Programmed Death Ligand 1: A Poor Prognostic Marker in Endometrial Carcinoma
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Histological Examination
2.3. Immunohistochemistry (IHC) Staining Method
2.4. Evaluation of Antibodies Staining
2.5. Statistical Analysis
3. Results
3.1. Demographic Data
3.2. PD-L1 Expression Analysis
3.2.1. Endometrial Carcinoma versus Non-Neoplastic Endometrium
3.2.2. Age (≥60 versus <60 Years)
3.2.3. Ethnic Groups
3.2.4. Type 1 versus Type 2 Endometrial Carcinoma
3.2.5. Grade and Stage of Tumour
3.2.6. Survival (Alive versus Died of Disease)
3.2.7. Types of Therapy
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- National Cancer Institute: SEER Stat Fact Sheets: Endometrial Cancer. Available online: https://seer.cancer.gov/statfacts/html/corp.html (accessed on 12 November 2019).
- The Malaysian National Cancer Registry Report (MNCR) 2007–2011. 2016. Available online: https://kpkesihatan.com/2016/12/07/the-malaysian-national-cancer-registry-report-mncr-2007-2011/ (accessed on 15 February 2020).
- Longoria, T.C.; Eskander, R.N. Immunotherapy in endometrial cancer-an evolving therapeutic paradigm. Gynecologic oncology research and practice. Gynecol. Oncol. Res. Pract. 2015, 2, 11. [Google Scholar] [CrossRef] [PubMed]
- Lortet-Tieulent, J.; Ferlay, J.; Bray, F.; Jemal, A. International Patterns and Trends in Endometrial Cancer Incidence, 1978–2013. J. Natl. Cancer Inst. 2018, 110, 354–361. [Google Scholar] [CrossRef] [PubMed]
- Bokhman, J.V. Two pathogenetic types of endometrial carcinoma. Gynecol. Oncol. 1983, 15, 10–17. [Google Scholar] [CrossRef]
- Setiawan, V.W.; Yang, H.P.; Pike, M.C.; McCann, S.E.; Yu, H.; Xiang, Y.B.; Wolk, A.; Wentzensen, N.; Weiss, N.S.; Webb, P.M.; et al. Type I and II endometrial cancers: Have they different risk factors? J. Clin. Oncol. 2013, 31, 2607–2618. [Google Scholar] [CrossRef] [PubMed]
- Lacey, J.V., Jr.; Sherman, M.E.; Rush, B.B.; Ronnett, B.M.; Ioffe, O.B.; Duggan, M.A.; Glass, A.G.; Richesson, D.A.; Chatterjee, N.; Langholz, B. Absolute risk of endometrial carcinoma during 20-year follow-up among women with endometrial hyperplasia. J. Clin. Oncol. 2010, 28, 788–792. [Google Scholar] [CrossRef]
- Mo, Z.; Liu, J.; Zhang, Q.; Chen, Z.; Mei, J.; Liu, L.; Yang, S.; Li, H.; Zhou, L.; You, Z. Expression of PD-1, PD-L1 and PD-L2 is associated with differentiation status and histological type of endometrial cancer. Oncol. Lett. 2016, 12, 944–950. [Google Scholar] [CrossRef]
- Fleming, G.F.; Brunetto, V.L.; Cella, D.; Look, K.Y.; Reid, G.C.; Munkarah, A.R.; Kline, R.; Burger, R.A.; Goodman, A.; Burks, R.T. Phase III trial of doxorubicin plus cisplatin with or without paclitaxel plus filgrastim in advanced endometrial carcinoma: A Gynecologic Oncology Group Study. J. Clin. Oncol. 2004, 22, 2159–2166. [Google Scholar] [CrossRef]
- Pectasides, D.; Pectasides, E.; Economopoulos, T. Systemic therapy in metastatic or recurrent endometrial cancer. Cancer Treat. Rev. 2007, 33, 177–190. [Google Scholar] [CrossRef]
- Pardoll, D.M. The blockade of immune checkpoints in cancer immunotherapy. Nat. Rev. Cancer 2012, 12, 252–264. [Google Scholar] [CrossRef]
- Dong, H.; Zhu, G.; Tamada, K.; Chen, L. B7-H1, a third member of the B7 family, co-stimulates T-cell proliferation and interleukin-10 secretion. Nat. Med. 1999, 5, 1365–1369. [Google Scholar] [CrossRef]
