scholar] [CrossRef]
- De Bernardo, M.; Capasso, L.; Tortori, A.; Lanza, M.; Caliendo, L.; Rosa, N. Trans epithelial corneal collagen crosslinking for progressive keratoconus: 6 months follow up. Contact Lens Anterior Eye 2014, 37, 438–441. [Google Scholar] [CrossRef] [PubMed]
- Viswanathan, D.; Kumar, N.L.; Males, J.J.; Graham, S.L. Relationship of Structural Characteristics to Biomechanical Profile in Normal, Keratoconic, and Crosslinked Eyes. Cornea 2015, 34, 791–796. [Google Scholar] [CrossRef] [PubMed]
- Sedaghat, M.; Bagheri, M.; Ghavami, S.; Bamdad, S. Changes in corneal topography and biomechanical properties after collagen cross linking for keratoconus: 1-year results. Middle East Afr. J. Ophthalmol. 2015, 22, 212–219. [Google Scholar] [CrossRef]
- De Bernardo, M.; Capasso, L.; Lanza, M.; Tortori, A.; Iaccarino, S.; Cennamo, M.; Borrelli, M.; Rosa, N. Long-term results of corneal collagen crosslinking for progressive keratoconus. J. Optom. 2015, 8, 180–186. [Google Scholar] [CrossRef] [PubMed]
- Toprak, I.; Yaylali, V.; Yildirim, C. Visual, Topographic, and Pachymetric Effects of Pediatric Corneal Collagen Cross-linking. J. Pediatric Ophthalmol. Strabismus 2016, 54, 84–89. [Google Scholar] [CrossRef] [PubMed]
- Shafik Shaheen, M.; Lolah, M.M.; Piñero, D.P. The 7-Year Outcomes of Epithelium-Off Corneal Cross-linking in Progressive Keratoconus. J. Refract. Surg. 2018, 34, 181–186. [Google Scholar] [CrossRef]
| Mean (SD) Median (Range) | Preoperative | 3 Months Postop | 6 Months Postop | p-Value |
|---|---|---|---|---|
| LogMAR UDVA | 0.66 (0.58) 0.52 (0.01 to 2.00) | 0.56 (0.51) 0.40 (0.10 to 2.00) | 0.66 (0.68) 0.38 (0.00 to 2.00) | 0.449 |
| Manifest sphere (D) | −0.75 (2.48) 0.00 (−6.25 to 3.75) | −0.40 (2.57) 0.00 (−6.50 to 3.25) | −1.41 (2.80) −0.75 (−6.50 to 1.25) | 0.949 |
| Manifest cylinder (D) | −2.97 (2.00) −2.25 (−9.00 to −0.50) | −2.63 (1.35) −2.50 (−4.75 to 0.00) | −2.59 (1.06) −3.00 (−4.75 to −1.00) | 0.465 |
| Manifest SE (D) | −2.24 (2.47) −2.25 (−7.00 to 2.88) | −1.72 (2.48) −1.38 (−7.50 to 2.50) | −2.70 (2.61) −2.13 (−7.50 to 0.75) | 0.538 |
| LogMAR CDVA | 0.09 (0.09) 0.09 (−0.04 to 0.30) | 0.12 (0.11) 0.10 (0.00 to 0.32) | 0.10 (0.11) 0.02 (0.00 to 0.28) | 0.438 |
| CCT (µm) | 496.42 (34.99) 495.00 (444 to 564) | 461.92 (36.82) 463.00 (414 to 520) | 468.73 (46.69) 484.00 (377 to 531) | 0.006 Preop—3 months 0.006 3–6 months 0.084 Preop—6 months 0.024 |