- Philips, G.K.; Atkins, M. Therapeutic uses of anti-PD-1 and anti-PD-L1 antibodies. Int. Immunol. 2014, 27, 39–46. [Google Scholar] [CrossRef] [PubMed]
- Massi, D.; Brusa, D.; Merelli, B.; Ciano, M.; Audrito, V.; Serra, S.; Buonincontri, R.; Baroni, G.; Nassini, R.; Minocci, D.; et al. PD-L1 marks a subset of melanomas with a shorter overall survival and distinct genetic and morphological characteristics. Ann. Oncol. 2014, 25, 2433–2442. [Google Scholar] [CrossRef]
- Iacovelli, R.; Nolè, F.; Verri, E.; Renne, G.; Paglino, C.; Santoni, M.; Rocca, M.C.; Giglione, P.; Aurilio, G.; Cullurà, D.; et al. Prognostic role of PD-L1 expression in renal cell carcinoma. A systematic review and meta-analysis. Target. Oncol. 2016, 11, 143–148. [Google Scholar] [CrossRef] [PubMed]
- McLaughlin, J.; Han, G.; Schalper, K.A.; Carvajal-Hausdorf, D.; Pelekanou, V.; Rehman, J.; Velcheti, V.; Herbst, R.; LoRusso, P.; Rimm, D.L. Quantitative assessment of the heterogeneity of PD-L1 expression in non–small-cell lung cancer. JAMA Oncol. 2016, 2, 46–54. [Google Scholar] [CrossRef] [PubMed]
- Herzog, T.; Arguello, D.; Reddy, S.; Gatalica, Z. PD-1 and PD-L1 expression in 1599 gynecological malignancies: Implications for immunotherapy. Gynecol Oncol. 2015, 137, 204–205. [Google Scholar] [CrossRef]
- Pinto, A.; Mackrides, N.; Nadji, M. PD-L1 Expression in carcinosarcomas of the gynecologic Tract: A potentially actionable biomarker. Appl. Immunohistochem. Mol. Morphol. 2018, 26, 393–397. [Google Scholar] [CrossRef] [PubMed]
- Vanderstraeten, A.; Luyten, C.; Verbist, G.; Tuyaerts, S.; Amant, F. Mapping the immunosuppressive environment in uterine tumors: Implications for immunotherapy. Cancer Immunol. Immun. 2014, 63, 545–557. [Google Scholar] [CrossRef] [PubMed]
- Gandini, S.; Massi, D.; Mandalà, M. PD-L1 expression in cancer patients receiving anti PD-1/PD-L1 antibodies: A systematic review and meta-analysis. Crit. Rev. Oncol. Hemat. 2016, 100, 88–98. [Google Scholar] [CrossRef]
- Allred, D.C.; Clark, G.M.; Elledge, R.; Fuqua, S.A.; Brown, R.W.; Chamness, G.C.; Osborne, C.K.; McGuire, W.L. Association of p53 protein expression with tumor cell proliferation rate and clinical outcome in node-negative breast cancer. J. Natl. Cancer Inst. 1993, 85, 200–206. [Google Scholar] [CrossRef]
- Liu, C.Q.; Xu, J.; Zhou, Z.G.; Jin, L.L.; Yu, X.J.; Xiao, G.; Lin, J.; Zhuang, S.M.; Zhang, Y.J.; Zheng, L. Expression patterns of programmed death ligand 1 correlate with different microenvironments and patient prognosis in hepatocellular carcinoma. Br J Cancer. 2018, 119, 80–88. [Google Scholar] [CrossRef]
- Birtalan, E.; Danos, K.; Gurbi, B.; Brauswetter, D.; Halasz, J.; Kalocsane Piurko, V.; Acs, B.; Antal, B.; Mihalyi, R.; Pato, A.; et al. Expression of PD-L1 on immune cells shows better prognosis in laryngeal, oropharygeal, and hypopharyngeal cancer. Appl. Immunohistochem. Mol. Morphol. 2018, 26, e79–e85. [Google Scholar] [CrossRef] [PubMed]
- Drakes, M.L.; Mehrotra, S.; Aldulescu, M.; Potkul, R.K.; Liu, Y.; Grisoli, A.; Joyce, C.; O’Brien, T.E.; Stack, M.S.; Stiff, P.J. Stratification of ovarian tumor pathology by expression of programmed cell death-1 (PD-1) and PD-ligand- 1 (PD-L1) in ovarian cancer. J. Ovarian Res. 2018, 11, 43. [Google Scholar] [CrossRef]