| KM (D) | 45.44 (2.00) 44.88 (42.16 to 48.82) | 44.36 (3.01) 43.51 (39.49 to 49.26) | 45.47 (2.80) 45.62 (41.40 to 50.27) | 0.311 |
| Q45 | −0.58 (0.79) −0.39 (−2.00 to 0.51) | −0.96 (0.84) −1.08 (−2.17 to 0.36) | −0.81 (1.17) −0.40 (−2.91 to 1.12) | 0.846 |
| Q8 | −0.67 (0.32) −0.68 (−1.35 to −0.16) | −0.69 (0.47) −0.81 (−1.13 to 0.51) | −0.63 (0.38) −0.51 (−1.37 to −0.17) | 0.115 |
| Total RMS (µm) | 4.92 (3.73) 4.13 (1.07 to 14.39) | 3.65 (1.48) 4.09 (1.25 to 6.20) | 4.45 (3.27) 4.33 (1.04 to 13.65) | 0.115 |
| Astigmatism RMS (µm) | 2.74 (1.14) 2.91 (0.78 to 4.53) | 2.69 (1.17) 2.47 (0.73 to 4.14) | 2.68 (1.34) 3.04 (0.78 to 4.88) | 0.135 |
| Primary spherical aberration (µm) | 0.02 (0.36) 0.04 (−0.53 to 0.56) | −0.10 (0.30) −0.06 (−0.61 to 0.36) | 0.15 (0.37) 0.12 (−0.52 to 0.78) | 0.311 |
| Primary coma RMS (µm) | 1.64 (0.97) 1.49 (0.26 to 3.71) | 1.21 (0.95) 0.98 (0.04 to 3.05) | 1.53 (1.04) 1.45 (0.12 to 3.33) | 0.223 |
| Spherical-like RMS (µm) | 0.75 (0.36) 0.64 (0.23 to 1.51) | 0.67 (0.36) 0.57 (0.27 to 1.36) | 0.84 (0.47) 0.77 (0.18 to 1.65) | 0.030 Preop—3 months 0.015 3–6 months 0.084 Preop—6 months 0.722 |
| Coma-like RMS (µm) | 1.85 (1.00) 1.60 (0.38 to 3.98) | 1.65 (0.86) 1.41 (0.43 to 3.19) | 1.86 (0.88) 1.87 (0.59 to 3.05) | 0.057 |
| Mean (SD) Median (Range) | Preoperative | 3 Months Postop | 6 Months Postop | p-Value |
|---|---|---|---|---|
| Aant (mm2) | 43.27 (0.26) 43.22 (42.93 to 43.81) | 43.16 (0.27) 43.08 (42.85 to 43.79) | 43.24 (0.32) 43.15 (42.83 to 43.75) | 0.205 |
| Apost (mm2) | 44.60 (0.51) 44.41 (43.94 to 45.64) | 44.56 (0.57) 44.30 (44.00 to 45.83) | 44.72 (0.48) 44.49 (44.24 to 45.71) | 0.338 |
| Atot (mm2) | 104.07 (2.00) 103.50 (100.97 to 107.68) | 102.98 (2.26) 102.59 (100.30 to 106.79) | 103.87 (1.81) 102.95 (101.37 to 106.77) | 0.001 Preop—3 months 0.006 3–6 months 0.999 Preop—6 months 0.003 |
| CV (mm3) | 24.61 (1.95) 24.16 (21.38 to 27.97) | 22.65 (1.91) 22.81 (20.37 to 26.32) | 23.62 (1.89) 23.28 (19.70 to 26.87) | <0.001 Preop—3 months <0.001 3–6 months 0.816 Preop—6 months 0.003 |
| AAD (mm) | 0.0098 (0.0091) 0.0080 (0.0000 to 0.0400) | 0.0084 (0.0066) 0.0068 (0.0000 to 0.0200) | 0.0113 (0.0139) 0.0061 (0.000 to 0.0500) | 0.779 |
| PAD (mm) | 0.21 (0.09) 0.17 (0.10 to 0.39) | 0.23 (0.11) 0.20 (0.10 to 0.51) | 0.26 (0.13) 0.28 (0.09 to 0.45) | 0.076 |
| AMTPD (mm) | 1.04 (0.30) 1.03 (0.60 to 1.75) | 1.14 (0.39) 1.22 (0.30 to 1.93) | 1.28 (0.41) 1.29 (0.66 to 2.18) | 0.717 |
| PMTPD (mm) | 0.97 (0.27) 0.94 (0.58 to 1.62) | 1.08 (0.37) 1.16 (0.26 to 1.86) | 1.19 (0.41) 1.20 (0.60 to 2.08) | 0.717 |
| Mean (SD) Median (Range) | Preoperative | 3 Months Postop | 6 Months Postop | p-Value |
|---|---|---|---|---|
| VOLmct (mm3) | ||||
| Radius | ||||
| 0.1 mm | 0.015 (0.001) 0.015 (0.014–0.017) | 0.013 (0.001) 0.013 (0.011–0.015) | 0.014 (0.002) 0.014 (0.010–0.016) | 0.002 |
| 0.2 mm | 0.060 (0.004) 0.060 (0.054–0.069) | 0.054 (0.005) 0.053 (0.043–0.060) | 0.054 (0.007) 0.056 (0.042–0.063) | 0.002 |
| 0.3 mm | 0.134 (0.087) 0.134 (0.122–0.156) | 0.121 (0.012) 0.120 (0.098–0.136) | 0.122 (0.015) 0.126 (0.095–0.142) | 0.002 |
| 0.4 mm | 0.241 (0.016) 0.239 (0.217–0.279) | 0.216 (0.021) 0.214 (0.176–0.243) | 0.219 (0.026) 0.225 (0.170–0.253) | 0.002 |
| 0.5 mm | 0.377 (0.024) 0.374 (0.340–0.437) | 0.335 (0.034) 0.323 (0.277–0.381) | 0.344 (0.040) 0.353 (0.268–0.397) | 0.002 |
| 0.6 mm | 0.545 (0.035) 0.541 (0.491–0.631) | 0.490 (0.047) 0.485 (0.403–0.551) | 0.497 (0.057) 0.510 (0.391–0.574) | 0.002 |
| 0.7 mm | 0.745 (0.049) 0.738 (0.670–0.862) | 0.670 (0.064) 0.664 (0.555–0.753) | 0.680 (0.078) 0.697 (0.539–0.784) | 0.002 |
| 0.8 mm | 0.977 (0.064) 0.968 (0.877–1.130) | 0.880 (0.083) 0.871 (0.734–0.988) | 0.894 (0.100) 0.914 (0.709–1.030) | 0.002 |
| 0.9 mm | 1.243 (0.082) 1.230 (1.112–1.435) | 1.121 (0.104) 1.109 (0.941–1.256) | 1.139 (0.126) 1.162 (0.905–1.310) | 0.002 |
| 1.0 mm | 1.542 (0.103) 1.526 (1.378–1.779) | 1.392 (0.126) 1.376 (1.179–1.557) | 1.416 (0.154) 1.441 (1.128–1.626) | 0.002 |
| 1.1 mm | 1.877 (0.127) 1.855 (1.675–2.163) | 1.694 (0.151) 1.675 (1.449–1.891) | 1.725 (0.185) 1.753 (1.378–1.979) | 0.002 |
| 1.2 mm | 2.248 (0.153) 2.221 (2.000–2.588) | 2.030 (0.178) 2.004 (1.750–2.265) | 2.070 (0.218) 2.097 (1.653–2.371) | 0.002 |
| 1.3 mm | 2.655 (0.183) 2.623 (2.357–3.052) | 2.400 (0.206) 2.371 (2.085–2.685) | 2.449 (0.254) 2.477 (1.961–2.802) | 0.001 |
| 1.4 mm | 3.100 (0.215) 3.063 (2.747–3.558) | 2.803 (0.237) 2.769 (2.458–3.145) | 2.863 (0.292) 2.900 (2.297–3.267) | <0.001 |
| 1.5 mm | 3.585 (0.251) 3.542 (3.168–4.109) | 3.241 (0.269) 3.204 (2.873–3.647) | 3.317 (0.331) 3.369 (2.668–3.778) | <0.001 |
| VOLaap (mm3) | ||||
| Radius | ||||
| 0.1 mm | 0.016 (0.001) 0.016 (0.014–0.018) | 0.014 (0.001) 0.014 (0.012–0.016) | 0.014 (0.004) 0.015 (0.000–0.017) | <0.001 |
| 0.2 mm | 0.062 (0.004) 0.062 (0.056–0.071) | 0.057 (0.005) 0.057 (0.047–0.064) | 0.059 (0.006) 0.060 (0.047–0.067) | <0.001 |