- Pulko, V.; Harris, K.J.; Liu, X.; Gibbons, R.M.; Harrington, S.M.; Krco, C.J.; Kwon, E.D.; Dong, H. B7-h1 expressed by activated CD8 T-cells is essential for their survival. J Immunol. 2011, 187, 5606–5614. [Google Scholar] [CrossRef]
- Marinelli, O.; Annibali, D.; Aguzzi, C.; Tuyaerts, S.; Amant, F.; Morelli, M.B.; Santoni, G.; Amantini, C.; Maggi, F.; Nabissi, M. The Controversial Role of PD-1 and Its Ligands in Gynecological Malignancies. Front. Oncol. 2019, 9, 1073. [Google Scholar] [CrossRef] [PubMed]
- Sungu, N.; Yildirim, M.; Desdicioglu, R.; Başaran Aydoğdu, Ö.; Kiliçarslan, A.; Tatli Doğan, H.; Kiliç Yazgan, A.; Akyol, M.; Erdoğan, F. Expression of immunomodulatory molecules PD-1, PD-L1, and PD-L2, and their relationship with clinicopathologic characteristics in endometrial cancer. Int. J. Gynecol. Pathol. 2019, 38, 404–413. [Google Scholar] [CrossRef] [PubMed]
- Panawala, L. Difference between Monoclonal and Polyclonal Antibodies. 2017. Available online: https://www.researchgate.net/publication/320707210_Difference_Between_Monoclonal_and_Polyclonal_Antibodies (accessed on 15 February 2020).
- Wang, X.; Teng, F.; Kong, L.; Yu, J. PD-L1 expression in human cancers and its association with clinical outcomes. Onco Targets Ther. 2016, 9, 5023. [Google Scholar]
- Wang, Q.; Liu, F.; Liu, L. Prognostic significance of PD-L1 in solid tumor: An updated meta-analysis. Medicine 2017, 96, e6369. [Google Scholar] [CrossRef]
- Zhang, S.; Minaguchi, T.; Xu, C.; Qi, N.; Itagaki, H.; Shikama, A.; Tasaka, N.; Akiyama, A.; Sakurai, M.; Ochi, H.; et al. PD-L1 and CD4 are independent prognostic factors for overall survival in endometrial carcinomas. BMC Cancer 2020, 20, 127. [Google Scholar] [CrossRef]
- Engerud, H.; Berg, H.F.; Myrvold, M.; Halle, M.K.; Bjorge, L.; Haldorsen, I.S.; Hoivik, E.A.; Trovik, J.; Krakstad, C. High degree of heterogeneity of PD-L1 and PD-1 from primary to metastatic endometrial cancer. Gynecol. Oncol. 2020, 157, 260–267. [Google Scholar] [CrossRef]
- Yamazawa, K.; Seki, K.; Matsui, H.; Kihara, M.; Sekiya, S. Prognostic factors in young women with endometrial carcinoma: A report of 20 cases and review of literature. Int. J. Gynecol. Cancer. 2000, 10, 212–222. [Google Scholar] [CrossRef]
- Kanopiene, D.; Vidugiriene, J.; Valuckas, K.P.; Smailyte, G.; Uleckiene, S.; Bacher, J. Endometrial cancer and microsatellite instability status. Open Med. (Wars) 2014, 10, 70–76. [Google Scholar] [CrossRef] [PubMed]
- Kythreotou, A.; Siddique, A.; Mauri, F.A.; Bower, M.; Pinato, D.J. PD-L1. J. Clin. Pathol. 2018, 71, 189–194. [Google Scholar] [CrossRef] [PubMed]
- Nishimura, H.; Nose, M.; Hiai, H.; Minato, N.; Honjo, T. Development of lupus-like autoimmune diseases by disruption of the PD-1 gene encoding an ITIM motif-carrying immunoreceptor. Immunity 1999, 11, 141–151. [Google Scholar] [CrossRef]
- Couzin-Frankel, J. Breakthrough of the year 2013. Cancer Immunother. Sci. 2013, 342, 1432–1433. [Google Scholar]
- Cancer Research Institute. Available online: https://www.cancerresearch.org/scientists/immuno-oncology-landscape/fda-approval-timeline-of-active-immunotherapies (accessed on 12 January 2020).
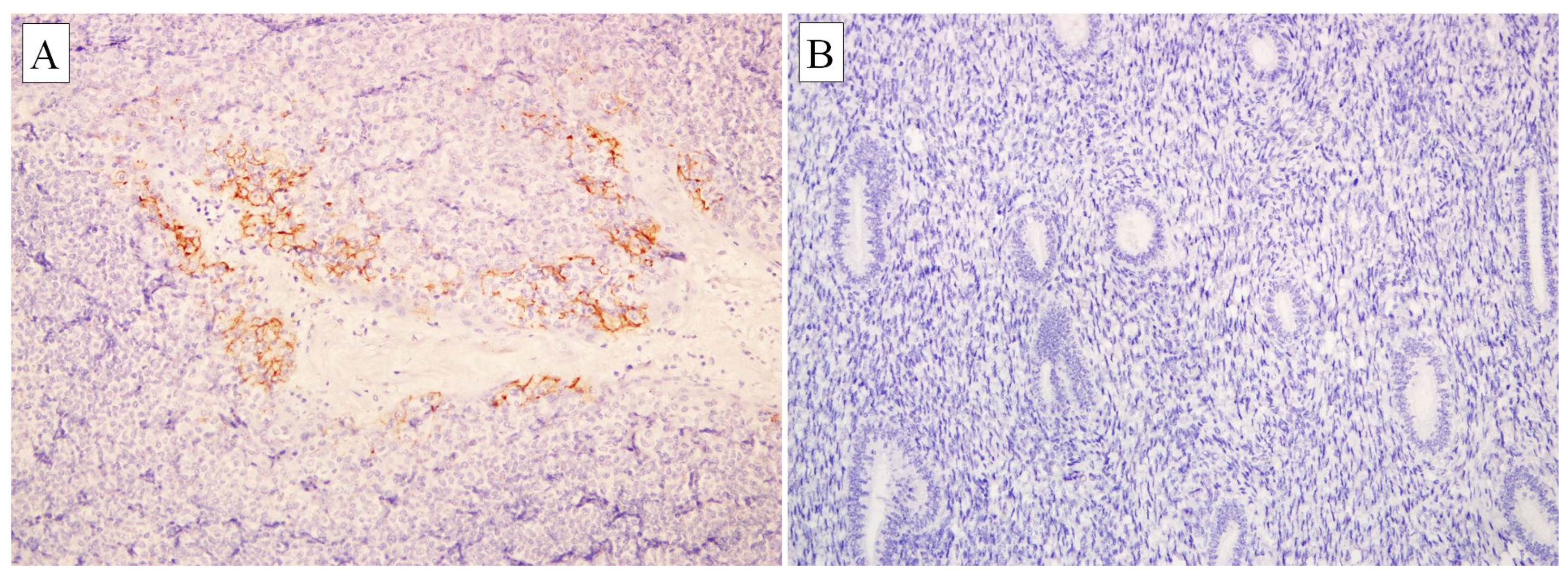
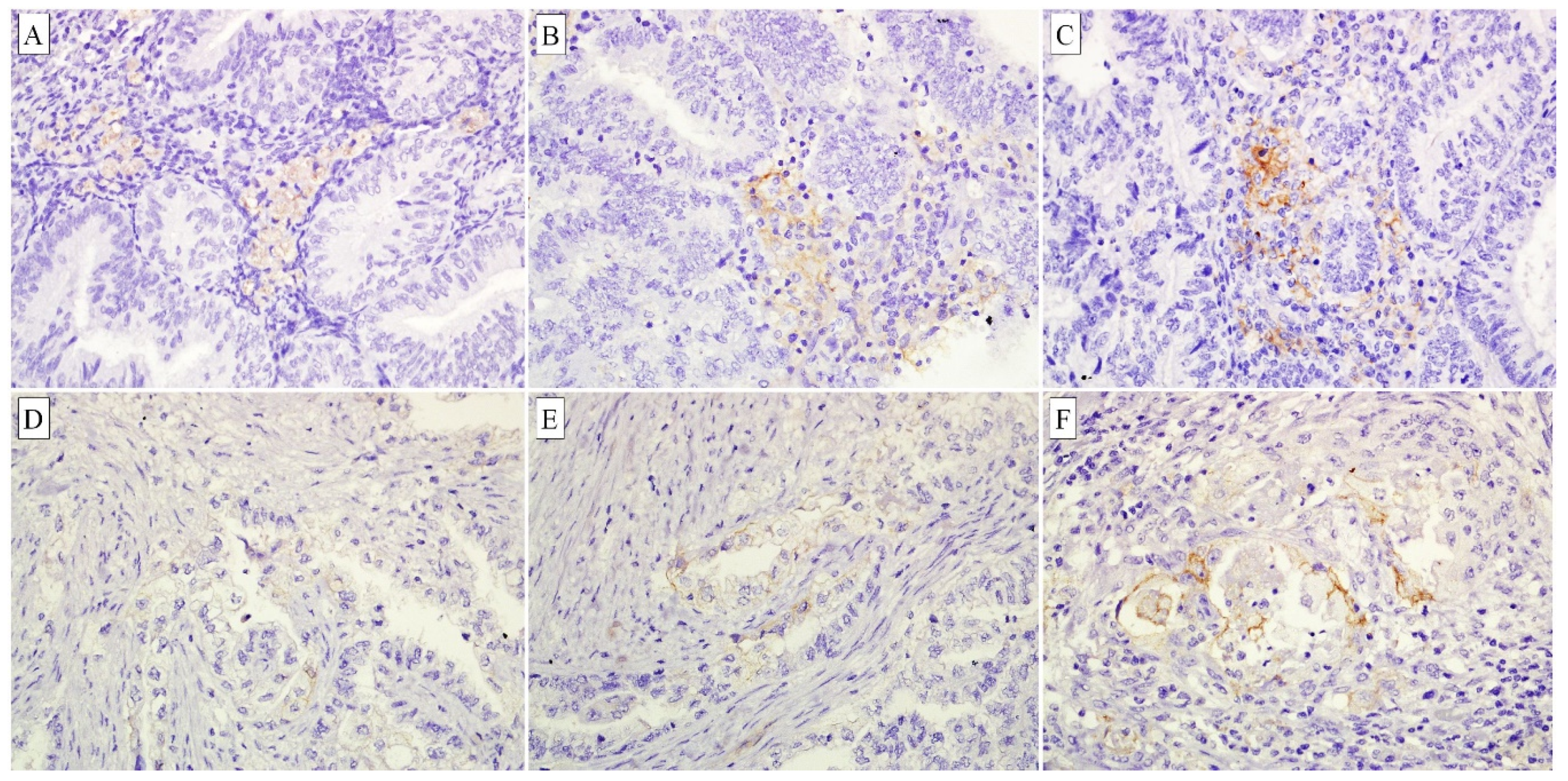
| Parameters | n (%) | PD-L1 Tumour Cells | p Value | PD-L1 Immune Cells | p Value | |
|---|---|---|---|---|---|---|
| Age | Mean ± SD (years) | 53.5 ± 12.0 | ||||
| <60 | 36 (61) | 7 (19.4) | 0.047 * | 24 (66.7) | 0.432 | |
| ≥60 | 23 (39) | 10 (43.5) | 13 (56.5) | |||
| Race | Malay | 32 (54.2) | 10 (31.3) | 19 (59.4) | ||
| Chinese | 15 (25.4) | 5 (33.3) | 0.575 | 10 (66.7) | 0.847 | |
| Indian | 12 (20.3) | 2 (16.7) | 8 (66.7) | |||
| Types of endometrial carcinoma | Endometrioid | 51 (86.4) | ||||
| Serous | 3 (5.1) | |||||
| Clear cell | 2 (3.4) | |||||
| Mixed Mucinous | 2 (3.4) 1 (1.7) | |||||
| Type 1 Type 2 | 52 (88.1) 7 (11.9) | 14 (26.9) 3 (42.9) | 0.40 | 33 (63.5) 4 (57.1) | 1.0 | |
| Grade | 1 | 34 (57.6) | 5 (14.7) | 18 (52.9) | ||
| 2 | 9 (15.3) | 4 (44.4) | 0.01 * | 8 (88.9) | 0.038 * | |
| 3 | 16 (27.1) | 8 (50) | 11 (68.8) | |||
| Stage | 1 | 41 (69.5) | 10 (24.4) | 25 (61.0) | ||
| 2 | 7 (11.9) | 2 (28.6) | 0.57 | 4 (57.1) | 0.512 | |
| 3 | 6 (10.2) | 3 (50) | 6 (100) | |||
| 4 | 5 (8.5) | 2 (40) | 2 (40) | |||
| Survival | Alive | 44 | 7 (15.9) | 0.001 * | 27 (61.4) | 0.767 |
| DOD | 15 | 10 (66.7) | 10 (66.7) | |||
| PD-L1 positive | Immune cells | 37 (62.7) | ||||
| Tumour cells | 17 (37.3) |
| Types | Total Number of Cases (n) | PD-L1 Positive in Immune Cells n (%) | p Value | PD-L1 Positive in Tumour Cells n (%) |