| 0.3 mm | 0.140 (0.010) 0.140 (0.125–0.160) | 0.128 (0.011) 0.129 (0.107–0.144) | 0.132 (0.014) 0.135 (0.105–0.150) | <0.001 |
| 0.4 mm | 0.250 (0.017) 0.249 (0.223–0.284) | 0.228 (0.019) 0.229 (0.191–0.256) | 0.234 (0.024) 0.240 (0.188–0.268) | <0.001 |
| 0.5 mm | 0.391 (0.027) 0.390 (0.349–0.445) | 0.356 (0.030) 0.358 (0.300–0.401) | 0.367 (0.037) 0.375 (0.296–0.419) | <0.001 |
| 0.6 mm | 0.565 (0.039) 0.562 (0.503–0.643) | 0.514 (0.042) 0.515 (0.435–0.578) | 0.530 (0.053) 0.541 (0.430–0.604) | <0.001 |
| 0.7 mm | 0.770 (0.053) 0.767 (0.685–0.877) | 0.701 (0.057) 0.700 (0.597–0.789) | 0.722 (0.073) 0.737 (0.589–0.825) | <0.001 |
| 0.8 mm | 1.008 (0.069) 1.004 (0.897–1.149) | 0.917 (0.074) 0.914 (0.786–1.033) | 0.946 (0.094) 0.965 (0.774–1.080) | <0.001 |
| 0.9 mm | 1.280 (0.088) 1.274 (1.137–1.459) | 1.164 (0.093) 1.157 (1.005–1.312) | 1.201 (0.119) 1.222 (0.982–1.371) | <0.001 |
| 1.0 mm | 1.585 (0.109) 1.578 (1.406–1.807) | 1.441 (0.114) 1.430 (1.253–1.624) | 1.488 (0.147) 1.512 (1.215–1.699) | <0.001 |
| 1.1 mm | 1.925 (0.133) 1.916 (1.705–2.195) | 1.750 (0.137) 1.732 (1.533–1.974) | 1.807 (0.177) 1.834 (1.476–2.066) | <0.001 |
| 1.2 mm | 2.300 (0.159) 2.288 (2.034–2.624) | 2.089 (0.163) 2.062 (1.842–2.362) | 2.158 (0.211) 2.187 (1.759–2.466) | <0.001 |
| 1.3 mm | 2.710 (0.188) 2.695 (2.394–3.094) | 2.461 (0.190) 2.424 (2.184–2.784) | 2.544 (0.247) 2.574 (2.071–2.906) | <0.001 |
| 1.4 mm | 3.156 (0.220) 3.140 (2.784–3.604) | 2.867 (0.218) 2.821 (2.562–3.242) | 2.965 (0.286) 2.996 (2.407–3.392) | <0.001 |
| 1.5 mm | 3.642 (0.255) 3.621 (3.206–4.158) | 3.308 (0.248) 3.251 (2.984–3.743) | 3.422 (0.328) 3.453 (2.779–3.922) | 0.001 |
| VOLpap (mm3) | ||||
| Radius | ||||
| 0.1 mm | 0.015 (0.001) 0.015 (0.014–0.018) | 0.014 (0.001) 0.014 (0.011–0.016) | 0.014 (0.015) 0.014 (0.011–0.016) | <0.001 |
| 0.2 mm | 0.062 (0.004) 0.062 (0.056–0.070) | 0.056 (0.005) 0.056 (0.045–0.062) | 0.057 (0.006) 0.058 (0.044–0.065) | <0.001 |
| 0.3 mm | 0.139 (0.009) 0.140 (0.125–0.159) | 0.125 (0.011) 0.125 (0.102–0.140) | 0.129 (0.014) 0.131 (0.099–1.467) | <0.001 |
| 0.4 mm | 0.247 (0.016) 0.248 (0.222–0.282) | 0.223 (0.020) 0.221 (0.182–0.249) | 0.229 (0.024) 0.233 (0.178–0.261) | <0.001 |
| 0.5 mm | 0.386 (0.025) 0.389 (0.348–0.442) | 0.350 (0.031) 0.346 (0.286–0.389) | 0.359 (0.037) 0.365 (0.280–0.409) | 0.002 |