|---|---|---|---|---|
| Endometrial carcinoma | 59 | 37 (62.7) | <0.001 * | 17 (28.8) |
| Non-neoplastic endometrium | 32 | 0 (0) | NA |
| Types of Tumour/Grading | No. | Immune Cells | Tumour Cells | Stage | Survival Status | ||
|---|---|---|---|---|---|---|---|
| Positive (%) | Intensity | Positive (%) | Intensity | ||||
| Type I | |||||||
| Endometroid G1 | 1 | 0 | - | 10 | Moderate | 1 | DOD |
| 2 | 0 | - | 1 | Moderate | 1 | Alive | |
| 3 | 0 | - | 0 | - | 2 | Alive | |
| 4 | 0 | - | 0 | - | 1 | Alive | |
| 5 | 0 | - | 0 | - | 1 | Alive | |
| 6 | 35 | Moderate | 0 | - | 1 | Alive | |
| 7 | 0 | - | 0 | - | 1 | Alive | |
| 8 | 10 | Strong | <1 | Strong | 1 | Alive | |
| 9 | 10 | Moderate | 0 | - | 1 | Alive | |
| 10 | <1 | Weak | 1 | Strong | 3 | Alive | |
| 11 | 0 | - | 0 | - | 1 | Alive | |
| 12 | 0 | - | 0 | - | 1 | Alive | |
| 13 | 0 | - | 0 | - | 1 | Alive | |
| 14 | 0 | - | 0 | - | 1 | Alive | |
| 15 | 1 | Strong | 0 | - | 1 | Alive | |
| 16 | 5 | Strong | 0 | - | 1 | Alive | |
| 17 | 0 | - | 0 | - | 2 | Alive | |
| 18 | <1 | Weak | 0 | - | 1 | Alive | |
| 19 | 10 | Strong | 0 | - | 1 | Alive | |
| 20 | 5 | Weak | 0 | - | 1 | Alive | |
| 21 | 10 | Strong | 0 | - | 1 | Alive | |
| 22 | 1 | Strong | 1 | Strong | 1 | Alive | |
| 23 | <1 | Strong | 0 | - | 1 | Alive | |
| 24 | 0 | - | 0 | - | 1 | Alive | |
| 25 | 2 | Strong | 0 | - | 1 | Alive | |
| 26 | 40 | Strong | 0 | - | 2 | Alive | |
| 27 | 0 | - | 0 | - | 1 | Alive | |
| 28 | 0 | - | 0 | - | 1 | Alive | |
| 29 | 0 | - | 0 | - | 1 | Alive | |
| 30 | <1 | Moderate | 0 | - | 2 | Alive | |
| 31 | 2 | Strong | 0 | - | 3 | Alive | |
| 32 | 15 | Strong | 0 | - | 1 | Alive | |
| 33 | 0 | - | 0 | - | 1 | Alive | |
| G2 | 1 | 30 | Strong | 10 | Strong | 4 | DOD |
| 2 | 1 | Moderate | 10 | Moderate | 2 | Alive | |
| 3 | 10 | Strong | 10 | Strong | 1 | DOD | |
| 4 | 1 | Moderate | 10 | Moderate | 1 | DOD | |
| 5 | <1 | Weak | 0 | - | 2 | Alive | |
| 6 | 0 | - | 0 | - | 4 | DOD | |
| 7 | 35 | Moderate | 0 | - | 1 | Alive | |
| 8 | <1 | Moderate | 0 | - | 1 | Alive | |
| 9 | 5 | Weak | 0 | - | 2 | DOD | |
| G3 | 1 | 15 | Moderate | 10 | Strong | 1 | DOD |
| 2 | 0 | - | <1 | Weak | 1 | Alive | |
| 3 | 5 | Strong | 30 | Strong | 1 | DOD | |
| 4 | <1 | Weak | 0 | - | 3 | Alive | |
| 5 | <1 | Weak | 0 | - | 1 | Alive | |
| 6 | 0 | - | <1 | Weak | 1 | Alive | |
| 7 | 1 | Weak | 0 | - | 1 | Alive | |
| 8 | 10 | Strong | 1 | Weak | 4 | DOD | |
| 9 | 5 | Strong | 0 | - | 1 | Alive | |
| Mucinous | 1 | 0 | - | 0 | - | 1 | Alive |
| Type II | |||||||
| Serous | 1 | <1 | Weak | <1 | Strong | 3 | DOD |
| 2 | 0 | - | 0 | - | 4 | DOD | |
| 3 | 0 | - | 0 | - | 4 | DOD | |
| Clear cell | 1 | 1 | Weak | 0 | - | 1 | Alive |
| 2 | 15 | Strong | <1 | Strong | 3 | DOD | |