| 0.6 mm | 0.557 (0.037) 0.561 (0.501–0.638) | 0.505 (0.045) 0.499 (0.416–0.561) | 0.518 (0.053) 0.527 (0.407–0.591) | 0.002 |
| 0.7 mm | 0.761 (0.050) 0.765 (0.682–0.870) | 0.690 (0.061) 0.681 (0.572–0.767) | 0.707 (0.073) 0.722 (0.560–0.807) | 0.002 |
| 0.8 mm | 0.997 (0.066) 1.001 (0.893–1.141) | 0.904 (0.078) 0.892 (0.755–1.006) | 0.927 (0.094) 0.949 (0.740–1.057) | 0.002 |
| 0.9 mm | 1.267 (0.084) 1.271 (1.133–1.449) | 1.148 (0.098) 1.132 (0.966–1.280) | 1.178 (0.120) 1.209 (0.949–1.344) | 0.001 |
| 1.0 mm | 1.570 (0.105) 1.574 (1.401–1.795) | 1.423 (0.120) 1.403 (1.207–1.588) | 1.461 (0.147) 1.504 (1.186–1.667) | 0.001 |
| 1.1 mm | 1.908 (0.128) 1.911 (1.670–2.181) | 1.729 (0.143) 1.704 (1.479–1.933) | 1.776 (0.178) 1.828 (1.454–2.027) | 0.001 |
| 1.2 mm | 2.281 (0.154) 2.283 (2.027–2.608) | 2.067 (0.169) 2.037 (1.785–2.315) | 2.124 (0.211) 2.180 (1.749–2.424) | 0.001 |
| 1.3 mm | 2.690 (0.182) 2.690 (2.385–3.075) | 2.438 (0.196) 2.401 (2.121–2.733) | 2.507 (0.246) 2.567 (2.062–2.862) | 0.001 |
| 1.4 mm | 3.136 (0.214) 3.133 (2.774–3.586) | 2.842 (0.225) 2.797 (2.492–3.186) | 2.926 (0.283) 2.989 (2.402–3.334) | 0.001 |
| 1.5 mm | 3.620 (0.250) 3.614 (3.196–4.138) | 3.280 (0.256) 3.228 (2.911–3.683) | 3.379 (0.328) 3.445 (2.765–3.854) | 0.001 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alifa, R.; Piñero, D.; Velázquez, J.; Alió del Barrio, J.L.; Cavas, F.; Alió, J.L. Changes in the 3D Corneal Structure and Morphogeometric Properties in Keratoconus after Corneal Collagen Crosslinking. Diagnostics 2020, 10, 397. https://doi.org/10.3390/diagnostics10060397
Alifa R, Piñero D, Velázquez J, Alió del Barrio JL, Cavas F, Alió JL. Changes in the 3D Corneal Structure and Morphogeometric Properties in Keratoconus after Corneal Collagen Crosslinking. Diagnostics. 2020; 10(6):397. https://doi.org/10.3390/diagnostics10060397
Chicago/Turabian StyleAlifa, Ramón, David Piñero, José Velázquez, Jorge L. Alió del Barrio, Francisco Cavas, and Jorge L. Alió. 2020. "Changes in the 3D Corneal Structure and Morphogeometric Properties in Keratoconus after Corneal Collagen Crosslinking" Diagnostics 10, no. 6: 397. https://doi.org/10.3390/diagnostics10060397
APA StyleAlifa, R., Piñero, D., Velázquez, J., Alió del Barrio, J. L., Cavas, F., & Alió, J. L. (2020). Changes in the 3D Corneal Structure and Morphogeometric Properties in Keratoconus after Corneal Collagen Crosslinking. Diagnostics, 10(6), 397. https://doi.org/10.3390/diagnostics10060397