| Mixed | 1 | 1 | Moderate | 0 | - | 1 | DOD |
| 2 | 0 | - | 2 | Moderate | 2 | DOD | |
| References | PD-L1 Expression | ||
|---|---|---|---|
| Tumour-infiltrating Immune Cells | Tumour Cells | Normal Endometrium | |
| Present study (2019) | 62.7% | 28.8% | 0% |
| Vanderstraeten et al., 2014 [19] | NA | 70–80% | 81% |
| Mo et al., 2016 [8] | 60% | 17.3% | 14.3% |
| Sungu et al., 2019 [27] | 36.2% | 36.2% | NA |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chew, M.; Wong, Y.P.; Karim, N.; Mustangin, M.; Alfian, N.; Tan, G.C. Programmed Death Ligand 1: A Poor Prognostic Marker in Endometrial Carcinoma. Diagnostics 2020, 10, 394. https://doi.org/10.3390/diagnostics10060394
Chew M, Wong YP, Karim N, Mustangin M, Alfian N, Tan GC. Programmed Death Ligand 1: A Poor Prognostic Marker in Endometrial Carcinoma. Diagnostics. 2020; 10(6):394. https://doi.org/10.3390/diagnostics10060394
Chicago/Turabian StyleChew, Mianxin, Yin Ping Wong, Norain Karim, Muaatamarulain Mustangin, Nurwardah Alfian, and Geok Chin Tan. 2020. "Programmed Death Ligand 1: A Poor Prognostic Marker in Endometrial Carcinoma" Diagnostics 10, no. 6: 394. https://doi.org/10.3390/diagnostics10060394
APA StyleChew, M., Wong, Y. P., Karim, N., Mustangin, M., Alfian, N., & Tan, G. C. (2020). Programmed Death Ligand 1: A Poor Prognostic Marker in Endometrial Carcinoma. Diagnostics, 10(6), 394. https://doi.org/10.3390/diagnostics10060394





